MICROBIO UNIT 5
1/235
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
236 Terms
how are lichens both a mutualism and microbial?
they are a mutualistic relationship between a fungus and an alga (cyanobacterium), where the fungus provides a structure for the phototrophic alga partner to grow within
do lichens have a microbiome?
yes, bacteria and archaea live on the lichen
what is a rhizosphere?
the root system of plants that interacts with the complex microbial ecosystem in the soil
what is a phyllosphere?
the leaf system of plants that is covered in microbes
vertical vs horizontal transmission of plants
vertical: colonized seeds form colonized plants
horizontal: plants are infected/colonized by their encounter with the environment
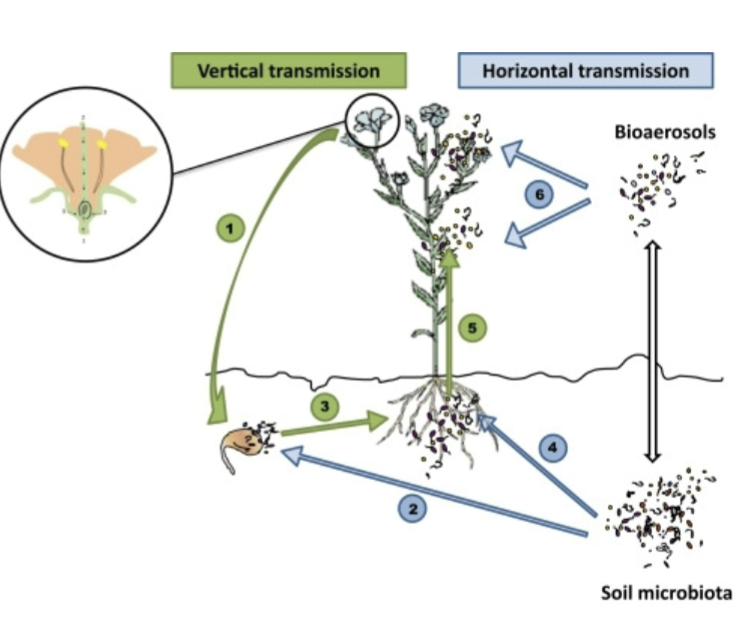
how is a plant’s microbiome beneficial to plants?
enhances nutrient absorption, stimulates growth, protect from pathogens and toxins
what are legumes?
plants with seeds in pods
why can legumes grow in nitrogen-poor soils?
because of their mutualistic relationship with nitrogen-fixing bacteria, which is one of the most important symbioses known. They have root nodules that provide nitrogen to both plant and soil.
how do the root nodules help provide nitrogen to the plant and soil?
free oxygen is bound up by O2 binding protein leghemoglobin that serves as an oxygen buffer to protect nitrogenases from free oxygen
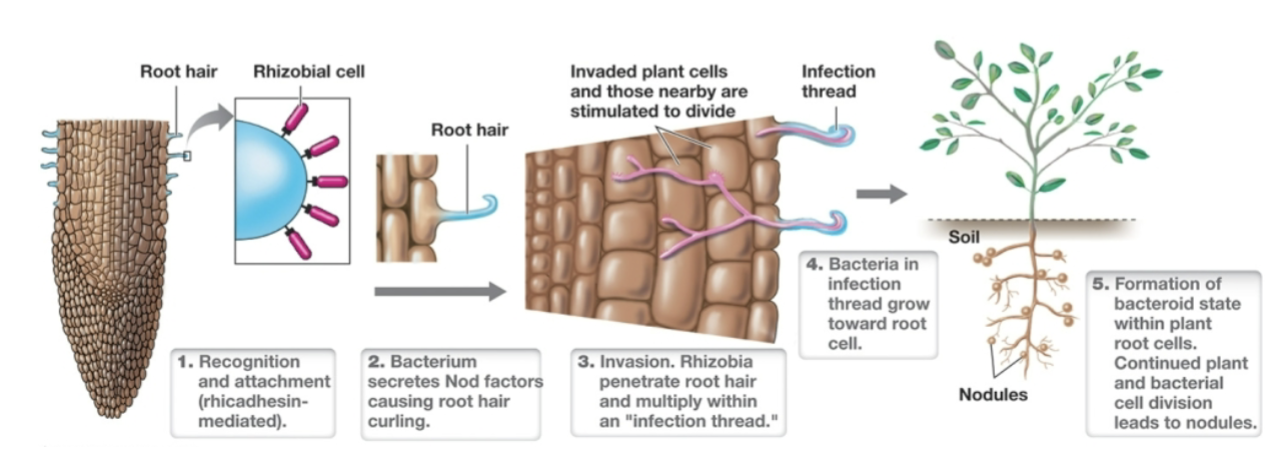
Would you expect a legume that lacked leghemoglobin to have access to more or less usable nitrogen? Why?
less nitrogen, because leghemoglobin would not be there to act as an oxygen buffer
what percent of the nitrogen fixed on earth each year is fixed in root nodules?
