Energy & Enzymes
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Metabolism
the sum total of all chemical reactions occurring in a biological system at a given time
Free energy
the usable energy for work
Anabolic reactions
complex molecules are made from simple molecules; energy is required (ex: condensation)
Catabolic reactions
- complex molecules are broken down to simpler ones; energy is released (ex: hydrolysis, breaking down food)
- the energy released can be stored in ATP
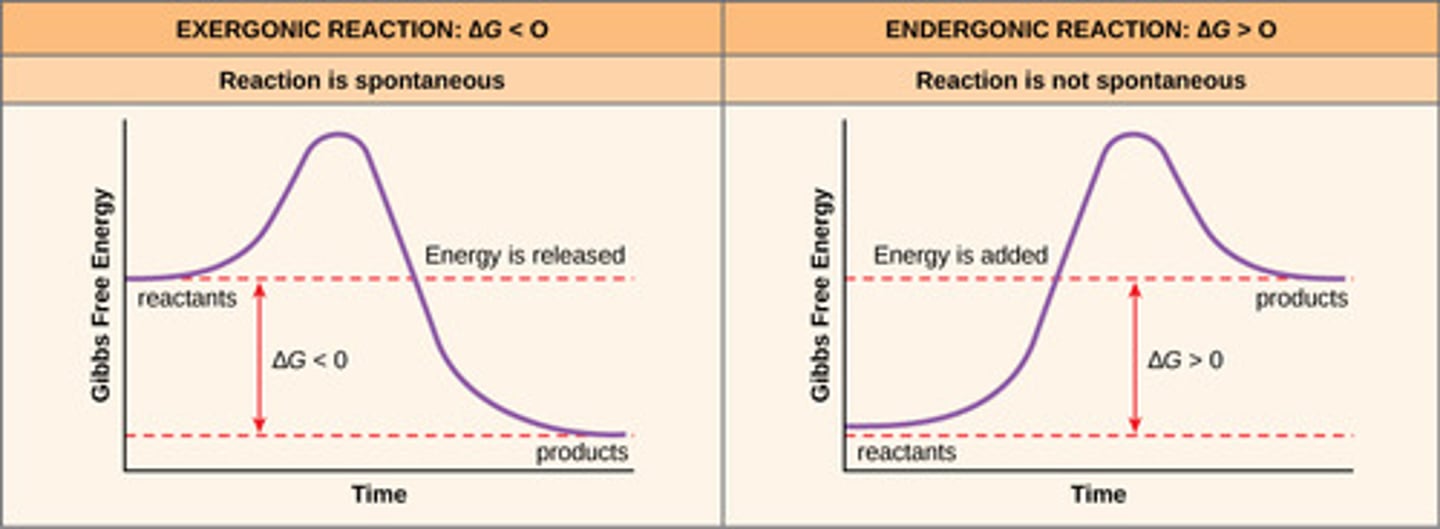
Diagram/graph both exergonic and endergonic reactions and outline their properties.
Endergonic reactions: consume free energy
- pos. delta G
- anabolism (condensation)
- must be coupled to an exergonic reaction to overcome the energy barrier
