Chem/phys day 1
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
what equation would you use for this question:
What is the magnitude of the electric field used in the MALDI-MS imaging device described in the passage?
E = V/d; voltage/distance
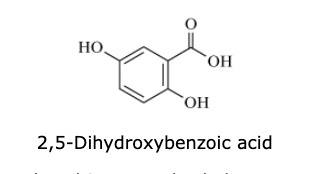
In MALDI, a plate containing the sample is coated with 2,5-dihydroxybenzoic acid (DHB) (structure shown).
Which reaction leads to the formation of DHB whose structure is shown in the passage?
carboxylation of hydroquinone
when trying to find organic chemistry questions always do what first
look at the functional groups
What property must be different between ions in order for them to separate? (MALDI-MS)
their mass-to-charge ratios
what am i solving for in this scenario:
what is one of the values of the electromagnetic energy delivered during one pulse by the ionizing radiation?
work
original equation: power = w/t
work = PT
proteolytic cleavage is what type of reaction
a hydrolysis
photon energy is directly proportional to
frequency. + bond energy (ΔH) = + frequency
Based on the ray diagram and distances shown in Figure 1, the focal length of the lens is:
1/f = 1/do + 1/d1
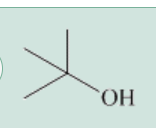
what is this?
a tertiary alcohol
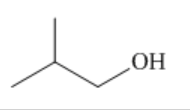
what is this?
a primary alcohol
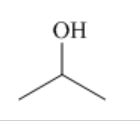
what is this?
a secondary alcohol
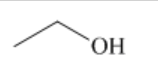
what is this?
a primary alcohol
d
d
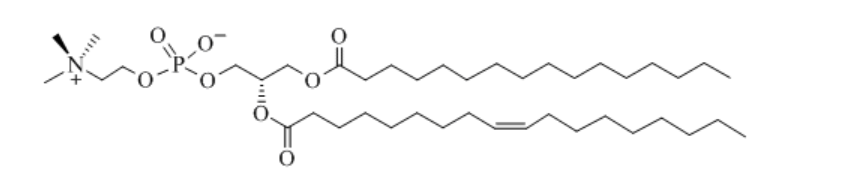
what is this -
a phospholipid specifically a phosphatide
can be difficult to detect since they do not absorb visible light
liposomes
The inside of a liposome is most similar to what type of environment?
aqueous
what does this situation imply:
Compound 2 formed new liposomes with an average size
Compound 2 is under thermodynamic control
what does this imply:
Compound 1 were stable to mixing
Compound 1 is under kinetic control
Kinetic control
product forms fastest, may get “stuck”
Thermodynamic control =
system can rearrange to the most stable, lowest-energy state
Assume the hydrolysis of ATP proceeds with ΔG′° = –30 kJ/mol.
ATP + H2O → ADP + Pi
Which expression gives the ratio of ADP to ATP at equilibrium, if the [Pi] = 1.0 M? (Note: Use RT = 2.5 kJ/mol.)
what equation would you use for this
ΔG∘= - RTlnK
What is the electronic configuration of the Co(II) center found in vitamin B12?
[Ar] 3d⁷
Which cation is most likely to be found in place of Fe(II) in the square planar binding domain of hemoglobin?
Co²⁺
The ATP-dependent phosphorylation of a protein target is catalyzed by which class of enzyme?
transferase
Colored molecules usually have:
more conjugation (many connected double bonds)
KM is
when the kinetics experiment reaches half the maximum velocity
kcat →
how fast the enzyme catalyzes the reaction (turnover rate)
Tm →
thermal stability (higher = more stable)
the bond that links monosaccharides together in an oligosaccharide is a special type of acetal linkage known as a
glycoside bond
what kind of curve is Michaelis–Menten
hyperbolic
relating voltage, current, and resistance
ohms law. V = IR
Which amino acids CAN hydrogen bond?
Polar / charged: Ser, Thr, Asn, Gln, Asp, Glu, Lys, Arg, His
What does kcat measure?
Catalytic turnover (rate) → how fast enzyme converts substrate → product
What does Tm measure?
Protein stability (folding) → temperature where protein unfolds
If mutations ↓ kcat MUCH more than Km → what’s affected?
Catalysis > binding
Hybridization change in nucleophilic attack on carbonyl?
Why?
sp2→sp3→sp2
carbonyl (sp²)
nucleophile attacks → tetrahedral (sp³)
collapse → carbonyl reforms (sp²)
What does base (NaOH) do in extraction?
Deprotonates acids → charged → aqueous layer
Where do neutral molecules go?
organic layer (ether)
Why does amide stay in organic layer?
it is neutral and not easily protonated/deprotonated
What type of ions replace Fe²⁺ best?
Transition metals with similar charge/size
What makes a compound colored?
Extended conjugation (delocalized electrons)
More conjugation → what happens?
absorbs visible light → appears colored
To measure kcat, substrate concentration should be:
saturating to ensure enzyme is always working at max rate
1 mM × 1 mL =
μmol
0.1 mM × 1 mL =
0.1 μmol = 100 nmol
μM/s → what does it mean?
μmol/L/s
2 μM/s for 60 s → concentration change?
2×60=120μM
Then multiply by volume (1 mL = 10⁻³ L):
120μM×10-3 = 1.2 μM×10-7
1 μmol =
10⁻⁶ mol
1 nmol =
10⁻⁹ mol
0.1 μmol = ? nmol
100 nmol
1 × 10⁻⁷ mol = ? nmol
100 nmol
1 mL = ? L
10−3L