Chem Final Exam
5.0(3)
Studied by 80 peopleCard Sorting
1/96
Earn XP
Description and Tags
All exam multiple choice put into one study tool. There may be typos....
Last updated 4:24 PM on 4/25/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
1
New cards
A substance that can be broken down into two or more simpler substances by chemical methods is called a
compound
2
New cards
The nucleus of an atom contains
Protons and neutrons only
3
New cards
Which of the following states of matter has a definite shape?
solid
4
New cards
which two gases make up more than 95 percent of an inhaled breath?
O2 and N2
5
New cards
Which substance is NOT considered to be an air pollutant?
N2
6
New cards
Currently, the primary source of sulfur dioxide emissions into the atmosphere is
coal burning power plants
7
New cards
Which statement is correct?
UV-C is the most energetic of the three forms of UV light
8
New cards
The ozone hole is most prominent on the earth over
Antartica
9
New cards
What is special about the South Pole vs the North Pole that leads to ozone depletion only at the South Pole?
Polar stratospheric clouds form almost exclusively at the South Pole
10
New cards
Light behaves like
both a particle and a wave
11
New cards
Provide the number of protons, neutrons, and electrons for an aluminum atom with a mass of 27
13 protons, 14 neutrons, and 13 electrons
12
New cards
What differentiates a compound from a mixture of two or more elements?
A compound does not exhibit the individual properties of the elements of which it is composed
13
New cards
What are the three pillars of sustainability?
Environmental, social, and economic
14
New cards
What two factors are considered when determining the risk assessment for air pollutants?
Toxicity and exposure
15
New cards
On a periodic table, the columns with elements with similar properties are called
a group
16
New cards
A gasolines octane rating is a measure of the
purity of the gasoline
17
New cards
In the petroleum industry, what does a refinery do?
It separates crude oil into fractions consisting of compounds with similar boiling points
18
New cards
A polar covalent bond is created when
two atoms share their bonding electron unequally
19
New cards
predict the products of this reaction: KOH(Acid) + HNO3(Base)---→
KNO3 + H2O
20
New cards
Argon, which comprises almost 1 percent of the atmosphere, is approximately 27 times more abundant than CO2, but does not contribute to global warming. Which explanation accounts for the fact?
Single atoms do not have bonds, therefore do not vibrate
21
New cards
A chemical reaction accompanied by a release of energy is called a ____ reaction.
Exothermic
22
New cards
Which substance has the highest PH?
A lye solution
23
New cards
How many atoms are present in 0.375 mol of Palladium (Pd) atoms?
2\.26x10^23
24
New cards
Which covalent bond is the most polar?
H--O
25
New cards
The fact that carbon (C) is less electronegative than Nitrogen (N) means that in a C--N bond, the
shared electrons are closer to the N atoms than to the C atom
26
New cards
In an exothermic chemical reaction
Heat is released as the reaction proceeds
27
New cards
Which of the following has the highest boiling point?
Octane
28
New cards
Every increase of one PH unit indicates
10 times less hydrogen ions in the solution
29
New cards
Find the molar mass of ethanol, C2H6O (use periodic table for masses)
46\.07 g/m0l
30
New cards
The carbon cycle refers to the
movement of carbon through living organisms, the atmosphere, the sea, and the earth.
31
New cards
Numbers 1 through 4 are used to identify four different elements. Based on periodic table trends, which number identifies the element that is expected to have the greatest tendency to attract a shared pair of electrons?
3
32
New cards
The energy of motion is called _____ energy.
Kinetic
33
New cards
The pH of rain water falling through an unpolluted atmosphere is closest to
5\.4
34
New cards
1\.0 calorie is defined as exactly 4.184 J. Therefore 10.000 calories are exactly
41\.84 J
35
New cards
Which process plays the most important role in the greenhouse effect?
Energy radiated by the earth is absorbed by the atmosphere
36
New cards
What percentage of earths water is in the oceans?
97 percent
37
New cards
Which reaction most accurately represents the dissociation of nitrous acid (HNO2) in water?
HNO2 + H2O ---> H3O^+ +NO2^-
38
New cards
The process by which a solution is heated to its boiling point and the vapors are condensed an collected is known as
Distillation
39
New cards
the energy stored in the chemical bonds of fossil fuels is a form of ____ energy.
Potential
40
New cards
Which common process on earth is endothermic?
The production of oxygen by photosynthesis
41
New cards
Petroleum (crude oil) is a complex mixture of thousands of substances, the majority of which are
hydrocarbons
42
New cards
What is the mass of exactly 5 moles of Sulfur (atomic mass= 32.07 g/mol)
160\.35g
43
New cards
The first law of thermodynamics states that
energy is neither created or destroyed
44
New cards
bases produce which ions in an aqueous solution?
