U1 Chemistry of Life
1/63
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
4 elements required to build biological molecules (carbs, proteins, lipids, nucleic acids)
Oxygen (O)
Carbon (C)
Hydrogen (H)
Nitrogen (N)
Element in proteins and nucleic acids
Nitrogen (N)
Element in nucleic acids and some lipids
Phosphorus (P)
Smallest units of an element
Atoms made of protons (+) , neutrons (O), and electrons (-)
Trace elements
Elements required by an organism only in very small quantities (Fe, I, and Cu)
Isotopes & Radiometric dating
Atoms with same number of protons but different amount of neutrons
Radiometric dating = artifacts dated by examining the rate of decay of carbon-14 in it
Chemical bonds
Atoms of a compound are held by:
Ionic bonds
Covalent bonds
Hydrogen bonds
Ionic bonds
When electrons are transferred from one atom to another
Ions = the atoms become negatively or positively charged depending on which one lost or gained the electron
Covalent bonds
Nonpolar covalent bonds = electrons are shared equally between atoms
Polar covalent bonds = Electrons are shared unequally between atoms
Single covalent bond = 1 pair of electrons is shared between 2 atoms
Double covalent bond = 2 pairs of electrons are shared
Triple covalent bond = 3 pairs of electrons are shared
Bonds in water
Hydrogen bonds
Electrons are unequally shared (Oxygen is more electronegative) (polar)
Negative oxygen of one water is attracted to the positive hydrogens
Hydrogen bonds
Intermolecular attraction
Weak that form when H atom covalently bonded to an electronegative atom is also attracted to another electronegative atom
Strong in large numbers
Great solvent to dissolve things
Special properties of water molecules because of hydrogen bonds
Cohesion and adhesion
Surface tension
High heat capacity (takes more energy to increase temperature of water than others)
Expansion on freezing
Cohesion
Water’s strong tendency to stick together
Adhesion
Water’s tendency to stick to other substances
(ex: two glass cups stuck together by the film of water between them)
Capillary action
Water’s ability to rise up the roots, trunks, and branches of trees (up the xylem)
Works because of both cohesion and adhesion
Surface tension
Water molecules stick together because of cohesion and let light things sit atop the surface
Acidic solutions
If you dissolve an acid in water, it will release many hydrogen ions (H+)
Has lots of hydrogen ions
Basic solutions (Alkalines)
If you add it to water, it will release many hydroxide ions (OH-)
Don’t release hydrogen ions (H+)
Slippery
pH scale
Measures the acidity or alkalinity of a solution
1 through 14
1 = acidic
7 = neutral
14 = basic
Formula for pH
pH = -log [H+]
H+ and pH have an inverse relationship
pH graphing/scaling
Logarithmic
pH = 3 is 10 times more acidic than pH = 4
1 pH increase = x10 decrease in hydrogen ion concentration
Organic compounds vs Inorganic compounds
Organic: Chemical compounds that contain a skeleton of carbon atoms surrounded by hydrogen atoms and other elements
Inorganic: Molecules that don’t have both carbon and hydrogen
Properties of carbon
Can bind with other carbons, N, O, and H
Monomers vs Polymers
Monomers: Individual parts that make up macromolecules that have chains of monomers
Polymers: Macromolecules that are made up of repeating monomers
Dehydration Synthesis (condensation reaction)
Forms polymers
Water molecule is lost to build a larger compound
Hydrolysis
Polymers broken down into monomers
Water is added to separate the two monomers
Carbohydrates
(CH2O)n —→ C, H, and O usually in a 1:2:1 ratio
Categorized as either monosaccharides, disaccharides, or polysaccharides
Saccharides = sugar
Monosaccharides
Simplest sugars and energy sources for cells
Most common: glucose, fructose, galactose, ribose, deoxyribose
Glucose (C6H12O6)
Most abundant monosaccharide
6 carbon sugar
Used in cellular respiration to convert into energy and in photosynthesis for plant food
Structure: Either a 6-cabon ring with many OHs and Hs OR a straight-chain with a carbon backbone and Hs and OHs attached to the side
Fructose
Monosaccharide
Common sugar in fruits
Structure: Either a 6-carbon ring with some OHs OR a straight-chain with a carbon backbone with OHs and Hs attached to the side
How to number carbons on rings/chains
Rings: First carbon is attached to 2 Oxygens (glucose) OR CH2OH which is to the bottom right of the lone oxygen (then clockwise)
Chains: First carbon is closest to the side with the double bond to O
Glycosidic Linkage
When 2 monosaccharides are joined by dehydration synthesis (-H of sugar combines with -OH from another sugar)
Creates a disaccharide
Common disaccharides
Maltose = two glucose molecules
Sucrose = table sugar
Lactose = in dairy products
Polysaccharides
Made of repeated units of monosaccharides (Branched or unbranched chains)
Common: Starch, cellulose, glycogen
Glycogen and Starch
Sugar storage molecules
Glycogen = sugar in animals
Starch = sugar in plants
Cellulose
Made of β-glucose
Function: structural support in cell walls of plants
Chitin
Polymer of β-glucose molecules
Structural molecule in walls of fungus and in the exoskeletons of arthropods (bugs)
Protein
Functions: structure, function, and regulation of tissues and organs
Monomers: Amino acids
Amino Acids
Monomers of proteins
Made of C, H, O, and N atoms
20 different common amino acids
Structure:
Central carbon
Amino group (-NH2)
Carboxyl group (-COOH)
Hydrogen
R-group
R-groups (side-chains) of amino acids
Vary in:
Composition (C, H, O, N, and S)
Polarity (polar, nonpolar)
Charge (o, +, -)
Shape (long, short, ring)
Affects whether it’s hydrophobic or hydrophilic
3 categories of amino acids
1) Hydrophobic (nonpolar & uncharged)
2) Hydrophilic (polar & uncharged)
3) Ionic (polar & charged)
Dipeptide
2 amino acids joined (carboxyl group to an amino group)
Joined by a peptide bond
Polypeptide
Made of a string of amino acids
1st stage before it is twisted and folded to make a 3D protein
New amino acids are always added on the carboxyl end of the chain
Peptide termini
N-terminus/amino terminus = end of the peptide with an amino group
C-terminus/carboxyl terminus = end with a carboxyl group
Generally, all peptides have an N- and C-terminus
Protein stages
1) primary structure - linear sequence of the amino acids
2) secondary structure - polypeptide twists
3) tertiary structure - folds 3D
4) quaternary structure (sometimes) - multiple polypeptide chain interactions to make a protein
Primary structure of a protein
Linear sequence of amino acids in a polypeptide chain
Secondary structure of a protein
Polypeptide:
Twists (forms a coil called an alpha helix)
OR
Zigzags (pattern called beta-pleated sheets)
Depends on the different R-groups interacting with each other
Tertiary structure of a protein
Far away amino acids interact with each other (because the helix or sheet structure brings different groups closer together)
Often locked into a stable 3D shape
Hydrophobic amino acids are on the inside of the protein
Hydrophilic are on the outside
Covalent disulfide bonds between two cysteine amino acids stabilize it sometimes
Covalent disulfide bonds
A bond between two cysteine amino acids that sometimes occurs to stabilize the tertiary structure of a protein.