- less favorable, nonspontaneous
Exergonic reactions: release free energy
- neg. delta G
- catabolism (hydrolysis)
- more favorable, spontaneous
How do enzymes reduce activation energy and aid in or carry out reactions?
Enzymes lower the activation energy, increasing the rate of chemical reactions. They stabilize the structure of the transition state.
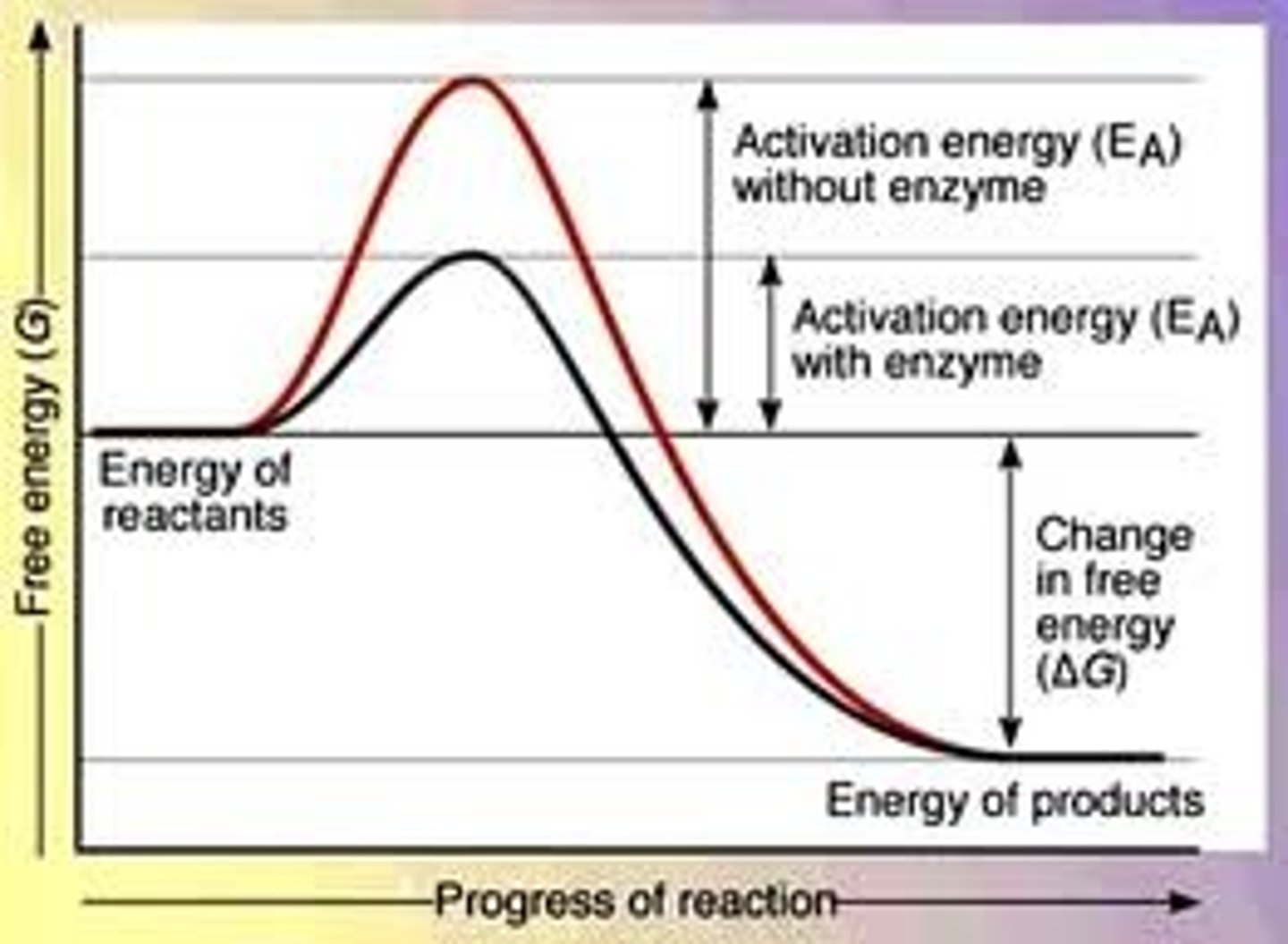
Activation energy
the amount of energy required to start the reaction
How does the structure of ATP "store" energy?
ATP has 3 phosphate groups. It stores energy within the bonds between phosphate groups, especially in the pyrophosphate bond between the 2nd and 3rd groups.
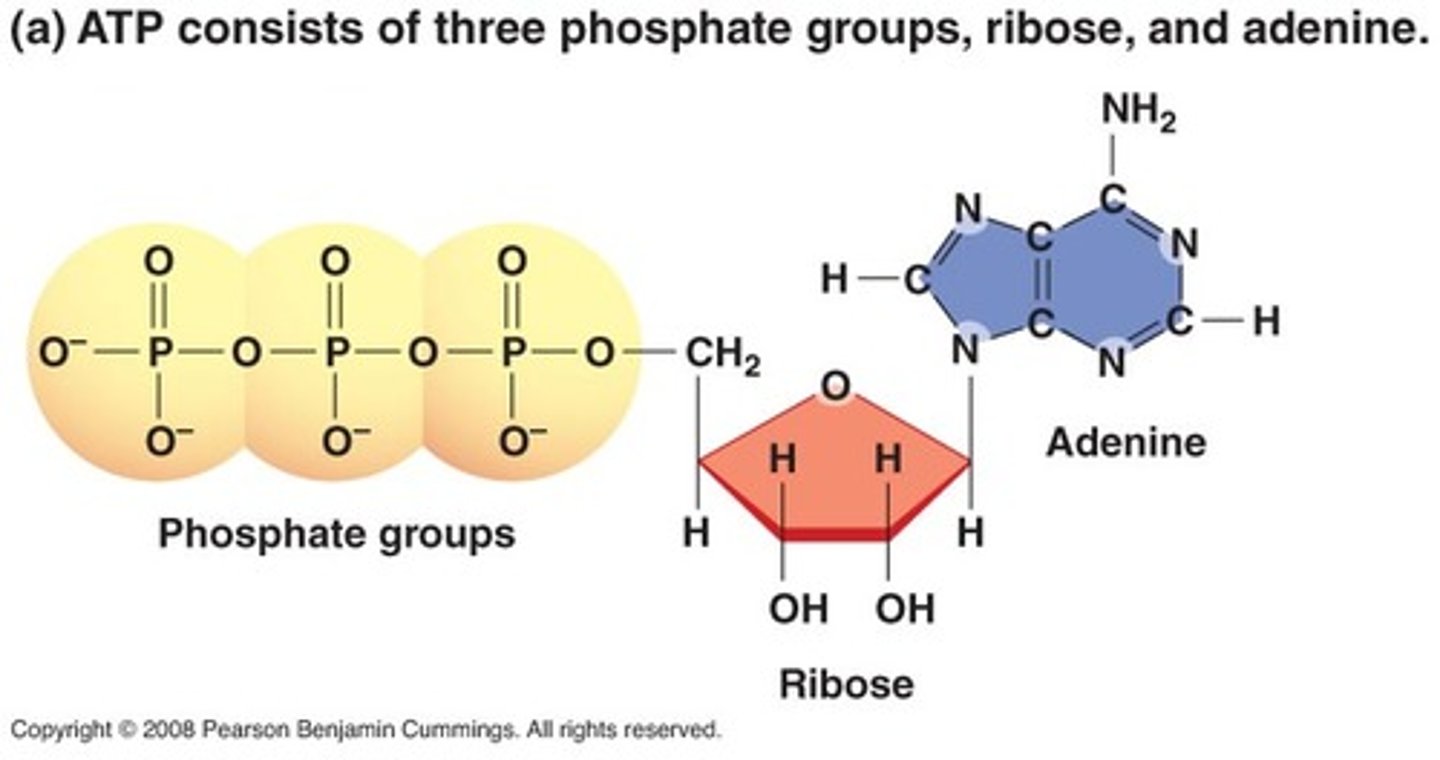
How is energy stored in ATP?
