OCHEM Ch. 3 Conformations and Stereochemistry
1/40
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Newman Projection
used to relate stability of different conformations of molecules
Drawing Newman Projections
look down C-C bond
see how the substituents are arranged
front carbon looks like Y with closed circle
wedge goes to top right
dash goes to top left
back carbon looks like upside down Y with open circle
wedge goes to bottom right
dash goes to bottom left
anti-periplanar (anti)
most stable
dihedral angle = 180°
lowerst energy confirmation and torsional strains
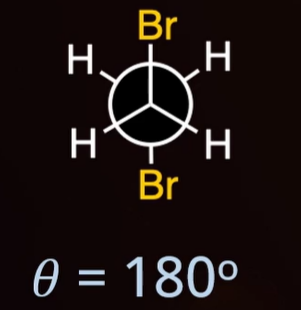
eclipsed
dihedral angle = 120°
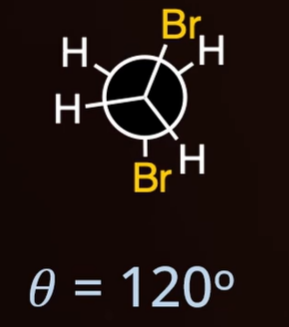
gauche
dihedral angle = 60°
steric strain
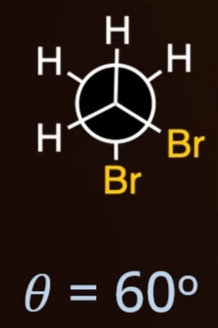
steric strain
repulsion experienced between two bulky groups forced close together in space
anti-periplanar 180°
gauche 60°
fully eclipsed 0°
torsional strain
eclipsed (0°) vs staggered (60°)
fully eclipsed
least stable
dihedral angle = 0°; groups are overlapping
highest energy conformation and torsional strain
staggered conformations are ______ stable than eclipsed
more
ideal C-C-C bond angle to minimize angle strain
109.5°
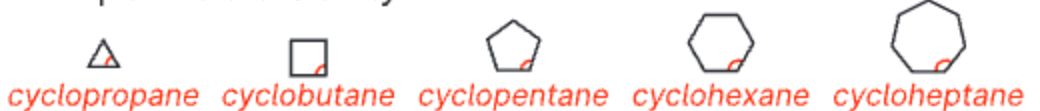
cyclopropane = 60°
cyclobutane = 90°
cyclopentane = 108°
cyclohexane = 120°
cycloheptane = 129°
most stable (lowest energy) conformation of cyclohexane is
chair conformation

cyclohexane chair conformation
low-energy
tetrahedral geometry and C-H bonds are staggered
each carbon has 1 axial and 1 equtorial substituent
axial substituents
bonds that go straight up or down the page
equatorial substituents
bonds that are at an angle upwards or downwards
most stable because they avoid 1,3-diaxial interactions (steric strain)
when a chair is flipped
all axial substituents become equatorial and all equatorial substituents become axial
all of the substituents that were pointed up, remain pointed up, and the substituents that were pointed down, remain pointed down
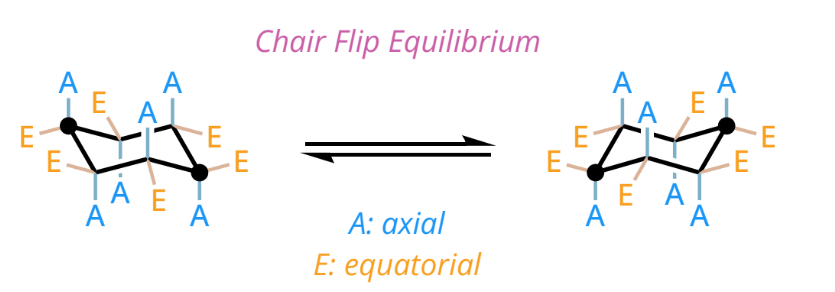
most stable chair conformation
bulky groups in EQUATORIAL positions due to less steric strain
equatorial most stable because they avoid 1,3-diaxial interactions (steric strain)
bulkier substituents in equatorial position are heavily favored
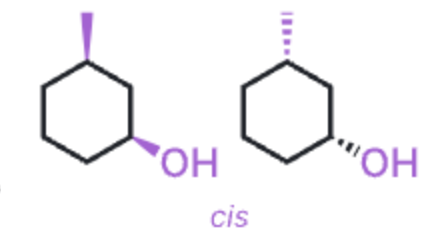
cis disubstituted rings
substituents pointing in the same directions
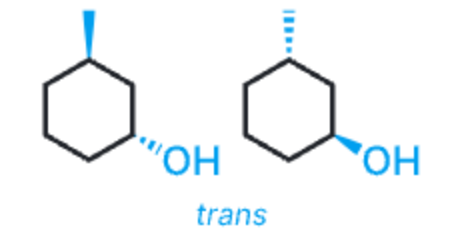
trans disubstituted rings
substituents point in opposite directions
stereocenter
any point within a molecule that can give rise to stereoisomers
