ORGANIC CHEM
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
List the order of priority of IUPAC groups
Carboxylic Acid
Ester
Acid Halide
Amide
Nitrile (CN)
Aldehyde
Ketone
Alcohol
Phenol
Amine
Alkene
Alkyne
Alkane
Ether
Alkyl Halide
Nitro (NO)
“Crazy Exes Always Act Nasty After Kissing At Parties And After-hours, Everyone Acts Naughty.”
pH < pka
protonate
pH = pka
protonate
pH > pka
deprotonate
Do SN1s and E1’s have rearrangements
Yes
Do SN2s and E2’s have rearrangements
No
Does SN2’s have inversions?
Yes
Does SN1’s have inversions?
No (± major/minor)
Does E2 care about sterochem?
Yes (Anti)
Does E1 care about sterochem?
No
Name 3 things about activating groups (e- density, EDG/EWG, o/p/m)
increasing e- density
EDG
o/p
Name 3 things about deactivating groups (e- density, EDG/EWG, o/p/m)
decreasing e- density
EWG
-F, -Cl, -Br, -I (deactivators)
m
Which 4 reagents do Michael (1,4) Addition?
CN
HNR2
R2CuLI
HSR
Which 4 reagents do Direct (1,2) Addition?
NaBH4
LiAlH4
-MgX
-Li
What is the hybridization of pi bonds in ethene?
2p-2p
What is the hybridization of sigma bonds in ethene?
sp2-sp2
What is the hybridization of the C-H bonds in ethene?
1s-sp2
Which is more acidic, EDG or EWG?
EWG > EDG
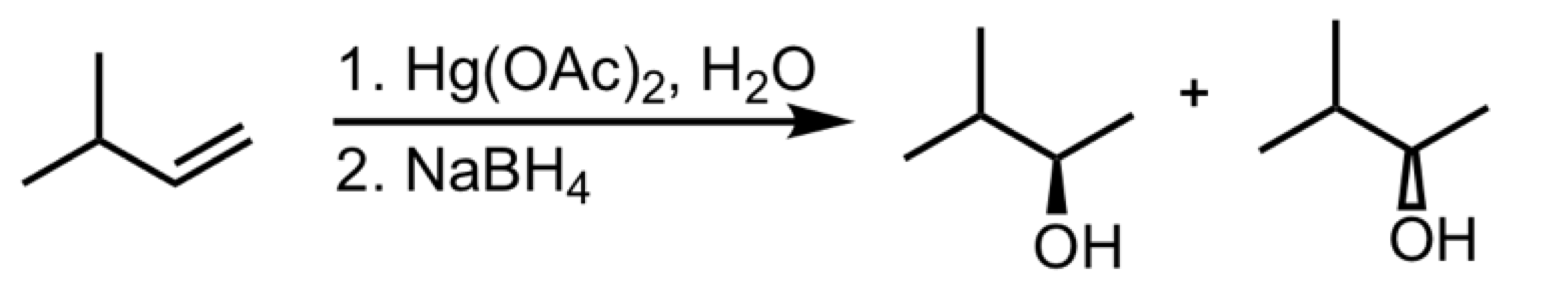
Oxymercuration-Demurcuration
Ozonolysis (Reducing Conditions)
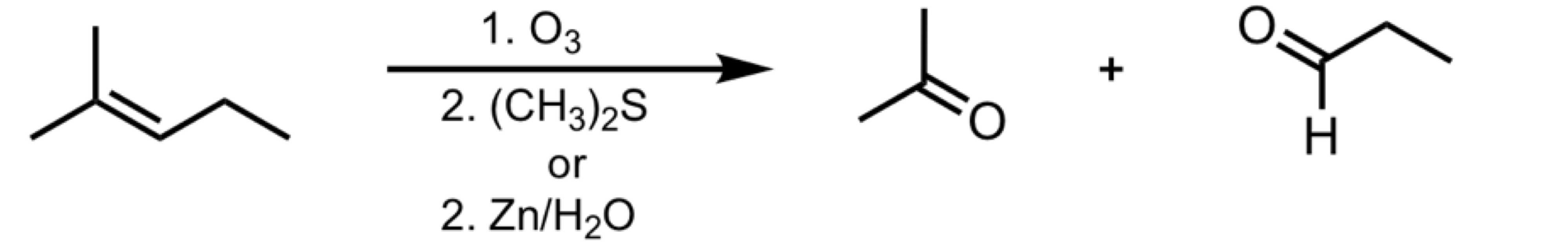