OH-
45
New cards
Electronegativity
is a measure of an atoms attraction for an electron in a chemical bond
46
New cards
Which atmospheric component does not contribute to global warming?
N2
47
New cards
The property of matter that determines the direction of heat flow is the
temperature
48
New cards
Which is a fossil fuel?
Natural Gas
49
New cards
The greenhouse effect refers to the process by which
atmospheric gases trap and return infrared radiation radiated by earth
50
New cards
Which of the following are forms of kinetic energy?
electrical energy
51
New cards
Which of these statements about biodiesel is NOT true?
biodiesel has been withdrawn from several states due to health concerns
52
New cards
What is the effect of the absorption of infrared energy on matter?
it increases the vibration of chemical bonds
53
New cards
Which shows the Lewis structure of water with the correct partial charge and nonbonding electrons?
A
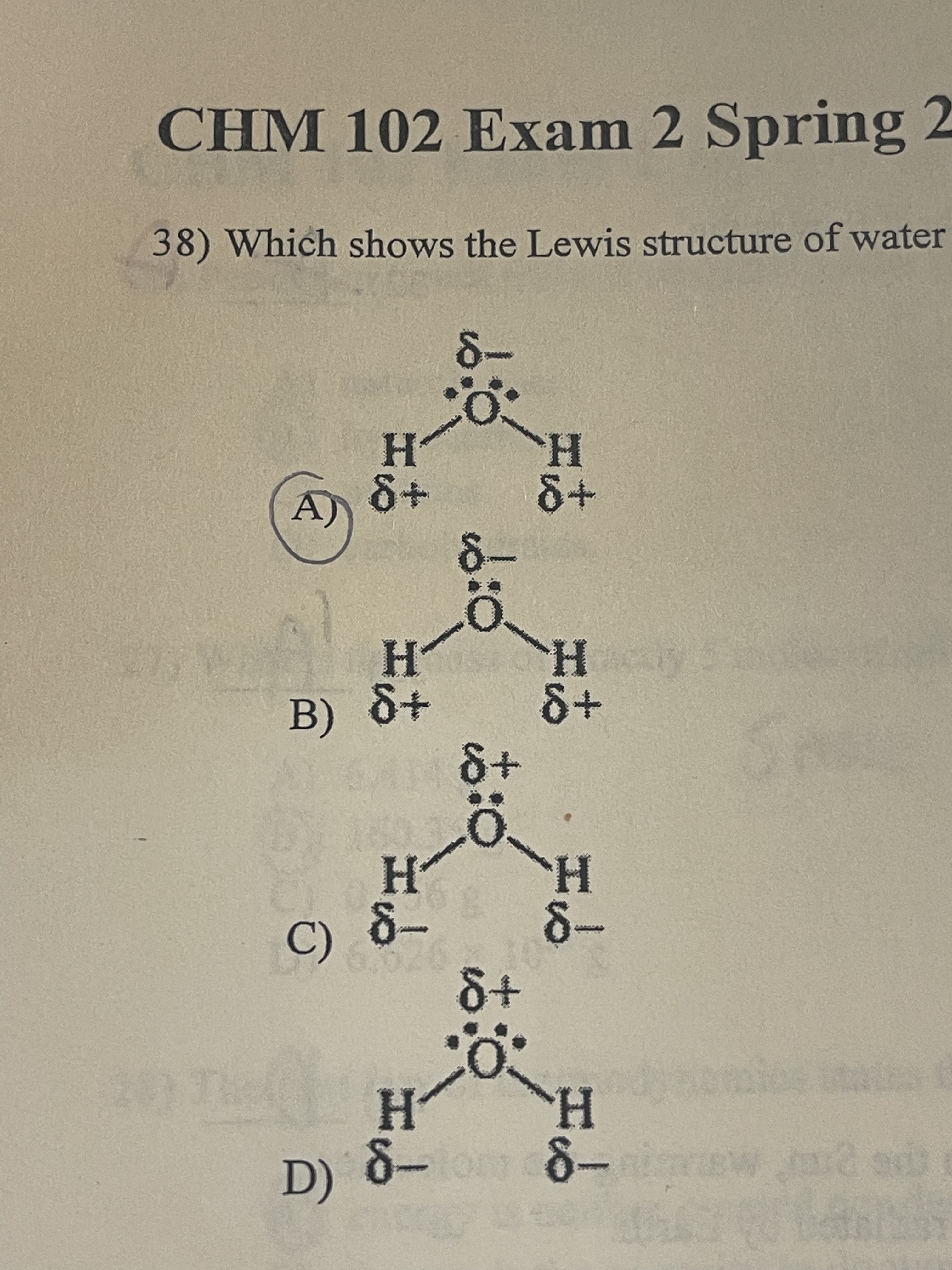
54
New cards
Neither N2 or O2 are greenhouse gases because
their overall electric charge distribution does not change when they vibrate
55
New cards
Why is water a liquid at room temp instead of a gas?