10-20%
how do mycorrhizae promote plant growth?
they are mutualistic fungi that improve nutrient absorption and overall plant growth
are mycorrhizae eukaryotic or prokaryotic
they are eukaryotic
how does the micorrhizae fungus benefit from the relationship with plants?
the plant can perform photosynthesis, unlike the fungi, so the host plant provides energy, feeding it carbohydrates and lipids that it produces from photosynthesis
what is bioremediation?
using microorganisms to clean up polluted environments
bioaugmentation vs biostimulation
bioaugmentation: adding exogenous bacteria to a spill site
biostimulation: providing additional nutrients for existing bacteria to grow
which form of bioremediation is more commonly used?
biostimulation
describe the mutualism between the Hawaiian bobtail squid and A. fischerii.
the squid supplies nutrients to the bioluminescent A. Fischeri bacteria in the specialized light organ structure. The transmission of bacteria cells is horizontal, and the bacteria emits light (glow) that camoflages the squid from predators.
what discovery did the relationship between the bobtail squid and A. fischerii lead to?
quorum sensing, because bacteria only glow when at high numbers.
would a strain of A. fischerii that lacked quorum sensing receptors be able to participate in this mutualism?
no, because the squid’s defense mechanisms would prevent the relationship from being established.
how do chemolithotrophic bacteria form the foundation of the food chain near deep sea vents?
where there is no sunlight in the deep sea, chemolithotrophic prokaryotes that utilize reduced inorganic materials emitting from vents form endosymbiotic relationships with vent invertebrates. vent tube worms harbor several features that facilitate growth of endosymbionts (e.g. specialized hemoglobin, high blood CO2 content)
how do symbiotic fungi benefit cicadas and aphids?
cicadas eat sap (which is AA deficient) almost exclusively, and most cicadas have an internal symbiont that makes their amino acids for them
aphids have heritable symbionts of insects. primary symbionts required for host to reproduce.
what determines the nature of the microbial-host interaction?
the pathogenicity of the microbe and the resistance of the given host
what is the disease triangle? What factors affect this interaction?
pathogenicity of microbe: virulence factors, nature of pathogen
resistance of host: immune status, age, nutrition
environment - how pathogens and hosts interact
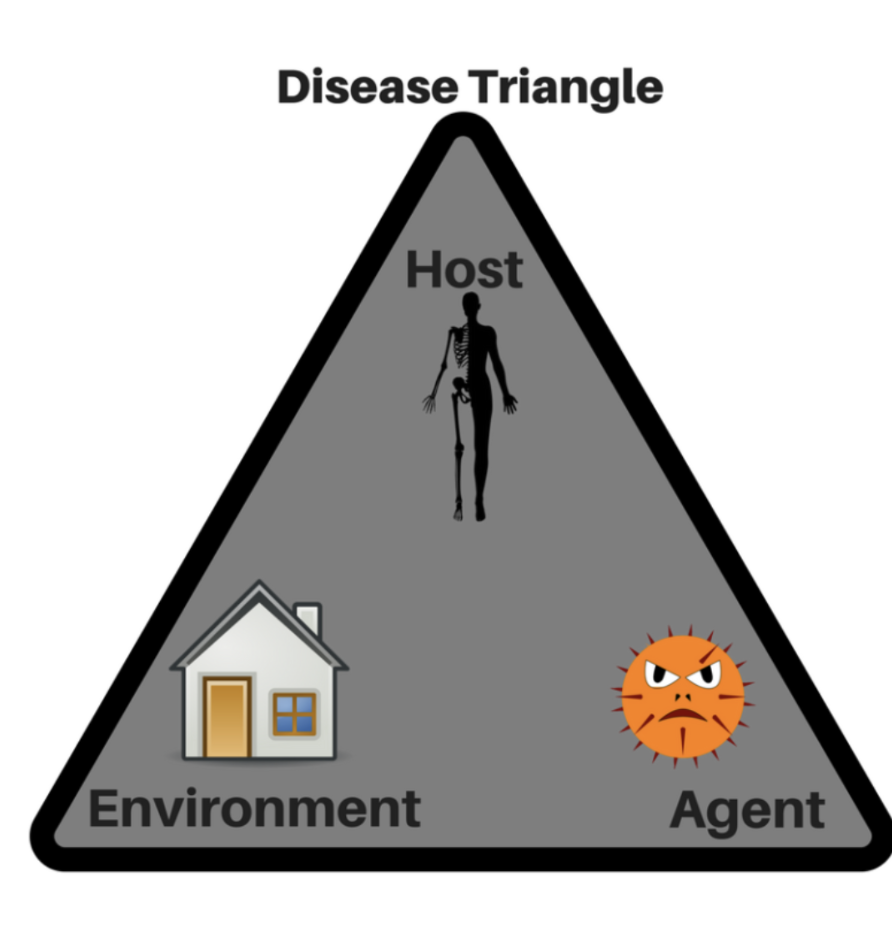
how do COVID-19 precautions influence the disease triangle?
they disrupt the interaction between the agent (SARS-CoV-2), the host (humans), and the environment.
how many microbes are in the human microbiome?