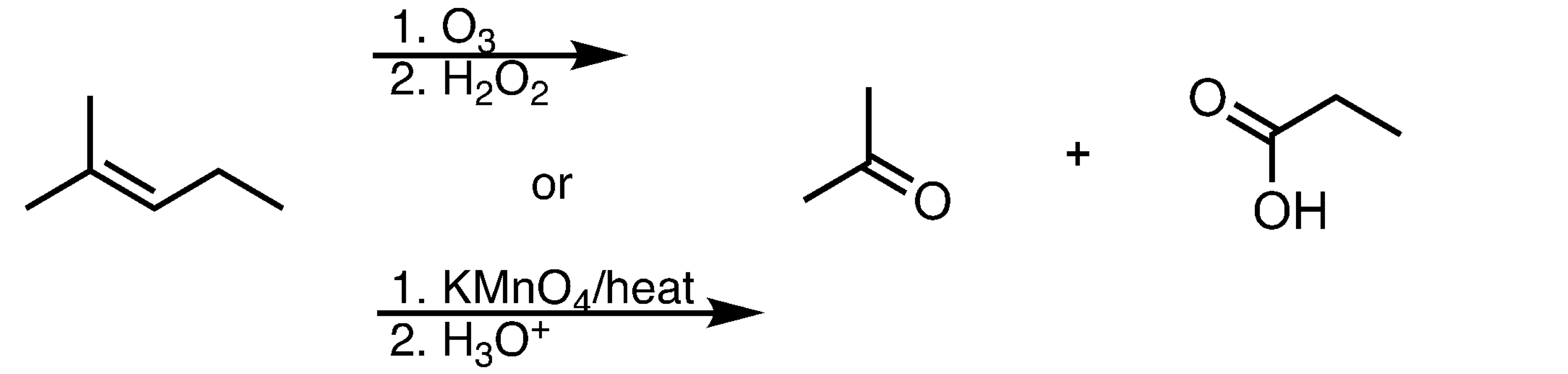
Ozonolysis (Oxidizing Conditions)
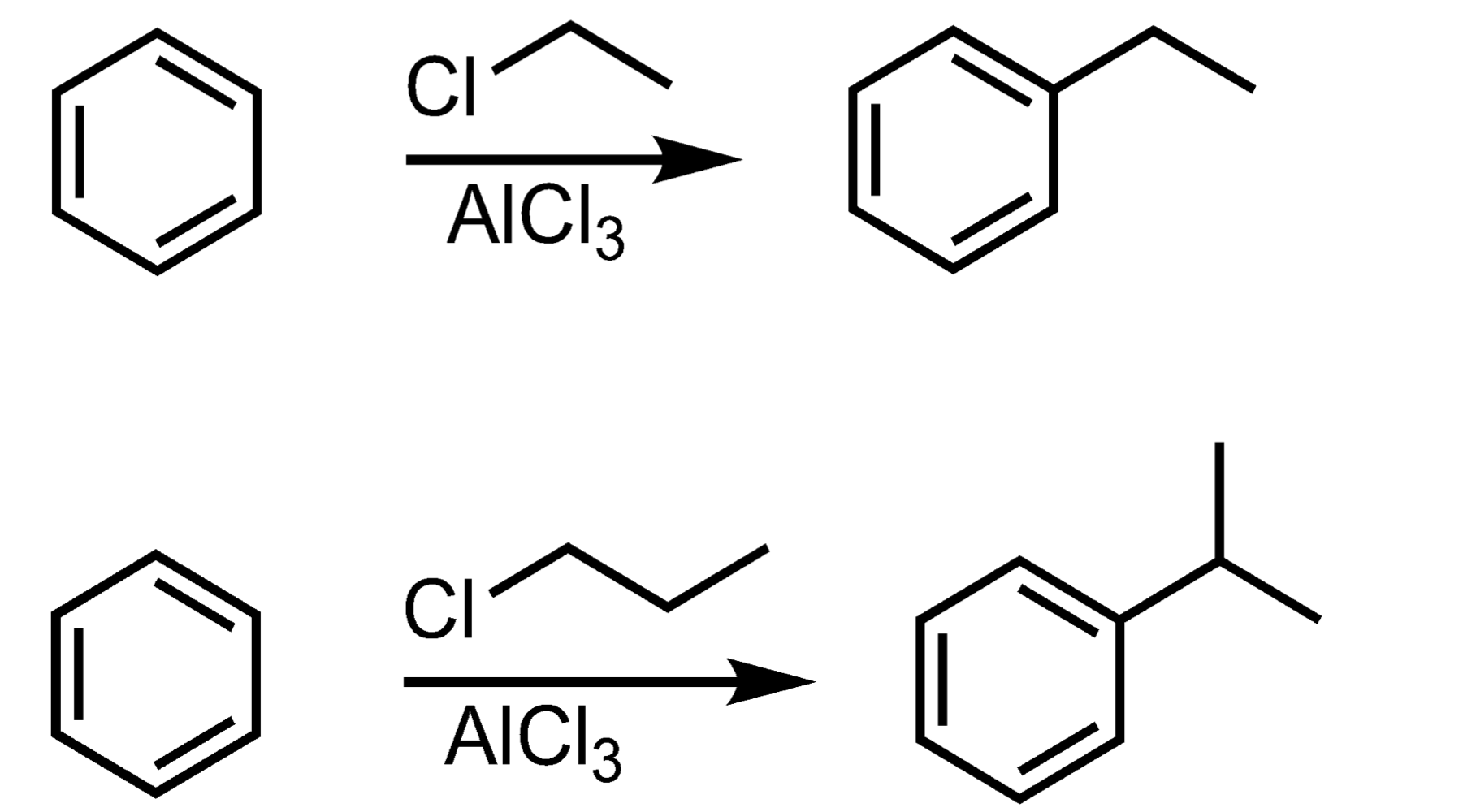
Friedel-Crafts Alkylation
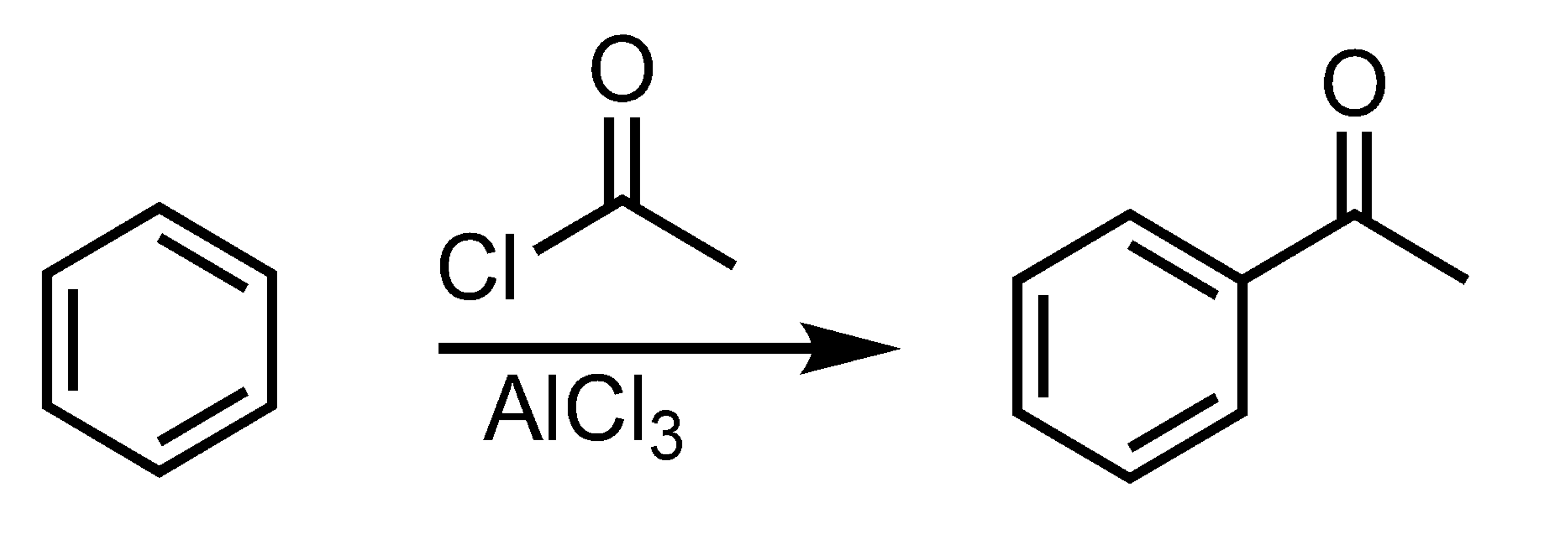
Friedel-Crafts Acylation
Wolff-Kishner Reduction
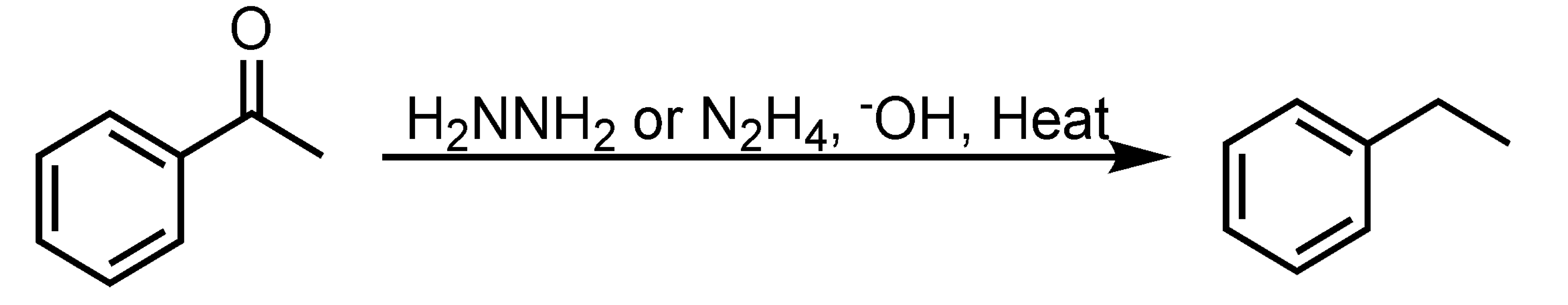
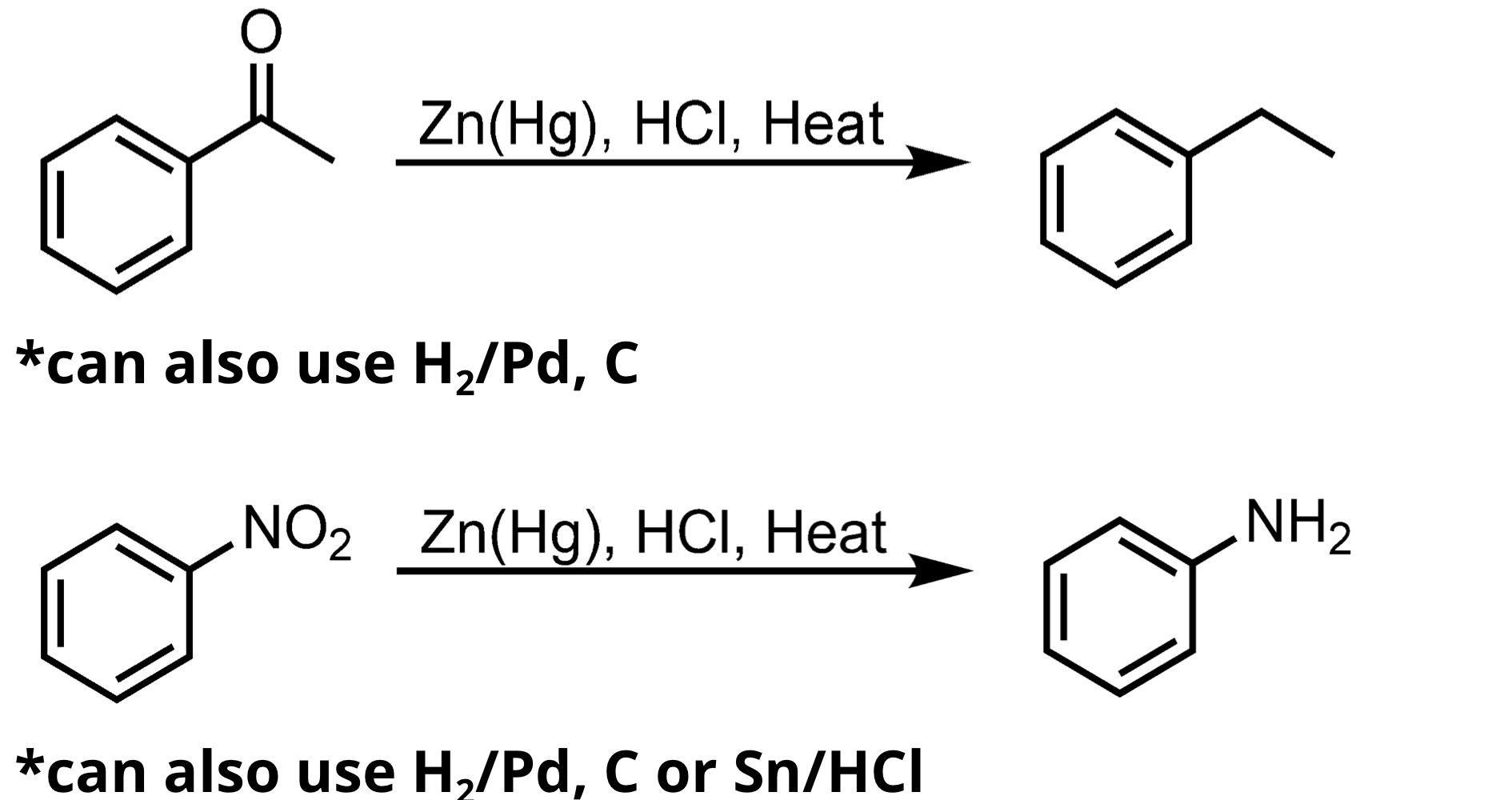
Clemmenson Reduction
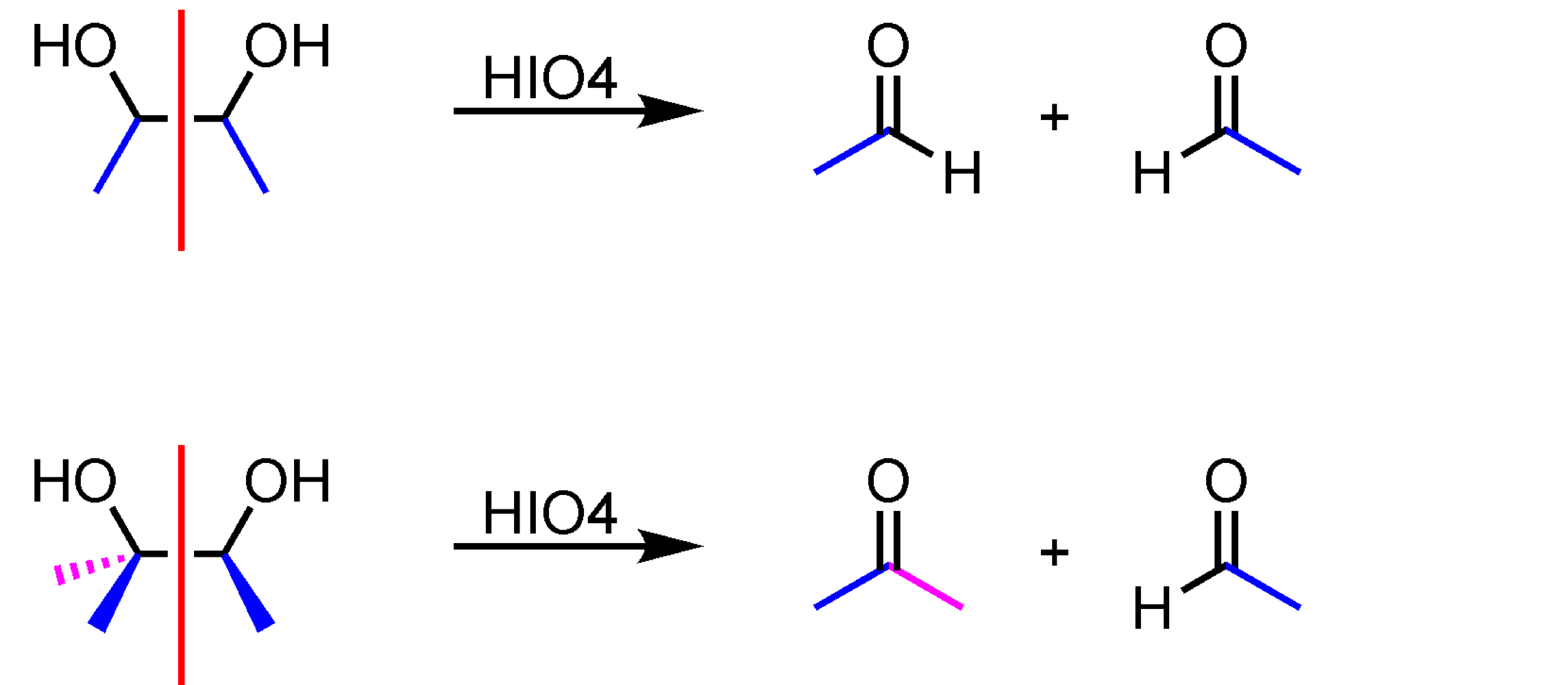
Oxidative Cleavage
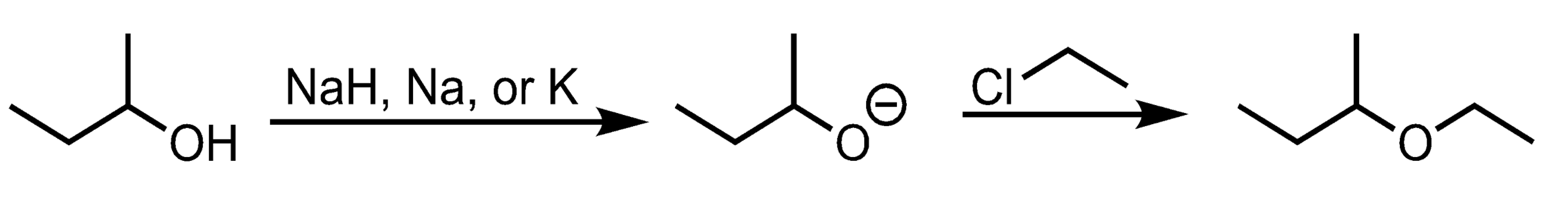
Williamson Ether
