colour by design
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
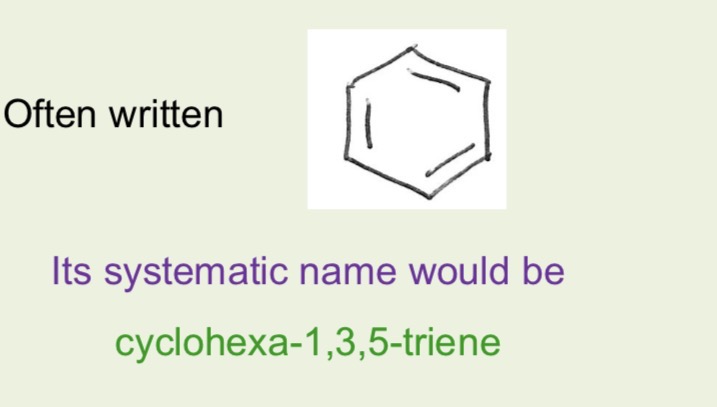
What is the molecular formula of benzene
C6H6
What is kekule drawing and systematic name of benzene and how did kekule predict benzene structure
In benzene bond angle around each carbon is 120 degrees and molecule if planar- kekules structure predicts this as around each C there are 3 sets of electrons which all repel so get as far apart as possible giving planar triangle shape with bond angles 120 degrees
Problems proposed with kerkules structure
if it was correct benzene would react like an alkene and decolourise aqueous bromine, but there is no reaction
If correct benzene should have 2 different carbon-carbon bond lengths because double bonds are shorter than single but all 6 are equal in length in between expected lengths
If correct there would be 2 different isomers of 1,2-dimethylbenzene but there’s only one
Change in enthalpy calculations using bind enthalpies and assuming kekule structure came out wrong and benzene does not give out as much energy as it should so it’s more stable
How does the hydrogenation show benzene is more stable than the kekule structure said it should be
If benzene has kekule structure with 3 c double bonds c the change in enthalpy for its hydrogenation would be 360KJ/moo 3X that for the hydrogenation of 1 C double bond C but its gives our a lot less energy then expected so benzene is more stable than the kekule structure which is confirmed as more severe conditions needed for hydrogenation:
higher temp and pressure and a special finely divided form of the nickel catalyst
What is the true structure of benzene
Each C uses 3 of its 4 outer electrons to bond:
single bonds are all sigma bins and form the sigma framework
This leaves one outer electron on every C each in a p- orbital at 90 degrees to the planar sigma framework.
Instead of overlapping in pairs giving 3 separate pi bonds
All 6 p- orbitals overlap sideways so the 6 electrons, one from each carbon, are spread out evenly so they are shared by all 6 carbon atoms in the ring
This spreading out of electrons is called delocalisation.
The 6 delocalised electrons form a delocalised donut shaped pi cloud of negative charge above and below the ring of carbons and is represented as

Why is the actual delocalised structure of benzene more stable than the kekule structure
As electrons repel so the more they spread out the more stable.
