EOC- Chem 1 Review
1/25
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
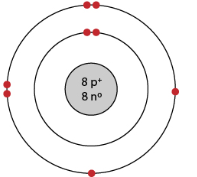
The model shows an oxygen atom, which consists of 8 protons, 8 neutrons, and 8 electrons. Choose THREE statements that describe two electrons being added to the oxygen atom.
Multiple choice
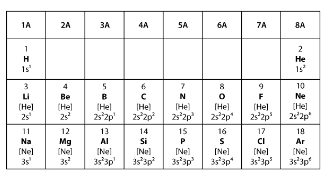
Covalent bonds form when two elements share electrons to make a complete outer shell of electrons. Ionic bonds form when one element donates one or more electrons to another element to make a complete outer shell of electrons. Which statement BEST explains the type of bond that will form between two elements from group 6A in the model?
The two elements will form a covalent bond because both elements will share a pair of electrons in order to have full outer shells.
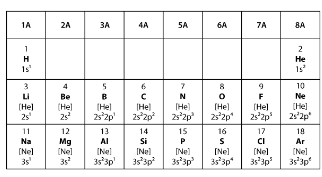
An atom is in its most stable configuration when it has 8 electrons in its outer shell. When forming an ionic bond, one atom gives up electrons to empty its outer shell while the atom it bonds with gains the electrons the first atom gives up. Atoms that are members of group 2A have two electrons in their outer shell, so they can easily give up these two electrons. An atom from group 2A forms an ionic bond with an atom from another group. Which group is the second atom MOST LIKELY from?
group 6A
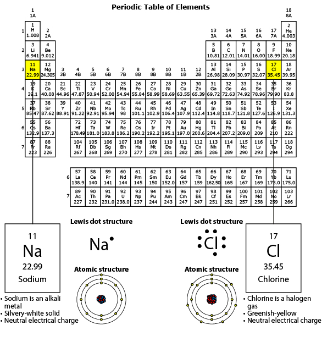
The class plans to perform another investigation using sodium. For this investigation, the teacher will react solid sodium with oxygen gas to produce sodium oxide. Which BEST predicts how sodium oxide will form?
One oxygen atom should bond with two sodium atoms. Each sodium atom will donate one electron to the oxygen atom to fulfill its octet.
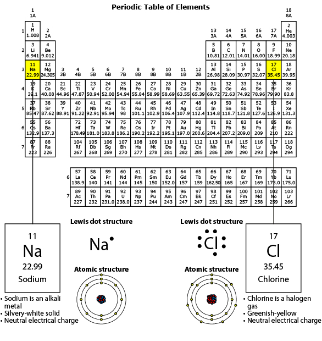
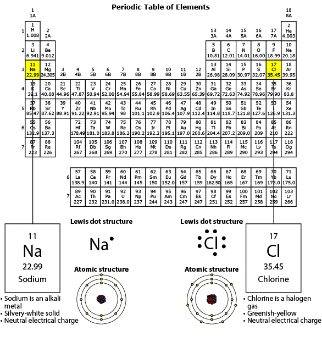
Which BEST describes how the reaction affects the charges of the ions in the product?
Donating an electron causes the sodium atom to have fewer electrons than protons, which causes the ion to have a positive charge. Receiving the electron causes the chlorine atom to have more electrons than protons, which causes the ion to have a negative charge.
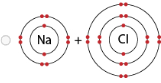
Using the information the students collected, which model MOST LIKELY represents the sodium chloride product?
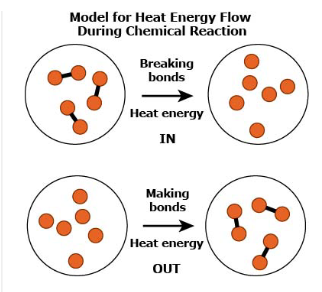
A hand warmer is a small packet that people can put into their gloves or mittens to keep their hands warm during cold weather. Sasha learns that the iron powder in a hand warmer undergoes a chemical reaction with oxygen when exposed to air. Choose TWO statements that explain why Sasha’s hands stay warm when she holds a hand warmer.
multiple choice
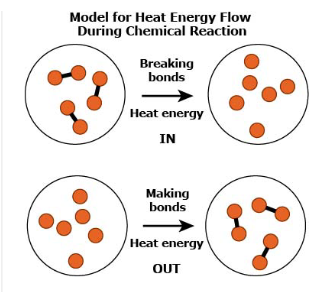
Which claim about why atoms bond to form stable molecules is BEST supported by Sasha’s model?
A stable molecule has less energy than the individual atoms.
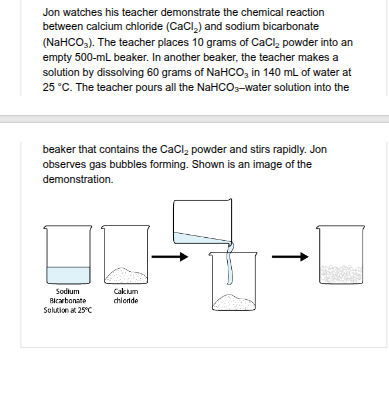
Jon thinks about ways he could increase the rate of the chemical reaction between calcium chloride (CaCl ) and sodium bicarbonate (NaHCO ). Which change would MOST LIKELY increase the rate of the chemical reaction?
use a sodium bicarbonate-water solution that has a temperature of 30 degrees Celsius instead of 25.
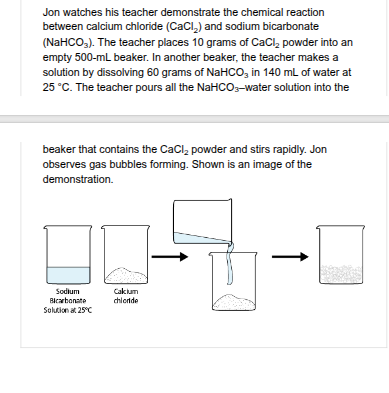
Which statement BEST explains why this change would increase the rate of the chemical reaction?
Molecular collisions would occur more often and with more energy.
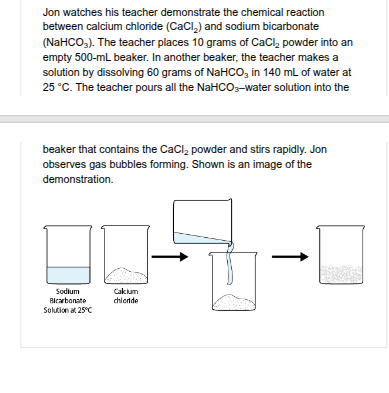
Jon does an experiment in which he varies the amount of calcium chloride (CaCl ) powder used for the chemical reaction with sodium bicarbonate (NaHCO ). He plans to observe whether the amount of CaCl powder has an effect on reaction rate. Make the most logical prediction for the results of his experiment using the drop-down menus.
1- increase 2-collide more often with other reactant molecules.