approximately 10^13. They are not evenly distributed — different bacteria classes are present in different ratios depending on system (e.g. skin, urogenital tract, gastrointestinal tract)
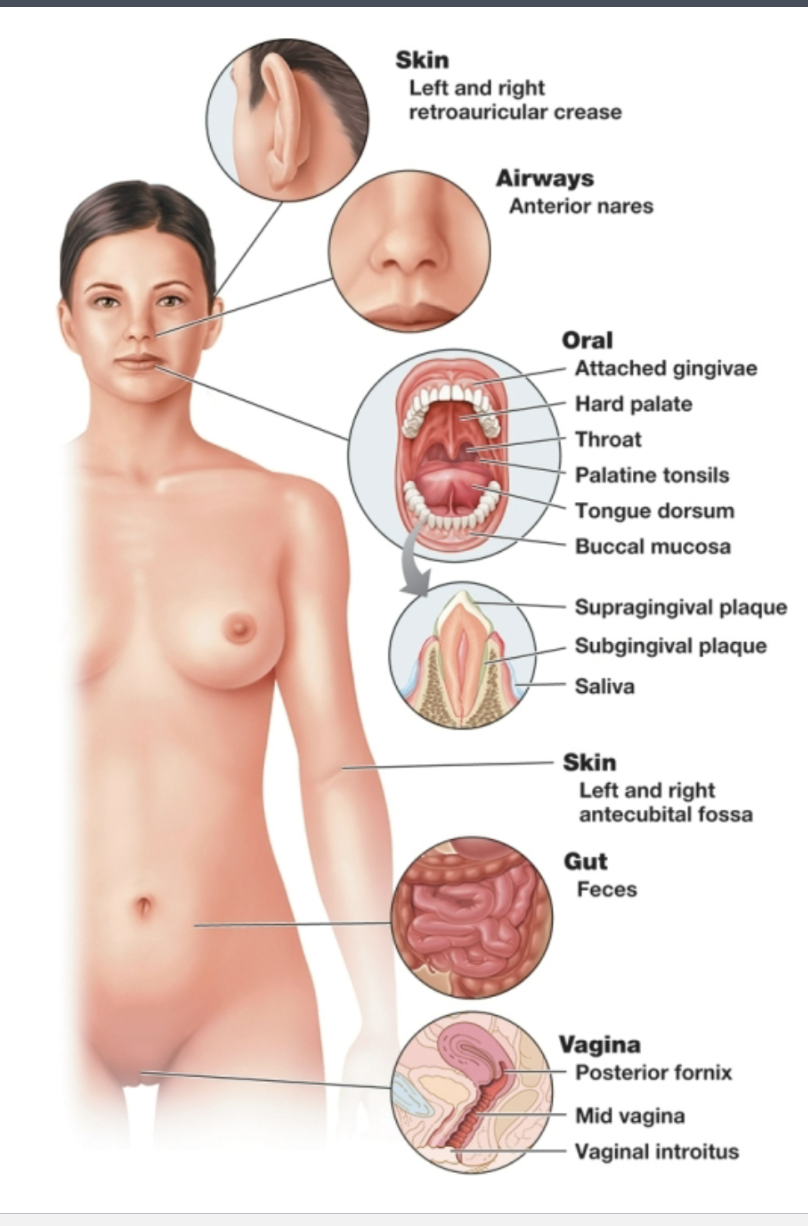
metabolites of ____ microbes play an important role in host health
intestinal
how do people acquire normal microbiota?
probiotics and prebiotics can help improve gut microbiota and diversity
what are examples of microbial metabolites in the gut that influence host physiology?
vitamin production, modification of steroids, amino acid biosynthesis
what is the pH of the stomach?
very acidic (pH ~2)
what disease is caused by Helicobacter pylori?
Helicobacter pylori cause gastric ulcers by burrowing into the protective mucus of the stomach (survives at pH 1)
what factors can decrease the acidity of the stomach?
malnutrition, medication, genetics
what are the 3 phyla of the gut microbiota?
Firmicutes, Bacteriodetes, Proteobacteria
what phyla are normally present in humans?
mostly Firmicutes, Bacteriodetes, or a mix of the two
what is poor microbial diversity associated with, health-wise?
gut dysbiosis and poor GI function and disease
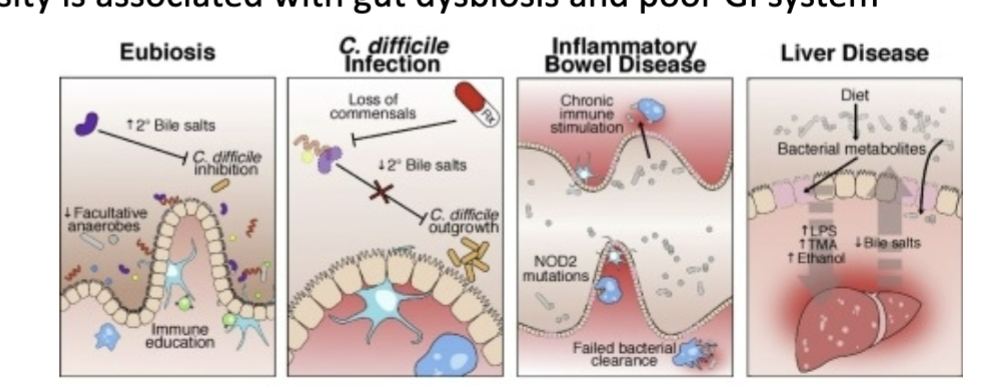
define gut microbiome dysbiosis:
chronic inflammation of the gut and disruption of homeostasis (dysbiosis)
what is Clostridium difficile? How does it occur?
a disease that causes severe diarrhea, colon inflammation, and dangerous intestinal conditions. It is a spore former that usually results from antibiotic treatments that cause dysbiosis.