ATP (adenosine triphosphate) captures and transfers free energy. It is an activated carrier, a molecule that is able to degrade then release free energy upon their degradation.
Condensation reaction
- energetically unfavorable
- produces water
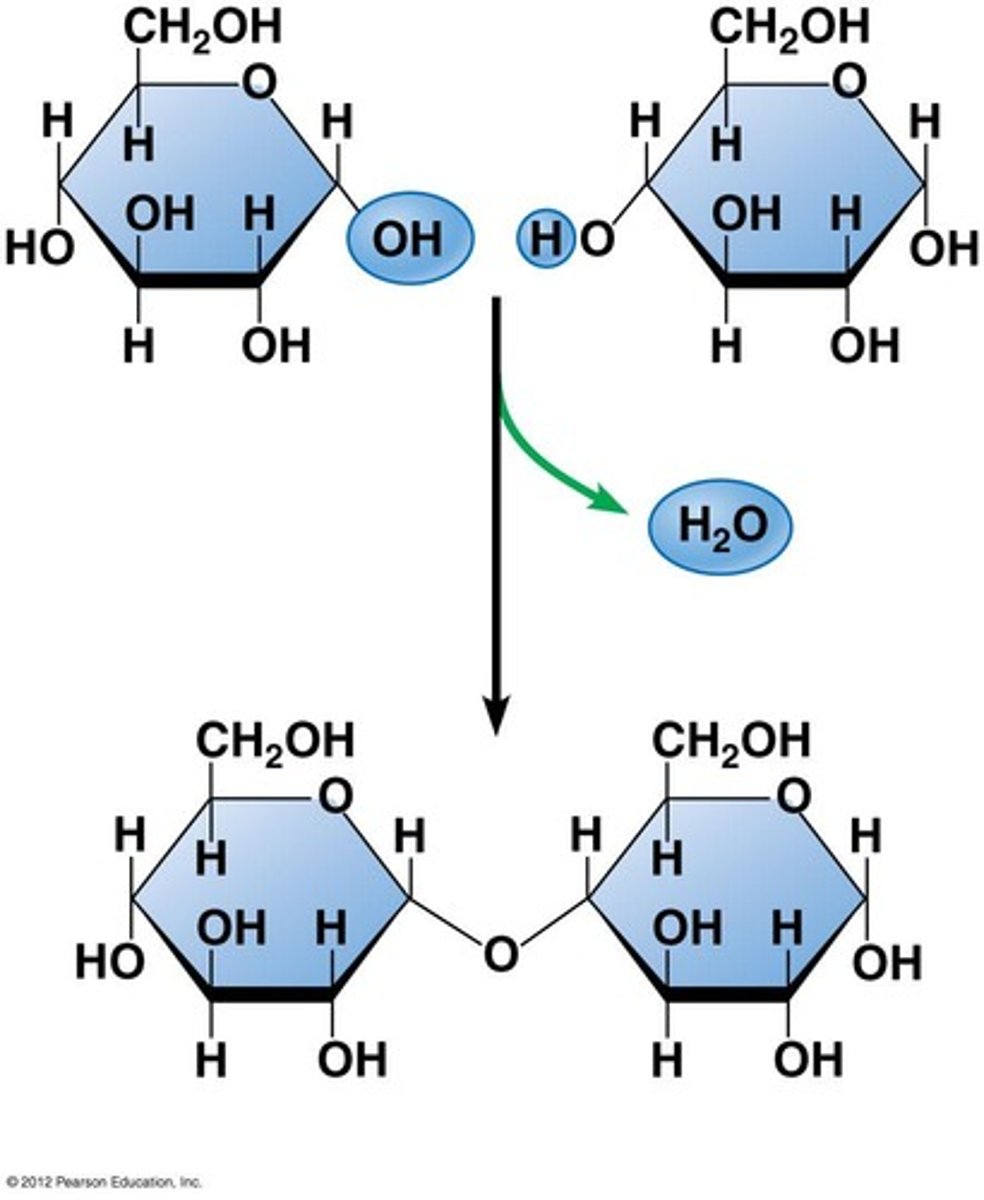
Hydrolysis reaction
- energetically favorable
- requires water
Metabolic reactions involve:
energy changes
1. energy to build things - anabolic reaction (i.e. macromolecules)
2. breaking down things - catabolic reaction (i.e. macromolecules) to generate energy
Energy
the capacity to do work, or the capacity for change
The energy released in catabolic reactions is used to drive anabolic reactions (usually indirectly) to...