Self-Aldol Condensation
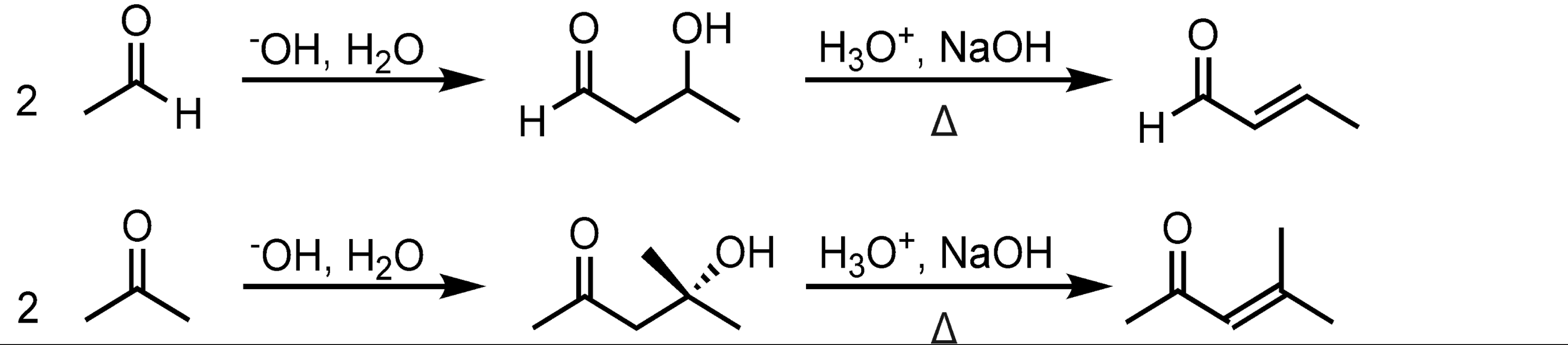
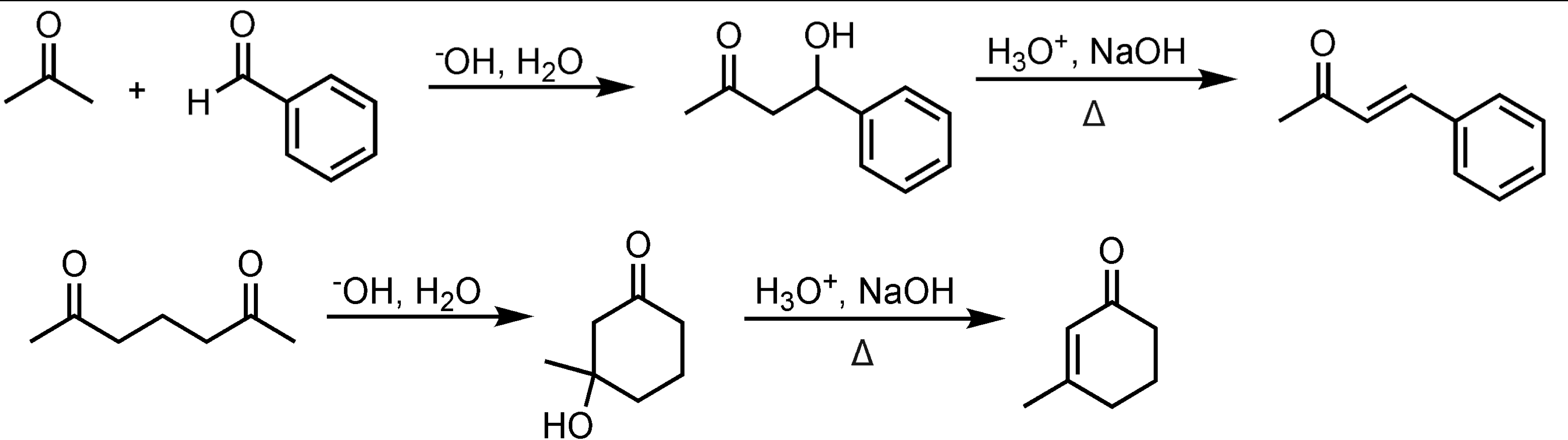
Mixed Aldol Condensation
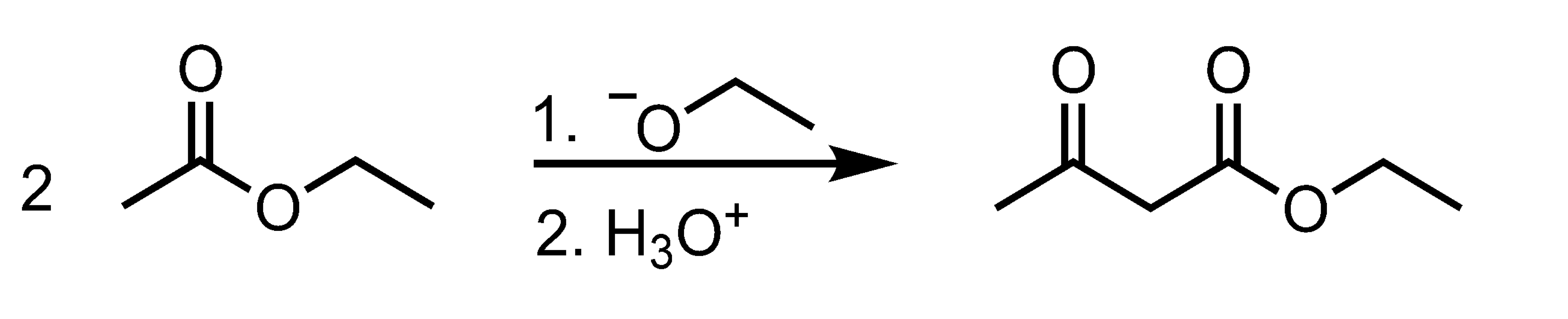
Self-Claisen Condensation
Mixed-Claisen Condensation
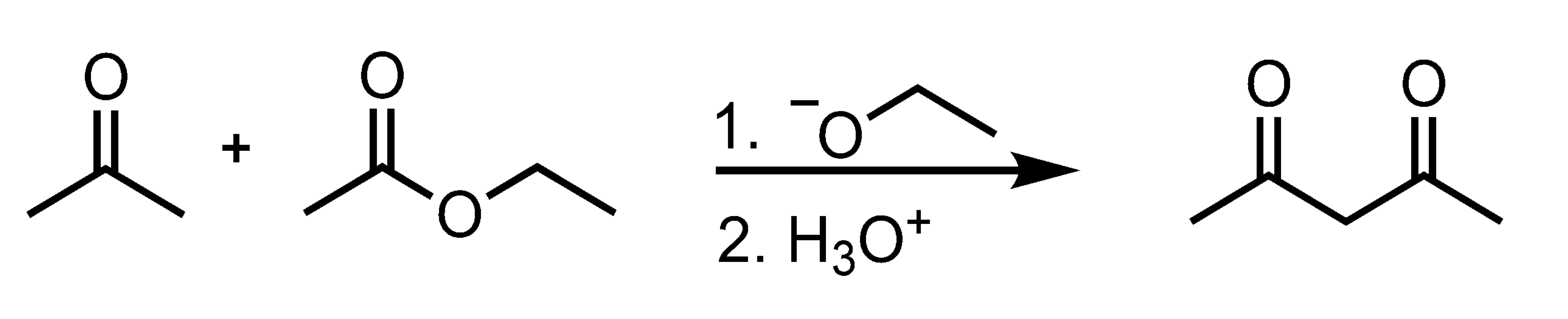
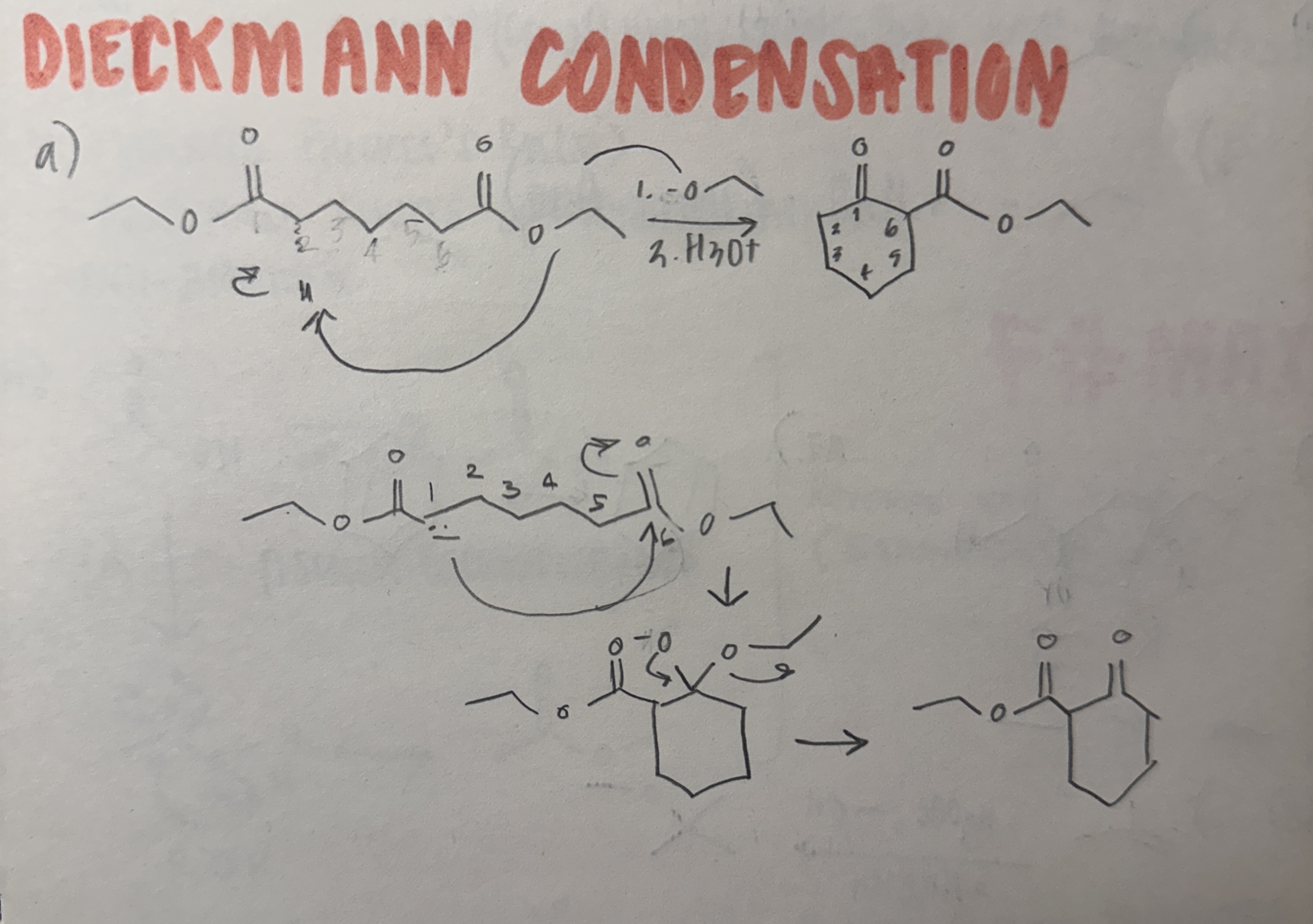
Dieckmann Condensation
What are 5 factors of thermodynamic and kinetic products
Kinetic
Low Ea
Less Stable
Low Temperature
High Energy
High Product