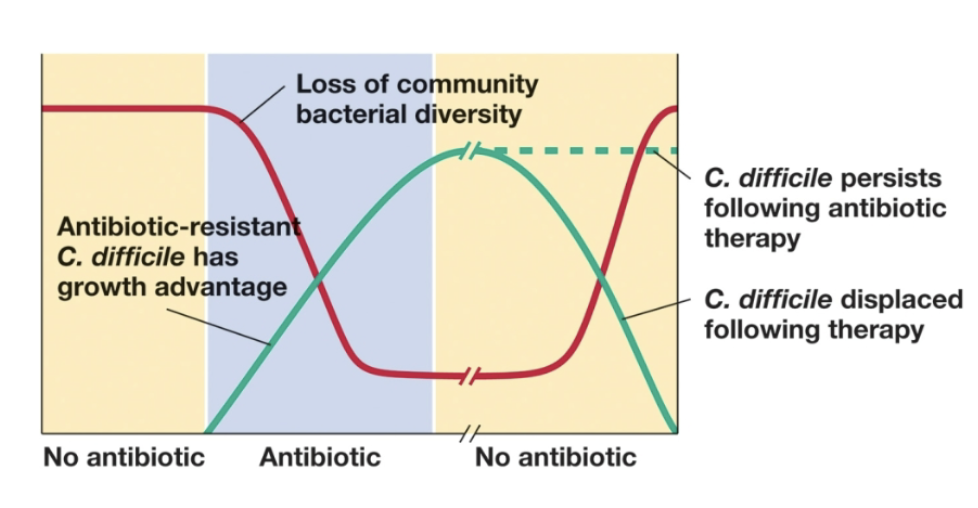
what is the usual effective treatment for Clostridium difficile?
fecal transplants have a ~90% effectiveness because they introduce healthy, diverse bacteria back to the gut.
what other disorders are associated with gut dysbiosis?
chronic inflammation of gut
IBD (inflammatory bowel disease)
is the microbiota of a patient with IBD more or less diverse on average than a person without this disorder?
patients with IBD have a LOWER gut microbiome diversity
what are gnotobiotic mice?
mice that lack normal microbiota
why are mouse models used to study gut host-microbe interactions?
they have a short life cycle and well-defined genetic lines, and can be raised in a germ-free environment.
what gut microbiome tests are mice used in?
antibiotic therapy
strict dietary control
fecal transplants to humanize mouse microbiota
germ-free environment (gnotobiotic)
behavioral studies
probiotics vs prebiotics vs synbiotics
probiotics: live microorganisms that improve microbiota balance and exert scientifically proven health
prebiotics: non-digestible food additives that promote growth of “good” bacteria. fuel existing bacteria in your gut.
synbiotics: combine probiotics with prebiotics that promote their growth
what is an infection?
a situation in which an organism is established and growing in a host, whether or not the host is harmed
what are pathogens?
microbial parasites that cause disease, or tissue damage in a host
pathogenicity
ability of a parasite to inflict damage on the host
what is adhesion?
the enhanced ability of microbes to attach to host tissues
what are adhesins?
glycoproteins or lipoproteins found on pathogen’s surface that enable it to bind to host cells
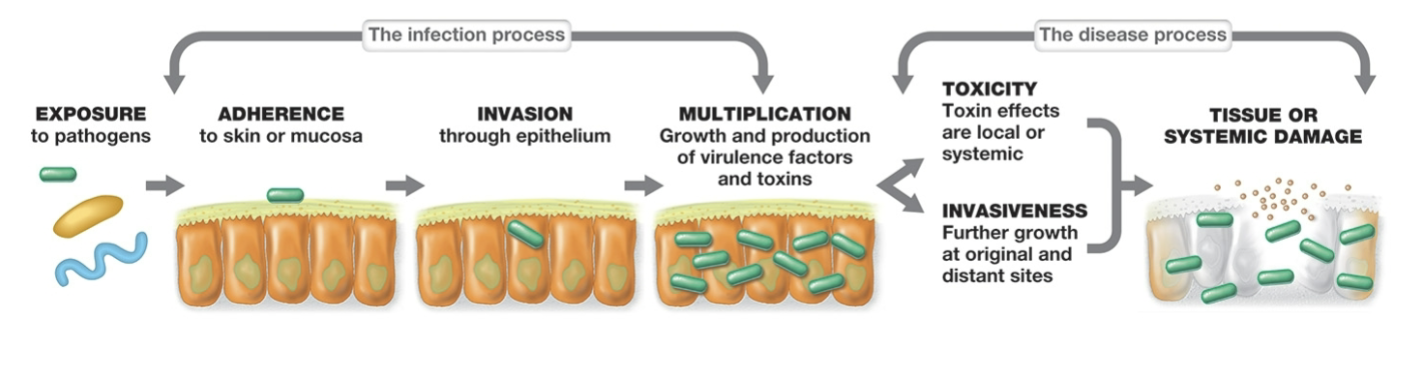
Diagram the infection process from adherence to disease.
exposure to pathogens —> adherence to skin or mucosa —> invasion through epithelium —> multiplication (growth and production of virulence factors and toxins) → toxicity or invasiveness —> tissue or systemic damage
what are the 3 different mechanisms of adhesion?
capsules — provide stickiness and immune invasion
adhesions — specific attachment
fimbraie or pili — specific attachment
how does toxicity differ from invasiveness?
invasiveness is the ability of a pathogen to grow in host tissue at densities that inhibit host function (ability to spread rapidly)
toxicity is the capacity of an organism to cause harm to humans
how does bacteremia differ from septicemia? which is more severe?
bacteremia: presence of bacteria in the bloodstream
septicemia: bloodborne systemic infection
septicemia is more severe because it may lead to massive inflammation, septic shock, and death
You are comparing 2 pathogens: one with an LD50 of 100 and one with an LD50 of 10,000. Which pathogen is more virulent?