do biological work
Catabolic and anabolic reactions are often...
linked
In order for some/all of the chemical bonds to be broken as reactant -> product so that new bonds in the product can form, ...
1) the molecule must be contorted into an unstable state -> transition state (higher energy state)
2) activation energy must be added for the molecule to reach the transition state (enzymes help to overcome this barrier)
Enzymes can increase reaction rates by...
1 million to 10^17 times.
The 3-D shape and composition of the enzyme...
determines the specificity (function).
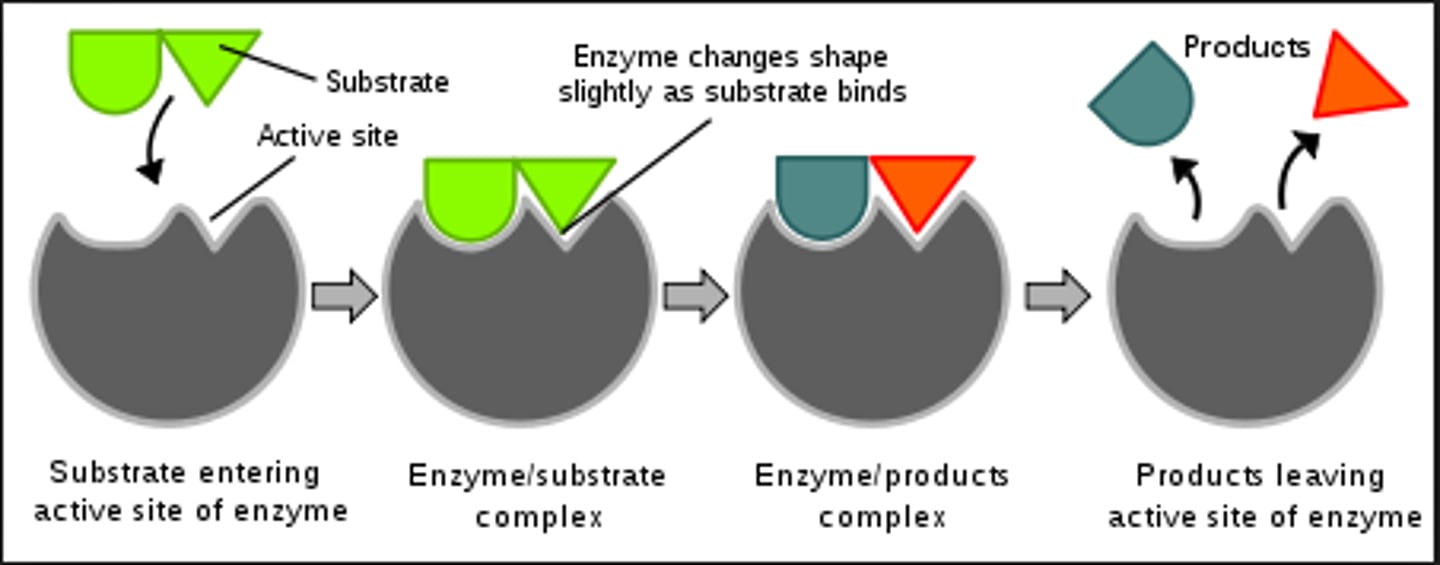
Many enzymes participate directly in...
the catalytic process.
