Inorganic reaction mechanisms
1/71
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
What does it mean for a reagent compound to be stable?
has a thermodynamic energy state that is more favorable than the product, lower ΔG for reagents compared to products
What does it mean for a reagent compound to be labile?
undergoes rapid ligand exchange; low ΔG between reagents and intermediate
Effective nuclear charge (decreases/increases) across a period and (decreases/increases) down a group.
Effective nuclear charge (decreases/increases) across a period and (decreases/increases) down a group.
As Zeff increases, size (decreases/increases), ionisation energy (decreases/increases), electron affinity (decreases/increases).
As Zeff increases, size (decreases/increases), ionisation energy (decreases/increases), electron affinity (decreases/increases).
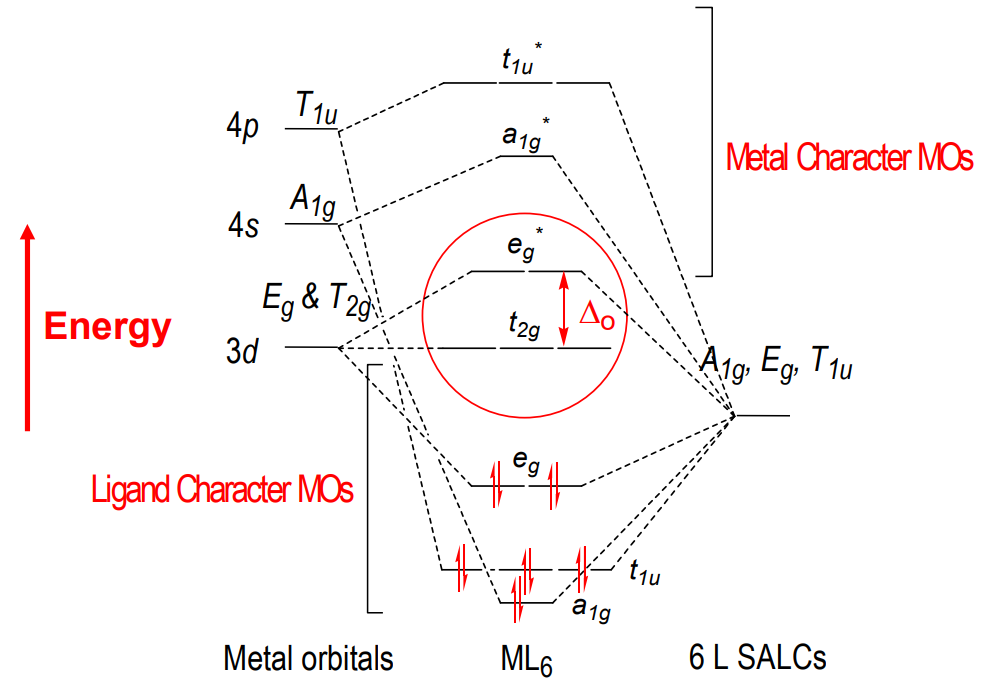
Octahedral metal complex orbitals
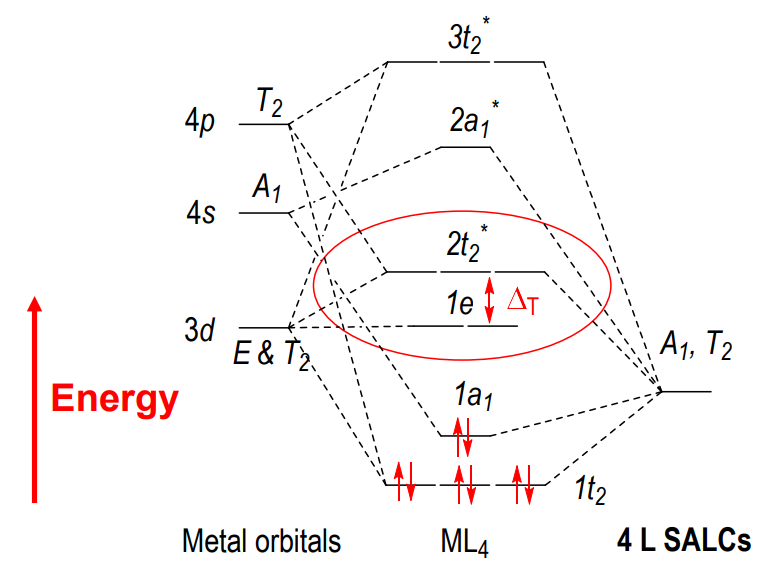
Tetrahedral metal complex orbitals
LFSE formula
LFSE= [ne(-0.4) + ne(0.6)]Δ + P
number of electrons in t2g orbitals
number of electrons in eg orbitals
number of orbitals in which paired electrons exist
Spectrochemical series
I- < Br- < S2– < SCN– < Cl- < NO3- < F- < OH- < C2O42- < OH2 < NCS- < CH3CN < py < NH3 < en < bipy < phen < NO2- < PPh3 < CN- < CO
*strong field ligands have increased sterics or has C or N donor (EXCEPT NO3-)
What spin configuration do tetrahedral complexes usually have?
high spin
How do 4d and 5d orbitals compare to 3d orbitals and how does it affect ligand interactions?