When benzene reacts it gives out less energy than the kekule structure would because of benzenes extra stability due to the delocalisation
How do we draw benzene rungs fused together
Not represented with circles in rings as they are not separate delocalised systems so drawn as alternating double and single bonds though are really extended delocalised systems
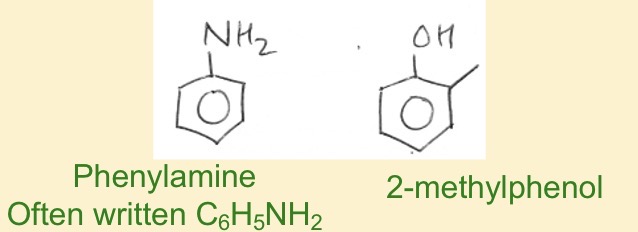
When is the benzene ring classed as phenyl
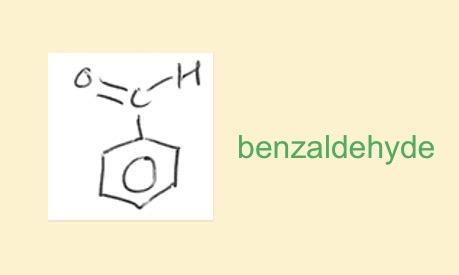
What do you call a benzene ring with aldehyde attached and draw it
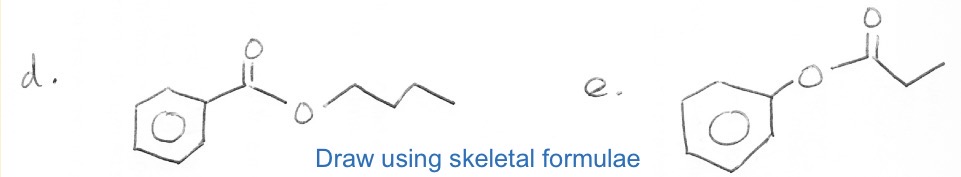
Draw butyl benzoate and phenyl propanoate
D is butyl benzoate
E is phenyl propanoate
What sort of reacting species are attracted to delocalised electrons in benzene attracted
The regions of negative charge attract electrophiles
Definition of elcetrophile
A positive ion or s+ part of a molecules which reacts by accepting a pair of electrons forming a covalent bond
What other family of organic molecules react with electrophiles and is the reaction easier or harder with arenes
Alkenes do elctrophillic addition
Reaction is harder with arenes and quicker with alkenes because the negative charge in pi cloud of an alkene is more concentrated whereas in an arene it is more spread out due to delocalisation. Reacting with benzene ring involves disrupting the stable delocalised ring and the activation energy is higher so arenes need a more powerful electrophiles and more severe reaction conditions
Why do arenes not do electrophillic addition
As the stable delocalised structure would be lost
What type of reaction do arenes undergo
Electrophillic substitution where the stable delocalised ring is retained. These reactions are relatively slow as the first step in the reaction has to disrupt the delocalisation which requires large amount and often a catalyst is needed to create an electrophile powerful enough to attack a benzene ring. Electrophiles used will need a full positive charge and delta plus is not powerful enough.
Chlorination of benzene
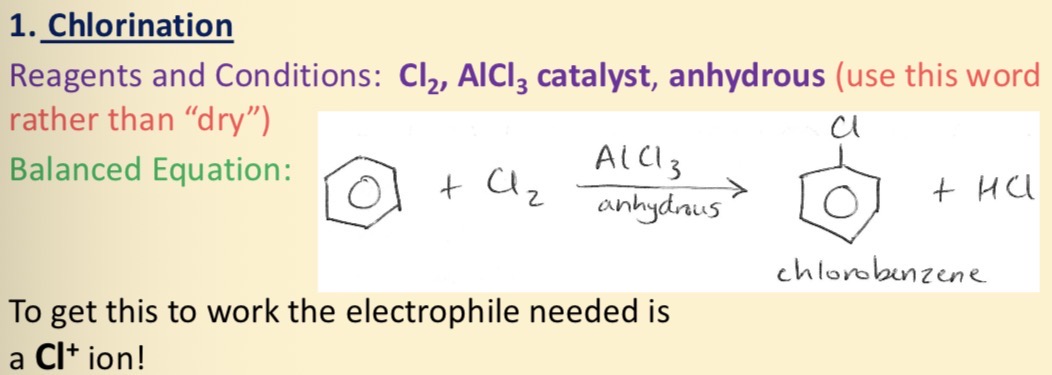
In chlorination of benzene why do we use AlCl3 catalyst and why is it covalent
Covalent because Al3+ ions are small and highly charged and they pull back electrons from cl- ions. In AlCl3 Al still needs 2 more electrons for noble gas configuration so AlCl3 is excellent electron pair acceptor.