Thomas studies each chemical reaction and sorts the reactions into two categories according to Le Chatelier’s principle. The two categories are: reactions that maximize the amount of product formed at low pressure and reactions that maximize the amount of product formed at high pressure. Drag and drop the chemical reactions into the appropriate box.
heat + CaCO2(S) = CaO(S) + CO2(g)
N2(g)+3H2(g)=2NH3(g) + heat

Thomas studies each chemical reaction and sorts the reactions into two categories according to Le Chatelier’s principle. The two categories are reactions that maximize the amount of product formed at low temperature and reactions that maximize the amount of product formed at high temperature. Drag and drop each chemical reaction into the appropriate box.
N2(g)+3H2(g)=2NH3(g)+heat ; 2SO2(g)+O2(g)=2SO3(g)+heat ; CO(g)+2H2(g)=CH3OH(g)+heat
heat + CaCO3(s)=CaC(s)+CO2(g)
Aluminum metal (Al) reacts with hydrogen chloride (HCl) to produce aluminum chloride (AlCl) and hydrogen gas (H). Drag and drop the numbers into the boxes that balance the chemical equation for this reaction.
Al + 2HCl = 3AlCl3 + 4H2
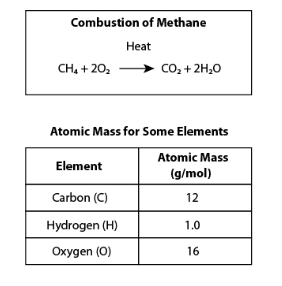

