Breast Cancer, Perimenopause, Menopause and Pelvic Health
1/96
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
What is the most significant risk factor for developing breast cancer?
Age, with risks rising until the early 60s and peaking in the 70s.

What are the three primary histologic categories of breast cancer?
Ductal carcinoma, lobular carcinoma, and nipple (Paget disease).
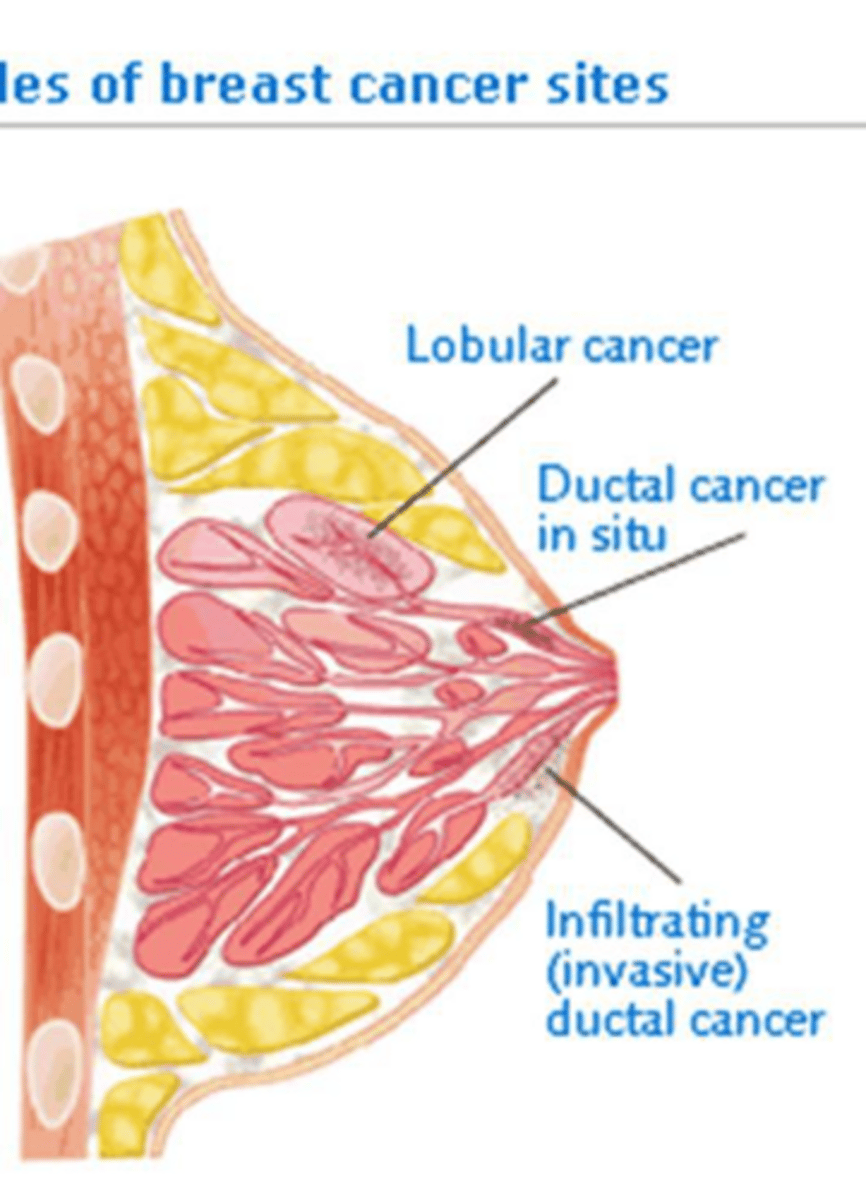
What are common physical characteristics of a malignant breast mass?
Size >2cm, immobility, poorly defined/irregular margins, firmness, skin dimpling, nipple retraction, and bloody discharge.
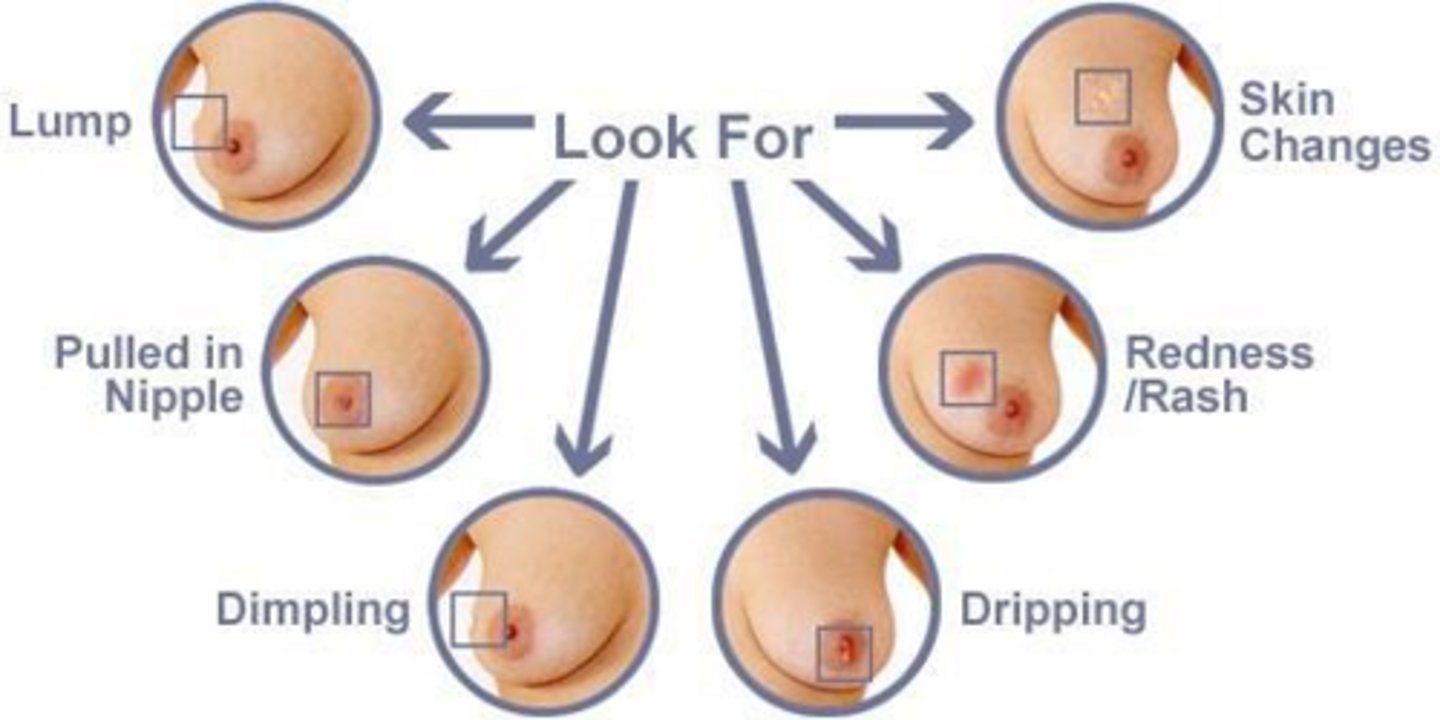
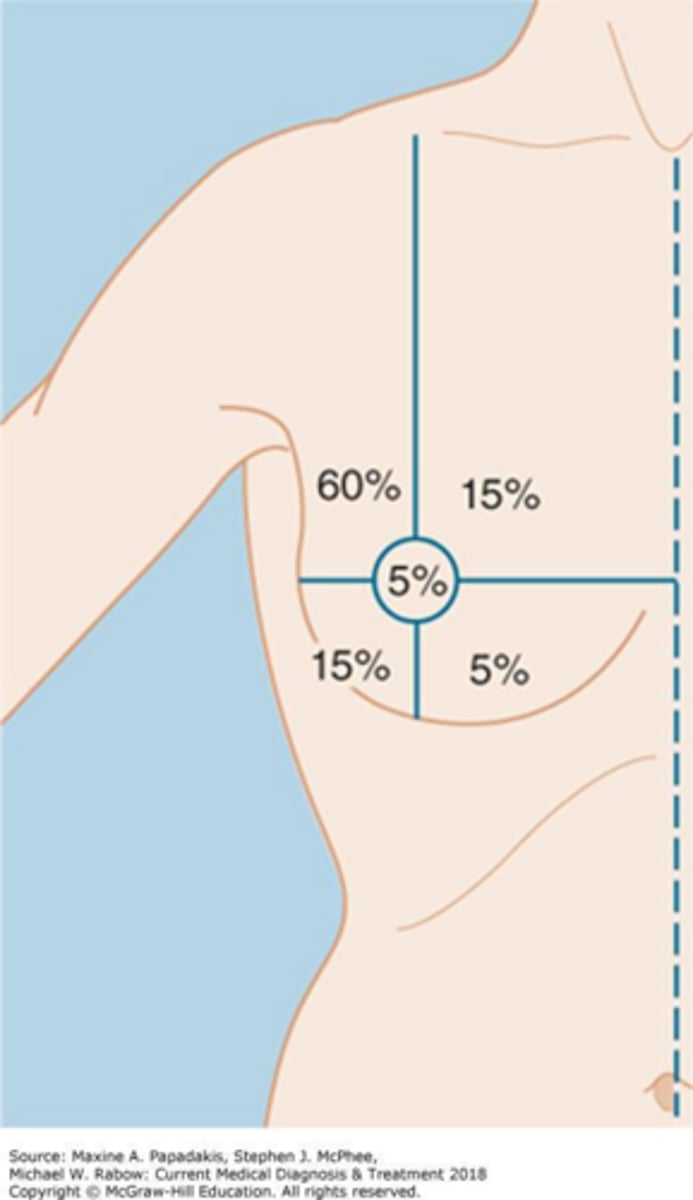
What is the most common location for breast cancer within the breast?
The upper outer quadrant.
What are the strongest genetic predictors for breast cancer?
BRCA1 and BRCA2 gene mutations.

How does nulliparity or a first live birth after age 35 affect breast cancer risk?
It increases the risk by approximately 40%.
What are the recommended screening intervals for a female with a BRCA1/2 mutation aged 30-75?
Annual mammogram plus annual breast MRI.
What is the recommended age range for a Risk-Reducing Salpingo-Oophorectomy (RRSO) in BRCA1 carriers?
Age 35-40.
What is the first-line diagnostic approach for a patient presenting with a breast mass?
Diagnostic mammogram and ultrasound.