AlCl3 takes an electron pair from the cl2 molecules and the cl-cl bond breaks by heterolytic fission giving Cl+ ion which is a powerful electrophile which attacks the benzene ring
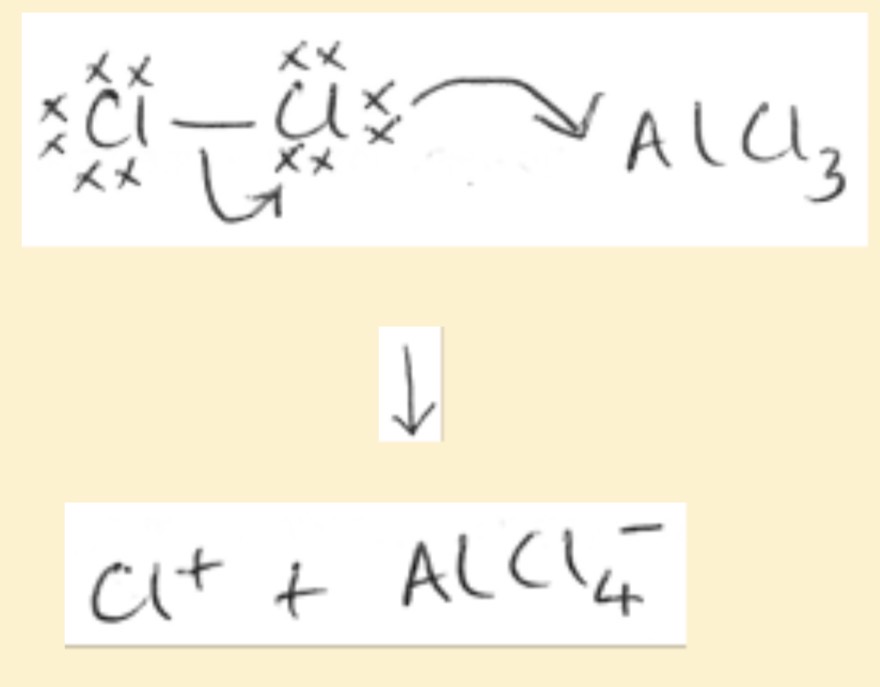
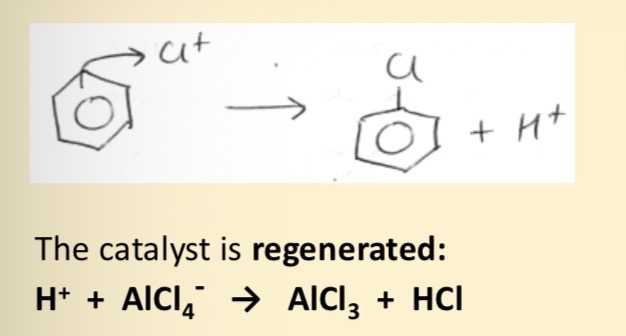
Equation of AlCl3 catalyst being regenerated
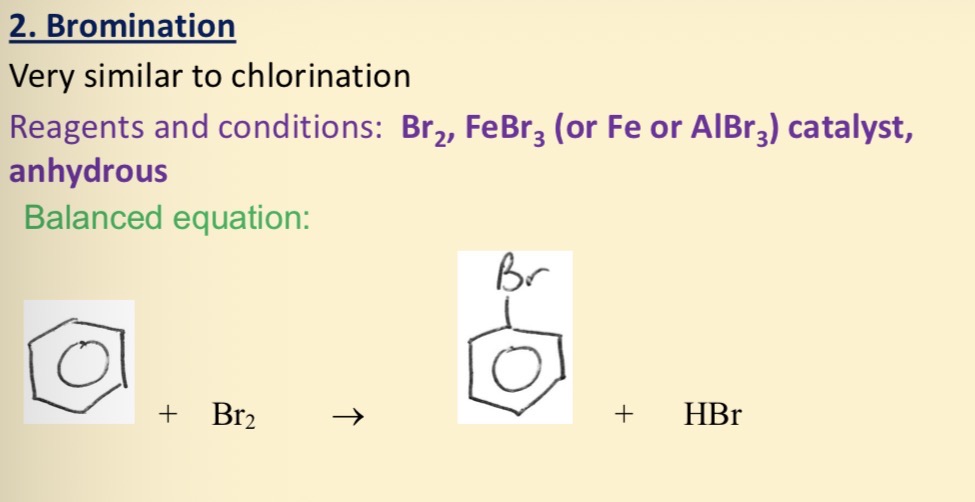