LD50 means lethal dose to kill 50% of an infected test population
virulence and LD50 have an inverse relationship, so a LOWER LD50 means the pathogen is more virulent. Therefore, the LD50 of 100 is more virulent.
virulence is often determined by the ____ of the pathogen
genetics
(e.g. Salmonella. several genes that direct invasion are clustered togther on the chromosome as pathogenicity islands)
what virulent traits does Salmonella have?
it has several genes that direct invasion that are clustered together on the chromosome as pathogenicity islands
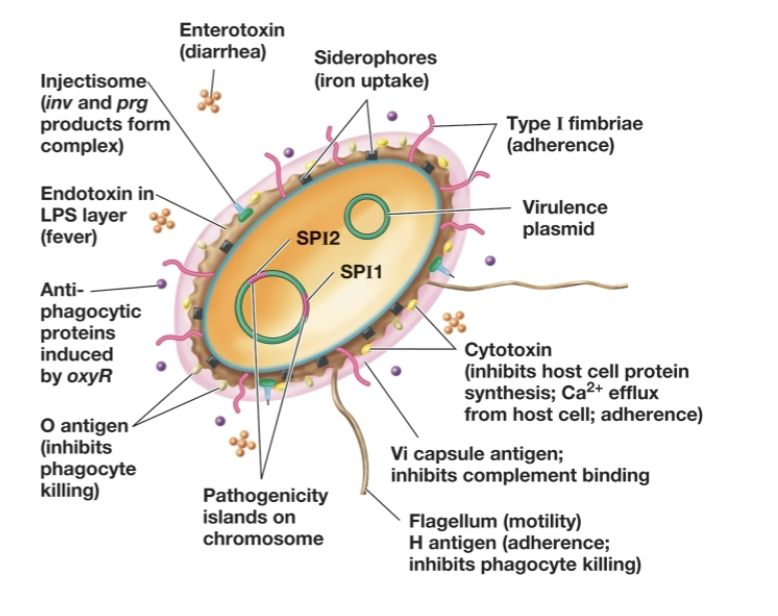
what are pathogenicity islands?
distinct segments of DNA within bacterial chromosomes that encode virulence factors
how does viral infection often lead to secondary bacterial infections, particularly respiratory pathogens?
The initial virus effectively terraforms the respiratory tract, altering the host’s physical, immunological, and metabolic landscape to create a highly permissive environment for opportunistic bacteria.
in other words, underlying conditions can predispose individuals to develop diseases.
what are opportunistic infections?
those caused by organisms that do not cause disease in healthy hosts
What are the different modes of disease transmission?
direct (person-to-person)
indirect (fomites, soil-borne infections)
horizontal
vertical (e.g. from insect to offspring)
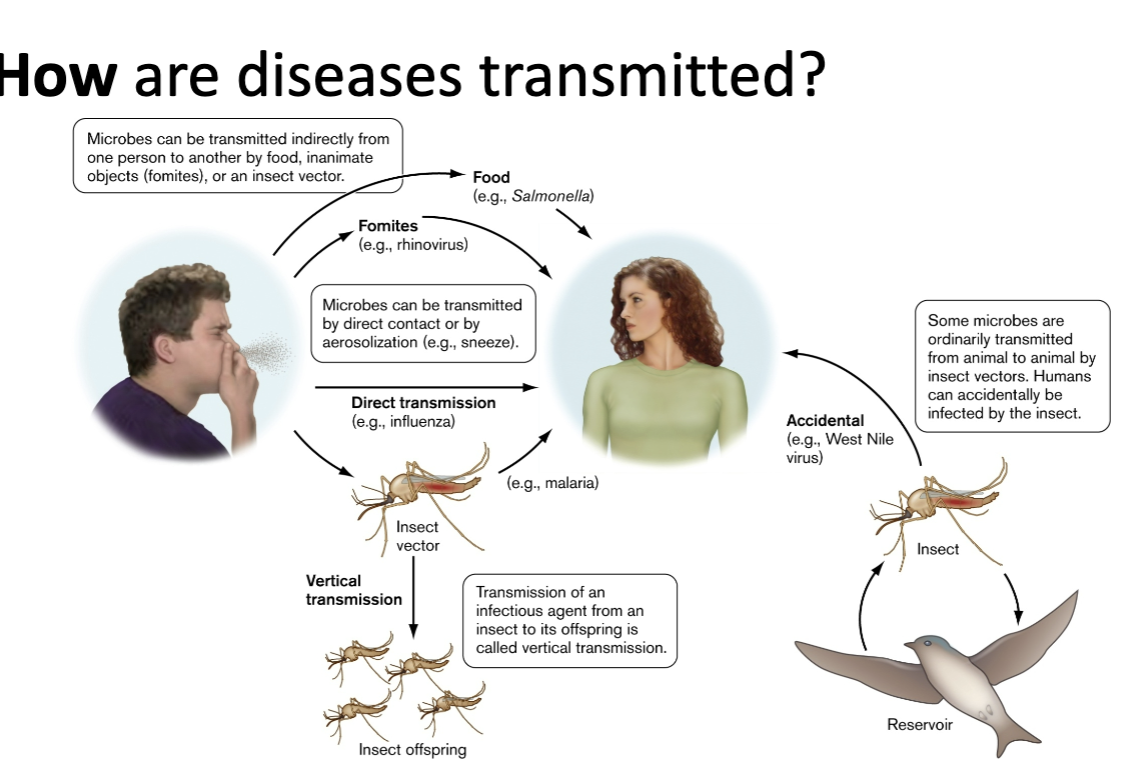
when offspring are born infected, is this vertical or horizontal transmission?
