Aldehydes AND KETONES
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Aldehydes Structure and General Formula
R-CHO (Carbonyl group at the chain end)
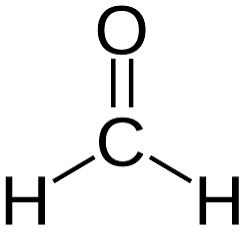
(Common and IUPAC)
Common: Formaldehyde | IUPAC: Methanal (HCHO).
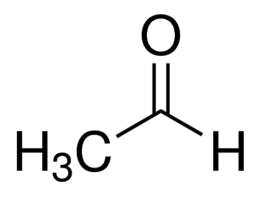
(Common and IUPAC)
Common: Acetaldehyde | IUPAC: Ethanal (CH3CHO).
Method of Preparation of Aldehydes
Oxidation of primary alcohol and hydration of alkynes
How to prohibit primary alcohol not to become carboxylic acid and
To form aldehyde
Must be distilled off immediately to prevent further oxidation into acid.
Oxidation of alcohol to obtain aldehyde
Use primary alcohol+ nasent oxygen with oxidising agent
(K2 CR2 07 ,H+) and get aldehyde+
Oxidation of Aldehydes
Easily oxidized to carboxylic acids using K2Cr2O7 or KMnO4.
Reduction of Aldehydes
Reduced to primary alcohols using LiAlH4 in dry ether. with dil H2SO4
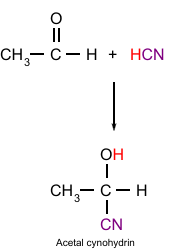
Addition of HCN to Aldehydes
Form hydroxypropanenitrate from ethanal
Form 2-hydroxy-2-methylpropanenitrate
aldehydes and Uses of Formalin
A 40% aqueous solution of methanal used as a disinfectant and preservative.

Chemical test for Aldehydes
Strong reducing agents
Nomenclature of aldehyde
Common name base on carboxylic acid
Indicated by Greek letter
IUPAC Name: name of parent hydrocarbon carbon
the suffix al instead of e
Use of Cinnamic aldehyde
Perfume of natural sweet or fruity scents

Aldehydes are___compound
versatile compound
Hydration of alkane to obtain aldehyde
Use ethyne and water with dil H2 SO4 at 60’C and Hg S O4 to obtain ethanol
Ketone Structure and General Formula
R-CO-R' (Carbonyl group in the middle)
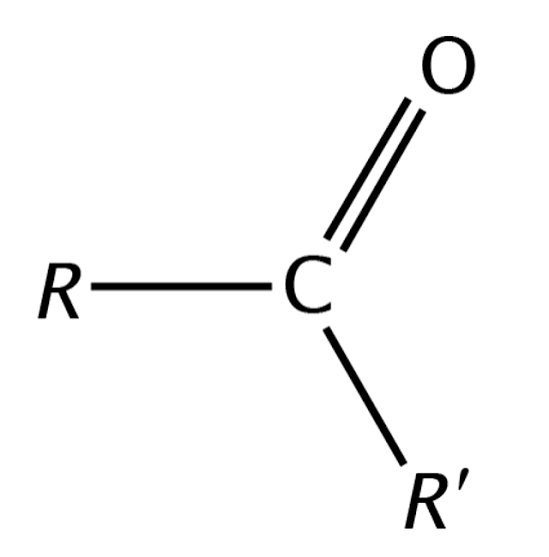
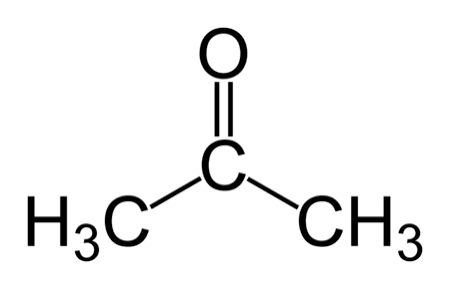
(Common and IUPAC)
Common: Acetone | IUPAC: Propanone (CH3COCH3).
Preparation of Ketones
Oxidation of secondary alcohols by using acidified K2Cr2O7.
Hydration of alkane by using dil H2 SO4 at 60’C and Hg S O4 or H2 S O4.
Ketone Oxidation
Do not oxidize easily but under vigorous condition
Reduction of Ketones
Reduced to secondary alcohols using LiAlH4 in dry ether.
Nomenclature of kentones
Common name: two alkyl +kentones but the first member is acetone
IUPAC name: derived from hydrocarbon like alkane but change to alkanone
Properties of kentones (state)
At room temperature,methanal is gas and ethanal is a volatile liquid.
Solubility of aldehyde and kentones
Methanal , ethanal and propanone are water soluble.
Use of Aldehyde
Versatile compound that can help make resins,dyes, organic compound.
40% of aqueous solution of methanal
Formalin

Formalin is used for
a desinfectant and a preservatives for biological specimen
Where kentones are used as popular solvents.
In making moderately polar substance, including waxes, paints, lacqures, varnishes, glues

The simple kentones (propane) acetone is used for?
an effective solvents in many nail polish removers, plastic cements, resins, and varni