Electrophilic Addition Reactions of Alkenes & Oxidation and Cyclopropanation Reactions of Alkenes
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
H — X
X = Cl, Br, I
Markovnikov orientation
Rearrangement can occur
Hydrohalogenation
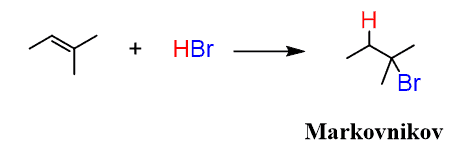
Hydrohalogenation
H — H (H2)
metal
Metal = Pt or Pd or Ni
Syn stereochemistry
No rearrangement
Catalytic Hydrogenation
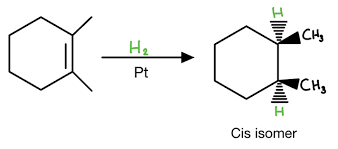
Catalytic Hydrogenation
X—X (X2) & H2O
X = Cl, Br
Markovnikov orientation
anti stereochemistry
No rearrangement
and enantiomer
Halohydrin Formation
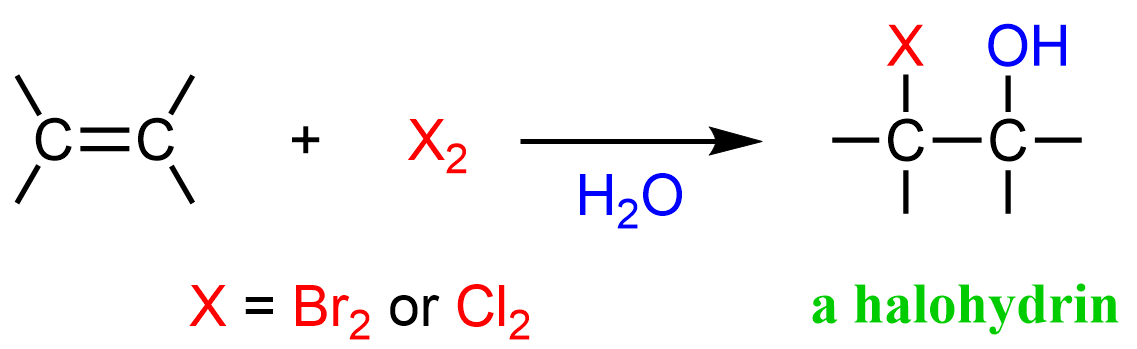
Halohydrin Formation
X—X (X2)
X = Cl, Br
anti stereochemistry
no rearrangement
and enantiomer
Halogenation
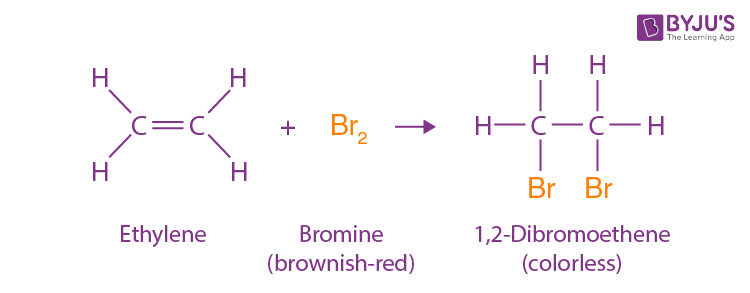
Halogenation
BH3 / THF
H2O2 / NaOH
anti-Markovnikov orientation
syn stereochemistry
no rearrangement
and enantiomer
Hydroboration-oxidation
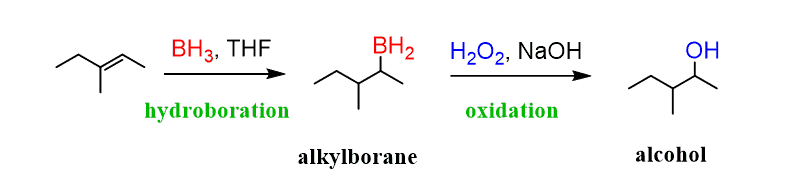
Hydroboration-oxidation
H—OR / Hg(OAc)2
NaBH4
Markovnikov orientation
