PATH 381 - Module 4: Inborn Errors of Metabolism
1/96
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
newborn screening (NBS)
test done shortly after birth to test for treatable diseases that usually show no symptoms in newborn period
screening program
follows brain to brain loop, including:
- lab tests
- providing parents with info
- infant treatment
benefits of NBS
early detection = early treatment = better long-term health outcomes
NBS tests
test small amount of blood within first days of life
Consequences of NBS (or not performing it)
- cost of missing a disease is huge (in suffering, financial terms)
- if untreated, infants can get learning disabilities, serious health problems, and can die, sometimes without ever being diagnosed
NBS time of screening
shortly after 24h of birth
NBS detectable conditions
over 1 in 300 newborns have detectable condition through NBS
NBS health conditions when disease is caught
most babies with serious but treatable conditins caught by NBS grow up healthy with expected development
Criteria for Offering NBS (10)
1. condition is an important health problem
2. there is an accepted treatment
3. facilities for diagnosis and treatment are available
4. recognizable latent/early symptomatic stage
5. suitable test/examination
6. test should be acceptable to population
7. natural history of condition (development from latent-disease) should be well understood
8. agreed policy on whom to treat as patients
9. cost of case funding (diagnosis and treatment) should be economically balanced
10. case finding should be a continuing process, not 'once and for all' project
Ontario's NBS System (3 stages)
Upstream of Testing:
- baby born, blood sample taken to NSO (newborn screening ontario) office at CHEO (childrens hospital eastern ontario)
At CHEO:
- sample processed by clinical, lab, clerical staff
- other roles: data mangement, research, education, quality assurance
Downstream of Testing:
- communication with important health advisory committees
- test results reported back to submitters
- communication remains between submitters and treatment centers for further testing, diagnosis, treatment, education, rehab
NBS diseases screened in Ontario
1. metabolic diseases: dsieases where can't break down substances in food, eg. fats/proteins/sugars
2. critical congenital heart disease (CCHID): affects amount of oxygen in blood
3. sickle cell disease: affects movement of oxygen in blood
4. severe combined immune disease (SCID): affects ability to fight infections
5. endocrine diseases: too much/too little of certain hormones
6. spinal muscular atrophy (SMA): muscle weakness and wasting
7. cystic fibrosis (CF): problems with breathing and growth
role of healthcare providers in NBS
- ensure all babies offered NBS
- NOT MANDATORY, but strongly recommended for all infants
- must fully explain NBS to parents
- explain that it identifies babies who need more testing, does not provide a diagnosis
prevalence
proportion of population with condition at a specific point in time (point prevalence) or during period of time (period prevalence)
prevalence equation
prevalence =
(total # with disease) / (population # at risk for disease)
incidence
rate of new cases/events during specific time period for population at risk
incidence equation
incidence =
(new cases) / (population x timeframe)
how can inborn errors in metaboolism (IEM)s be screened?
1. tandem mass spectrometry (MS/MS)
2. immunoassay
MS/MS: detection and process
- detection of 25 metabolic disorders in single process
- used dried blood spot (DBS) specimens collected by heel prick from newborns
- can separate, detect, quantify biomarkers
detection of DBS using MS/MS
- sample collected via heel prick on Guthrie card
- then use organic solvents and separate analytes via liquid chromatography
MS/MS procedure (6)
1. electrospray: once DBS extracted, converted into charged particles using electrospray
2. MS-1: mix of components introduced to first mass spectrometer through electrospray and compounds separated by mass/charge ratio
3. collision cell: analyte ion of interest passes into chamber, fragmented to charged molecules of smaller size
4. MS-2: smaller ions pass into second mass spectrometer, programmed to detect select ion
5. detector: combined, gives specificity to selectively detect small molecule analytes in complex bio matrix of DBS
6. data analysis: data collected and analyzed electronically, tells you how much biomarker present
how is MS/MS so specific
- detects charged particles in narrow mass range
- fragments molecule of charged particle further
- detects fragments within narrow mass range
mass spectrometry screens for what 3 IEMs?