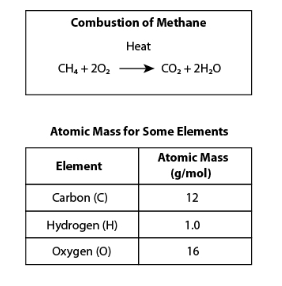
Determine whether each statement is supported by the balanced chemical equation for methane combustion. Choose Yes or No from the drop-down menu for each statement.
No
Yes
No
Yes
Yes
Yes
No
What are the coefficients needed to balance this chemical reaction to show that matter is conserved? Fe +Cl2 = FeCl3
2, 3, 2

Using Figure 1, classify each of the following items as to whether it is involved in alpha decay, beta decay, or gamma decay. Drag and drop each response under the appropriate category. Responses may be used more than once.

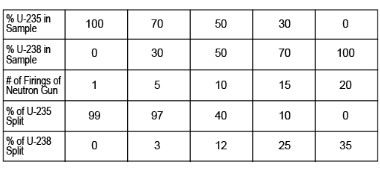
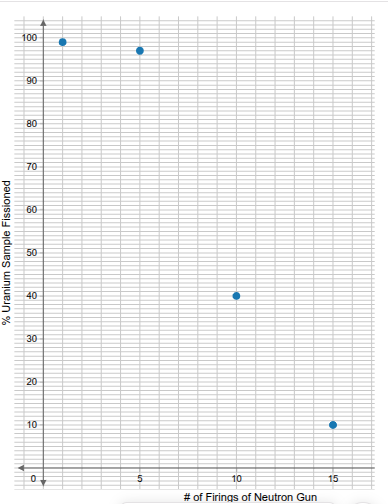
Examine the data in Figure 4 for the results of the tests conducted on uranium-235 and uranium-238. Select the isotope with the best overall efficiency; then use its data to create a scatter plot that illustrates the amount of that isotope successfully fissioned versus the number of firings of the neutron gun. In the graph, "Number of Firings of Neutron Gun" will be on the horizontal axis and "Percent of Uranium Sample Fissioned" will be on the vertical axis.

Use the drop-down menus to explain why copper is a good conductor of electricity and heat.
metallic
move freely
a good conductor of electricity
do not form
vibrate easily
Tia learns that the temperature of rock in Earth’s mantle varies depending on its location. Rock located close to the lithosphere is about 1,000 °C and rock located close to the outer core is about 3,700 °C. Tia uses the formula shown below to calculate the amount of heat that needs to be added to a 5-g piece of rock in order to raise its temperature from 1,000 °C to 3,700 °C . She uses 0.79 J/g °C for the specific heat of the rock.
10,665 joules

absorb more thermal energy
trap the radiant heat inside the pan
percent the transfer of heat out of the collector
Mitchell records the temperature of the system by using an infrared thermometer. Which statement BEST describes when Mitchell should record the final temperature data of the system?
when the temperature of the left beaker is the same as the temperature of the right beaker.

While planning the experiment, Mitchell researches the specific heat capacity of liquid water and finds it to be 4.18 J/g°C. Use the drop-down menus to describe how this is related to the heat and temperature changes in the experiment.
equal to
the same as
the same as
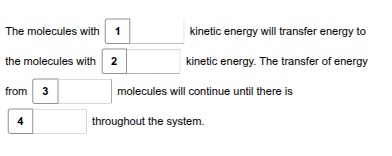
According to the second law of thermodynamics, use the drop-down menus to predict what will MOST LIKELY happen to the net flow of energy in Mitchell's investigation.
higher
lower
hot to cold
uniform energy
Which property of the liquids is being investigated by Julia and Phillip?
surface tension
Liquid | Boiling point (Celsius) | |
rubbing alcohol | 82.5 | |
water | 100 |
Julia and Phillip observe that the difference between the average numbers of drops of rubbing alcohol and drops of water is small. They perform another investigation to provide additional evidence. They observe the time it takes for the same amount of each of the two liquids to boil. Their data is shown. Which liquid's molecules have the strongest intermolecular force?
Water has the strongest intermolecular force because it has a higher boiling point than rubbing alcohol.