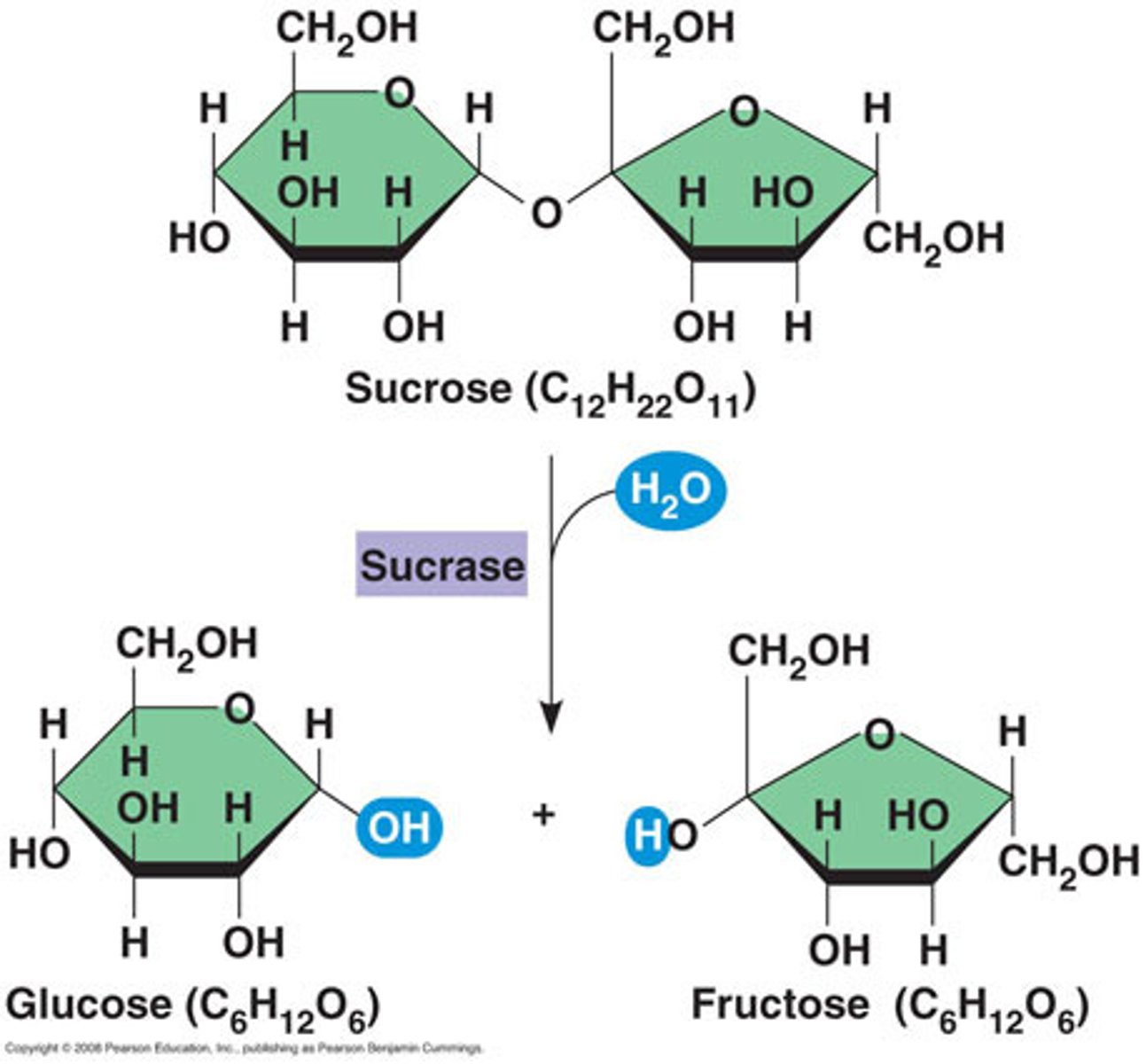
Carbohydrates are broken down by...
amylase, sucrase, lactase, or maltase.
Proteins are broken down by...
trypsin, pepsin, peptidase and others.
Lipids are broken down by...
lipases.
Sucrase
breaks down sucrose (disaccharide) into glucose and fructose (monosaccharides)
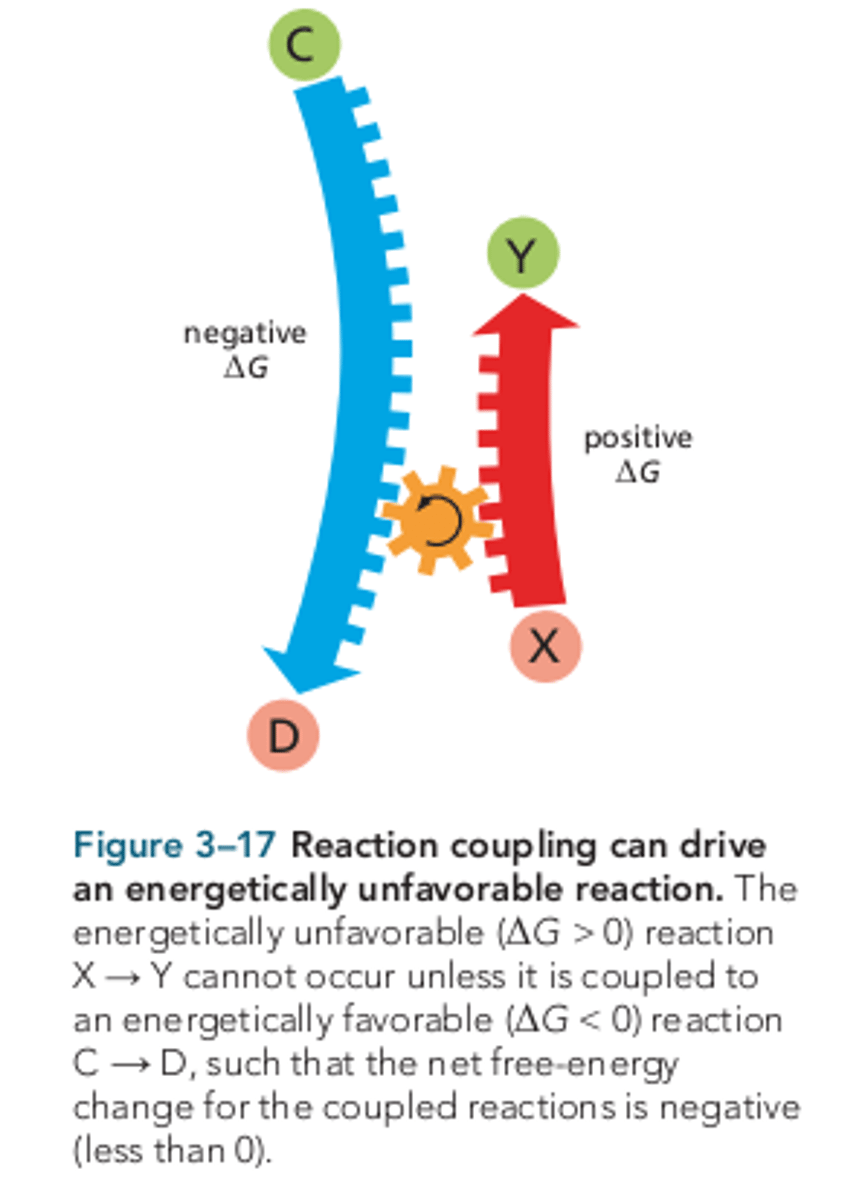
The energetically unfavorable reaction cannot occur...
unless it is coupled to an energetically favorable reaction, such that the net free-energy change for the pair of reactions is negative.