Thermodynamic
High Ea
More Stable
High Temperature
Low Energy
Low Product
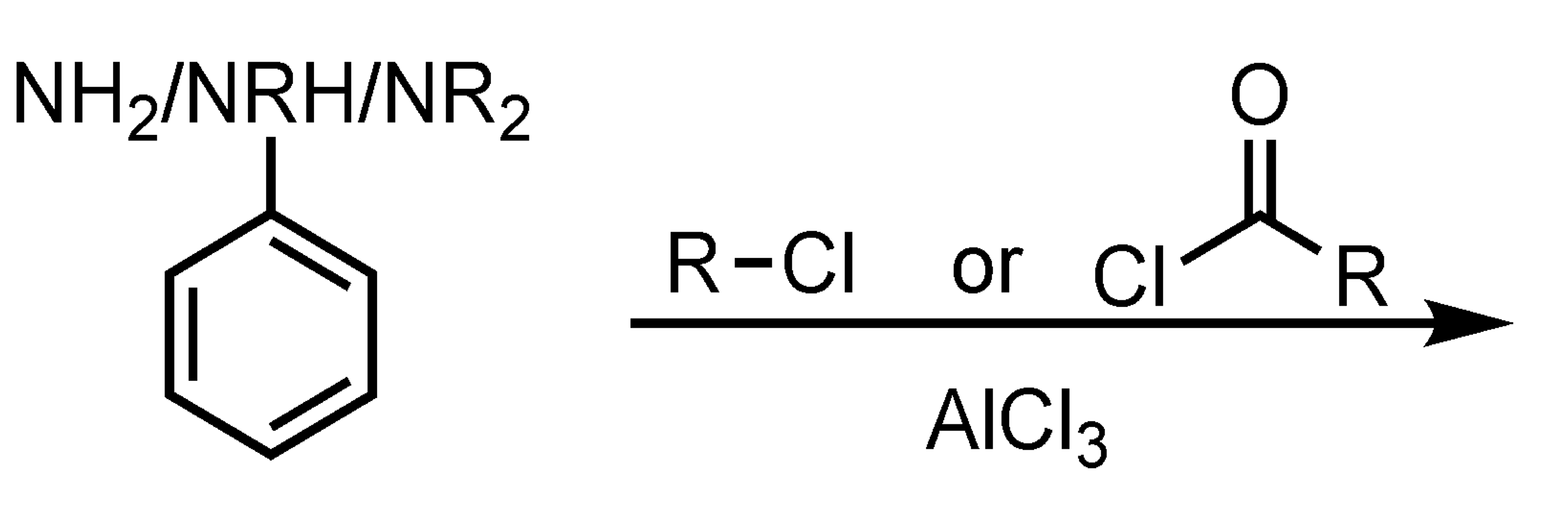
no reaction
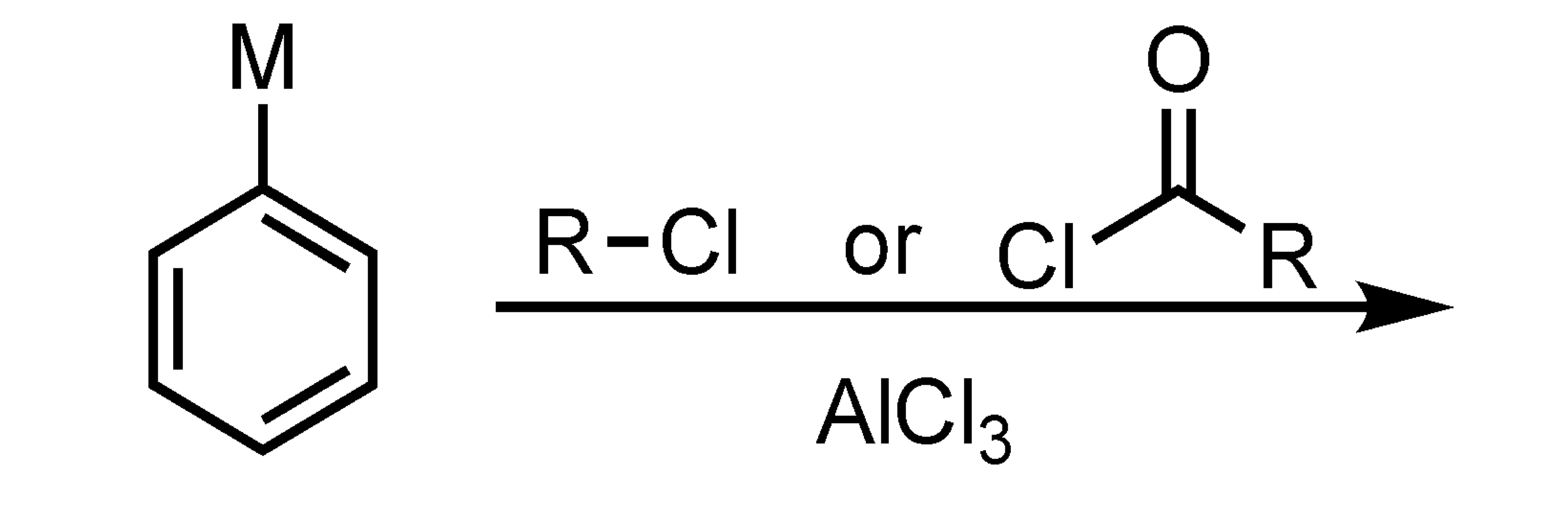
no reaction
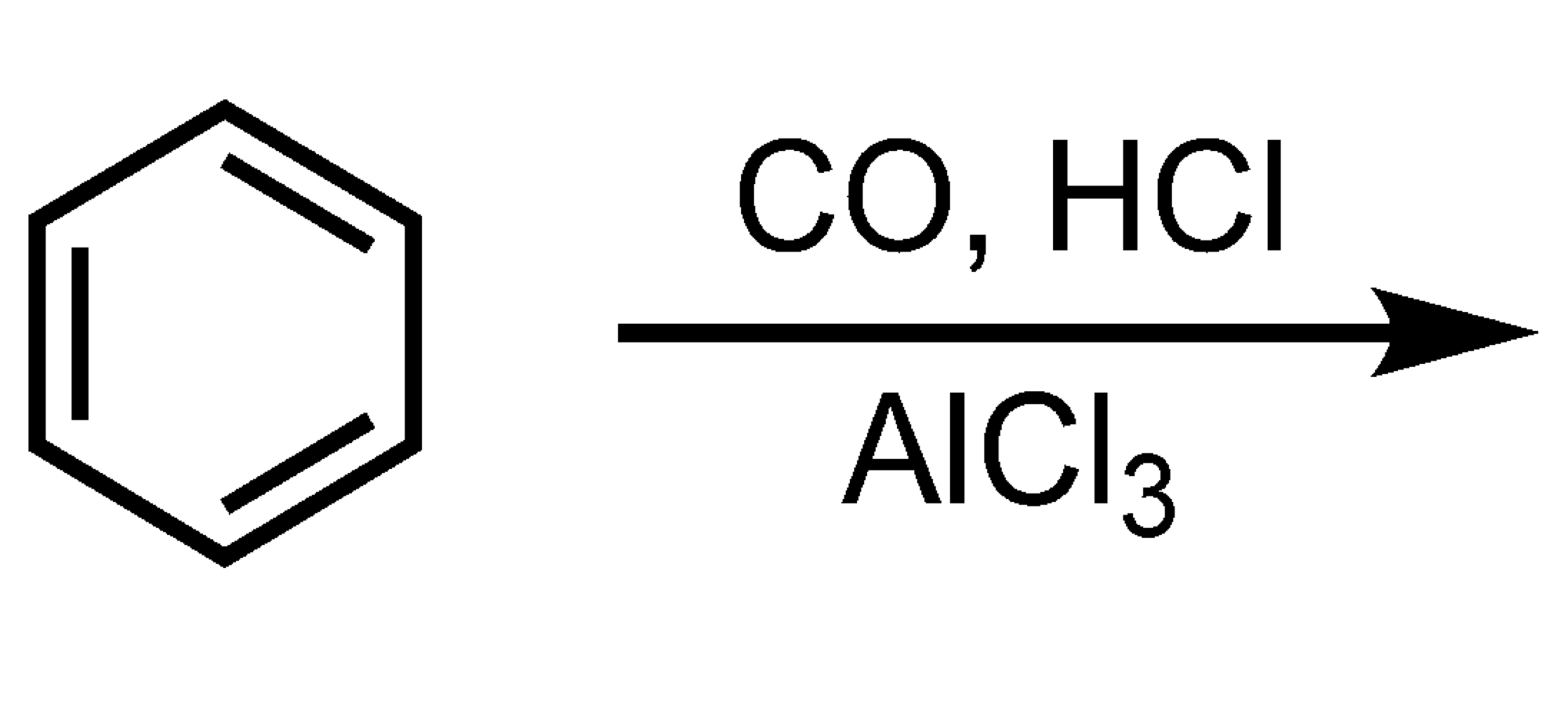
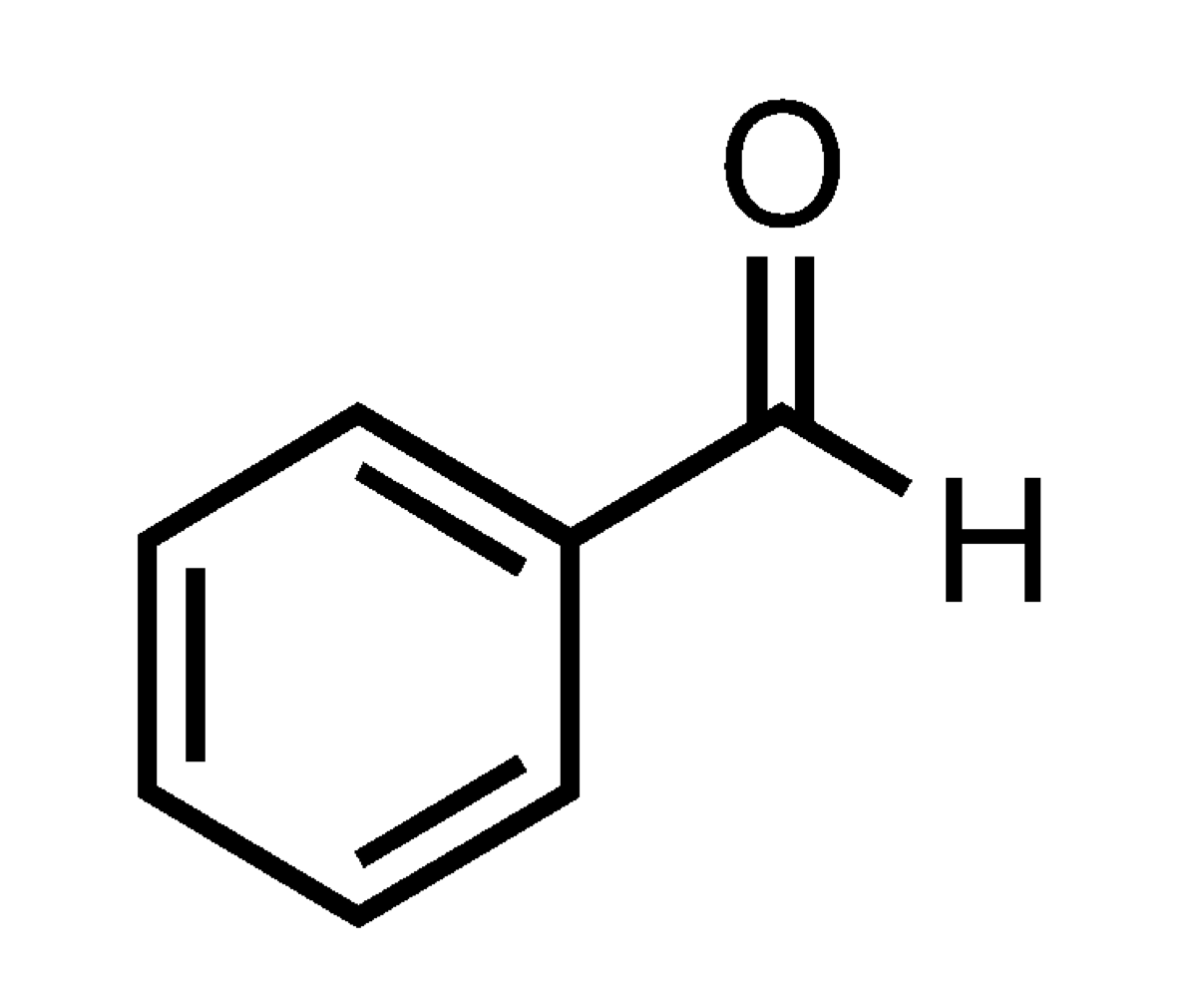
What type of acid or base is benzene?
Lewis Base
How do you calculate formal charge?
Formal Charge = (# valence e-) - (bonds + dots)
Hofmann Rearrangement
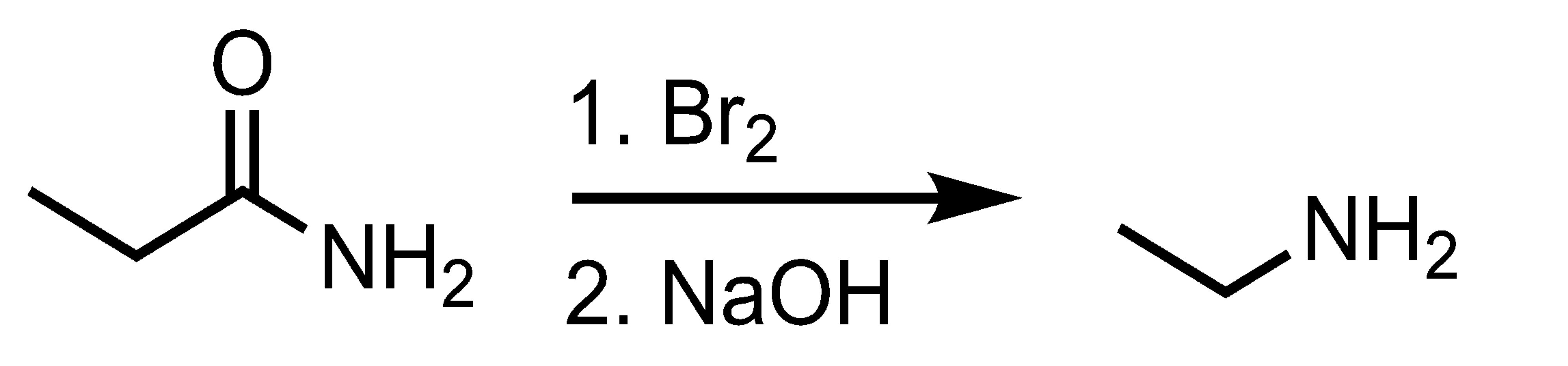