chiral center
sp3 hybridized carbon atom with 4 unique substituents
configuration of a chiral center is indicated by R/S designation
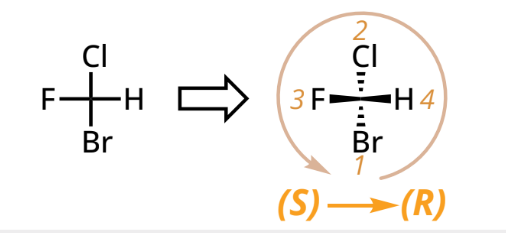
To assign R/S designation:
Locate chiral centers: sp3 hybridized carbon with 4 different groups
Assign priority (Cahn-Ingold-Prelog)
Higher atomic # = higher priority
Compare further atoms if direct ones are the same
Trace circle from 1 to 2 to 3 and Orient lowest priority (4):
On dash: assign R (clockwise) or S (CCW)
On wedge: assign then reverse designation
On line: swap pairs to move 4 to dash and assign as usual
Naming: (R)-, (S)-, or (2R,3S)- as prefix in IUPAC names
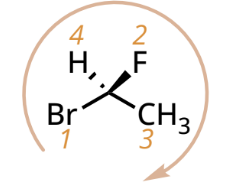
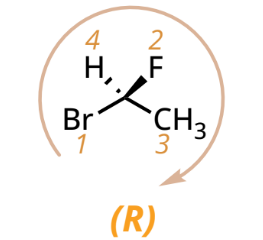
R/S designation with lowest priority group on a dashed bond
R: CW
S: CCW
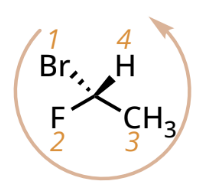
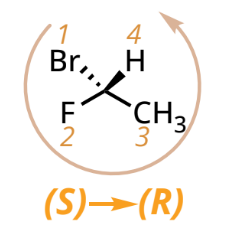
R/S designation with lowest priority group on a wedged bond
reverse the designation
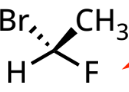
R/S designation with lowest priority group on a line
swap pairs to move 4 to dash and assign as usual
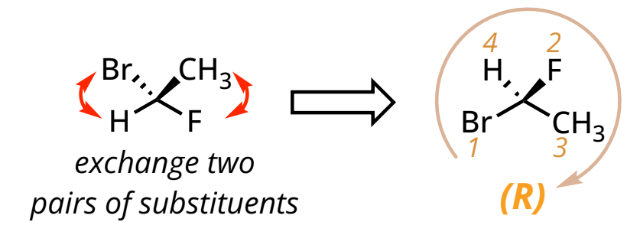
If there is more than one chiral center in the compound,
include locants before each R/S designation.
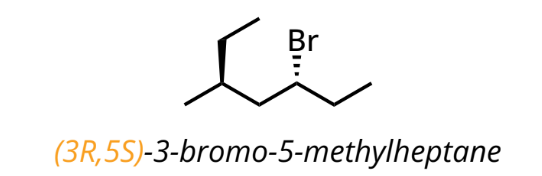
Stereoisomers
molecules with the same molecular formula and connectivity but different spatial arrangement
Enantiomers
stereoisomers that are non-superimposable mirror images of each other and possess the same physical properties
opposite configurations at all chiral centers
differ only in optical activity

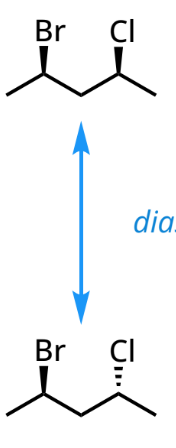
Diastereomers
stereoisomers that are non-superimposable, non-mirror images of each other and possess different physical properties
opposite configurations at some, but not all, stereocenters
need more than one stereocenter
2n rule
max # of stereoisomers
n = # chiral centers
ex: molecule with 3 chiral centers, 23=8 possible stereoisomers
constitutional / structural isomer
share the same molecular formula, but differ in the connectivity of their atoms
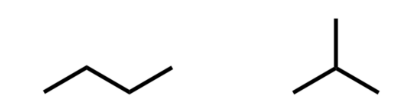
two ways to draw enantiomer
switch all wedges to dashes and all dashes to wedges
draw a reflection over an imaginary line of reflection
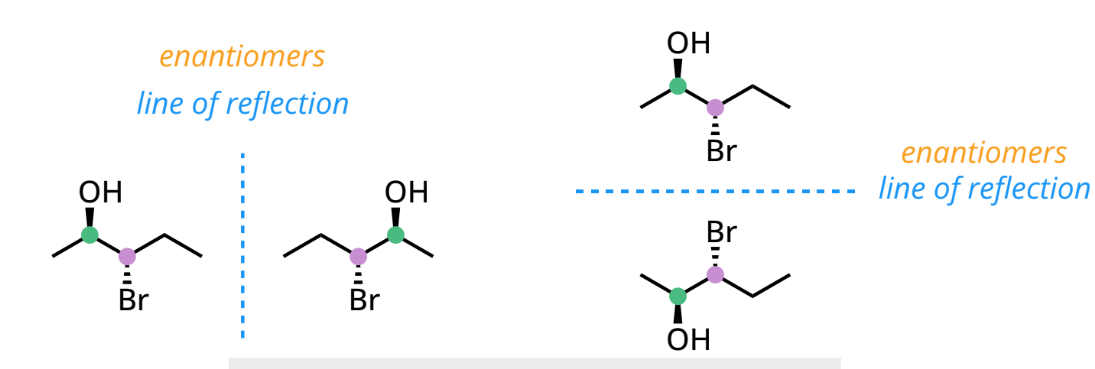
optical activity
chiral molecules rotate plane-polarized light
enantiomers: rotate light equally but in opposite directions
racemic mixture (50:50) = optically inactive
T/F: A molecule with one chiral center is always chiral.