ATP can be hydrolyzed to...
ADP and Pi, releasing a lot of energy for endergonic reactions.
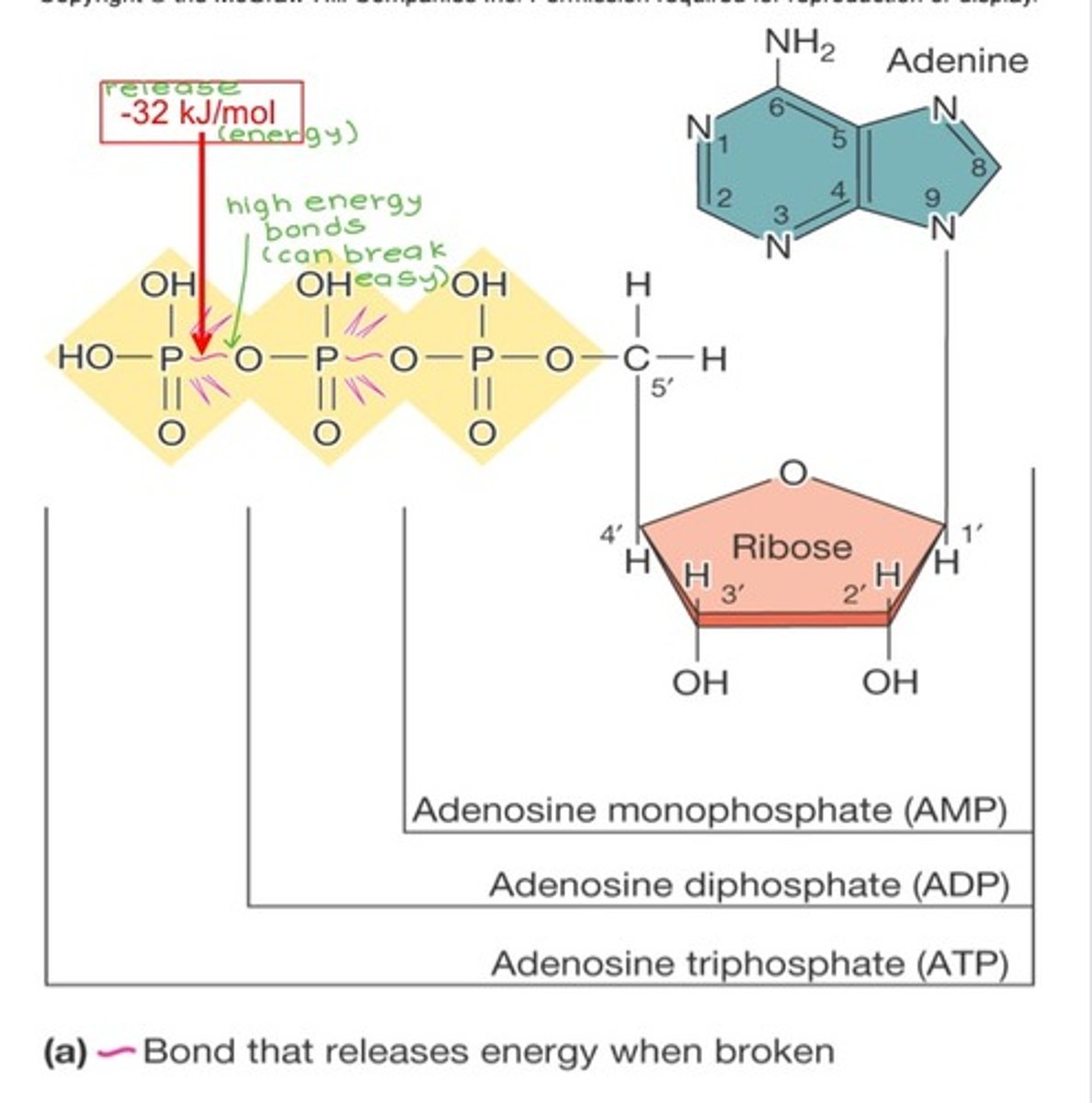
2 characteristics of ATP that account for the free energy released:
1) Phosphate groups have negative charges and repel each other - the energy needed to get them close enough to bond is stored in the P-O bond
2) The free energy of the P-O bond is much higher than the energy of the O-H bond that forms after hydrolysis (the free energy of ATP is higher than the free energy of ADP + Pi)