Water has hydrogen bonds between the molecules
56
New cards
The attractions between anions and cations throughout a crystal are known collectively as
ionic bonds
57
New cards
What atmospheric component is responsible for the natural acidity of rain?
Carbon dioxide
58
New cards
Any chemical or physical change that absorbs energy
is an endothermic process
59
New cards
The drawing shows two water molecules. Which statements is correct?
A: Covalent bond; B: hydrogen bond; A is stronger
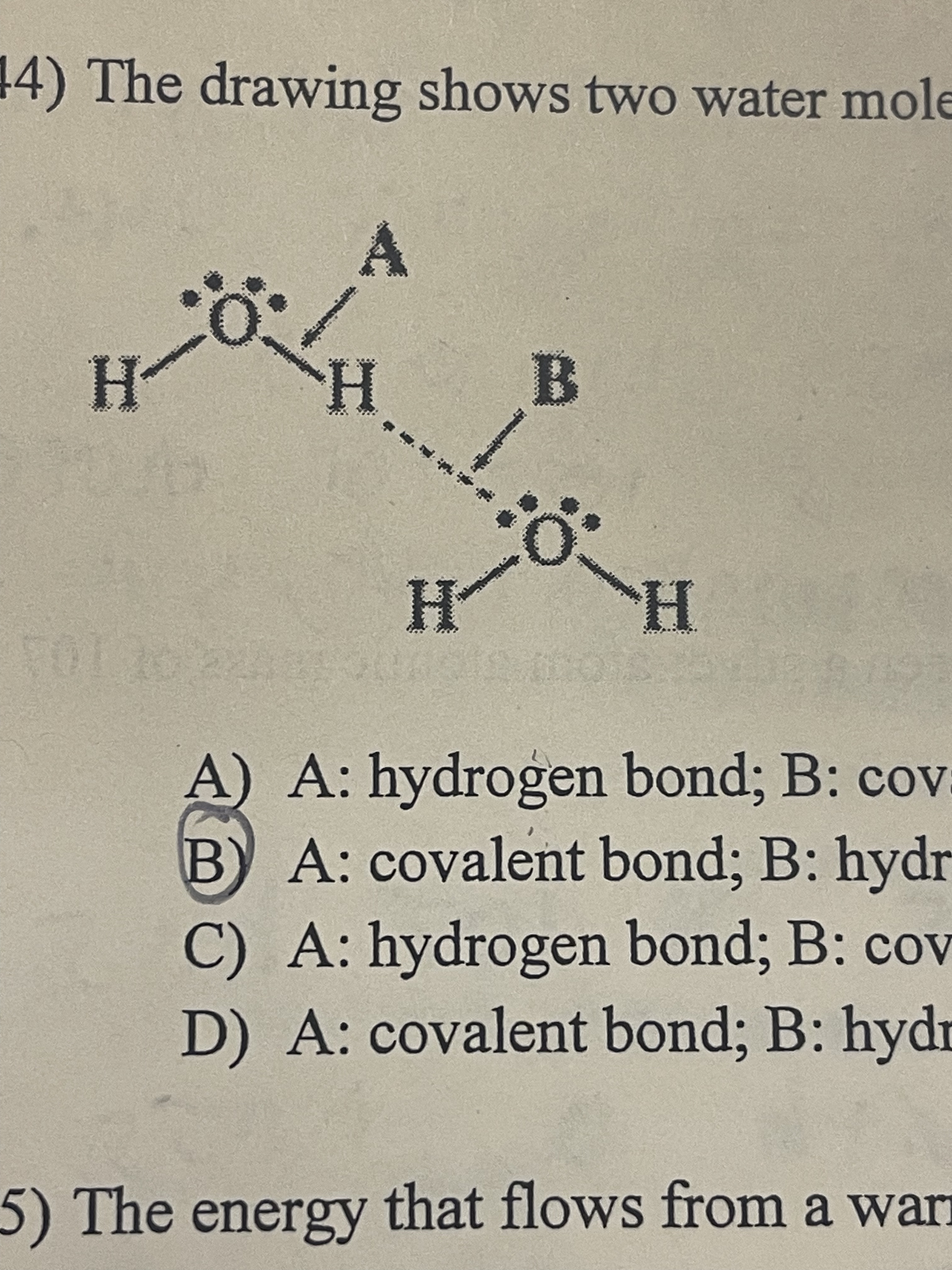
60
New cards
the energy that flows from a warmer body to a colder body is called
heat
61
New cards
From the information given, which statement is false?