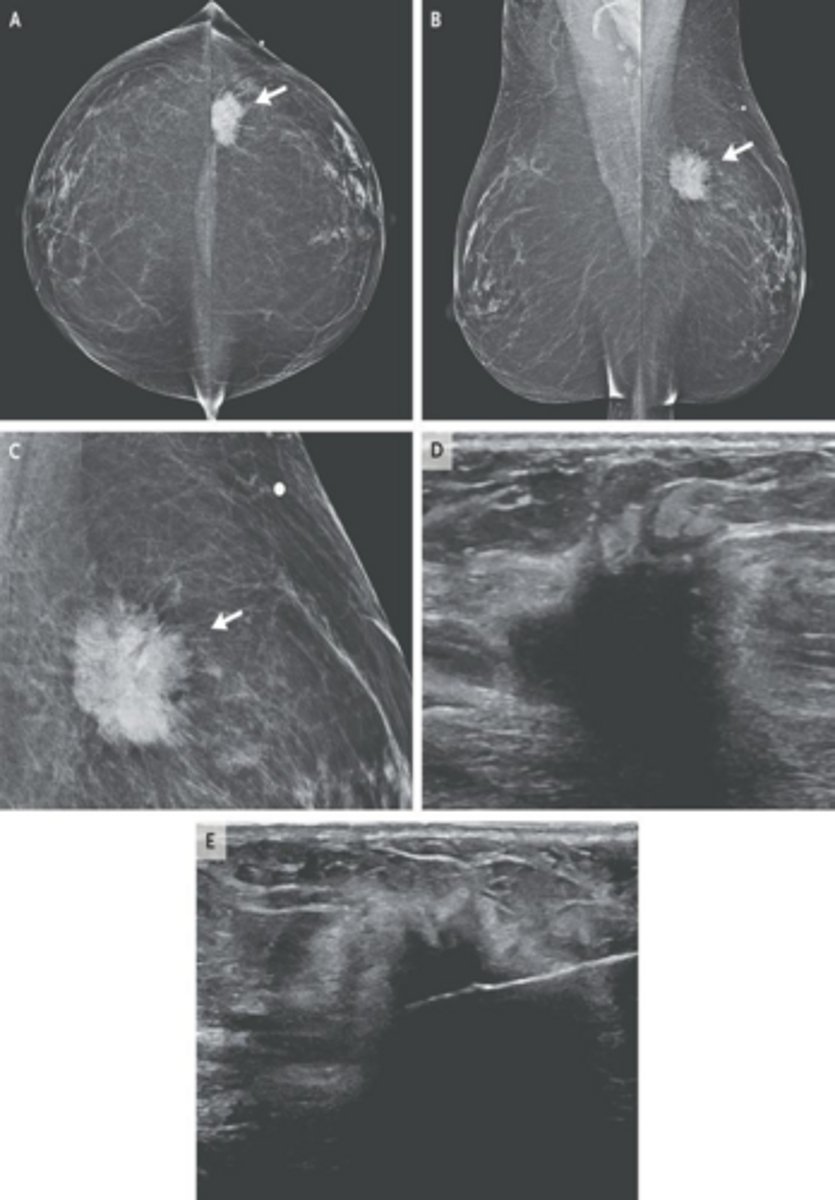
What is the gold standard for tissue diagnosis of a suspicious breast lesion?
Core biopsy guided by ultrasound.
What does the 'grade' of a breast tumor indicate?
It indicates how aggressive the tumor cells are and how likely they are to grow (Grade 1 is least aggressive, Grade 4 is fastest growing).
What are the three hormone receptor statuses typically analyzed in a breast cancer biopsy?
Estrogen receptors, progesterone receptors, and HER2 (human epidermal growth factor receptor 2).
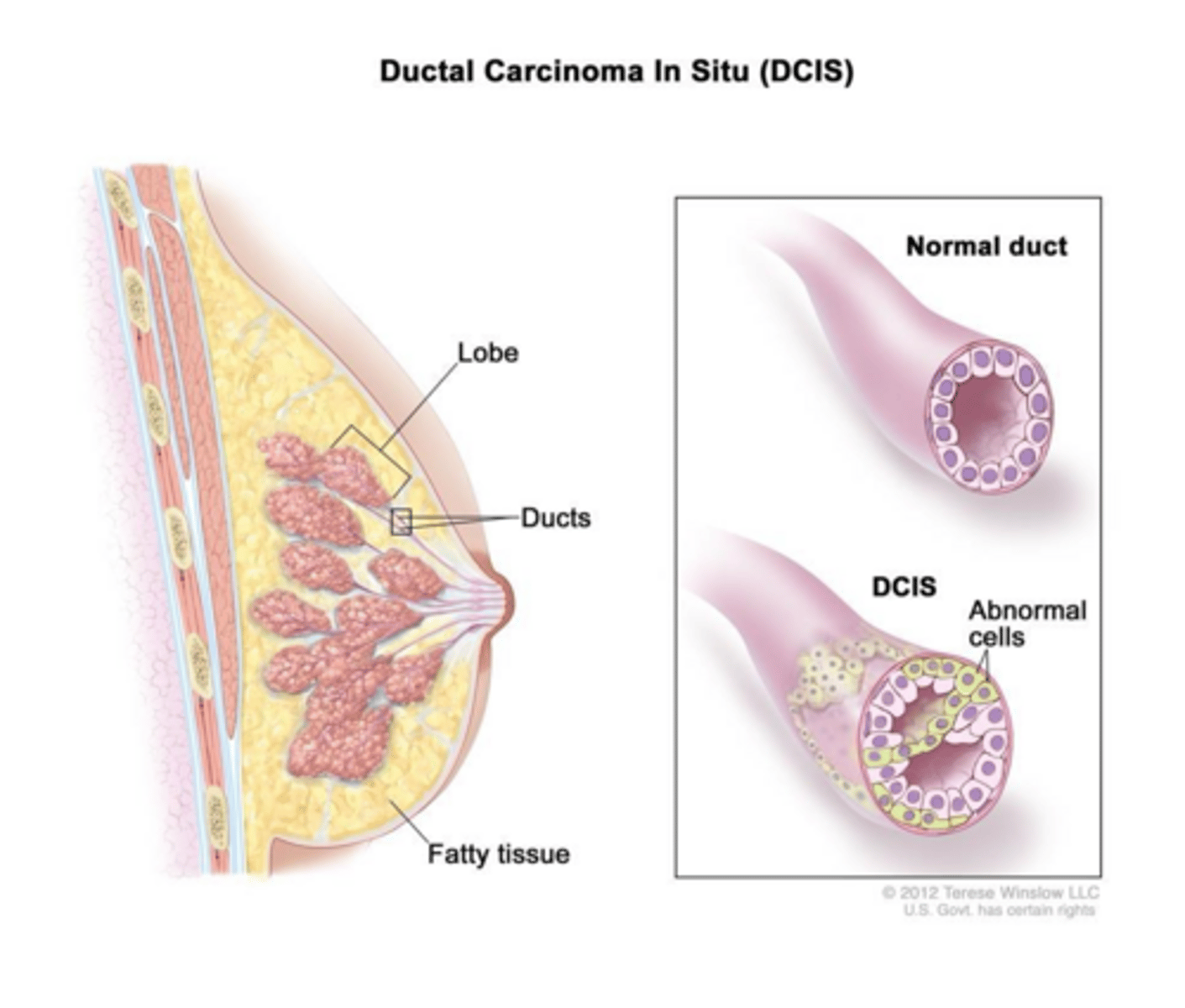
What is the defining characteristic of Ductal Carcinoma In Situ (DCIS)?