vertical
what is formite transmission?
indirect transmission of microbes by inanimate objects
what is active escape of pathogen from one host?
movement of pathogen to portal of exit
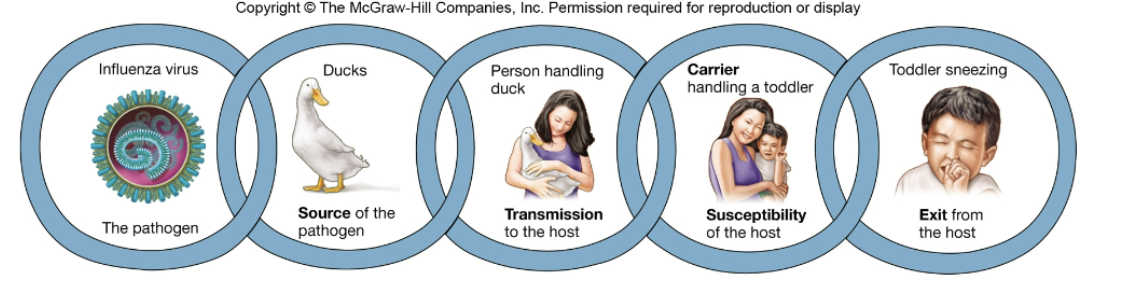
what is passive escape of pathogen from host?
excretion in feces, urine, droplets, blood, saliva, or desquamated cells.
how are the method of entry linked to the disease and symptoms caused?
the portal of entry of disease is tightly linked to the resulting disease, and the initial disease symptoms are almost always localized to the portal of entry
fecal-oral disease (where mouth is mode of entry) often are linked to what kinds of diseases/symptoms?
gastrointestinal symptoms (diarrhea, nausea, vomiting)
respiratory portal of entry (inhalation) is linked to what symptoms?
respiratory systems like coughing, sneezing, inflammation of respiratory tract
sexually transmitted pathogens are often found
in the urogenital tract or reproductive organs
what are three different main types of infection?
localized, systemic, focal
localized vs systemic vs focal infection:
localized: microbe enters body and remains confined to specific, limited site of infection
systemic:: generalized infection characterized by widespread distribution (usually transported through bloodstream or lymphatic system)
focal: hybrid—begins as localized, but circumstances allow it to spread to a secondary location where it is again confined.
how do Clostridium tetani and Streptococcus pneumoniae differ in their mode of infection and nature of disease?
Clostridium tetani is acquired from environmental exposure and is non-communicable, causing a focal infection. If the bacteria spores are allowed to germinate, they can secrete a potent neurotoxin that can cause potential death.
Streptococcus pneumoniae spreads through person to person contact via respiratory droplets (coughing or sneezing). opportunistic pathogen that starts as a localized infection and has potential to become systemic infection.
invasiveness requires a pathogen to break down host tissues. this is often done with _____ that attack host cells.
enzymes
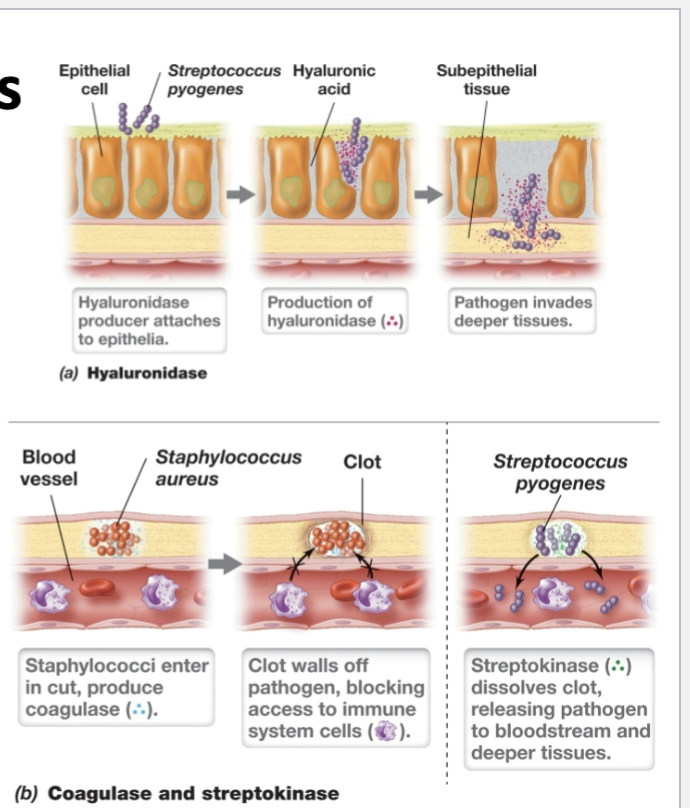
streptokinase vs coagulase vs hyaluronidase
streptokinase: dissolves blood clots
coagulase: triggers blood clotting
hyaluronidase: breaks down host tissues
what is toxicity?