They are larger/extended, leading to significant penetration of core electrons and increased ligand orbital overlap, lower pairing energy
TRUE OR FALSE: All 4d and 5d transition metal octahedral complexes are low spin
TRUE
What causes Jahn-Teller distortion?
uneven electronic occupation of the orbitals, especially the eg orbitals
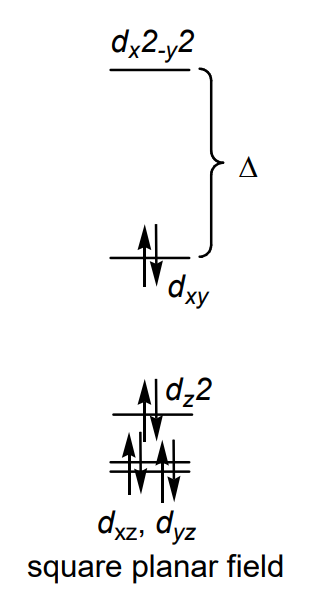
What is required for square planar complexes? (3) (Also commit figure to memory)
d8 electron configuration
heavy metals (4d or 5d)
strong field ligands (for 3d)
Organometallic compounds
contain a metal to carbon bond or interaction
Do organometallic or non-organometallic/classical complexes experience pi backbonding?
organometallic
What effects does pi backbonding have? (3)
the metal is electron rich (low formal oxidation states)
The metal to ligand bonds are more covalent
modifies ligand reactivity
Do octahedral complexes with pi accepting ligands have a greater or lower Δ and why?
greater (lower t2g) because pi acceptor ligand orbitals ABOVE dt2g also have t2g symmetry and can interact with the t2g d orbitals
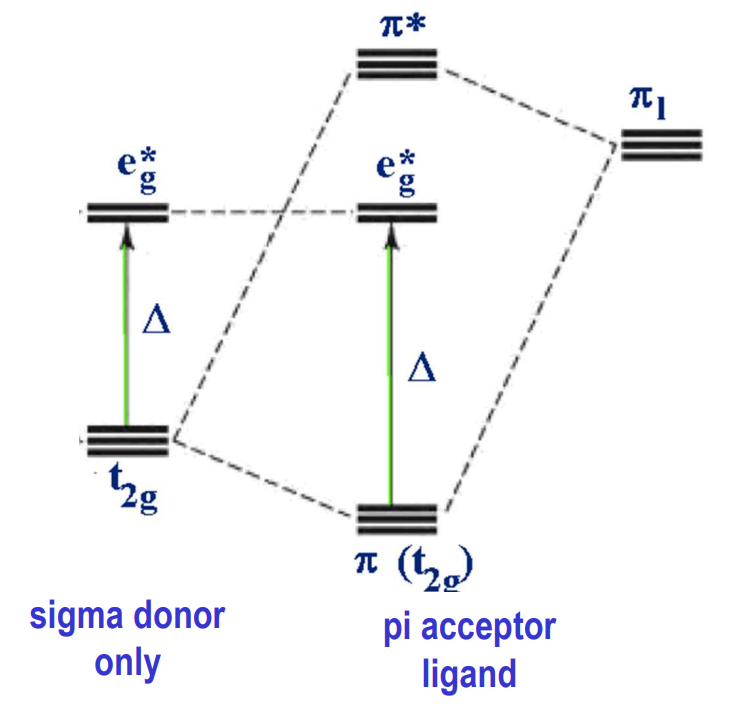
Do octahedral complexes with pi accepting ligands have a greater or lower Δ and why?
lower (raised t2g) because pi acceptor ligand orbitals BELOW dt2g also have t2g symmetry and can interact with the t2g d orbitals
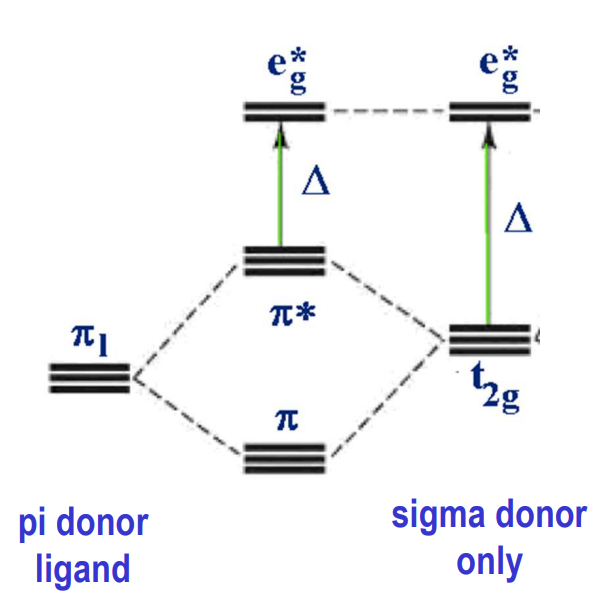
How does pi back bonding act on CO ligands in terms of a) reactivity and b) bonds?