Bromination of benzene
Nitration of benzene
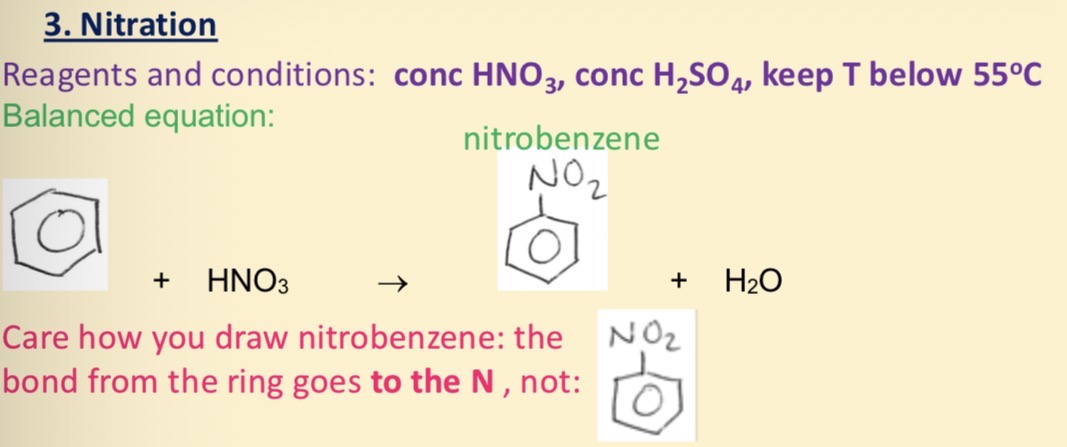
What electrophile needed for the nitration of benzene and what is it made from
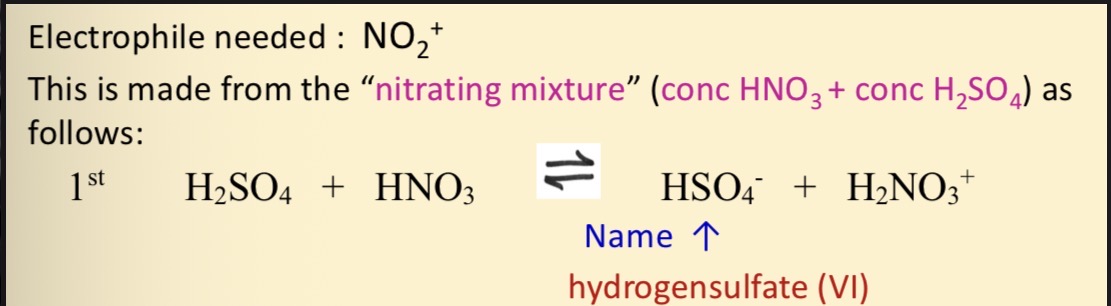
What is HNO3 acting as in this reaction

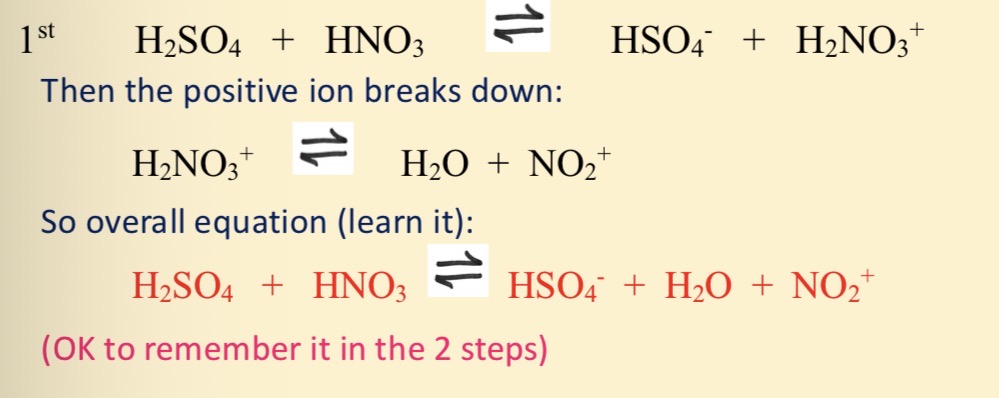
What is the overall equation to make the electrophile NO2 +