no rearrangement
Alkoxymercuration-demurcuration
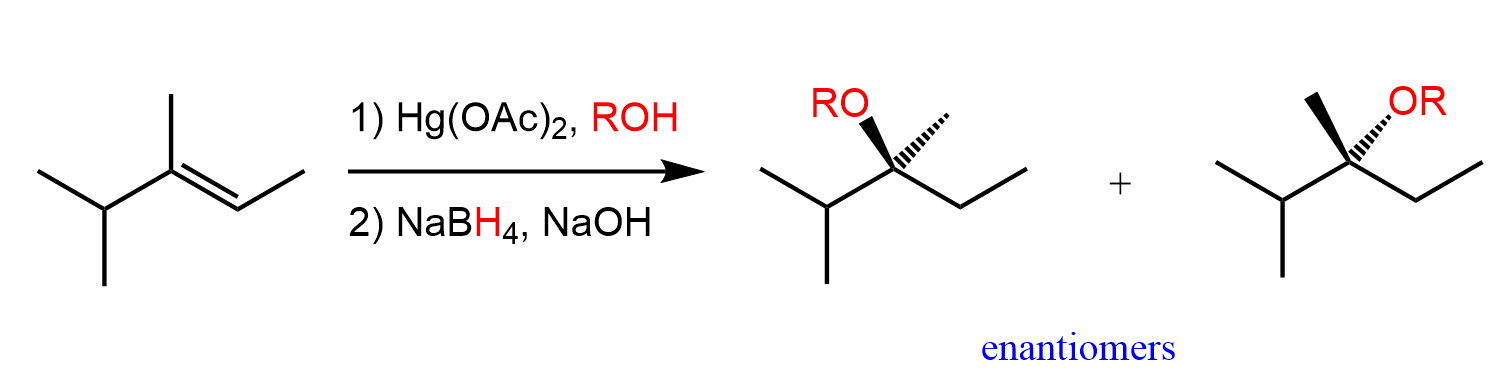
Alkoxymercuration-demurcuration
Hg(OAc)2, H2O
NaBH4
Markovnikov orientation
no rearrangement
Oxymercuration-demercuration
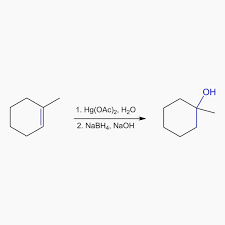
Oxymercuration-demercuration
H2O / H+ catalyst
Markovnikov orientation
rearrangement can occur
Hydration
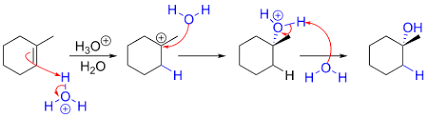
Hydration
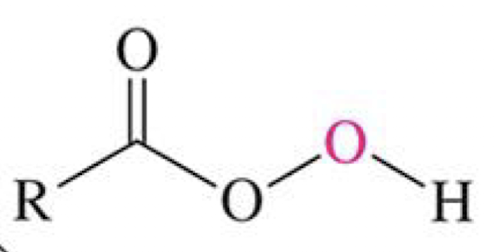
(in photo)
syn stereochemistry
no rearrangement
and enantiomer
Epoxidation
