L1(3.3b Mass transport) Harmoglobin
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms

Hemoglobin(quaternary structure is made up of…1?
•Each chain in hemoglobin has a …2? group which gives it its …3? colour
•Harmoglobin has an affinity for …4? (can carry 4 O, molecules)
• When haemoglobin becomes oxygenated, it is known as …5?
(1)4 globular proteins.
(2) 1 iron ion-(prosthetic group)
Haem 3. Red. 4. Oxygen
Oxyhaemoglobin
Affinity for oxygen and pO2
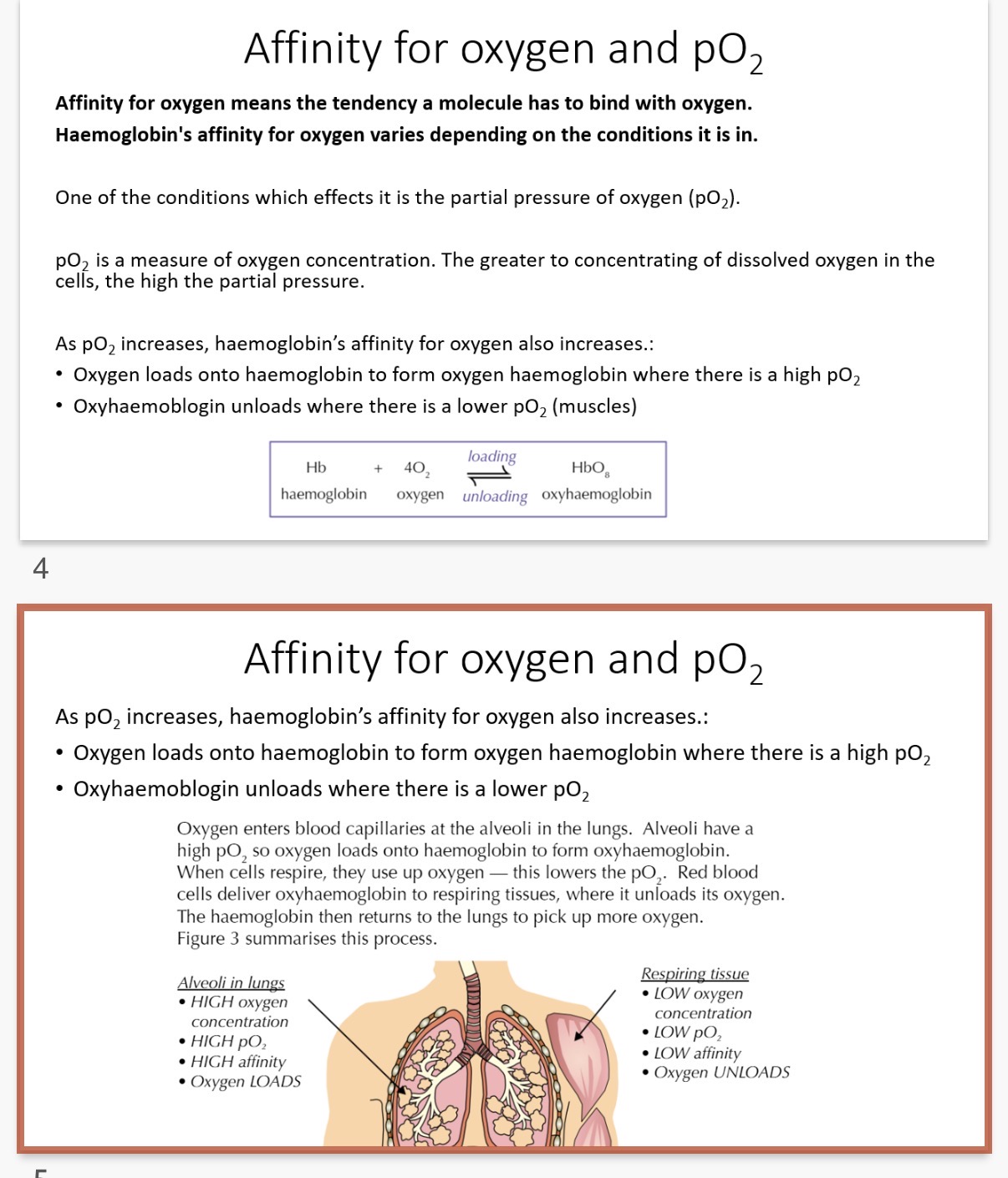
Affinity for oxygen means…1?