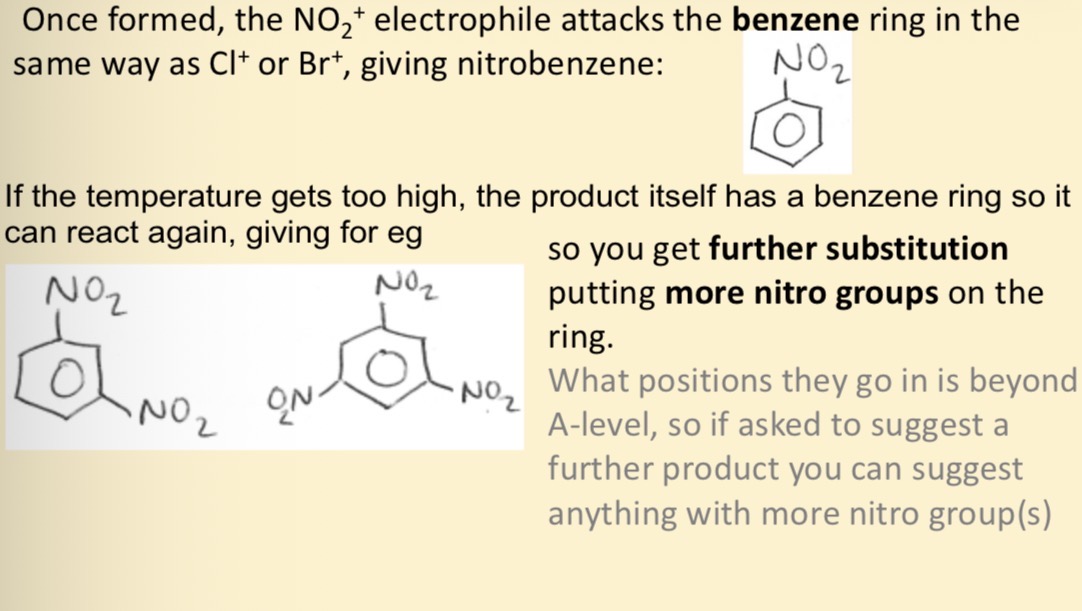
How does the NO2 + electrophile attack benzene and what happens if the temp gets too high
Sulphonation of benzene
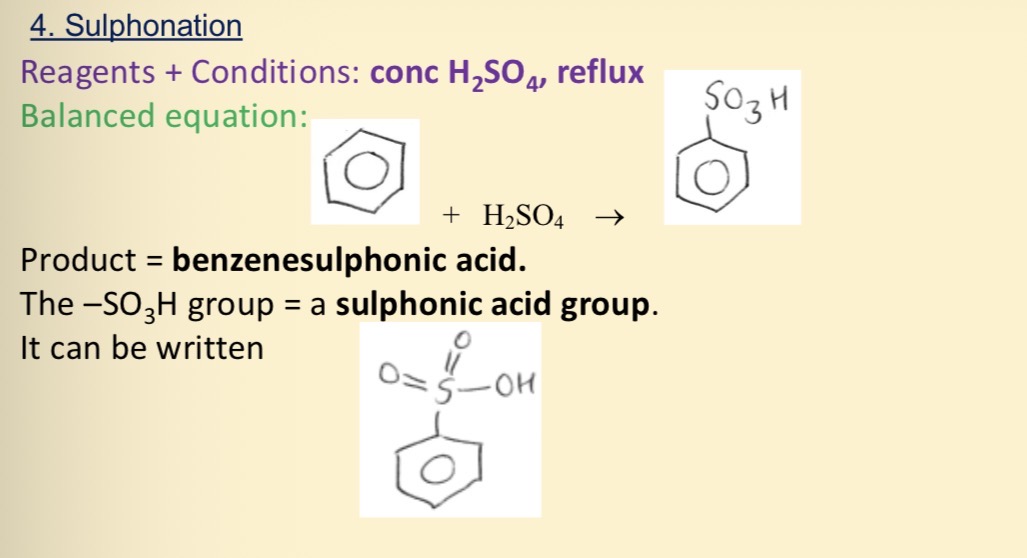
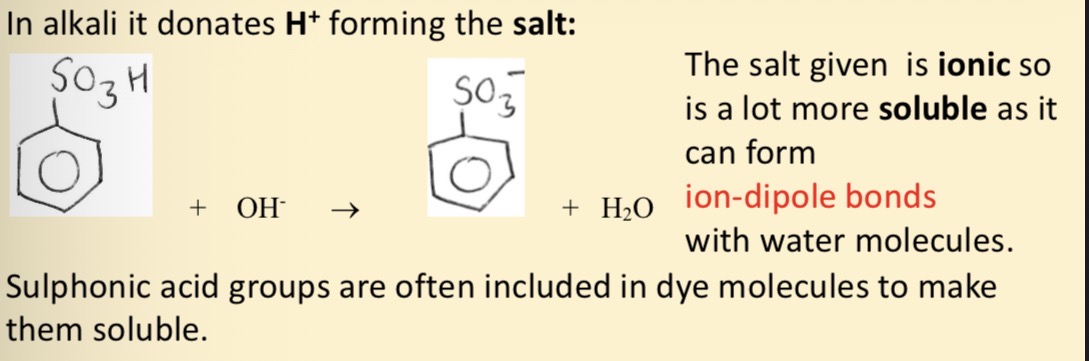
How does benzenesulphonic acid react in alkali
in friedel crafts alkylation what is alkylation and what type of molecule or species is needed as an electrophile
