RP 10 - organic
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
what is RP10
preparation of a pure organic solid, test of its purity, and preparation of a pure organic liquid
what is reflux
continuous boiling & condensing of a mixture
whens reflux used
to heat an organic mixture without losing reactants or products
purpose of anti bumping granuels
prevent vigorous or uneven boiling - by making small bubbles form instead of large ones
separating funnel purpose
to separate into two layers
higher density liquid (typically aqueous) = bottom layer
organic product layer = top layer
purifying organic liquid steps
put distillate of impure product in separating funnel
wash product
allow layers to separate, then run and discard aqueous layer
run organic liquid into conical flask
add 3 spatulas of drying agent
decant into distillation flask
distil to collect pure product
ways to wash the product (step 2)
sodium hydrogencarbonate solution, shaking and releasing pressure from CO2 produced - removes acid impurities by neutralisation
saturated NaCl solution - helps separate organic from aqueous layer
what should the drying agent be (step 5)
insoluble in the organic liquid and not react with it
what happens when the drying agent is added and examples
examples = anhydrous sodium sulphate, anhydrous calcium chloride
absorbs remaining water
liquid remains cloudy until all water drops have been removed
testing with melting point
pure = sharp, same as in quoted data books
impure = lower and melt over a range of degrees
measuring melting point ways
electronic melting point machine
practical set up - capillary tube strapped to a thermometer immersed in some heating oil
what happens in both cases
small amount of the salt put into capillary tube
heated up - slowly near melting point
compare experimentally determined value with one quoted in a data source
where can an error occur
if the temperature on the thermometer is not the same as the temperature in the sample tube
measuring boiling point to determine purity
distillation or boiling tube of sample in a heating oil bath
note pressure - changing pressure can change the boiling point
whys it not as accurate
several substances may have the same boiling point
how to get accurate measure of boiling point
thermometer should be above the level of the surface of the boiling liquid, and be measuring the temperature of the saturated vapour
recrystallisation method
dissolve impure compound in minimum volume of hot solvent
filter hot to remove insoluble purities
cool by inserting beaker in ice to recrystallise
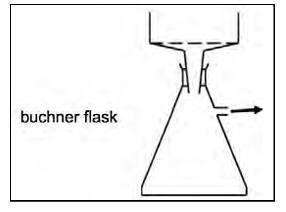
suction filtrate with buchner flask to separate crystals
wash crystals with distilled water
dry crystals between absorbent paper
whats an appropriate solvent (step 1)
dissolve both compound and impurities when hot
compound itself does not dissolve when cold
why is the minimum volume added (step 2)
to obtain saturated solution, and enable crystallisation on cooling
what does both do
means the desired compound is pure in the crystals formed
what does hot filtering do (step 2)
removes insoluble impurities
the heat prevents crystals forming during filtration
whys it cooled (step 3)
to increase the yield by ensuring all of the compound crystallises
what does the water pump in a buchner flask do (step 4)
reduces pressure and speeds up filtration
why are the crystals washed with distilled water (step 5)
to remove soluble impurities
why is water removed (step 6)
it would affect % yield
buchner flask diagram
how is yield lost
crystals lost when filtering or washing
some product stays in solution after recrystallisation
other side reactions occurring