Proliferation of malignant epithelial cells within the ductal system with an intact basement membrane (no stroma invasion).
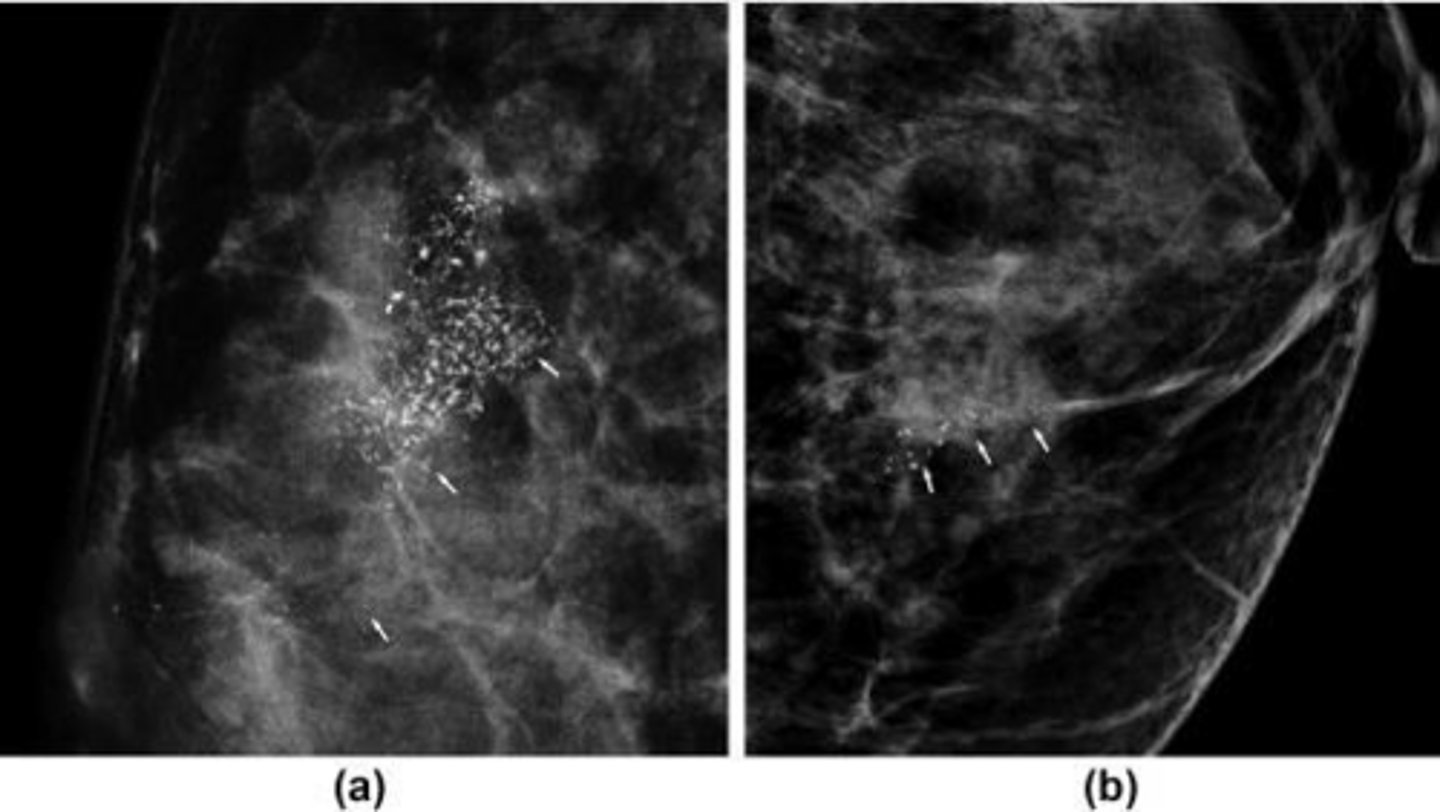
How does DCIS typically present on a screening mammogram?
As clustered microcalcifications.