Alkylation = putting an alkyl group (carbon chain) onto the ring
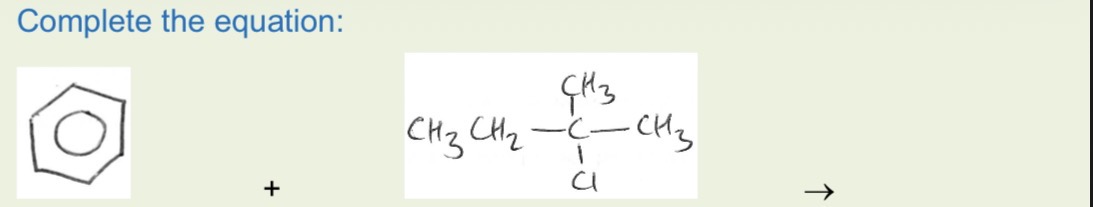
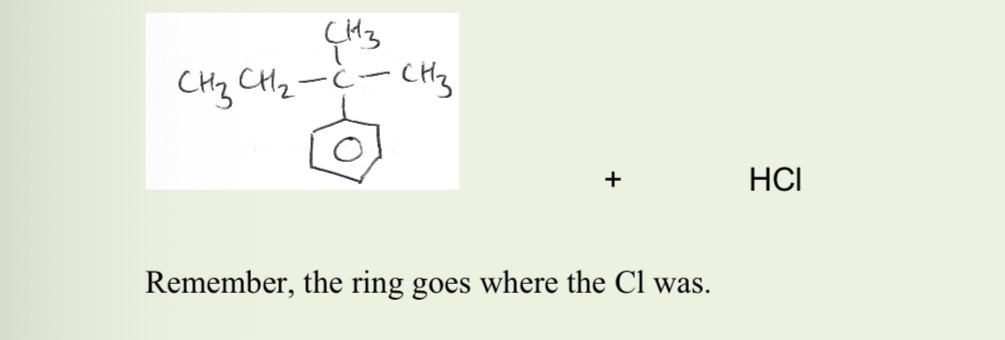
The molecule/species as an electrophile = carbocation (carbon chain with C+ where we want it to attach to benzene ring) which is made from the s+ C of haloalkane using AlCl3 catalyst.