•Haemoglobin's affinity for oxygen varies depending on the conditions it is in.
One of the conditions which effects it is the partial …2? of oxygen (pO2).
What is partial pressure? (The greater the concentration of dissolved oxygen in the cells, the …4? the partial pressure)
•As pO2, increases, haemoglobin's …5? for oxygen also increases.:
• Oxygen …6? onto haemoglobin to form oxyhaemoglobin where there is a …7? pO2
• Oxyhaemoglobin unloads where there is a …8? pO2 (muscles)
Oxygen enters blood capillaries at the …9? in the lungs. Alveoli have a high …10?, so oxygen …11? onto haemoglobin to form oxyhaemoglobin. When cells respire, they use up oxygen — this …12? the pO2.
Red blood cells deliver oxyhaemoglobin to …13? tissues, where it unloads its oxygen.
The haemoglobin then returns to the lungs to pick up more oxygen.
Note:(Figure 3 summarises this process.)
Why do respiring muscle tissues have low pO2?
• Note: As pO2 increases, the saturation of haemoglobin increases.
the tendency a molecule has to bind with oxygen.
Pressure
pO2 is a measure of oxygen concentration.
Higher. 5. Affinity. 6. Loads. 7.High
Lower. 9. Alveoli. 10. pO2. 11.loads. 12.lowers. 13. Respiring
Oxygen is being used for respiration to break down glucose and release energy.
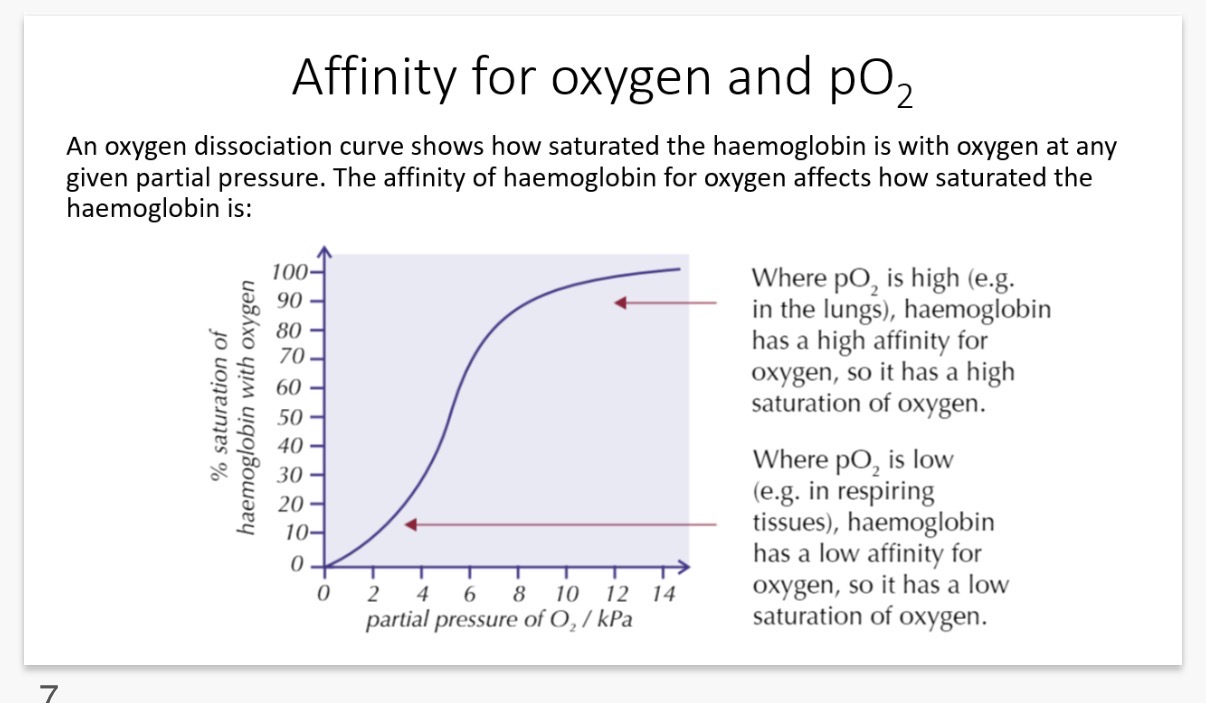
Read the info in the graph and then explain the shape of the S curved oxygen dissociation graph?