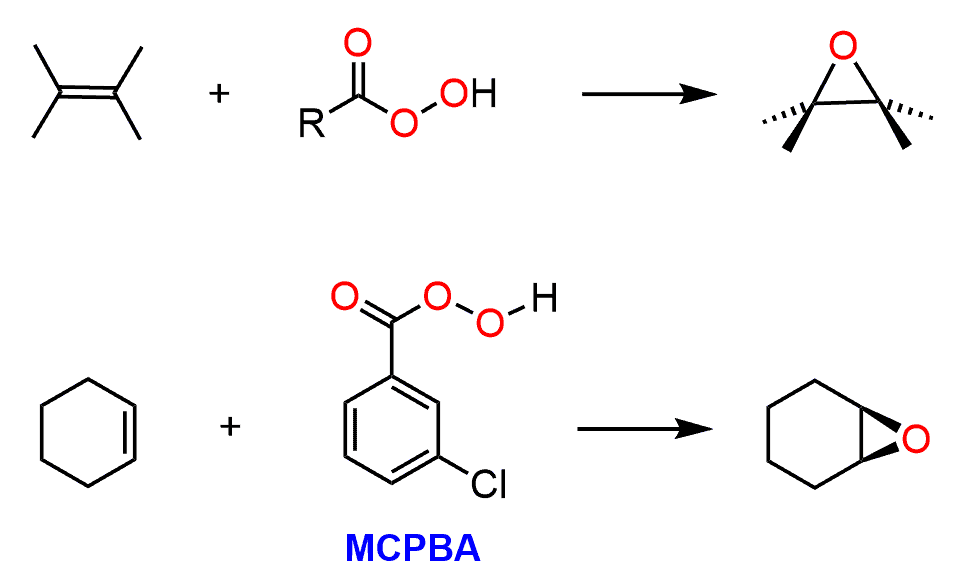
Epoxidation
CHBr3 / KOH and H2O
syn stereochemistry
no rearrangement
and enantiomer
Cyclopropanation by alpha elimination
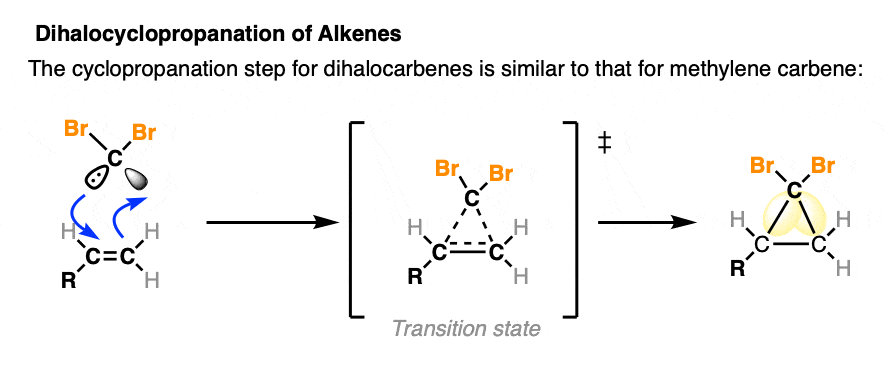
Cyclopropanation by alpha elimination
CH2I2, Zn, CuCl
syn stereochemistry
no rearrangement
and enantiomer
Simmons-Smith
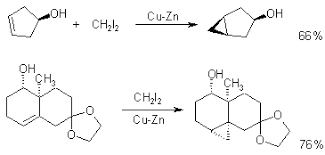
Simmons-Smith
O3
(CH3)2S
produces ketones and aldehydes
Ozonolysis
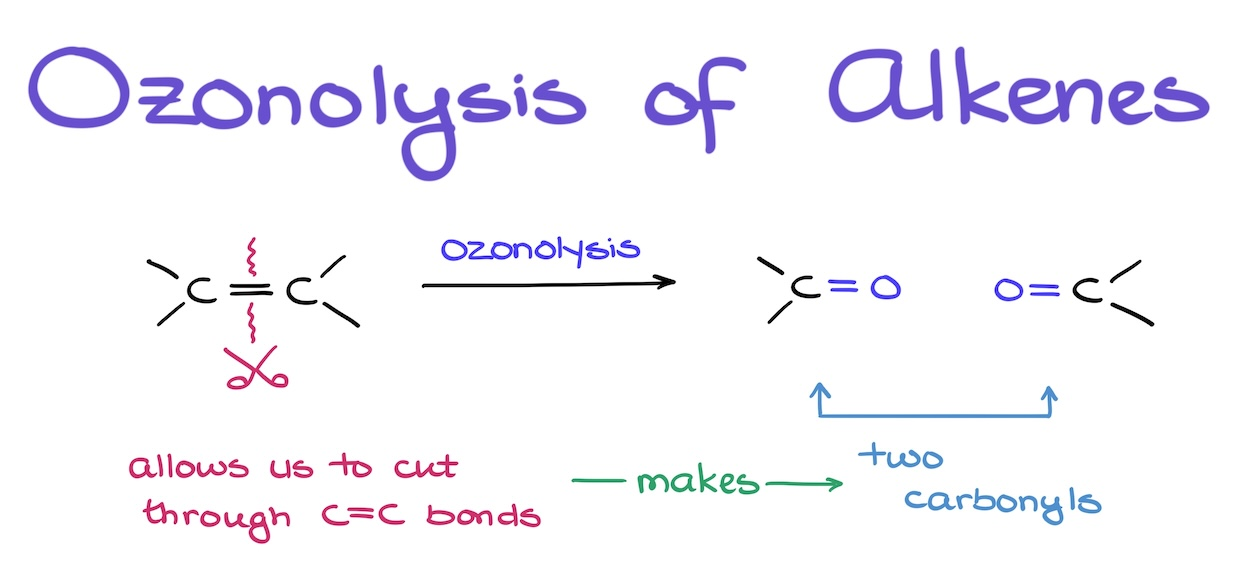
Ozonolysis
KMnO4, H2O, heat