a) The empty antibonding π* orbitals of CO accept electron density from the metal t2g orbitals making Cδ+ and Oδ-
b) weakening the carbonyl bond and strengthening the metal bond
How can electron donating/accepting capability of trans ligands to CO be rationalized?
A stronger electron-donating (less accepting) trans ligand increases electron density on the metal, enhancing CO backbonding, which weakens the C-O bond and lowers the infrared stretching frequency
Why does the large HOMO-LUMO gap between t2g and eg result in the complex being kinetically stable?
It is difficult to remove electrons from the t2g orbitals or add electrons to the eg orbitals to form low energy intermediates or transition states.
Why are 18 electron compounds stable?
all bonding molecular orbitals are filled, and antibonding unoccupied
What complexes deviate from the 18 electron rule?
d8 square planar organometallics: dx2-y2 orbital is very high in energy (16 electron configuration)
Early transition metal compounds: low number of metal valence electrons and difficulty avoiding steric conflicts
Electron counting (4 steps)
Treat metal and ligand as neutral
Count all valence electrons (4s, 3d) of the metal
Classify the ligands and include all electrons donated. Consider M-M bonding and bridging ligands
Adjust for the overall charge on the complex
What are the two types of ligand electron donors (neutral configurations)?
L type: 2 electron donor
X type: 1 electron/radical donor
What is the oxidation state of a) L and b) X type ligands?
a) 0
b) -1; also applies to LnX
TRUE OR FALSE: early transition metal complexes are unstable due to lower electron counts
SOMEWHAT FALSE: Very sterically demanding ligands can be used to stabilise them
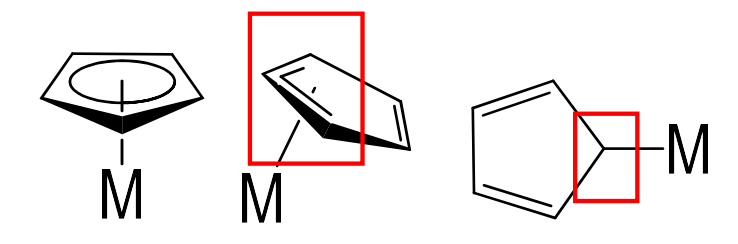
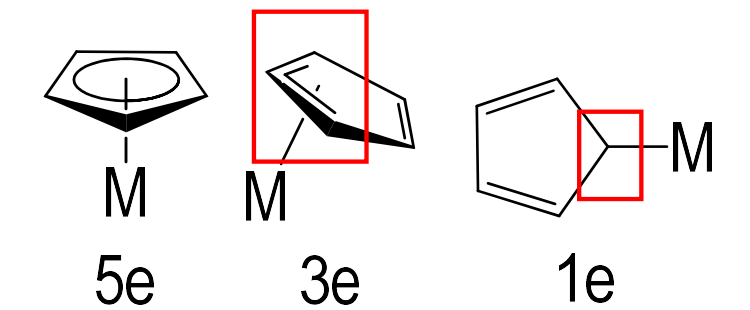
How many electrons do the following cyclopentadienyl orientations donate?
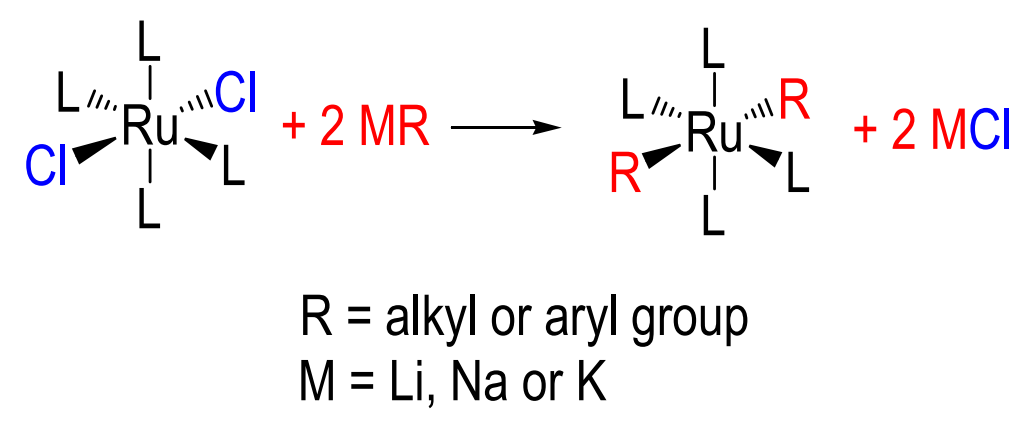
alkali metal halide elimination
How does length of the C=C bond in an η2 -alkene complex compare to the free alkene and why?
