Chemistry Ch. 9
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
mixtures be
heterogenous and/or homogenous
What does a homogenous mixture have
solutions and colloids
Solutions are
Light transparency
DOES NOT separate
Nonfilterable
Colloids are
Opaque to light (murky)
DOES NOT separate
Nonfilterable
Colloids ex
milk, smoke, blood
Solvents (major component of mixture) are usually liquid, but sometimes
not a liquid!
REMEMBER: ____ dissolves ____
like, like
stronger the attraction between solute and solvent
greater solubility
Dilution means
add solvent (water)
strong electrolyte
completely dissociate into ions when dissolved → conduct electricity

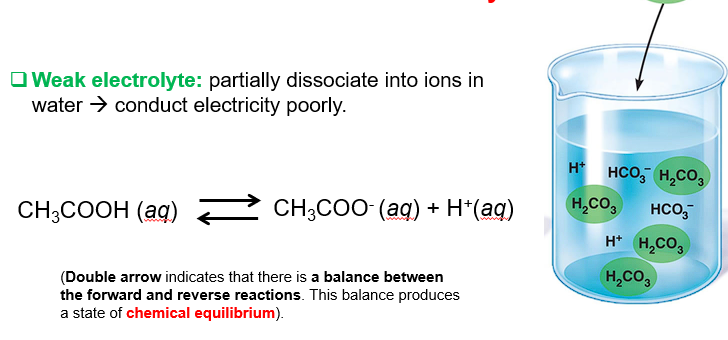
Weak electrolyte
Partially dissociate into ions → poorly conduct electricity
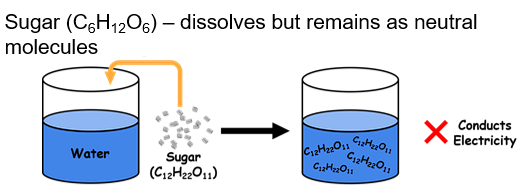
Non-electrolyte
DO NOT dissociate into ions when in water → NO electricity (ex: sugar, ethanol, acetone)
Solution properties (list)
Vapor pressure lowering
Boiling point elevation
Freezing point depression
Osmotic pressure
Vapor pressure lowering
Boiling point elevation
Freezing point depression
Osmolarity
Particle concentration
Osmotic Pressure
minimum pressure (outside force) needed to stop osmosis
Unit for osmolarity
osmol
What’s the point of osmosis?
maintain cell shape/function
Titration
Adding one solution slowly of known concentration (titrant) to known volume of another solution of unknown concentration until neutralization reached