Will EDG react faster or slower than a sole benzene ring?
Faster than a benzene ring because it increased electron density
Will EWG react faster or slower than a sole benzene ring?
Slower than a benzene ring because it decreases electron density
What order of substitution does radical bromination prefer?
3* > 2* > 1* > methyl
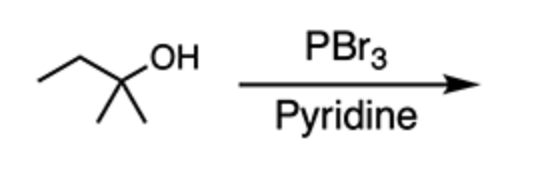
no reaction
Reduction of an acyl chloride with LTBA results in the formation of:
aldehyde
What is the difference between concentrated H2SO4 and dilute?
concentrated dehydrates and causes a double bond
dilute adds an -OH group
What is the order of least to most readily oxidized?
methyl < alcohol < aldehyde < COz
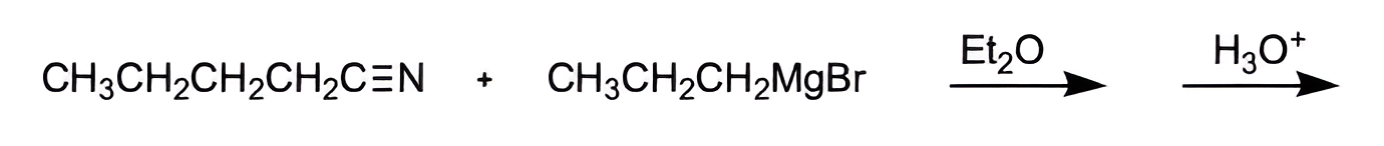
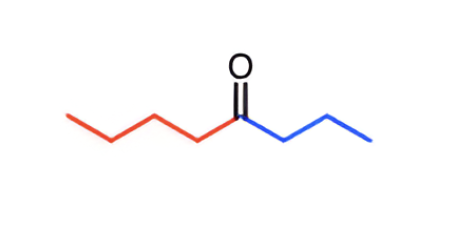
What is the order of most to least reactive of addition-elimination reactions?
carboxylic acid derivatives:
acid halide > anhydride > ester > amide