Note: when the curve is steepest, a small change in pO2 causes a big change in the amount of oxygen carried by the haemoglobin.
Explain the meaning of quaternary structure?
(1)When the partial pressure is low, haemoglobin has a lower affinity to oxygen and so there is a lower chance for oxygen to load onto haemoglobin to form oxyhaemoglobin.
(2) When the first O2 molecule binds, haemoglobins quaternary structure shape(protein shape) alters in a way that makes it easier for other O2 molecules to load onto haemoglobin.→ Therefore there is a rapid rise in oxyhaemoglobin saturation.
(3) When the partial pressure is at its highest, the graph gets less steep since all of the haemoglobins are occupied by (saturated with) oxygen.
The quaternary structure is the way multiple protein subunits are arranged and interact with each other to form a larger, functional protein complex.
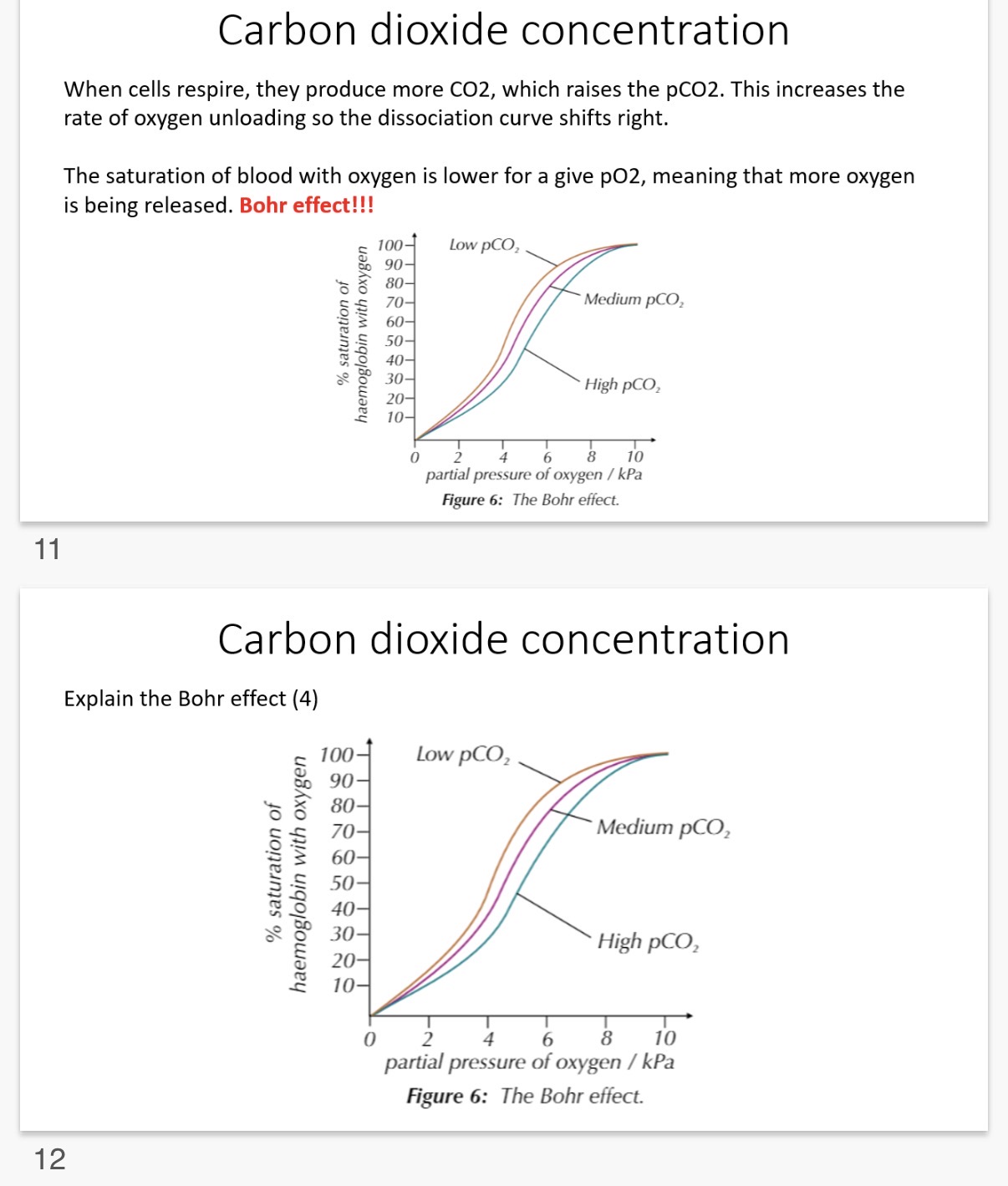
Carbon dioxide concentration
The partial pressure of carbon dioxide (pCO2) is a measure of the …1?