True
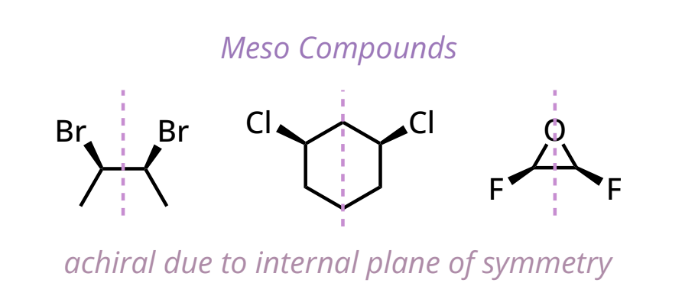
meso compound
have at least 2 chiral centers + internal plane of symmetry
achiral (optically inactive) despite having chiral centers
# of stereoisomers for meso compound: 2n - 1
Fischer Projections
depicts stereochemistry of chiral molecules
horizontal lines = wedges (out of plane)
vertical lines = dashes (into plane)
R/S designation determined by lowest priority group
R/S configuration of a stereocenter in a Fischer projection
assign priorities to each group based on atomic number
if lowest priority on vertical, assign R/S normally
if lowest priority on horizontal, assign → reverse
E configuration of alkene
higher priority groups on both carbons are on OPPOSITE sides of the double bond
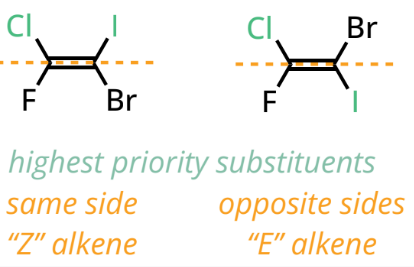
Z configuration of alkene
higher priority groups on both carbons are on the SAME side of the double bond
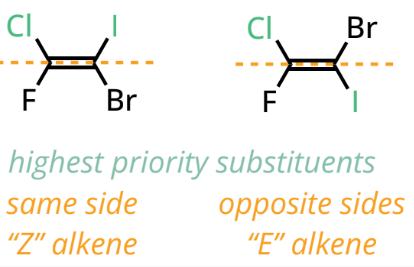
E/Z configuration for tied alkenes
If both atoms attached to one side of the alkene are identical, we assign priority by comparing the elements directly bonded to the atoms that are tied. We keep doing this until the first point of difference.
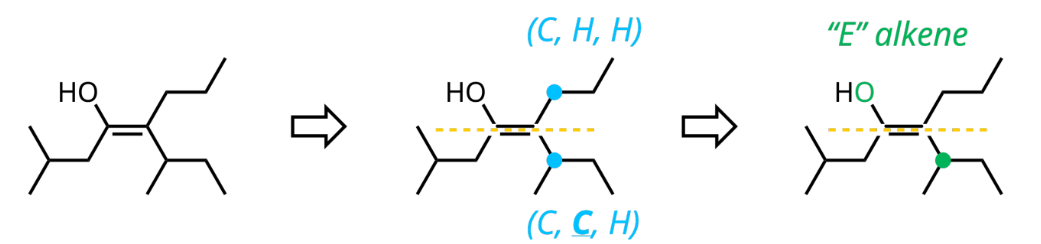
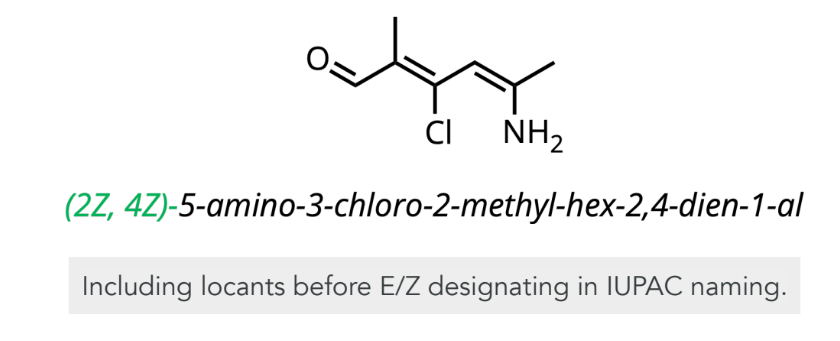
E/Z (Cis/Trans) Alkene Naming
include locants and stereochemistry at the beginning of IUPAC name