Biochemistry UIOWA Exam 3
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
How is carbon coming from glycolysis introduced into the citric acid cycle?
Pyruvate, the product of Glycolysis, is converted to Acetyl CoA by Pyruvate Dehydrogenase, which is the fuel for the CAC. irreversible
How does the compartmentalization of glycolysis and the citric acid cycle affect the pathways
Pyruvate has to be moved across the mitochondrial membrane, in the mitochondrial matrix it is converted to Acetyl CoA
What is the role of pyruvate dehydrogenase?
It decarboxylates pyruvate, creating CO2, NADH, and acetyl-CoA, It also does oxidative decarboxylation and NADH, ATP, and FADH2 are generated
What are the cofactors involved with pyruvate dehydrogenase?
Three catalytic coenzymes: TPP, Lipoic Acid, and FAD
Two sociometric coenzymes: CoA, NAD+
What are the three components of the pyruvate dehydrogenase complex?
E1: pyruvate dehydrogenase
E2: Dihydrolipoyl transacetylase
E3: Dihydrolipoyl dehydrogenase
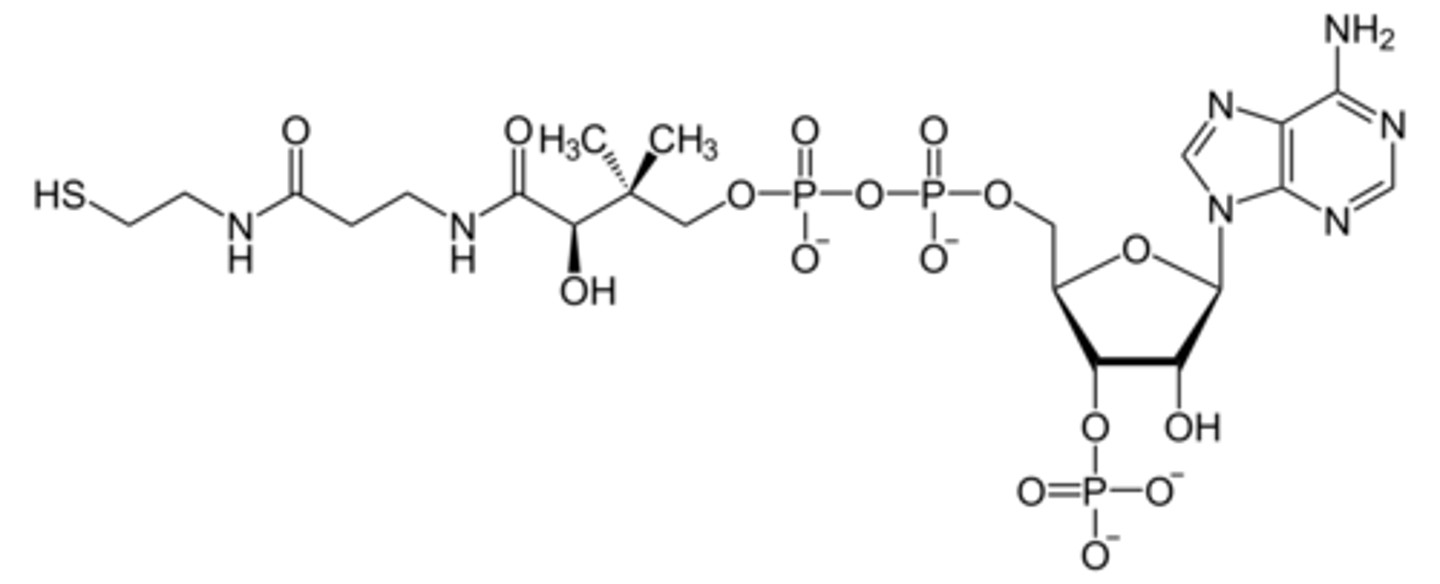
Acetyl CoA
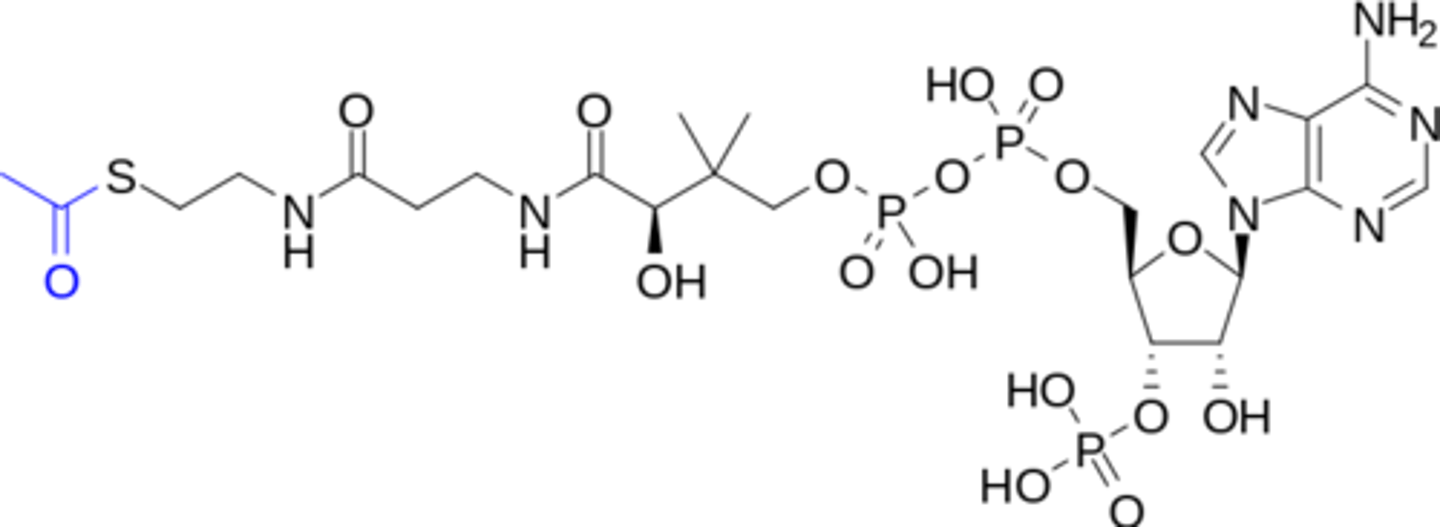
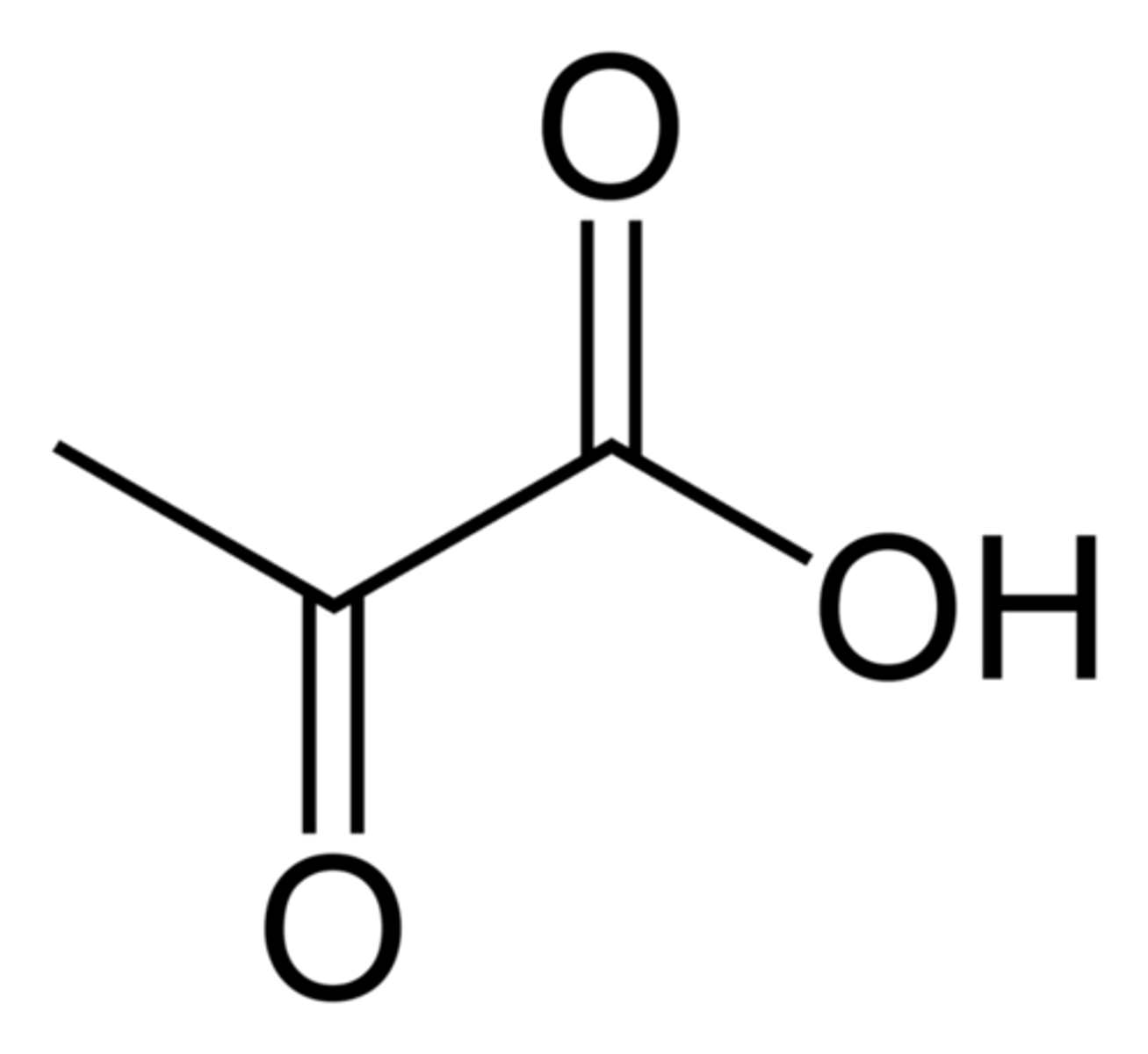
Pyruvate
Co-Enzyme A (CoA)
Effects of kinase on pyruvate dehydrogenase
Phosphorylates and inactivates E1
Effects of phosphatase on pyruvate dehydrogenase
Removes phosphate and activates complex
Effect of ATP, Acetyl CoA and NADH on pyruvate dehydrogenase
Inhibits
Effect of ADP and pyruvate on pyruvate dehydrogenase
stimulates
Citric Acid Cycle
oxidizes the acetyl fragment of acetyl-CoA to CO2, produces three NADH, one FADH2 and one ATP
What is the entry point reaction for Acetyl-CoA into the citric acid cycle
The condensation of the acetyl group with oxaloacetate (4 carbon compound) resulting in citrate (6 carbons)
Isocitrate synthase
Stage 1 of the citric acid cycle
formation of citrate and the two time oxidative decarboxylation of this 6 carbon compound to a four carbon compound Succinyl CoA produces 2 CO2 and 2 NADH
Stage 2 of the citric acid cycle
Regeneration of oxaloacetate; requires ADP and produces ATP, CoA, FADH2 and NADH
Regulation of isocitrate synthase
inhibited by NADH, succinyl-CoA, citrate, ATP (products)
regulation of isocitrate dehydrogenase
Activated by ADP
Inhibited by NADH and ATP
Regulation of a-ketoglutarate dehydrogenase
rate limiting step: inhibited by succinyl CoA, NADH, ATP
How does the Glyoxylate Cycle influence Glucose metabolism?
Enables plants and bacteria to convert fats into carbohydrates
loses reducing power
Where does the citric acid cycle occur?
The mitochondrial matrix
How is the compartmentalization of different steps in oxidative phosphorylation achieved?