Longer due to use of bonding electrons to coordinate metal
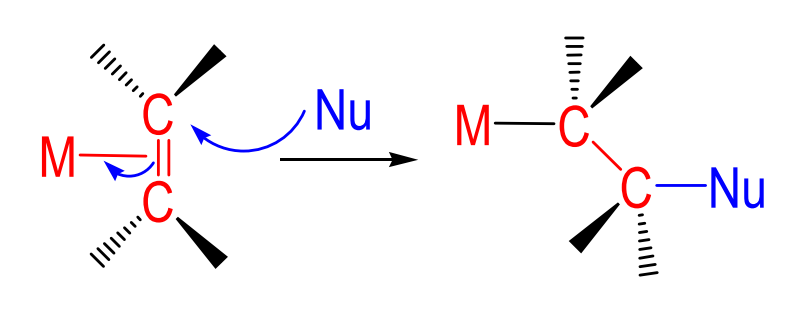
M-(η2-Ligand) + nucleo/electrophile → ?
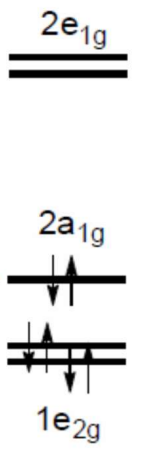
Bis(benzene) Chromium d-orbital splitting
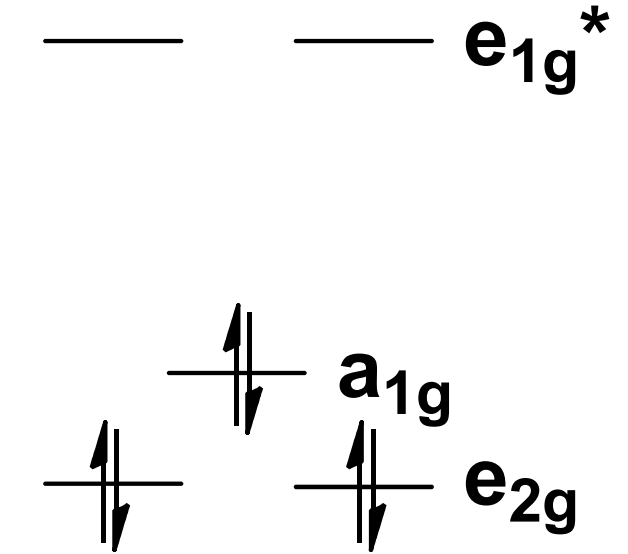
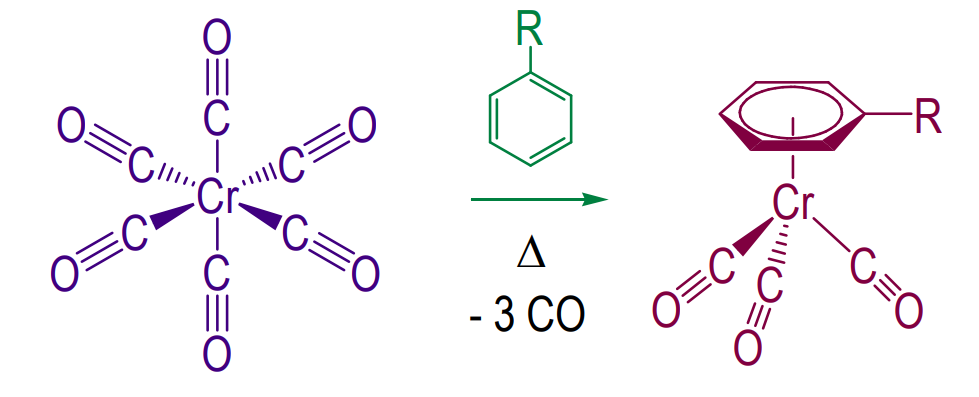
Piano Stool Complexes Preparation
Ferrocene d-orbital splitting
As Zeff increases for metallocenes, M-C bond length…
decreases
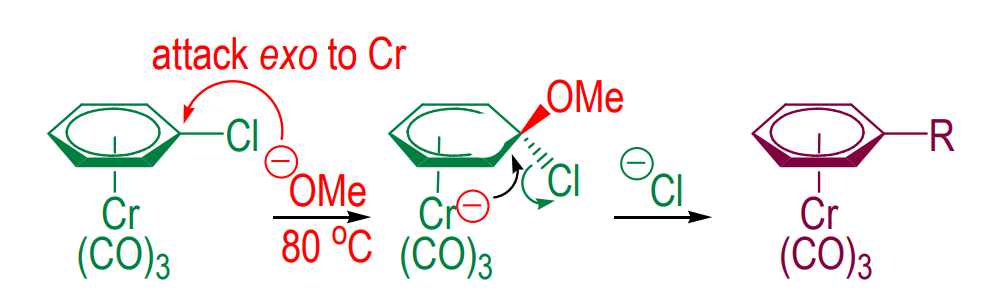