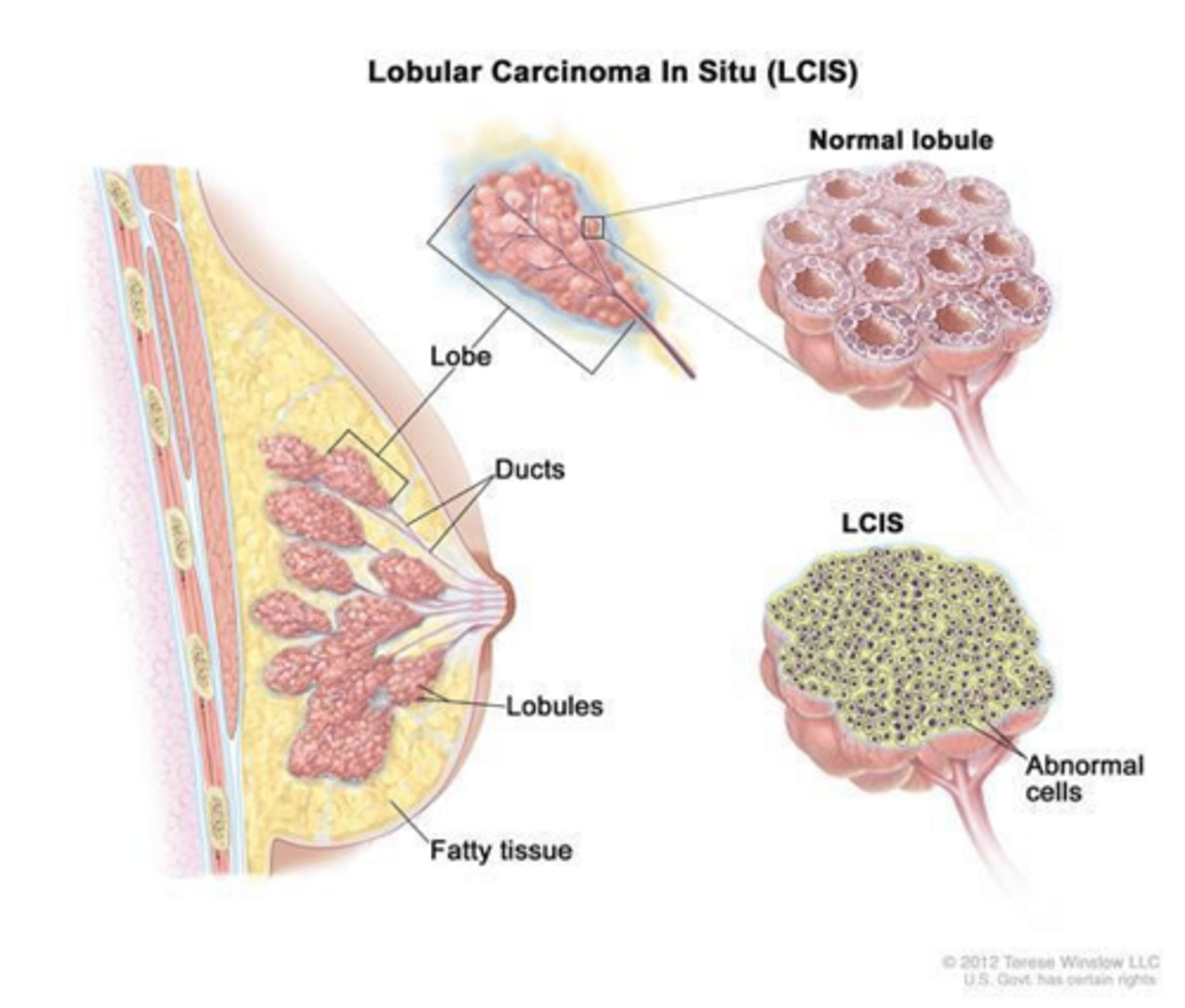
What is the clinical significance of Lobular Carcinoma In Situ (LCIS)?
It is a marker of increased bilateral breast cancer risk and a potential precursor to invasive carcinoma.
What is the most common form of invasive breast cancer?
Invasive ductal carcinoma (IDC), accounting for approximately 70-80% of cases.
What physical exam findings are classic for Invasive Ductal Carcinoma (IDC)?
A star-shaped, hard, fixed (immobile), and irregular mass.
What is the primary treatment for DCIS to ensure breast preservation?
Lumpectomy followed by radiation therapy.
Why is LCIS often difficult to detect?
It is typically an incidental finding on a biopsy performed for other reasons and is often not visible on mammography.
What chemoprevention is strongly recommended for patients with LCIS?
A SERM (Selective Estrogen Receptor Modulator) such as Tamoxifen.
What percentage of breast cancers are first noticed by the patient?
90%.
What are the non-modifiable biological risk factors for breast cancer mentioned in the notes?
Age >65, White race, Ashkenazi Jewish heritage, and inherited mutations like BRCA1/2.
How does high breast density affect breast cancer screening?
It is an independent risk factor and acts as a masking factor on mammograms.
What is the approximate risk reduction provided by a bilateral mastectomy for BRCA carriers?
Approximately 90-95%.
What is a sentinel lymph node?
The first lymph node to receive drainage from a primary tumor, making it the most likely site for initial cancer spread.
How is a sentinel lymph node identified during surgery?
By injecting blue dye or radioactive sulfur colloid around the tumor on the day of surgery, which then travels to the sentinel node.
What is the clinical significance of a positive sentinel lymph node biopsy?
It indicates the need for an axillary lymph node dissection.
What are the typical characteristics of Invasive Lobular Carcinoma (ILC)?
It is the second most common breast cancer, difficult to detect on mammograms, and is generally ER+ and HER2-.
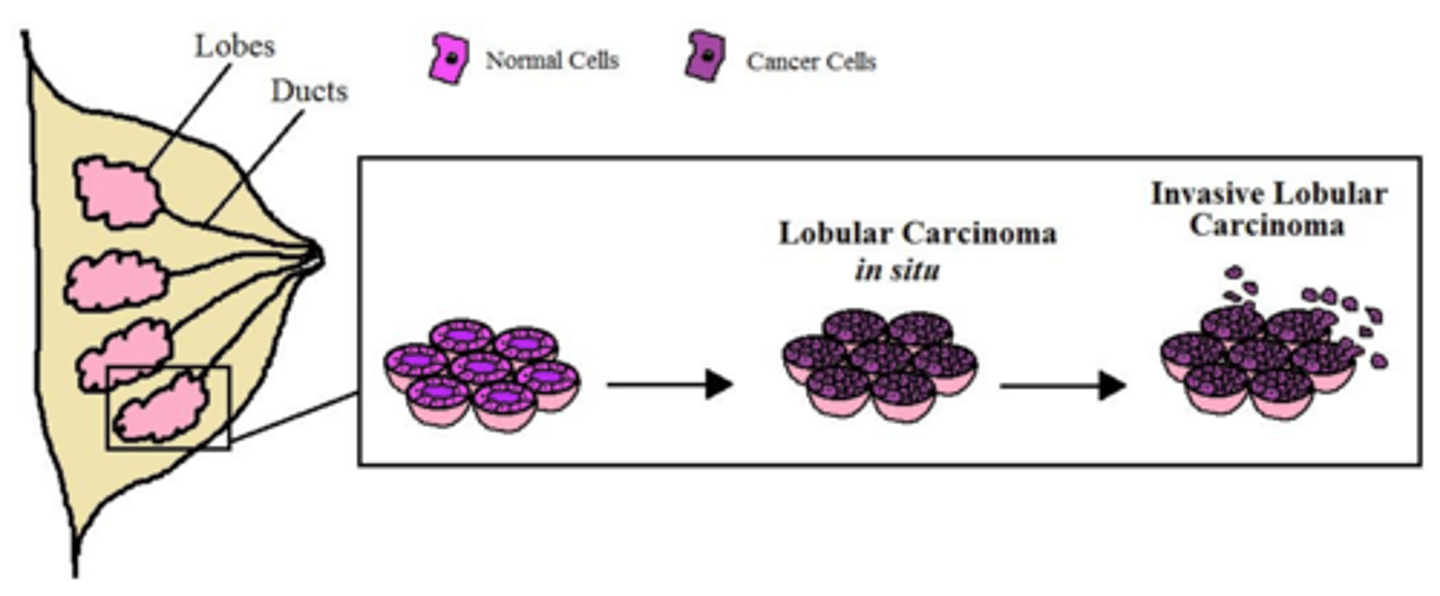
How does Invasive Lobular Carcinoma typically present on physical exam?