ability of an organism to cause disease by means of a toxin that inhibits host cell function or kills host cells
what are exotoxins?
proteins that are secreted by the pathogen
what are the three categories of toxins?
cytolytic toxins
AB toxins
superantigen toxins
how to cytolytic exotoxins work?
they degrade cytoplasmic membrane integrity, causing cell lysis and death
what are AB toxins?
vary greatly, but have similar structure that is made up of an active (A) domain and a binding (B) domain
what kind of toxin is diphtheria? how does it cause disease?
it is a AB exotoxin that blocks protein synthesis by the action of the A domain adding an ADP-ribosyl group to EFs, which prevents translation
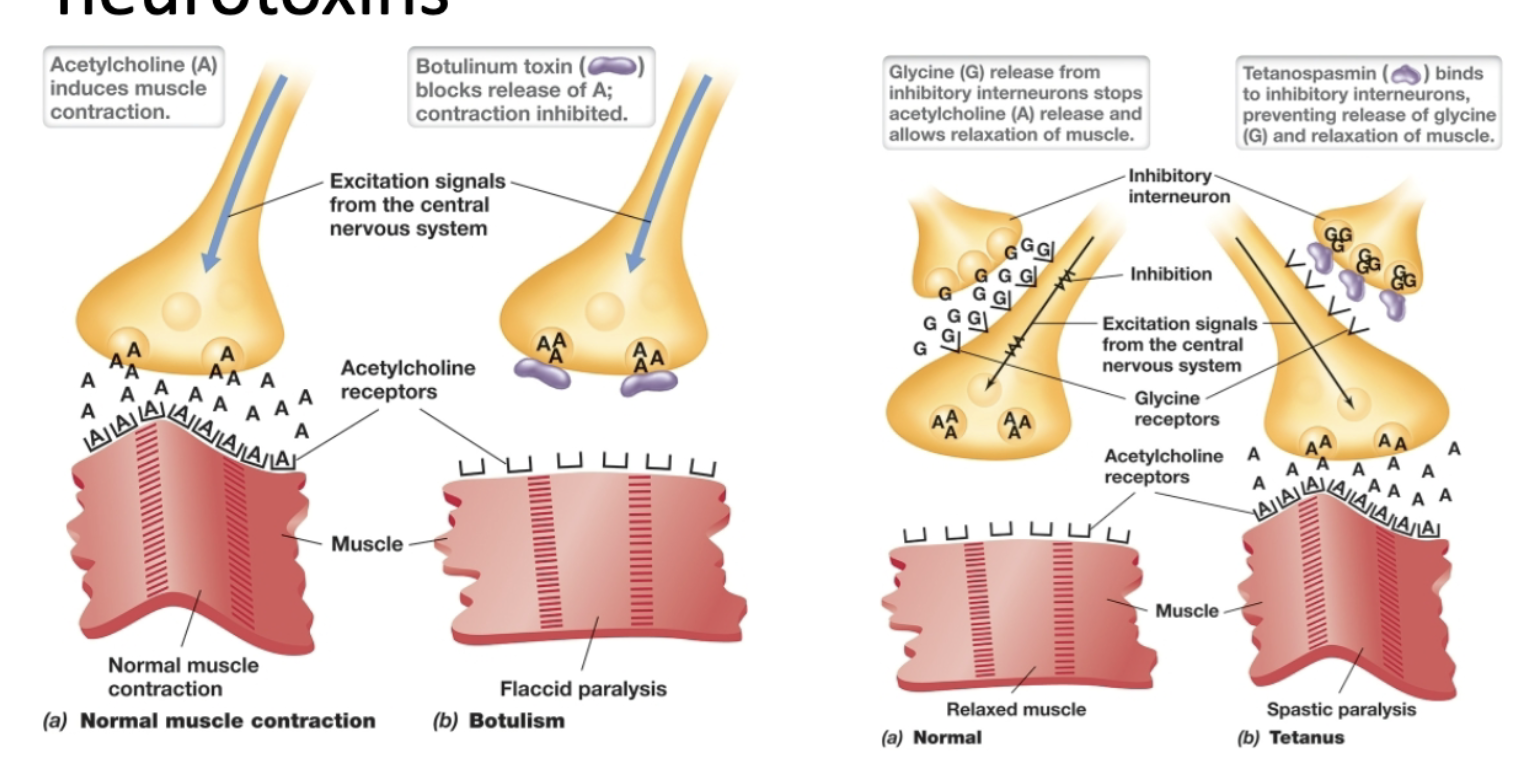
what kind of toxins are tetanus and botulism?