1. medium-chain acyl-coA dehydrogenase deficiency (MCADD)
2. phenylketonuria (PKU)
3. maple syrup urine disease (MSUD)
MCADD is what type of disorder
fatty acid oxidation disorder
MSUD is what type of disorder
branched-chain amino acid disorder
PKU is what type of disorder
amino acid disorder
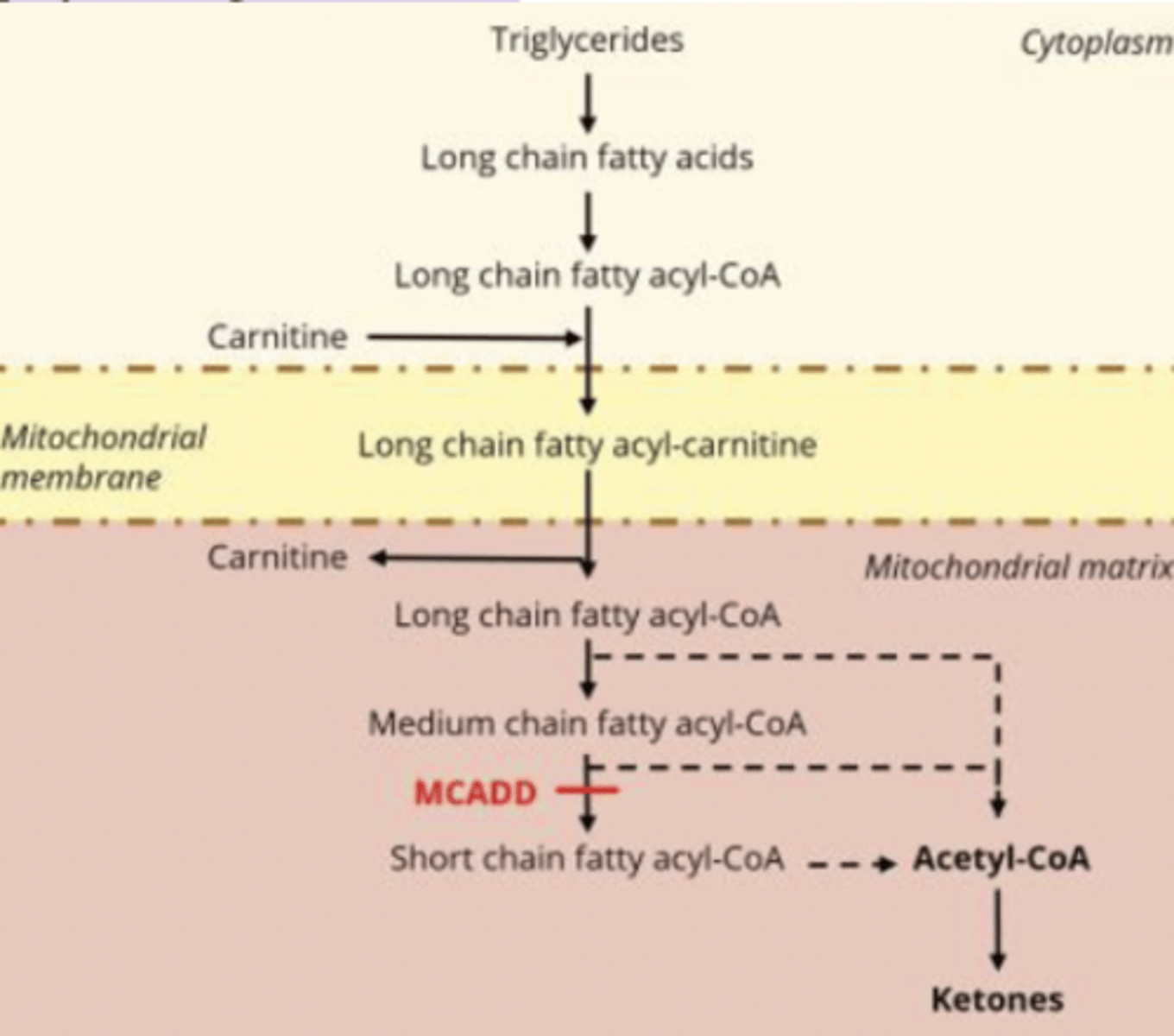
fatty acid beta oxidation steps
1. Transport: Fatty acids enter the cell
2. Activation: FACS adds CoA→ fatty acyl-CoA
3. Carnitine translocase: acyl-CoA → acylcarnitine (to cross IMM) → back to acyl-CoA in mitochondria
4. β-oxidation: produces NADH, FADH₂, and acetyl-CoA
5. TCA + ETC: acetyl-CoA → ATP via NADH & FADH₂
what is MCADD?