Often lacks a discrete mass; instead, the breast may feel 'full' or show skin induration/thickening.
What is the primary treatment for Invasive Lobular Carcinoma?
Lumpectomy with radiation or mastectomy, and it is highly responsive to hormonal therapy like tamoxifen.
What is the pathogenesis of Inflammatory Carcinoma?
An aggressive cancer where malignant cells block the dermal lymphatic vessels.
What are the hallmark physical findings of Inflammatory Breast Cancer?
Rapid breast enlargement, erythema, edema, warmth, and a 'peau d'orange' skin appearance.
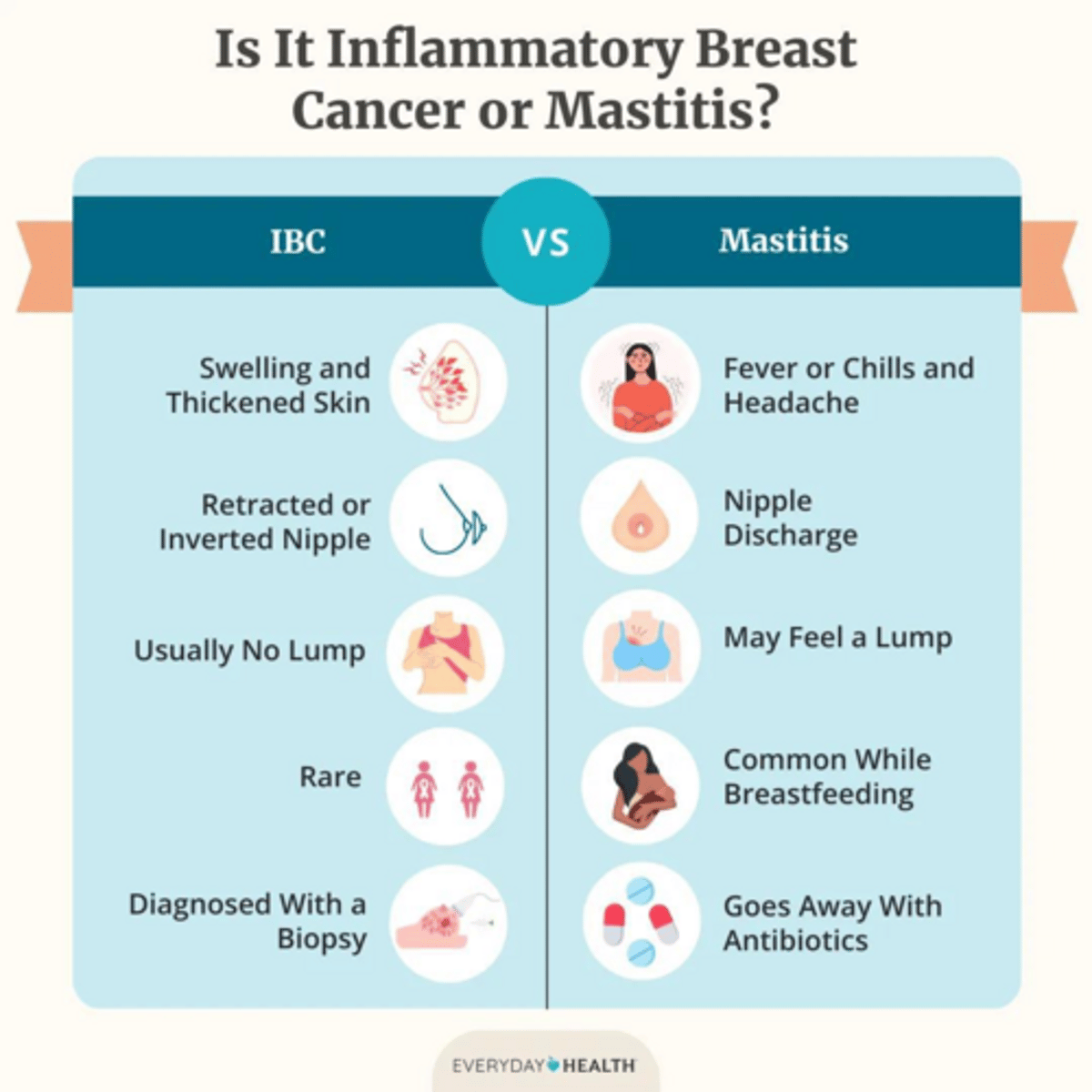
Why is Inflammatory Breast Cancer often initially misdiagnosed?
Its inflammatory changes (redness, swelling) are often mistaken for mastitis (infection).
What is the management protocol if a patient presents with suspected mastitis?
Start antibiotics; if there is no response, a biopsy must be performed to rule out inflammatory carcinoma.
What is the standard treatment sequence for Inflammatory Breast Cancer?
Systemic neoadjuvant chemotherapy first, followed by surgery and radiation.