Carbon is the most important element
62
New cards
What is meant by malnutrition?
The diet is lacking in proper nutrients, even though caloric intake may be sufficient
63
New cards
Which naturally occurring radioactive particles are negatively charged?
Beta Particles
64
New cards
Which statement describes what happens when oil and steer are mixed together?
Oil is non polar and water is polar and they do not mix
65
New cards
Which is the balanced equation for the fission of U-235?
235^ 92U + 1^ 0n --→ 87^ 35Br + 146^ 57La + 3 1^ 0n
66
New cards
What is an advantage of sous vide cooking?
The food is cooked to an exact temp
67
New cards
Higher altitudes have higher boiling points of water.
False
68
New cards
why is water a good solvent for cooking?
It has the ability to absorb significant amounts of heat before changing phase
69
New cards
Some smoothie bars have the option to add extra vitamin C to smoothies that already contain the daily recommended value. Vitamin C is a polar molecule. Why is it unnecessary to add more than 100 percent of vitamin C recommended values to food?
Vitamin C is water soluble and excess is passed as urine
70
New cards
The line drawings of two fatty acids are shown. Which structural feature do they have in common?
Carboxylic acid group (COOH)
71
New cards
In the famous equation E=MC^2, the symbols represent
E= energy M= mass C= speed of light
72
New cards
The half life of carbon-14 is 5730 years. If you started with 100.0 g of carbon-14, how much would remain after 4 half lives?
6\.25g
73
New cards
Which is NOT a polymer of glucose?
Cholesterol
74
New cards
What type of radiation is given off in this nuclear reaction? 210^ 84 Po --→ 206^ 82Pb + ____
Alpha
75
New cards
Which statement correctly describes starch and cellulose?
Starch and cellulose are both made from glucose but have different linkages
76
New cards
If you were worried about you cholesterol intake, which breakfast would you not eat?
Eggs fried in butter with bacon and white toast
77
New cards
Which naturally occurring radioactive particles are the size of a helium nucleus?
Alpha particles
78
New cards
What is the difference between a fat and an oil?
A fat is a solid, while an oil is a liquid at room temp
79
New cards
What factors determine how many calories per day a person needs?
health, level of activity, gender, age, and body size
80
New cards
Which is a common characteristic of both nuclear and conventional electric power plants?
steam turbines
81
New cards
Which butter has the highest percentage of polyunsaturated fat?
I can’t believe its not butter
82
New cards
Egg whites start out translucent and become white and solid only after heat is added. What type of change occurs when the egg turns white and solidify?
Chemical and physical
83
New cards
The basal metabolism rate (BMR) is
then minimum amount of energy required daily for your body to function
84
New cards
The Maillard reaction occurs at high temp and is a chemical reaction between proteins and sugars on the surface of the food.
True
85
New cards
More sugar can be dissolved in hot water than cold water because the kinetic energy of the water is increased when heat is added. This increased the umber of collisions with the sugar thus dissolving more of it.
True
86
New cards
Which if the following is NOT a benefit of using a vacuum bag when cooking sous vide?
The Maillard reaction can be achieved
87
New cards
What sort of energy is associated with energy from wind?
Kinetic energy
88
New cards
A chef in Denver and a chef in Tampa make the same recipe but he Denver chef has to modify the recipe. Why did the Denver chef have to modify the recipe?
The boiling point of water is lower at higher altitudes
89
New cards
All nine essential amino acids can be found in meat. By employing the nutritional principle of complementarity, a veteran gets all nine. What is complementarity?
combing foods that in their diets have all 9 essential amino acids
90
New cards
The fat soluble vitamins, A,D,E,K, are stored in lipid material. Which statement is false?
These vitamins do not have to be taken daily
91
New cards
Which statement describes how food is cooked using a microwave?
The food absorbs the microwaves and its molecules begin to rotate causing heat
92
New cards
Structurally, how are water soluble and fat soluble vitamins different?
water soluble vitamins contain polar groups, such as OH and fat soluble do not
93
New cards
A 120 pound person uses 3 calories per minute walking at a 20 minute/ mile pace. Approximately, how many miles would a person have to walk to burn off a scoop of ice cream (175 Calories)?
3\.0 Miles
94
New cards
Some short answers that weren’t super long are below
95
New cards
Water is a unique molecule. What are some properties of water that make it unusual?
When made into a solid it expands and floats.
96
New cards
Silver has an atomic number of 47, what is the difference between a silver atom atomic mass of 107 and a silver atom with Ana Tomic mass of 109?
The one with a mass of 107 has 60 neutrons and the one with a mass of 109 has 62 neutrons.
97
New cards
A student has 0.2 moles of NaCL and places it into a 500 mL flask. What its the molarity?
.2/500 mL x 1000mL/ 1L = .4 Molarity