- inherited disorder stopping body from breaking down certain fats and converting them into energy
- specifically a disorder of fatty acid oxidation
- can cause hypoglycemia
Medium-chain acyl-CoA dehydrogenase (MCAD)
flavoprotein required for first step in B-oxidation of medium-chain fatty acyl CoA to produce acetyl-coA (needed to make ATP, ketones)
MCAD deficiency (MCADD) biochemical pathology
no MCAD = no 𝛽-oxidation of medium-chain fatty acids = ↓ acetyl-CoA = ↓ketones, ↑medium-chain fatty acids = hypoketotic hypoglycemia
MCADD Symptoms
- if untreated → hypoglycemia → seizures, breathing difficulties, coma, etc
- but can be avoided with nutritional approach
GI symptoms can cause what in MCADD
GI symptoms (lost appetite, vomiting, diarrhea) can cause metabolic crisis in MCADD
what are metabolic crises in MCADD
- accummulation of potentially toxic acylcarnitine
- hypoketotic hypoglycemia
- Reye syndrom like episodes (swelling of liver and brain)
- seizures
- brain damage
- death (if undetected, 20-25% die in first metab crisis)
MCADD screening method
- heel prick to take drops of blood shortly after birth
- screen positive = more tests needed to know if baby has MCADD (does not mean baby has MCADD)
MCADD treatment
- very effective at preventing metab crises = babies live normal lives
- must avoid caloric starvation (prevents need to use ketosis for energy)
- patient must adapt diet, especially during stressful events
MCADD follow-up testing
- elevation of medium-chain acyl carnitine values = positive results
- testing includes plasma acyl carnitine analysis, urine organic acid analysis, urine acylglycine analysis
false pos/false neg for carnitine testing
- false positives not common, but can be seen in term infants at appropriate gestational age
- false negatives in newborns with low free carnitines
(eg. born to mother with low carnitine, mothers with MCADD, etc)