produces ketones and carboxylic acids
Oxidative Cleavage by KMnO4
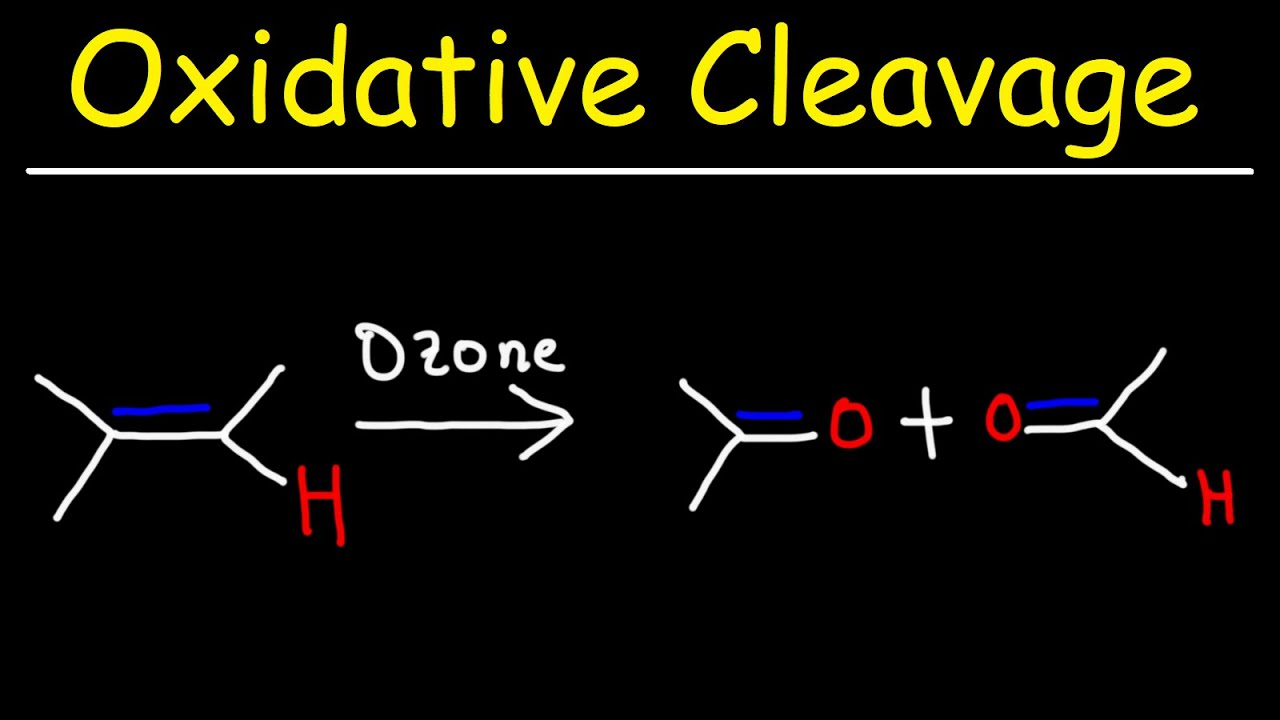
Oxidative Cleavage by KMnO4
OsO4 + H2O2 or KMnO4 + HO-
syn stereochemistry
no rearrangement
and enantiomer
Syn dihydroxylation
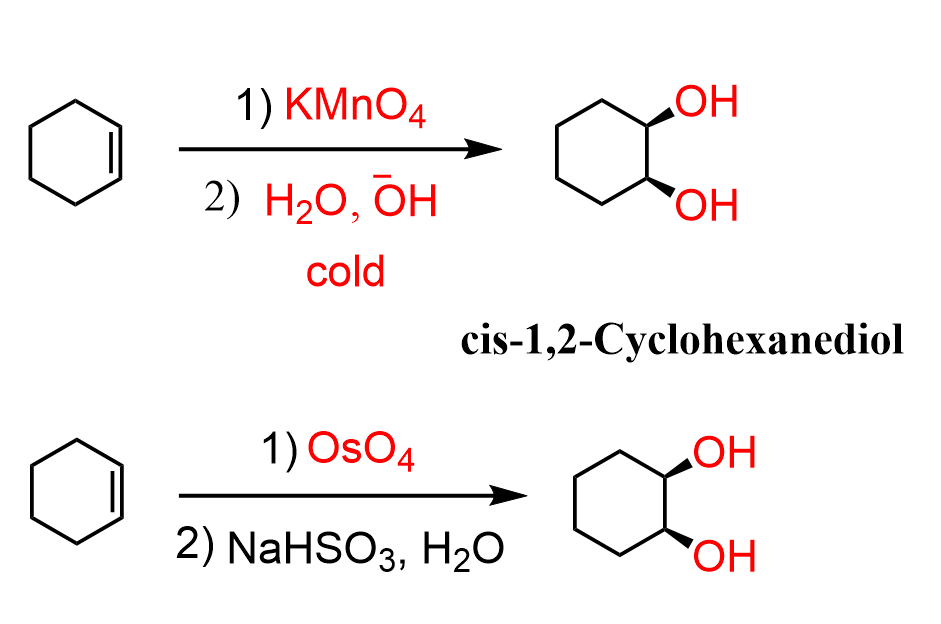
Syn dihydroxylation
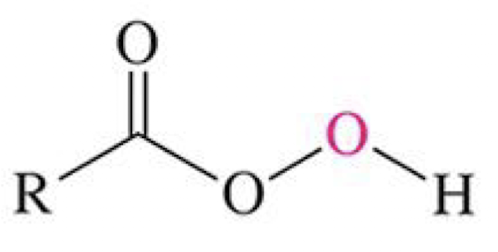
H+ catalyst, H2O, (look at picture)
from epoxide intermediate
anti stereochemistry
no rearrangement
and enantiomer
Anti dihydroxylation
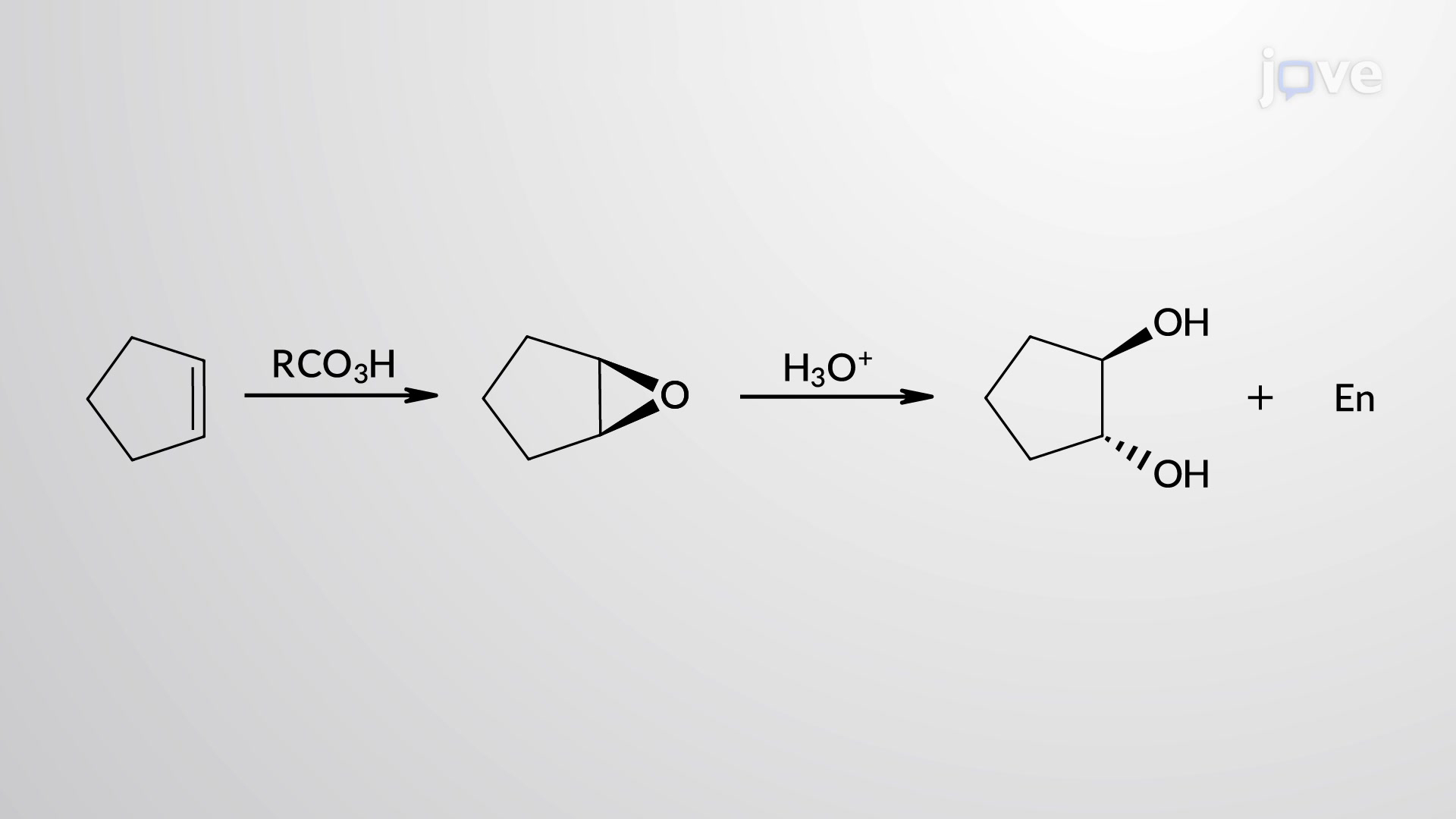
Anti dihydroxylation