CHEM Chapter 2 Study Help
1/107
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
108 Terms
What are atoms?
the smallest units of an element
How many atoms make up an element?
1
A compound contains…
the atoms of 2 or more different elements
Dalton’s Atomic Theory is that…
atoms don’t change identity in chemical reactions only how they’re joined does
What is the law of constant composition?
all samples of a substance contain the same elements in the same proportion by mass
What is the law of multiple proportions?
same elements make more than one compound → the mass of one combined with the fixed mass of the other are in a ratio of small whole numbers
What is the law of conservation of mass?
mass does not change (or can’t be detected to change) when a chemical reaction occurs
Name the subatomic particles!
electrons, protons & neutrons
Electrons are…
negatively charged
Protons are…
positively charged
Neutrons…
have no charge
What is the relative charge & mass of an electron?
relative charge: 1-
relative mass: 0
What is the relative charge & mass of a proton?
relative charge: 1+
relative mass: 1
What is the relative charge & mass of a neutron?
relative charge: 0
relative mass: 1
What does the nucleus of an atom contain?
the mass & a high positive charge (protons)
What does the “empty” space of an atom contain?
electrons
The charge of a proton is __ to the charge of an electron
=
Protons & neutrons make up about ____ of the nuclear mass respectively.
half
The atomic number is the number of ______ in the nucleus of the atom
protons
What determines the identity of an element?
# of protons
What symbol represents the atomic number?
Z
The mass number is the sum of the # of _____ + _____ in the nucleus.
protons; neutrons
What symbol represents the mass number?
A
Isotopes are…
atoms of an element that have nuclei containing different #’s of neutrons
What symbol is used to identify isotopes? (hint: this is not a formula, I just don’t have any other way to position it TT)
(A/Z)X
What does the X represent when used to identify isotopes?
the letter(s) symbol for the element (ex. H, He)
Z is often omitted (not included) from the symbol for isotopes. (T/F)
True
Ions are…
the resulting charged particles that come from atoms gaining or losing electrons
Cations have a ______ charge & form when an atom _____ one or more electrons.
positive; loses
Anions have a ______ charge & form when an atom _____ one or more electrons.
negative; gains
If an element has ions, where would the symbol for that be located?
in superscript following the element, represented as a number followed by a charge (+/-)
What happened to this calcium atom: Ca2+?
it lost 2 electrons
What happened to this chlorine atom: Cl-?
it gained 1 electron
How do you find the charge for an ion?
subtracting the electrons from the protons (p+ - e-)
What symbol represents the atomic mass unit?
u
The atomic mass unit is….
1/12 the mass of one 12C atom; the mass of a single proton/neutron
What is the atomic mass unit of one 12C atom?
12 u
What is the atomic mass unit of one 24Mg atom?
about 24 u
What is the atomic mass unit of one 4He atom?
4 u
When does the atomic mass unit become the mass number?
when rounded to a whole #
A ____________ measures the masses & abundances of isotopes.
mass spectrometer
Is the relative abundance of an element’s abundance consistent throughout nature?
Yes
Isotopic mass is…
the mass of a particular isotope of an element
To calculate the atomic mass, find the…
weighted average mass of the element’s natural isotopes
The formula for atomic mass is…
[(percentage of isotope A) x (isotopic mass A)] + (percentage of isotope B) x (isotopic mass B)] + …..
_______ & _______ both separately proposed the periodic table.
Dmitri Mendeleev; Lothar Meyer
On the periodic table elements with similar properties are arranged in the same ______.
column
The horizontal row on the periodic table is referred to as the…
period
The vertical column on the periodic table is referred to as the…
group or family
A metal is…
an element that’s shiny & conducts electricity well
Where are metal elements located on the periodic table?
the center & left
A nonmetal is…
an element that’s usually a nonconductor (doesn’t conduct electricity)
Where are nonmetal elements located on the periodic table?
the top right
A metalloid is…
an element that has both metal & nonmetal properties
Where are metalloid elements located on the periodic table?
the staircase separating metals & nonmetals
What groups are representative elements in?
A groups
What groups are transition metals in?
B groups
What groups are inner transition metals in?
lanthanides & actinides (2 groups at the bottom of the periodic table)
Alkali metals are _______ in group ____
soft, reactive metals; 1A
Alkaline earth metals are _______ in group ____
; 2A
Halogens are _______ in group ____
reactive nonmetals; 7A
Noble gases are _______ in group ____
stable, largely inert (nonreactive) gases; 8A
A molecule is…
a combo of atoms (usually nonmetals) joined so strongly they act as 1 particle
How many atoms do diatomic molecules have?
2
Homonuclear diatomics contain _____
2 of the same atom
Heteronuclear diatomics contain _____
2 different atoms
If all atoms are the same in a molecule the substance is a(n) _____
element
If 2 or more elements form a molecule it is a(n) _____
molecular compound
_________ form molecular compounds.
nonmetals
A molecular formula gives the…
# of every type of atom in a molecule
A structural formula shows…
how the atoms connect in the molecule
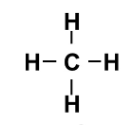
structural formula
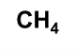
molecular formula
What is the molecular mass?
the sum of the atomic masses of all atoms represented in the molecular formula
How is molecular mass expressed?
atomic mass units (u)
What are the 7 homonuclear molecular compounds?
oxygen, nitrogen, fluorine, iodine, chlorine, bromine, hydrogen
Ionic compounds consist of a _______ _______ and a ______ ______
metal cation (+); nonmetal anion (-)
What formula is used for ionic compounds?
the empirical formula
All ionic compounds are initially ______.
neutral
Which groups on the periodic table have cations?
1A, 2A, some of 3B, and Aluminum in 3A
Which groups on the periodic table have anions?
7A, 6A, and Nitrogen in 5A
What charge does group 1A have?
1+
What charge does group 2A have?
2+
What charge does group 3A & 3B have?
3+
What charge does group 7A have?
1-
What charge does group 6A have?
2-
What charge does group 5A (Nitrogen) have?
3-
A polyatomic ion is…
a group of atoms with net charge & behave as a single particle
Show me ammonium.
NH4+
Show me nitrate.
NO3-
Show me carbonate.
CO32-
Show me phosphate.
PO42-
Show me sulfate.
SO42-
Show me sulfite.
SO32-
Show me hydroxide.
OH-
Chemical nomenclature is…
the organized system for naming compounds
For ionic compounds: the name of the ______ comes first, followed by the name of the _____
cation; anion
For ionic compounds: the ending of the element name for the ______ is changed to ______
anion; -ide
What shows the charge for metals that have more than one cation (usually transition metals)?
roman numerals in parentheses
What polyatomic ion represents ammonium?
NH4+