Phenylketonuria (PKU)
rare amino acid disorder caused by a genetic mutation preventing body from breaking down phenylalanine (can be found in foods and aspartame) → if individual eats these foods, Phe builds up in blood
PKU Screening Test
- blood test given to newborns 24-74 h after birth
- PKU test to see if high levels Phe in blood
- if high levels, order more confirmatory tests
PKU: Consequences
- high levels Phe can permanently damage nervous system and brain
- can cause seizures, psychiatric problems, learning disability
PKU: management
if identified early and follow special, low-protein/low-Phe diet, can prevent complications for the rest of their life
Branched Chain Amino Acids (BCAAs)
- Leucine
- valine
- isoleucine
- allo-isoleucine
how are BCAAs usually metabolized
through processed catalyzed by branched-chain 2-ketoacid dehydrogenase (BCKDH) complex
Maple Syrup Urine Disease (MSUD)
- BCAA disorder: deficiency of BCKDH complex
- genetic metabolic disorder, usually caused by mutations in BCKDHA, BCKDHB, DBT
what happens in MSUD
due to metabolic block = build up high conc of BCAAs (leucine) and toxic keto-acids if not on BCAA-restricted diet and during increasd protein catabolism
MSUD: incidence
1 in 185,000
MSUD: symptoms
- maple syrup odor (sweet smell) in urine and ear wax
- failure to thrive (delay in weight gain/growth)
- developmental delays
- seizures
- movement problems
MSUD: diagnosis
- early diagnosis and management essential to prevent permanent brain damage
- NBS by MS/MS shows elevated BCAA in blood before encephelopathic symptoms (affecting brain)
MSUD: treatment
restrict leucine in diet, supplement with isoleucine and valine
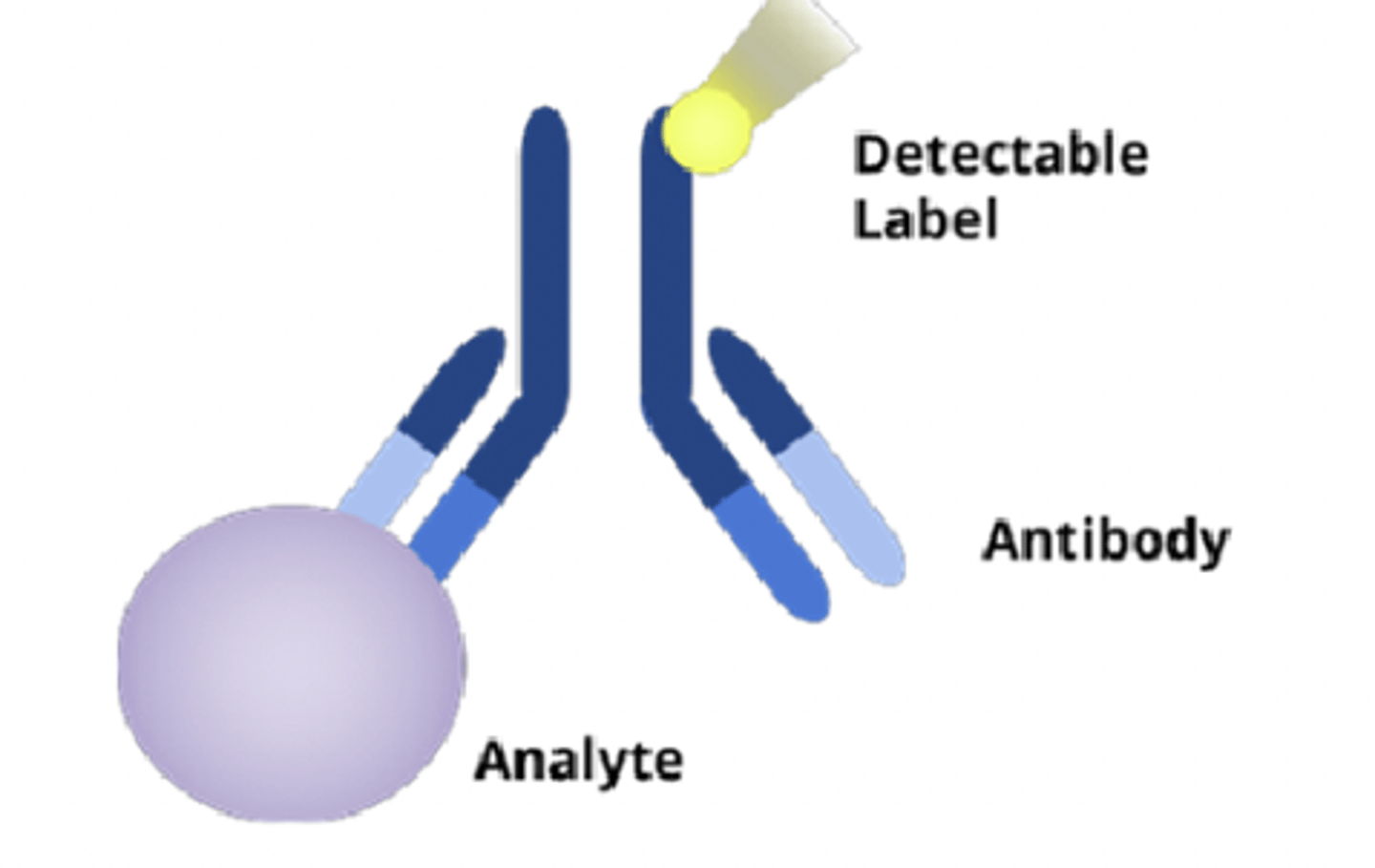
immunoassays
analytical technique using antibody-antigen rxn to quantify amount of analyte in sample
why do immunoassays use antibodies?