Nucleophilic substitution
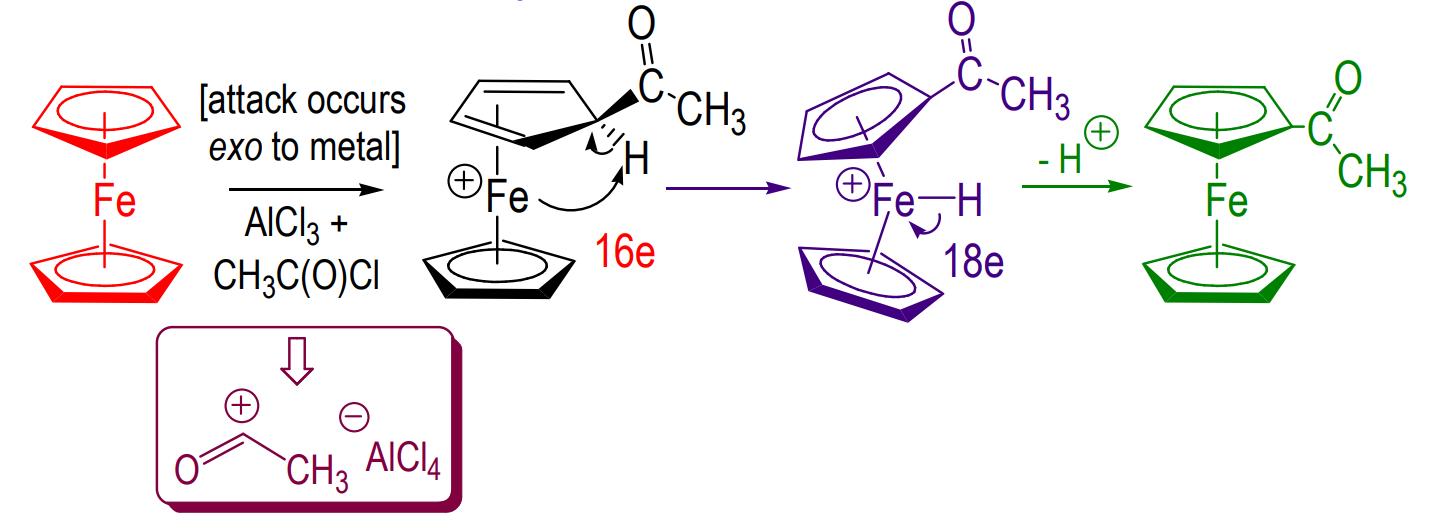
Friedel-Crafts acylation
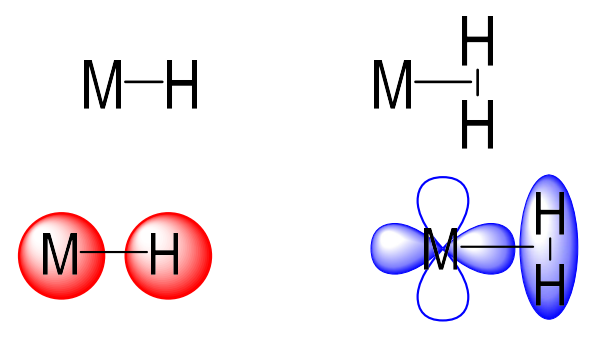
How does hydrogen bond to a metal
σ-lone pair donor (X type) or as dihydrogen (L type)
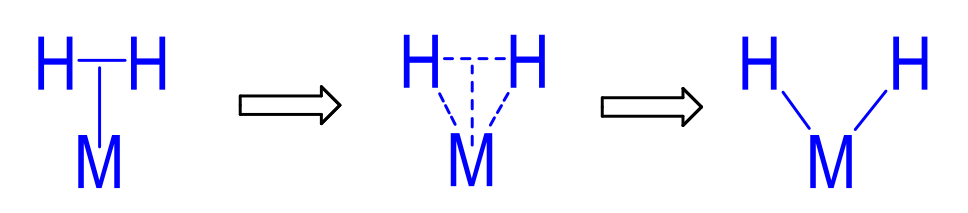
Metal Hydride Formation (2 steps)
bonding of H-H to metal centre
Backbonding from electron rich complex breaks H-H bond
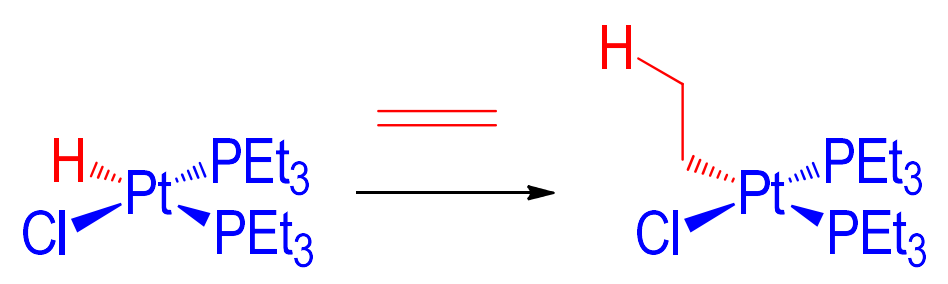
Rn-M-H (hydride) + RC=CR (alkene/alkyne) → ?