What is the pathogenesis of Paget's Disease of the breast?
Intraepithelial spread of malignant cells (Paget cells) into the epidermis of the nipple.
What are the classic symptoms of Paget's Disease?
Persistent itching, burning, and a scaling, eczematous rash on the nipple and areola.
How can you clinically distinguish Paget's Disease from common eczema?
Paget's typically involves the nipple first and spreads to the areola, and may present with bloody or purulent nipple discharge.
What is the association between Paget's Disease and underlying malignancy?
Over 90% of cases have an underlying DCIS or invasive breast cancer.
What is the standard of care for Paget's Disease?
Mastectomy is the standard of care, though conservative local excision or wedge resection may be used if the disease is localized.
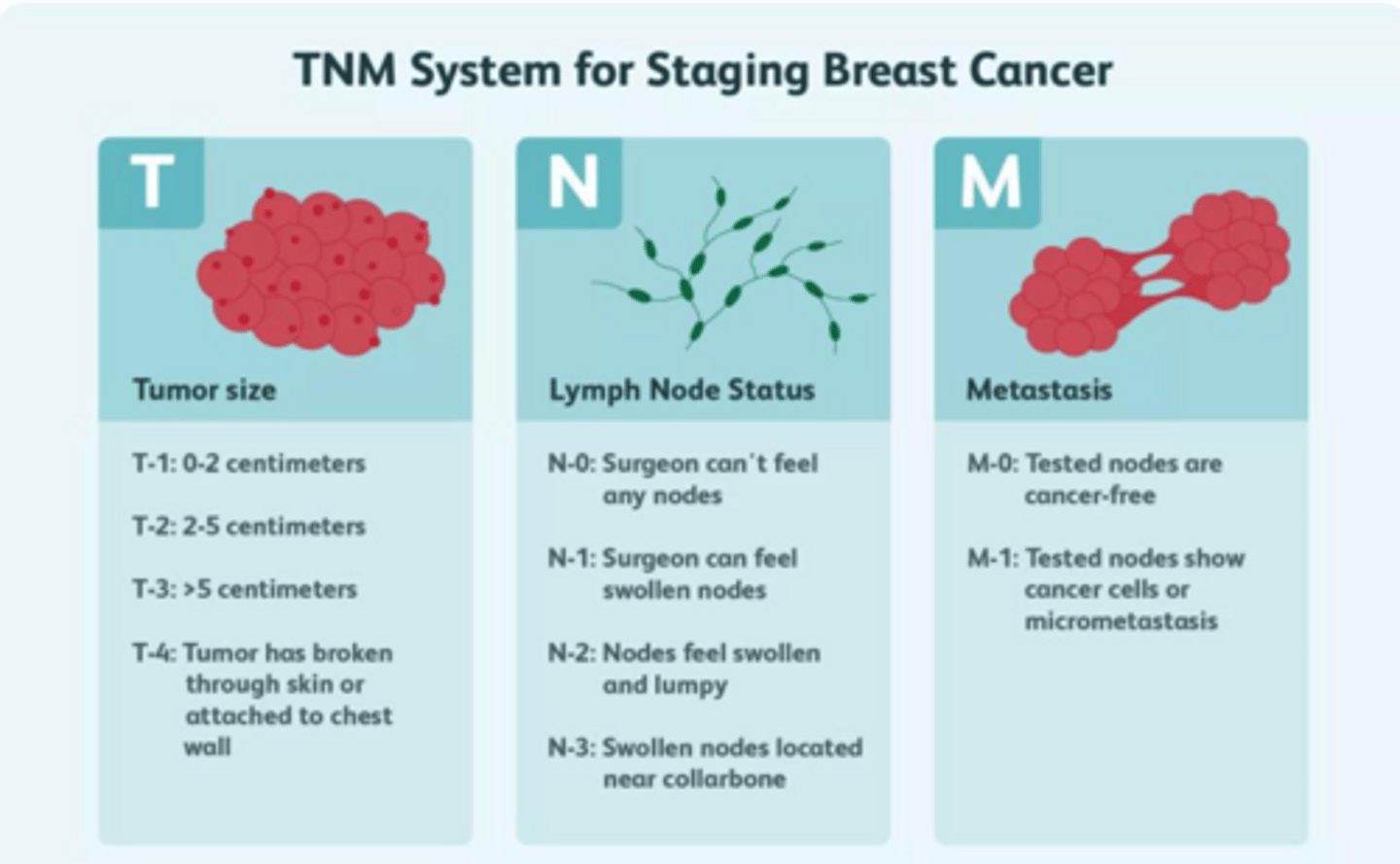
What does the AJCC TNM system stand for in breast cancer staging?
T (Tumor size), N (Lymph node involvement), and M (Metastasis to distant sites).
Which factor is considered the most reliable prognostic factor for breast cancer?
The stage of the cancer at diagnosis.
What is the most significant prognostic factor regarding lymph node status?
Axillary lymph node status.
What are the most common sites for breast cancer metastasis?
Bone, lung, brain, and liver.
How do ER/PR positive breast cancers differ from ER/PR negative cancers?
HR+ cancers tend to grow more slowly and have a better overall prognosis.
What is the function of Aromatase Inhibitors (AIs) in breast cancer treatment?
They block the peripheral conversion of androgens to estrogen and are used specifically in post-menopausal women.
What are Selective Estrogen Receptor Modulators (SERMs) used for?
They are used to treat HR+ breast cancer in both pre- and post-menopausal women (e.g., Tamoxifen).
What does HER2/neu protein expression indicate in breast cancer?
It indicates a more aggressive, fast-growing tumor.
What is the primary targeted therapy used for HER2-positive breast cancer?
Trastuzumab (Herceptin).
What characterizes Triple Negative Breast Cancer (TNBC)?
Tumors that lack hormone receptors (ER, PR) and HER2 expression; it is highly aggressive with a worse survival rate.
What is the most important prognostic factor for breast cancer?
The stage of the disease.