- antibodies are antigen-specific
- immunoassays use this antibody-antigen relationship to identify specific molecules
- biomarker/analyte acts as the antigen
- use detection label attached to antibody to quantify results
enzyme-linked immunosorbent assay (ELISA)
- antibody/antigen immobilized on plate
- target molecule detected using one or more specific antibodies
- final antibody in the sequence is enzyme-linked, and the enzyme converts a substrate into a measurable colored product
3 types of ELISA
1. direct
2. indirect
3. sandwich
direct ELISA
antigen immobilized on bottom of plate and enzyme-linked antibody directly binds antigen
indirect ELISA
- antigen immbolized on plate
- primary antibody binds antigen
- secondary antibody (enzyme-linked) binds primary antibody
sandwich ELISA
1. primary/capture antibody immobilized on plate
2. antigen (target added) and binds antibodies
3. detectore/secondary antibody (enzyme-linked) binds antigen
4. substrate added → enzyme converts to coloured product
5. colour intensity = amount of antigen present
what do all ELISAs have in common?
the last antibody binding is always conjugated to an enzymatic label that confirms binding
non-competitive assays
- eg. sandwich immunoassay
- biomarker/analyte captured between 2 capturing sites
- secondary antibody has detection label
- no analyte = no signal
competitive assay
- known amount of labeled analyte competes with the unlabeled analyte in sample for same antibody-binding site
- After binding, unbound analyte washed away
- Only labeled analyte bound to antibody produces a signal:
so less signal = more unlabeled (patient) analyte.
Therefore:
More signal → less unknown analyte
Less signal → more unknown analyte
congential hypothyroidism (CH)
baby born with defective thyroid gland = deficient thyroid hormone
potential causes oh CH
- gland not developed properly (missing, wrong location, too small)
- or look healthy but not working properly
early signs/symptoms of CH (6)
- puffy face
- swelling around eyes
- poor feeding
- constipation
- jaundice
- soft spot on head slow to close
thyroid hormones are essential for...
- CNS development (especially in first 3 years)
- regulating metabolic rate
- controlling heart, muscle, digestion, bone maintenance
CH: screening
- screening can prevent developmental delayes/failure to thrive
- DBS collected by heel prick
- results analyzed using immunoassay measuring TSH in mIU/L blood vol
CH: diagnosis and treatment
- if diagnosed early, easily treated with daily oral thyroid meds (TH replacement)
- early treatment can prevent delayed milestones, intellectual impairment, poor growth, hearing loss
TSH Screening Cut-Offs for CH
- many programs use standard TSH screening cut-off in 20-30 mIU/L range (newborns below cut-off = negative for CH)
- other programs use 2 cut-offs: standard and lower threshole (eg. 6 mIU/L)
- second group is low risk but still has follow up thyroid function testing
why is there a TSH cut-off debate
- TSH falls quickly after birth (in first hours after delivery, TSH is VERY high, but decreases over the first few days) → age of sample collection affects what is "normal"
- different labs use diff TSH assays → diff sensitivity and reference ranges
what is the reference TSH versus what level TSH is considered a significant risk of CH?
reference: 1.7-9.1 mIU/L
high-risk: 17-19.9 mIU/L
what does NBS for CH measure?
1. thyroxine (T4)
2. thyroid-stimulating hormone (TSH)
3. T4-binding globulin (TBG)
what is the diagnosis of CH/primary hypothyroidism confirmed by? (2)
1. decreased serum thyroid hormone (total/free T4) levels
2. elevated TSH
3 screening strategies for detecting CH
1. TSH as primary measurement:
(backup T4 test in infants with high TSH)
2. T4 as primary measurement:
(backup TSH tests in infants with low T4)
3. simultaneous measurements:
(measure T4 and TSH together)
true and false positives, true and false negatives
true positives: number patients correctly identified by test having disease (done in relation to gold-standard diagnostic)
false positives: number pts incorrectly identified by test having disease
true negative: number patients correctly identified by test as NOT having disease
false negative: number patients INcorrectly identified by test as not having diseiase
what are 2 metrics that can characterize how likely it is for one to be healthy vs affected given a test result?