•The pCO2 also affects oxygen …2?. •Haemoglobin gives up its oxygen more readily at a …3? pCO2 (it's a way we get more oxygen to cells during activity)
•When cells respire they produce …4? CO2, which raises the pCO2. This increases the rate of oxygen …5? so the dissociation curve shifts …6?.
•The saturation of blood with oxygen is …7? for a given pO2, meaning that more …8? is being released. Bohr effect!!!
Explain the Bohr effect? (4)
Describe and explain the effect of increasing carbon dioxide concentration on the dissociation of oxyhaemoglobin?
Concentration of CO2 in a cell
Unloading. 3.higher. 4. More
unloading. 6.right. 7. Lower
oxygen
(1) Increased Respiration: Actively respiring tissues produce more CO2. This increases the partial pressure of carbon dioxide (pCO2).
(2) Acidity and Shape: The CO2 dissolves to form carbonic acid, lowering the pH. This causes a conformational change (change in tertiary structure) of the haemoglobin molecule.
(3) Reduced Affinity: This structural change reduces haemoglobin's affinity for oxygen.
(4) Oxygen Unloading: Oxygen is released (dissociates) more readily to the respiring tissues at any given pO2.
It increases the dissociation of oxyhaemoglobin(1) because CO2 will dissolve and form carbonic acid which will lower the pH of the area and make it more acidic. This changes the tertiary structure of haemoglobin which lowers its affinity for oxygen, causing oxygen to be released more readily.
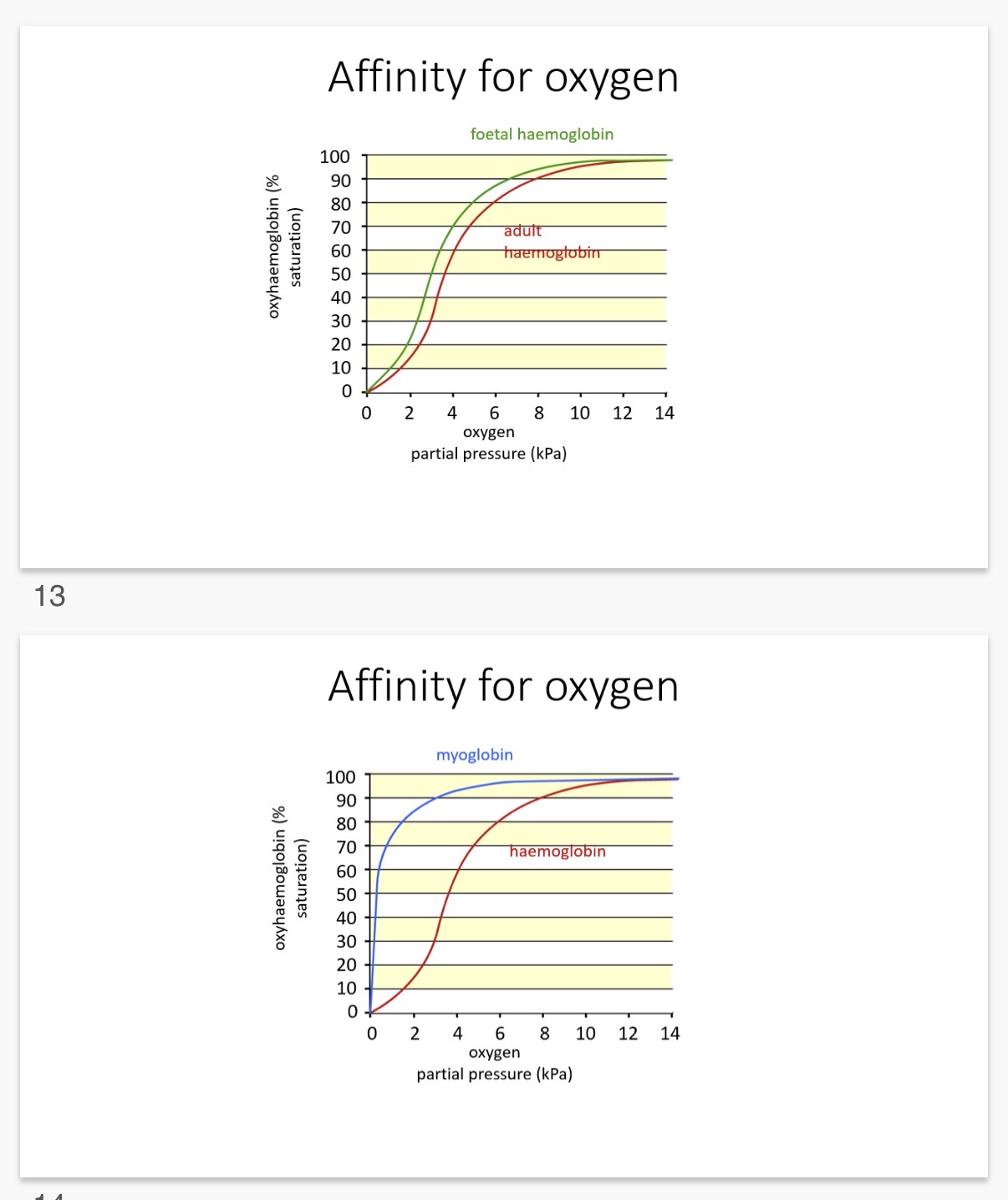
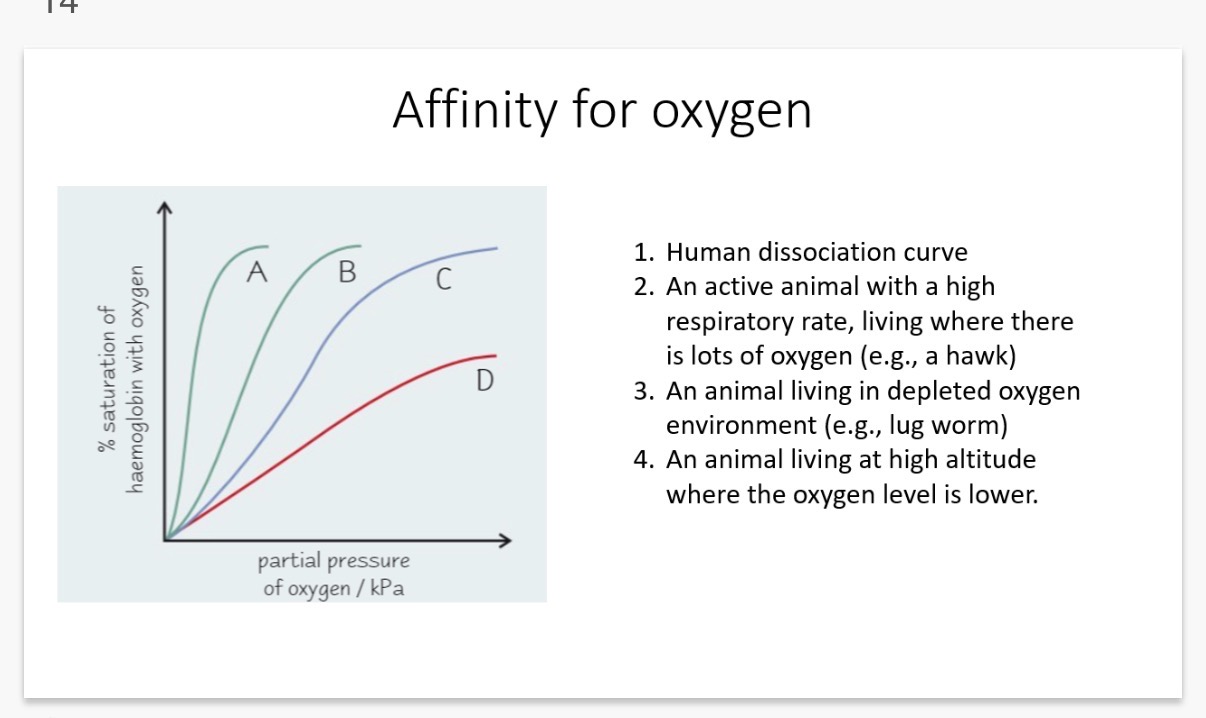
Answer image q?