Rn-M-RC-CR-H
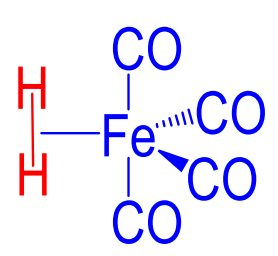
TRUE OR FALSE: Electron poor complexes can form metal hydride complexes if they have a heavy metal
FALSE: more likely to yield dihydrogen complex (pictured)
Incorporating H2 into metal complex (decreases/increases) acidity and (lengthens/shortens) the H-H bond
Incorporating H2 into metal complex (decreases/increases) acidity and (lengthens/shortens) the H-H bond
What changes does oxidative addition to a metal result in? (4)
+2 coordination number
Mn+ to M(n+2)+
n+2 electrons; usually tending to 18 → increased stabilisation
molecule generates a compound with X and Y cis to one-another
Oxidative Addition
where X and Y must have an electronegativity greater than the metal
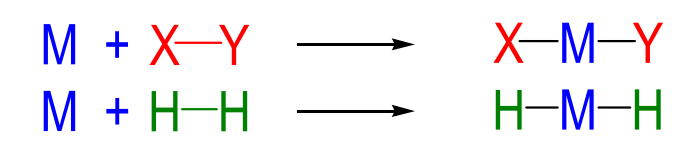
metal nucleophilic attack
SN2
common for polar molecules
trans addition
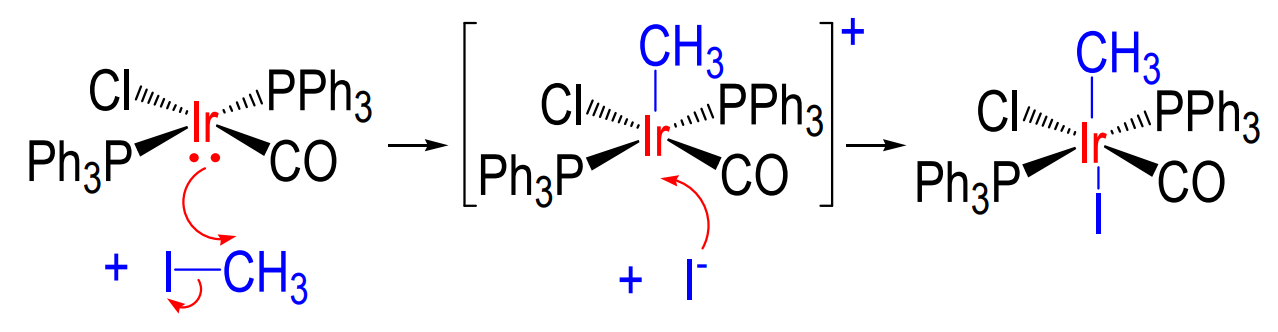
a) What is the mechanism for reductive elimination and b) what structural feature does it require?
a) exact opposite to oxidative addition
b) eliminated molecules cis to one another
TRUE OR FALSE: Oxidative addition and reductive elimination exist in an equilibrium
TRUE, BUT one direction is thermodynamically favoured
C-H bond activation
Irradiation or heat can leads to the loss of H2 producing a 16 electron intermediate (by reductive elimination) which can undergo oxidative addition of C-H bonds
1,1-migratory insertion
primarily with CO ligands
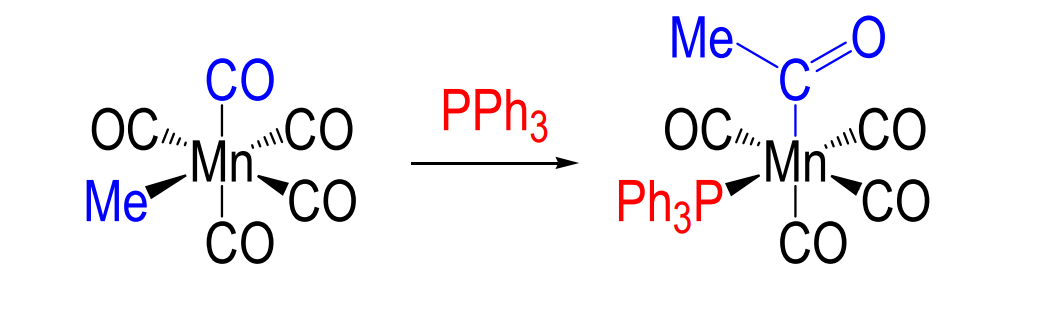
What effect does migratory insertion have?