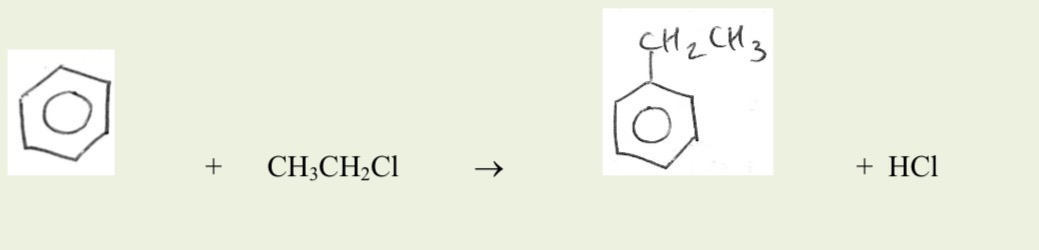
Reangents and conditions for Friedel- Crafts alkylation
The required chloroalkane with its Cl where the ring will go, AlCl3 catalyst, anhydrous, reflux
Balanced equation of friedel crafts alkylation
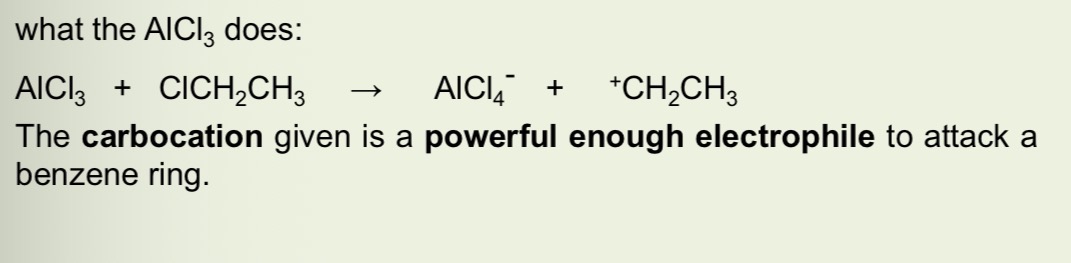
What does the AlCl3 catalyst do in friedel crafts alkylation alkylation
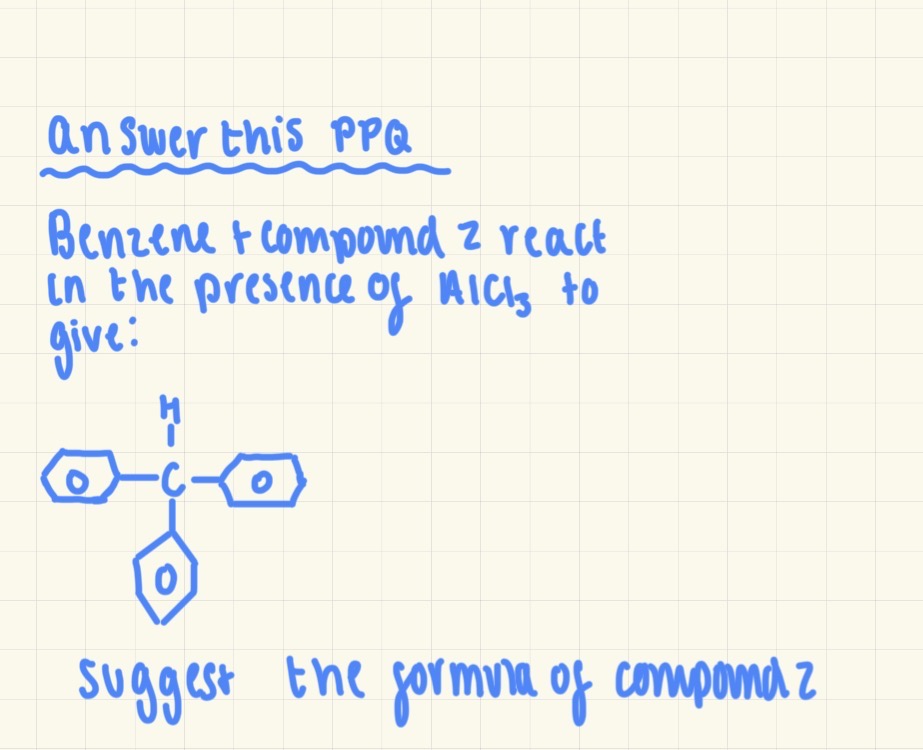
CHCl3