Cells use the energy released by ATP hydrolysis to...
fuel endergonic reactions (i.e. condensation).
Cells harness the energy released from exergonic reactions to...
fuel regeneration of ATP from ADP + Pi.
The interconversion of ATP and ADP with exergonic and endergonic reactions
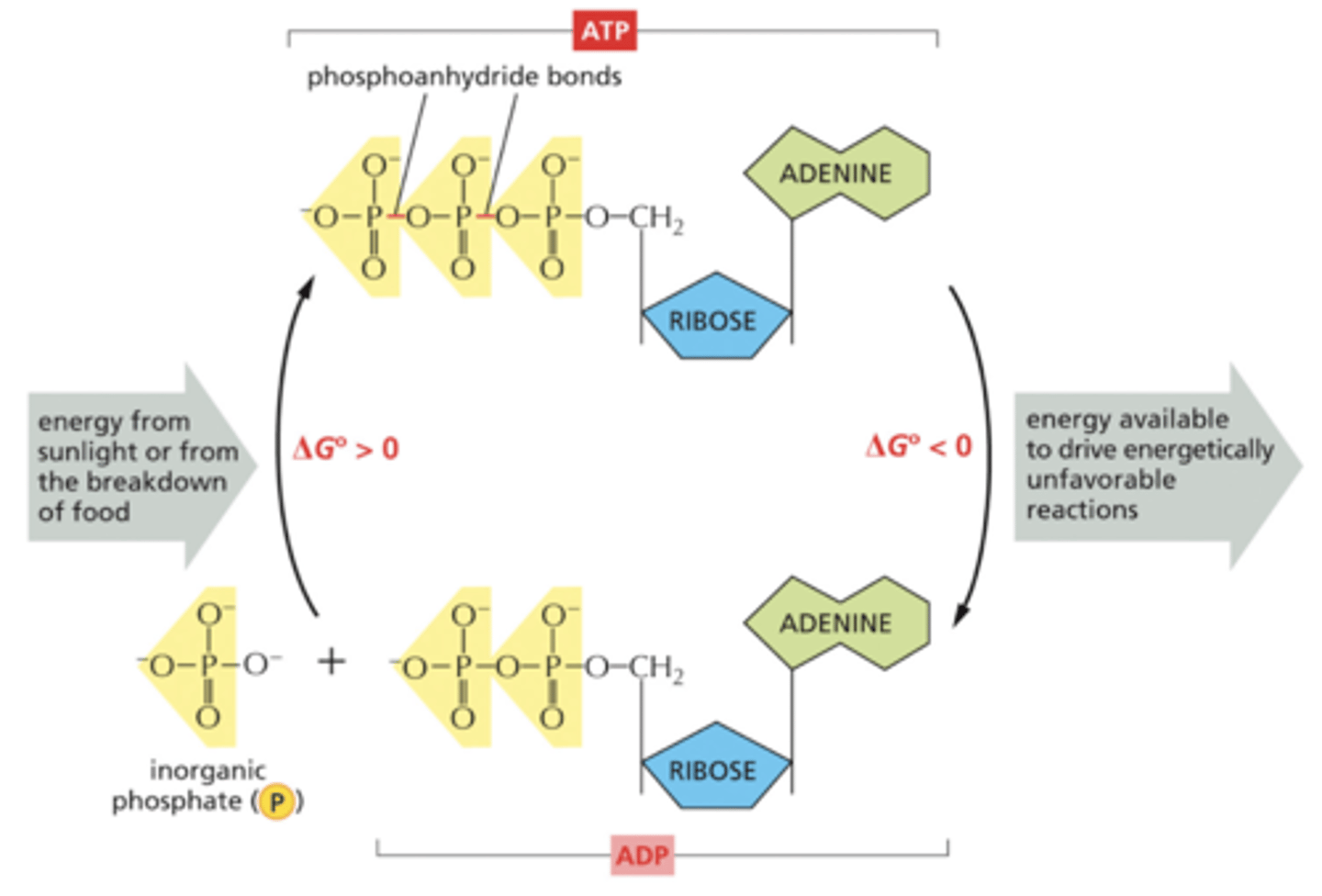
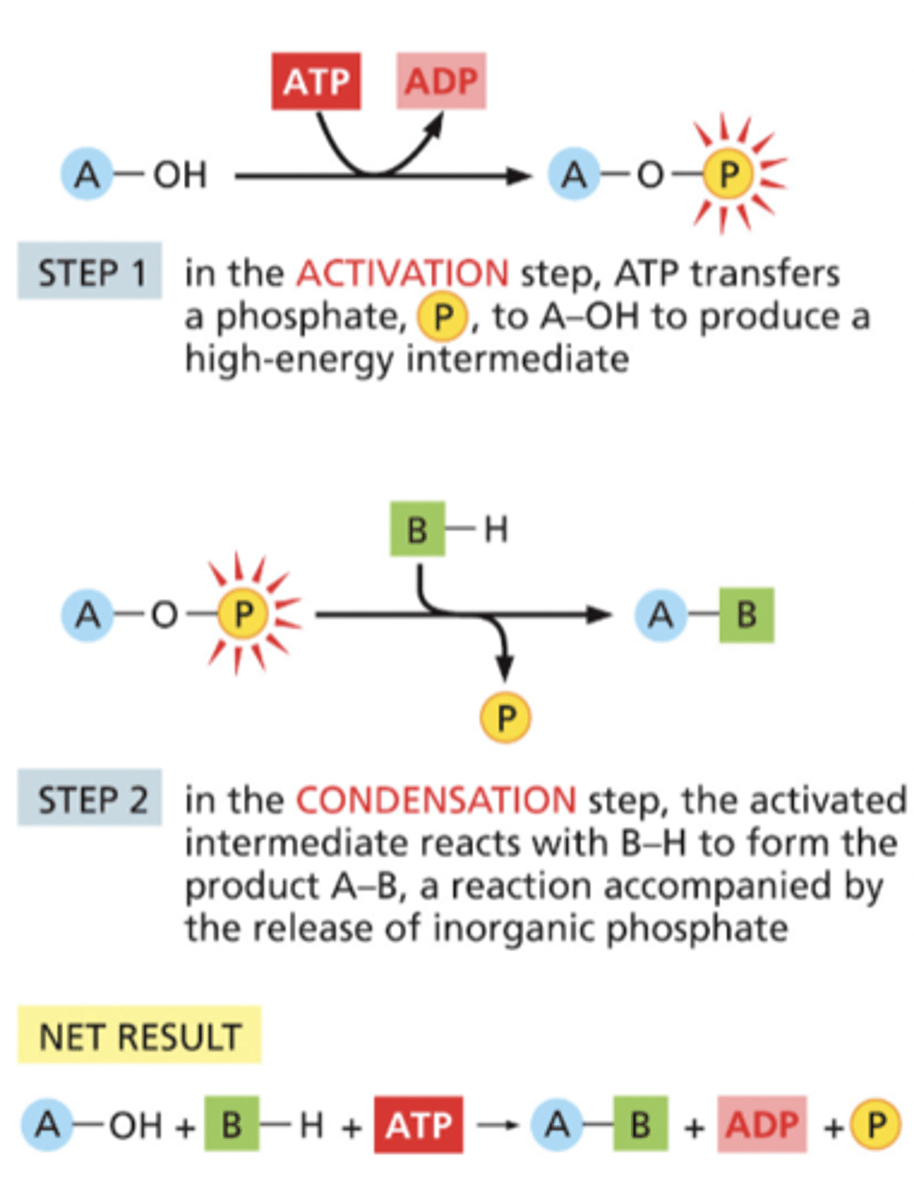
Reaction coupling
occurs for each condensation reaction in the synthesis of polysaccharides, nucleic acids, and proteins
Steps of reaction coupling
1) Activation Step: ATP transfers a phosphate, P, to A-OH to produce a high-energy intermediate
2) Condensation Step: the activated intermediate reacts with B-H to form the product A-B, a reaction accompanied by the release of inorganic phosphate
ACEN
Anabolic
Condensation
Endergonic
CHEX
Catabolic
Hydrolysis
Exergonic
Conversion of ADP to ATP must be coupled with...
an exergonic reaction
ATP and ADP contain ____________ in their structure
purine