AB neurotoxins
how do tetanus toxin and botulism toxins cause disease?
tetanospasmin toxin binds to inhibitory interneurons that prevents release of glycine, which allows relaxation of muscle. Thus, muscle is prevented from relaxing.
botulinum toxin blocks release of acetylcholine, which is a neurotransmitter that induces muscle contraction. Thus, muscle cannot contract.
why are superantigens deadly?
superantigens are proteins that activate many more T cells than a regular antigen would. they overstimulate the host immune response, which can lead to shock and death.
what are endotoxins?
not secreted, but are part of the outer membrane (lipopolysaccharide portion) of Gram negative cells. becomes a toxin when solubilized.
which are more toxic: endotoxins or exotoxins?
exotoxins
endotoxins vs exotoxins table

why can researchers make a toxoid for exotoxins but not endotoxins?
because of the heat stability of endotoxins
describe 3 different methods used by intracellular bacteria to avoid the vertebrate immune system
avoiding phagolysosomal fusion by secreting proteins
maturing in an acidic environment
escaping and dividing in the cytosol
which would you expect to see in an invasive microbe: streptokinase, coagulase, or hyaluronidase?
hyaluronidase
what is immunity?
the ability of an organism to resist infection
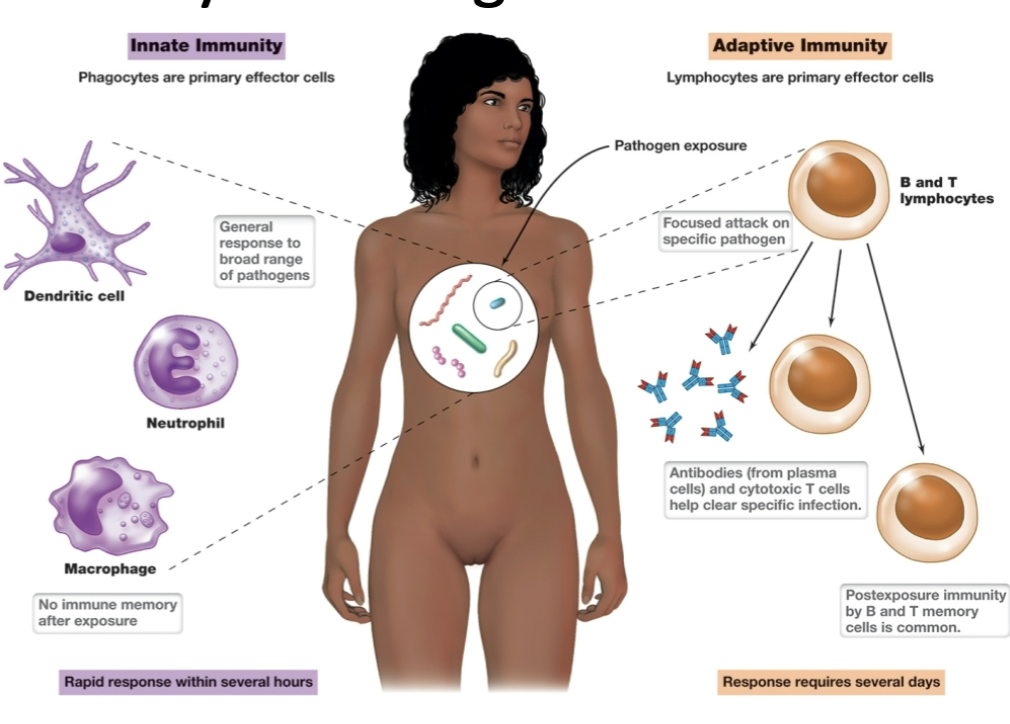
what is innate immunity?
the rapid ability to recognize and destroy pathogens or their products, not requiring previous exposure to pathogen.
what is adaptive immunity?
a slower-acting defense system where lymphocytes are the primary effector cells and develop immune memory for specific pathogen
innate vs adaptive immunity diagram
what is the first line of defense to infection?
physical barriers
what are the human body’s multiple structural barriers to infection?
stomach acid (pH 2 inhibits microbial growth) and lysozyme
intact skin (physical barrier, produces antimicrobial FA and anti-bacterial peptides)
flushing of respiratory and digestive systems
what are 2 similarities between barrier and physiologic defenses in the respiratory system and defenses in the gastrointestinal tract?
mucosal epithelium and tight junctions that form physical barrier from microbes
secretory immunity and antimicrobial peptides (AMPs)
what are 2 differences between barrier and physiologic defenses in the respiratory system and defenses in the gastrointestinal tract?
the respiratory tract relies on ciliated epithelial cells in the trachea to move microorganisms out of body, while GI tract uses peristalsis to propel opportunistic pathogens through system and out of body via defecation
respiratory maintains a stable, neutral pH, whereas GI tract has highly acidic environment of 1.5-3 to denature bacterial proteins
why is the route of infection important?
because different pathogens infect different tissues
what is “route of infection”
the different pathways that different pathogens use to infect different tissues (e.g. tetanus in wounds, Salmonella is ingested)
if the physical barriers are breached, what cells are the “first responders”?
erthythrocytes, lymphocytes, neutrophils, granulocytes, monocytes
tissue damage triggers:
recruitment of a large number of immune cells of the innate response