(Explanation for curve 1)
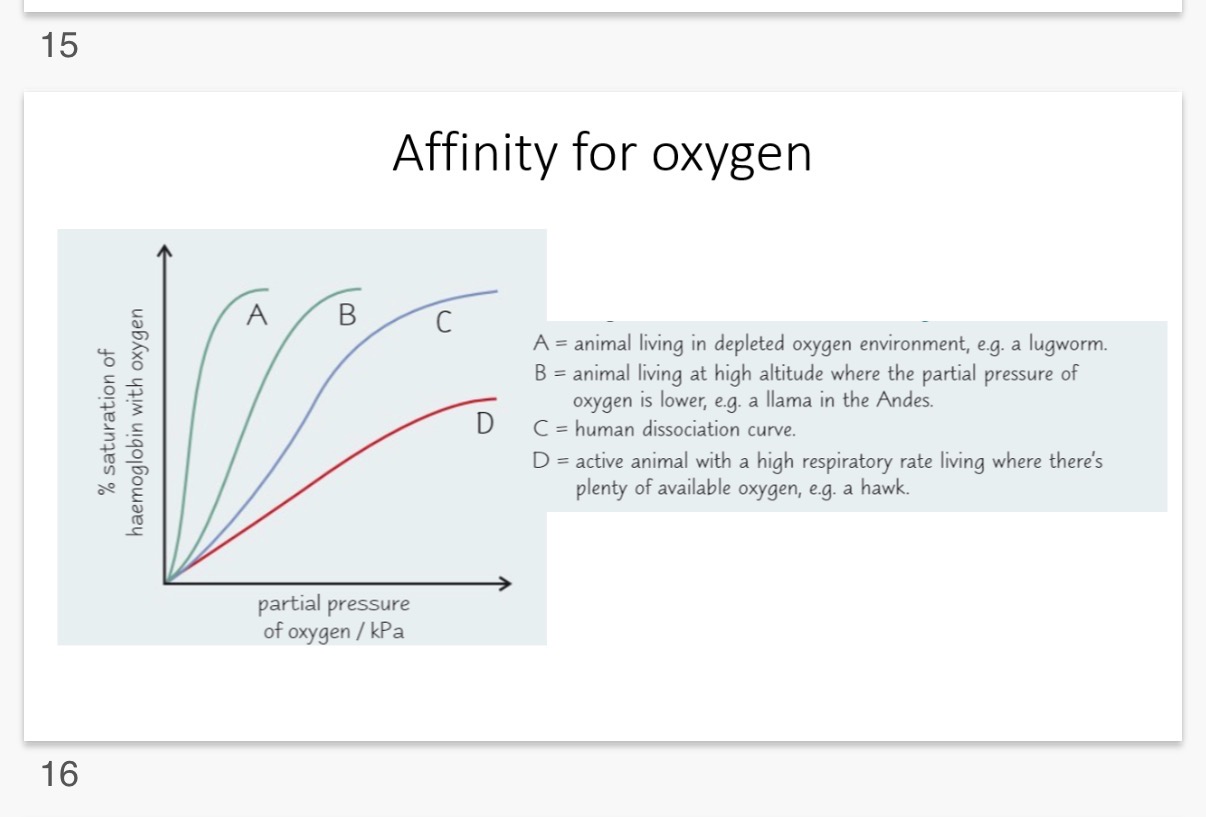
1. Curve A: High Affinity (The Lugworm)
• Environment: Oxygen-depleted (e.g., in muddy burrows where water contains very little O_2).
• Adaptation: Because there is so little oxygen available, the lugworm's haemoglobin must be extremely "greedy."
• Why Curve A: It reaches 100% saturation at a very low partial pressure of oxygen. It loads up on O_2 even when there is hardly any around.