What is the primary goal of treating Stage IV (metastatic) breast cancer?
Palliative care focused on symptom control, prolonging survival, and improving quality of life.
What is the standard of care for axillary staging in breast cancer surgery?
Sentinel lymph node biopsy.
What is the difference between lumpectomy and mastectomy regarding radiation?
Lumpectomy (breast-conserving surgery) requires radiation, whereas mastectomy involves removing the entire breast.
Which medication is used for premenopausal hormone receptor-positive breast cancer?
Tamoxifen, a Selective Estrogen Receptor Modulator (SERM).
How do aromatase inhibitors work in postmenopausal patients?
They block the peripheral conversion of androgens to estrogen in fat and muscle tissue.
What are the common side effects of Tamoxifen?
Hot flashes, increased risk of DVT/PE, and a small risk of endometrial cancer.
What are the common side effects of Aromatase Inhibitors?
Bone loss (osteoporosis) and significant joint/muscle aches (arthralgias).
What is the primary risk associated with Anthracyclines (e.g., Doxorubicin) in chemotherapy?
Cardiotoxicity.
What is the primary risk associated with Taxanes (e.g., Paclitaxel) in chemotherapy?
Peripheral neuropathy.
When is chemotherapy administered relative to endocrine therapy?
Chemotherapy is always administered before endocrine therapy if both are indicated.
How is menopause clinically defined?
The permanent cessation of menstruation, determined retrospectively after 12 months of amenorrhea.
What is the mean age of natural menopause?
51 years old.
What is the diagnostic FSH level for menopause?
An FSH level greater than 30 IU/L.
What is the primary hormonal event leading to menopause?
Oocyte depletion, which leads to a decline in follicular quantity and quality.
Which estrogen becomes dominant after menopause?
Estrone (E1), produced by peripheral aromatization of androstenedione in adipose tissue.
What is the most common vasomotor symptom of menopause?
Hot flashes and night sweats.
What is Genitourinary Syndrome of Menopause (GSM)?
A condition involving vaginal dryness, burning, and urinary symptoms due to urogenital atrophy.
What is the expected finding on a vaginal cytologic exam in a menopausal patient?
Predominantly parabasal cells.
How does menopause affect cardiovascular risk?
Hypoestrogenism leads to a shift in lipid profiles (increased LDL, decreased HDL) and loss of vascular elasticity, increasing ASCVD risk.
What is the approximate annual bone loss rate in the first 5 years post-menopause?
3-5%.
What is the 'Golden Rule' for the timing of Menopausal Hormone Therapy (MHT)?
MHT is most beneficial and carries the lowest risk when initiated within 10 years of menopause or before age 60.
How should MHT be prescribed for a woman with a uterus versus one without?
Women with a uterus require estrogen plus progestogen to prevent endometrial hyperplasia; women without a uterus require estrogen only.
What are the primary absolute contraindications for MHT?
Active breast cancer or estrogen-dependent neoplasia, history of VTE (DVT/PE), stroke, myocardial infarction, undiagnosed abnormal vaginal bleeding, and active liver or severe gallbladder disease.
What non-hormonal pharmacotherapy is considered the new standard for hot flashes?
Fezolinetant, an NK3 receptor antagonist.
What are the recommended screening guidelines for bone density (DXA) in postmenopausal women?
Baseline at age 65, or younger if the patient has high risk factors (e.g., high FRAX score).
Define a cystocele.
Bladder herniation through the anterior vaginal wall.
Define a rectocele.
Rectum herniation through the posterior vaginal wall.
Define an enterocele.
Small bowel descent into the vaginal vault, specifically through the Pouch of Douglas.
What is a paravaginal deficit?
Lateral detachment of the vagina from the pelvic wall.
What is the first-line treatment for all compartments of pelvic organ prolapse?
Pelvic floor muscle training (Kegels), vaginal pessaries, and lifestyle modifications like weight loss and high-fiber diets.
What is the surgical procedure for a cystocele?
Anterior colporrhaphy.
What is the surgical procedure for a rectocele?
Posterior colporrhaphy.
What is the clinical presentation of a rectocele?
Sensation of incomplete bowel movements and often a history of digital reduction to facilitate defecation.
How is stress urinary incontinence characterized?
Urethral hypermobility or intrinsic sphincter deficiency (mechanical failure).
What is the pathophysiology of urge urinary incontinence (OAB)?
Detrusor overactivity, characterized by involuntary bladder contractions during filling.
What does a Post-Void Residual (PVR) volume of > 200 mL suggest?
Overflow incontinence or bladder retention.
When is cystoscopy indicated in the evaluation of urinary incontinence?
When there is hematuria or suspicion of structural anomalies.
What is the role of urodynamic testing?
It measures bladder pressure and flow to differentiate between stress and urge incontinence when initial treatment fails.
What is the gold standard for treating recurrent postmenopausal UTIs?
Topical vaginal estrogen, which restores local flora and lowers vaginal pH.
What are the first-line pharmacologic treatments for recurrent UTIs?
Nitrofurantoin (Macrobid), TMP-SMX (Bactrim), or Fosfomycin.
What is the most common and effective surgical treatment for stress urinary incontinence?
Mid-urethral sling.
How does estrogen deficiency contribute to UTIs in postmenopausal women?
Loss of estrogen leads to a decrease in Lactobacilli and an increase in vaginal pH (> 5.0), allowing colonization by pathogens like E. coli.
What lifestyle modifications are recommended for managing urinary incontinence?
Weight loss, limiting caffeine and alcohol, and fluid management.
What is the primary goal of surgical intervention for an enterocele?
To re-anchor the upper vaginal vault and prevent hernia.
What physical exam maneuver is used to diagnose a cystocele?
Having the patient bear down (Valsalva) during a bimanual exam to observe the anterior vaginal bulge.
What is the recommended daily intake of Calcium and Vitamin D for women over 50?
1,200 mg of Calcium daily and 800-1,000 IU of Vitamin D daily.