Only cysteine and methionine have sulfur in their R-groups (and methionine is almost always the start amino acid)
Quaternary structure of a protein
Interaction between multiple different polypeptide chains (almost always in tertiary structure)
Incorrectly folded proteins
Only proteins that have folded correctly into a 3D structure can perform their intended function
Mistakes in amino acid chain can create nonfunctional, differently shaped proteins
Chaperone proteins (chaperonins)
Proteins that sometimes help other proteins fold properly and more efficiently
Lipids (elements, types, and function)
Consist of C, H, and O atoms in different ratios
Monomer: basically fatty acids (but technically lipids doesn’t have one)
Common examples: triglycerides, phospholipids, and steroids
Function: Structural components of cell membranes because they’re nonpolar, insulation, signaling, and energy storage
Triglycerides
Makes up fat storage in tissue
Made of a glycerol molecule (backbone) with 3 fatty acid chains attached to it
Fatty acid chain = Long chain of carbons where each carbon is covered in hydrogen with a carboxyl group end on one side
Saturated fatty acids
Hydrogens along its carbon chain or with a few gaps where double bonds replace a hydrogen
NO double bonds
Tend to form solids at room temp.
Generally linear molecules to be tightly packed (butter)
Unsaturated (monounsaturated) fatty acids
One double bond in the carbon chain
Tend to be liquid at room temp. if cis-double bonds (solid if not)
More kinked in structure so cannot pack as tightly (oil)
Unsaturated (polyunsaturated) fatty acids
Has many double bonds within the fatty acid
More double bonds = more unsaturation
Tend to be liquid at room temp. if cis-double bonds (solid of not)
More kinked in structure so cannot pack as tightly (oil)
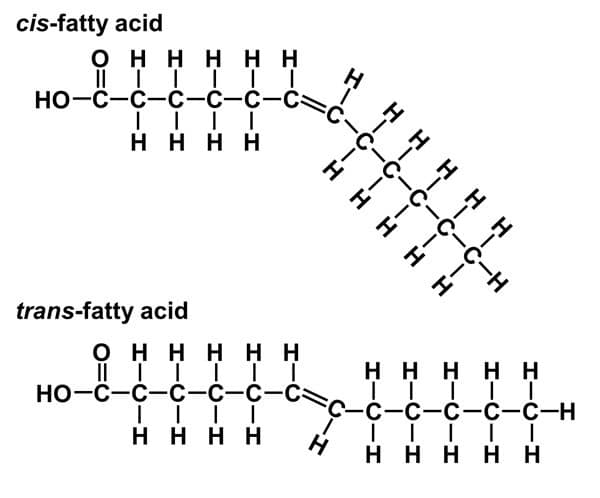
Cis-double bonds vs Trans-double bonds
Cis-double bonds = The hydrogens are either both above or both below the 2 double bonded carbons to which they are connected
Cis-double bonds are more kinked in their structure (around a 30 degree bend)
Trans-double bonds = The hydrogens are on opposite sides of the chain where they attach to the 2 double bonded carbons
Phospholipids
2 fatty acid “tails” = hydrophobic
Nonpolar —→ also don’t mix well with polar water
1 negatively charged phosphate “head” = hydrophilic
- drawn to + end of water
Amphipathic molecule = 1 side is hydrophilic and 1 is hydrophobic
Cholesterol
Four-ringed molecule and lipid in some membranes
Function: Increases membrane fluidity EXCEPT at high temps where it holds things together instead
Also helps make certain hormones
Nucleic acids [elements, monomers, types]
Elements: C, H, O, N (line proteins) AND Phosphorus
Monomers: nucleotides
Types: deoxyribonucleic acid (DNA) and ribonucleic acid (RNA)
DNA structure and function
Structure: Phosphate group, Adenine/Cytosine/Thymine/Guanine (nitrogenous base), and deoxyribose (5-carbon sugar without OH)
Function: Contains hereditary blueprints of all life
RNA structure and function
Structure: Phosphate group, Adenine/Cytosine/Uracil/Guanine (nitrogenous base), and ribose (5-carbon sugar including OH)
Function: Essential for protein synthesis
Elements in the 4 macromolecules
All contain C, H, and O
N = Proteins and nucleic acids
S = Proteins
P = Nucleic acids and sometimes lipids