PPV (positive predictive value) and NPV (negative predictive value)
What is PPV?
- ratio of true positives to all positive tests (including false pos)
- predict how likely it is for someone to truly have disease in case of positive test result
PPV equation
PPV = True Pos / (True Pos + False Pos)
(TP / all pos tests)
What is NPV?
- ratio of true negatives to all negative results (including false neg)
- predict how likely for someone to be truly healthy in case of negative test result
NPV equation
NPV = True Neg / (True Neg + False Neg)
(TN / all neg tests)
What is sensitivity?
- proportion of true pos tests out of all patients with a condition
- ability of a test to yield positive result for someone with the disease
sensitivity equation
sensitivity = (true pos) / (true pos + false neg)
What is specificity?
- proportion of true negatives out of all patients withOUT condition (healthy)
- ability of test to accurately identify healthy individuals
specificity equation
specificity = (true neg) / (true neg + false pos)
likelihood ratios (LRs)
- how much a result changes probability of disease
- used to undnerstand utility of diagnostic tests
- use spec and sensitivity to see how likely it is pt has disease
what is positive LR (LR+)?
likelihood that pos test result would be expected in a person with disease compared to likelihood that pos test would be expected in someone without disease
(bigger LR+ = greater likelihood of disease)
LR+ equation
LR+ = sensitivity / (1-specificity)
what is negative LR (LR-)? (and equation)