-1 coordination number
-2 electrons at metal center
1,2-Insertion
primarily with η2 -coordinated alkenes or alkynes
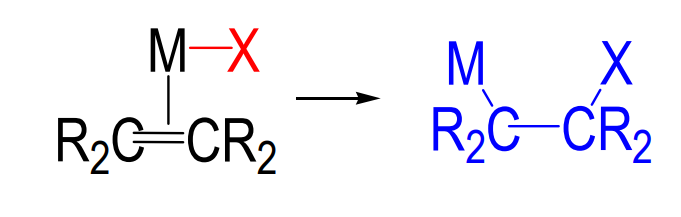
In what circumstances is β-elimination not common?
When the β-carbon to M has less than 2H
Wilkinson’s catalyst
[RhCl(PPh3 )3 ]; used to convert alkenes to alkanes with < 1 atm H2
Enantioselective reactions require a (__) substrate
Enantioselective reactions require a prochiral substrate
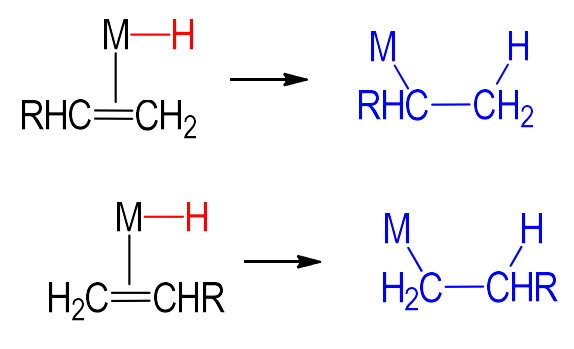
How can branching (pictured) be prevented?
partial replacement of a catalyst’s ligand to induce steric effects
How is the nature of the interactions between the main group metal ion and the water molecules described?
predominantly electrostatic, non-directional
Covalency of interactions transition (__) main group metals
Covalency of interactions transition > main group metals
How many H2O molecules in the first hydration sphere of metals?
typically six, except Li+ which has four due to its small size and low charge
What increases hydrated ionic radii?
increased charge density (small ion, large charge)
TRUE OR FALSE: neutral ligands result in more stable complexes for both transition and main group metals
FALSE: less stable for main group. Same effect as for large polarisable ions (eg. Br-)
stability of a complex with a Group 1 (__) Group 2 metal ion with same ligands
stability of a complex with a Group 1 < Group 2 metal ion
Put in order of complex stability with a) small donor ligands and b) multidentate ligands: Ca2+, Li+, Mg2+, Na+
Mg2+ > Ca2+ > Li+ > Na+
Ca2+ > Mg2+ > Na+> Li+
TRUE OR FALSE: chelating ligands provide stability
TRUE
Macrocyclic ligands form strong complexes with the Group(s) (__) elements
Macrocyclic ligands form strong complexes with the Group 1 elements
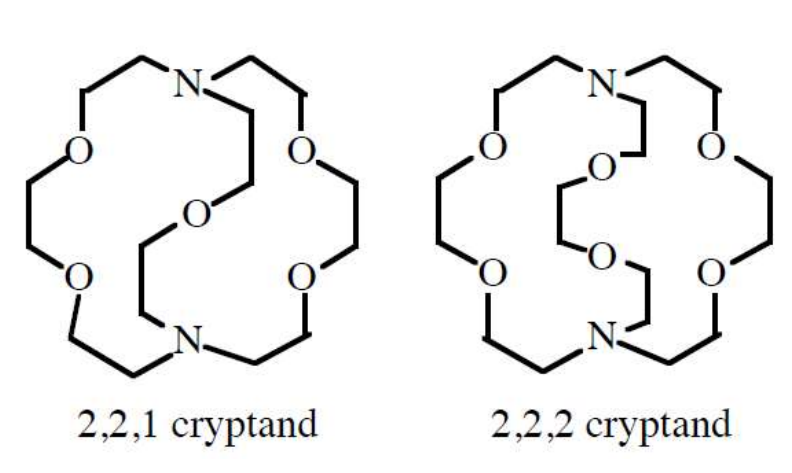
How can macrocyclic ligands be used for “selection”?