Mitochondria contains an outer membrane, inner membrane and matrix. Not even protons can pass through the inner membrane. The electron transport chain gives energy to pump protons across the inner membrane from the matrix to the inner membrane
Oxidative phosphorylation in the electron transport chain
The flow of electrons from NADH and FADH2 to O2
Redox potential
The tendency of a member of a redox pair to donate or accept electrons. It is increased in the electron transport chain by the flow of electrons to oxygen
How does the transfer of electrons along the electron transfer chain generate ATP?
It forms a proton gradient which powers the creation of ATP by ATP synthase. The purpose of the electron transport chain is to generate a proton gradient.
What determines the sequence of complexes used for electron transfer?
The redox potential, members of the electron transport chain are arranged so that electrons always flow ro components with more-positive electron reduction potentials (higher e- affinity)
Coenzyme Q (ubiquinone)
Carrier it transfers electrons from both complex 1 and 2 to complex 3
NADH oxidoreductase
Complex 1: Entrance of NADH, flow of 2 electrons pumps 4 protons from matrix to intermembrane space
Coenzyme Q-Cytochrome C oxidoreductase
Complex 3: catalyzes the transfer of electrons from QH2 to cytochrome c, 2 protons pumped into the intermembrane space.
cytochrome C reductase
Complex 4: catalyzes the transfer of electrons from reduced cytochrome c to oxygen. 8 protons removed, 2 H2O and 4 protons pumped into intermembrane space.
Role of O2 in the electron transport chain
terminal electron acceptor
How is toxicity from oxygen radicals mitigated?
superoxide dismutase and catalase help protect against ROS
Complex 2
non-transmembrane
How is a proton gradient converted to ATP?
ATP synthase, protons flow through a proton chanel powering ATP synthase. Formed from ADP and orthophosphate
sequence of events that happen during ATP synthesis
3 active sites, C ring rotates gamma subunit which interconverts B-subunits
ATP synthase B subunit loose conformation
binds ADP and Pi
ATP synthase B subunit T conformation
binds ATP, converts ADP and Pi
ATP synthase B subunit O (open) form
Releases ATP
Where is ATP generate?
ATP synthase within the inner mitochondrial membrane
How is ATP redistributed?
ATP-ADP translocase, coupled transport of ATP out and ADP in (antiporter)
How many ATP formed from the complete combustion of glucose?
30, 26 via oxidative phosphorylation
Glycerol 3-phosphate shuttle
Allows electrons from the cytoplasm to enter the electron transport chain
malate-aspartate shuttle
NADH from cytoplasm to the mitochondria in the heart and liver.
mitochondrial uncoupling
any process by which the electron transport chain is not used to drive ATP synthesis or other useful work such as net ion translocation, regulated uncoupling: brown fat
Regulation of the rate of oxidative phosphorylation
Rising levels of ADP speed up oxidative phosphorylation
What poisons affect oxidative phosphorylation?
Inhibitors and uncouplers,
What are the products of the light reactions
Converts light to chemical energy
General product of photosystem 1
reducing power NADPH, O2
General product of photosystem 2
Proton gradient
Calvin cycle
reduce CO2 to produce glucose
Where does photosynthesis occur?
Chloroplast
What are the three types of membrane in chloroplasts
Outer, inner, thylakoid
What are the three spaces within chloroplasts
Intermembrane space, stroma, lumen: interior of thylakoid
Thylakoid membrane
Redox reactions,
Which processes transfer energy from excited electrons?
Light creates a separation of charge which induces electron flow. Redox
How to antenna chlorophylls and other pigments contribute to light harvesting?
Use resonance energy transfer to transfer energy to the reaction center
Where does photosystem 1 transfer electrons
From H2O to NADPH
Where does photosystem 2 transfer electrons
Provides electrons to photosystem 1
How is the proton gradient produced by photosystem 2?
The cytochrome complex transfers electrons from plastoquinone (Q) to plastocyanin, pumps protons
Manganese center in photosystem 2
water oxidizing complex
Role of ferredoxin in photosynthesis
transfers electrons to NADP+ then NADPH
Cytochrome b6f complex
bridge for PS1 and PS2, QH2 enters is and loses its electrons to reduce plastocyanin. protons are pumped across the membrane to produce proton gradient.