likelihood that neg test is expected in someone with the disease compared to neg test expected in someone without disease (smaller LR- = lesser likelihood of disease)
LR- = (1-sensitivity) / specificity
What questions does each address? : Specificity/Sensitivity, PPV/NPV, LRs
specificity: is this test good at confirming a disease?
sensitivity: is this test good at confirming NO disease?
PPV: pt tested pos, what does this mean for pt?
NPV: pt tested neg, what does this mean for pt?
LR+: how much does the pos test result increase odds of having disease?
LR-: how much does neg result decrease odds of having disease?
what sensitivity/specificity metrics would be ideal in NBS? is this realistic?
- goal of NBS/any screening is to identify all with disease and exclude all without
- so ideal sens/spec would be 1.00
- hard to achieve this, and often compromise
compromise in NBS for test metrics
- hard to acheive a sens/spec of 1.00:
- compromise in setting cut-off values for pos and neg findings
- eg. sensitivity of 0.961 vs specificity 0.906 means test is better at ruling-IN a disease vs ruling-OUT
cut-off concentration levels
- used in NBS to determine which levels are considered healthy vs abnormal
- cut-off conctells clinician if marker conc indiciates high or low risk for a risease
- determining precise cut-off values very important and constantly re-evaluated
how are cut-off concentrations determined? (using DBS specimens) (6)
1. perform population study:
- test 100s-1000s of DBS samples from healthy (unaffected) newborns
- include manufactured controls (known normal/abnormal samples)
- test on every instrument that will be used clinically (accounts for inter-instrument variation)