matching the cavity size with the “ionic” radius of elements
macrocyclic effect
the cyclic ligands are pre-organised, so the entropy change on ligation is even more positive therefore favourable than for chelating ligands
f orbitals
there are 7 with 3 angular nodes each
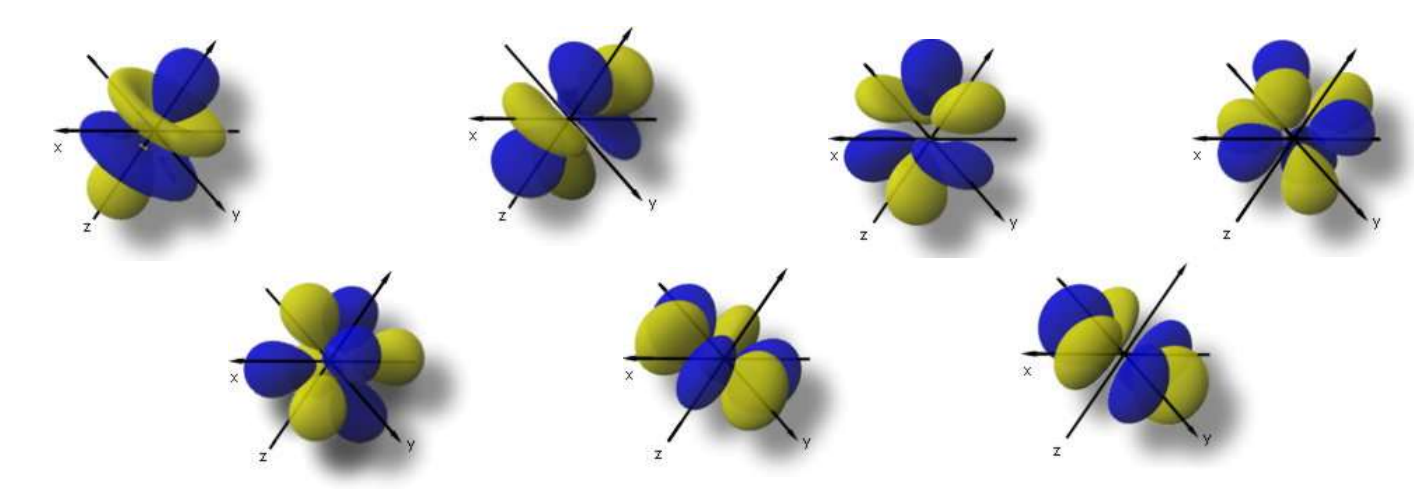
The term given to the f orbitals due to the electrons being kept away from the nucleus
non-penetrating
In what order are the orbitals after 4p filled?
5s, 4d, 5p, 4f
BUT no 4f2, 4f8 or 4f15;
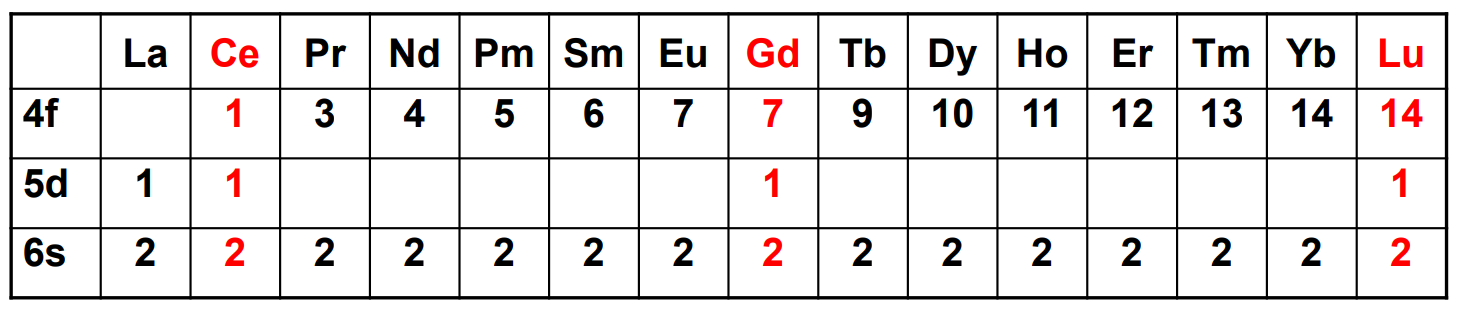
As charge on a lanthanide increases, the 4f energy (decreases/increases). Therefore, the maximum in the radial distribution function moves (away from/towards) the nucleus and the 4f orbitals become (more/less) core-like, which (does/does not) result in ligand orbital overlap.
As charge on a lanthanide increases, the 4f energy (decreases/increases). Therefore, the maximum in the radial distribution function moves (away from/towards) the nucleus and the 4f orbitals become (more/less) core-like, which (does/does not) result in ligand orbital overlap
The initial oxidation of lanthanides occurs from the electrons in which orbitals and which is the typical oxidation state?
6s, 3+