Plastoquinone (Q)
Similar to Coenzyme Q (ubiquinone). carrier of electrons to cytochrome complex
Plastocyanin
receives electrons in the cytochrome complex, provides them to PS1
Regulation of the light reactions
cyclic photophosphorylation: electrons redirected from photosystem 1 to cytochrome b6f complex. no production of O2 and NADPH.
Calvin Cycle
Fixes carbon dioxide into sugar, using energy from the light reactions, draws CO2 from the air
Fixation stage of the calvin cycle
Enzyme called Rubisco catalyzes reaction between ribulose 1,5-bisphosphate and CO2, producing two 3C molecules
Reduction stage of the calvin cycle
two 3-phosphoglycerides are reduced to make fructose 6 phosphate, requires 2 ATP and 2 NADPH
Regeneration stage of calvin cycle
regenerates ribulose 1,5-bisphosphate, requires ATP
Rubisco (enzyme)
Rate limiting step of calvin cycle, bound CO2 forms a carbamate with lysine 201
What are the starting products of the regeneration step in the calvin cycle
Hexose monophosphate pool, G1P, G6P, F6P
Stoichiometry of Calvin Cycle
6 cycles are required to make one 6 C glucose, 12 ATP total (2 percycle)
Starch
glucose polymer with a-1,4 and a-1,6 linkages
Sucrose
disaccharide of glucose and fructose
Regulation of the calvin cycle by light reactions
Rubisco is activated by light, conditions change to support the calvin cycle when given light
Regulation of the calvin cycle by thioredoxin
High levels of thioredoxin activate the enzymes in the calvin cycle
Photorespiration
Rubisco reacting with O2 with no fixation, requires 1 more ATP and releases ATP,
What is the structure of glycogen?
Polymer of glucose with a-1,4 linkages to form linear arrays and a-1,6 linkages to form branches (1 in 12)
Glycogen Degradation: glycogen phosphorylase
Activity 1: cleaves the glucose ends of glycogen chains (regulated and committed step)
Glycogen Degradation: glycogen phosphorylase b
less active non phosphorylated , T state.
Glycogen Degradation: glycogen phosphorylase a
more active, phosphorylated, R state.
What is the regulated and committed step of glycogen degradation?
The cleavage of the glucose ends of glycogen chains by glycogen phosphorylase.
Glycogen Degradation: Transferase
Activity 2: moves 3 glucose residues to another chain
Glycogen Degradation: a-1,6-glucosidase (debranching enzyme)
Activity 3: hydrolyses a-1,6 linkages
Glycogen Degradation: Hexokinase
Activity 4: Phosphorylates glucose released by debranching enzyme
Glycogen Degradation: Phosphoglucomutase
Activity 5: converts G1P to G6P
Glycogen Degradation: Glucose 6-phosphatase
Activity 6: dephosphorylates G6P in liver to allow transport into blood
glycogen phosphorylase regulation in muscles
Regulated by energy status
glycogen phosphorylase regulation in the liver
default is active unless free glucose is present
What is the role of UDP in glycogen synthesis?
It is the substrate for polymerization via a-1,4 linkages catalyzed by glycogen synthase
Glycogen synthesis: Glycogen synthase
catalyses the synthesis of glycogen, only forms a-1,4 linkages, regulatory enzyme
Glycogen synthesis: Branching enzyme
Breaks a-1,4 linkages and forms a-1,6 bonds
UDP-glucose pyrophosphorylase
catalyses the synthesis of UDP-glucose
What is the energy cost of glycogen synthesis
2 ATP
effects of epinephrine of glycogen synthesis and degradation
epinephrine activates degradation and deactivates synthesis, need more energy!
Protein phosphatase 1 (PP1)
Accelerates glycogen synthesis and decelerates glycogen breakdown, dephosphorylate glycogen synthase stimulating enzyme
Insulin
When blood glucose is high, insulin stimulates the synthesis of glycogen
Diabetes Type 1
caused by autoimmune destruction of beta cells in pancreas that make insulin
Diabetes Type 2
Body stops responding to insulin
McArdle's disease
cannot proper mobilize stored glycogen, extended exercise, myophosphorylase