2. analyze data:
- to determine if screen has good precision (reproducibility) and accuracy (closeness to true value)
- the test must clearly distinguish values near the cut-off (so borderline results are reliable)
3. set preliminary cut-off:
- use data to assign prelim cut-off by comparing healthy and positive specimen results collected
4. verify cut-off:
- use positive control specimens or residual DBS of a confirmed patient
5. compare cut-off:
- check that cut-off aligns with other labs using same assay and published literature
6. ongoing evaluation:
- NBS programs have standard operating procedure and regulatory requirements for establishing cut-off conc, that can be referred to and cited in situations where cut-off must be defended/changed
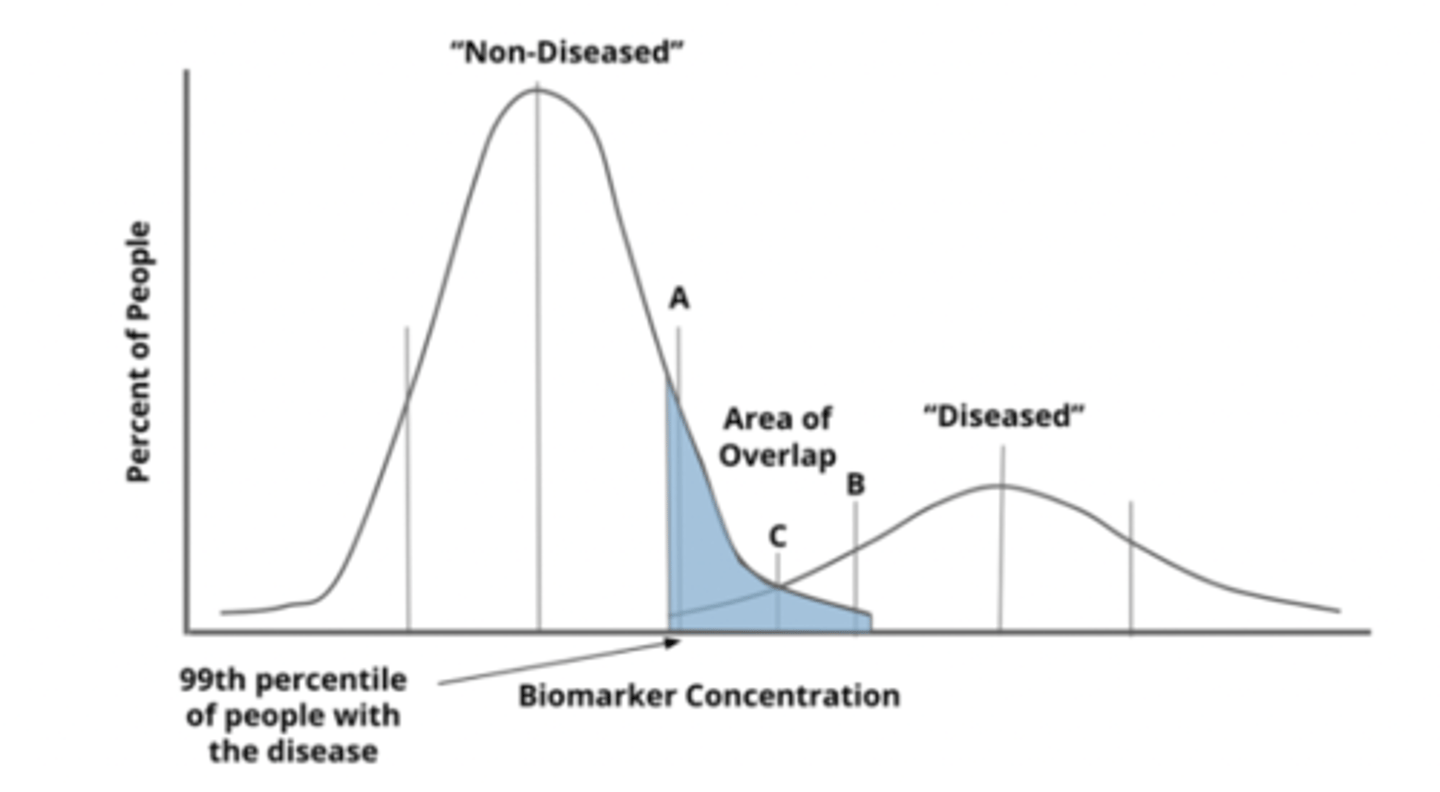
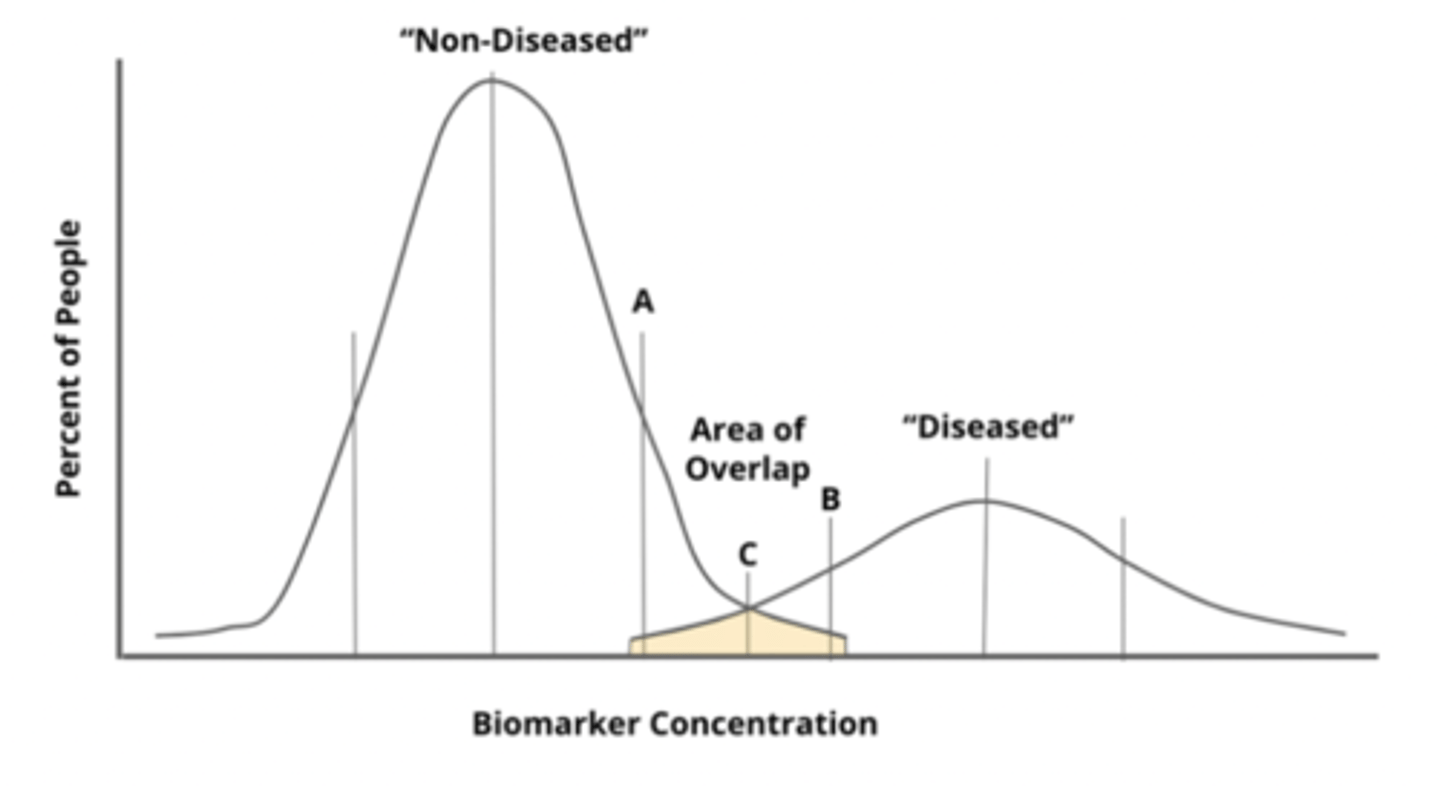
how can the range of those who have/don't have disease be represented?
population distribution
what scenarios are ideal vs realistic for population distributions? what does this mean?
ideal: large spread (no overlap) between 2 ranges
reality: there will be overlap, so must decide where in overlap you are going to place cut-off → based on what the test is used for
what would a high sensitivity cut-off be (on a population distribution)
- 99th percentile of diseased people (only 1% diseased missed = very low false neg = very high sensitivity)
- low specificity bc high false positives (ie. shaded region)
- good for an initial screening test to catch as many cases as possible, but must be followed up by confirmatory testing (to rule out false pos)
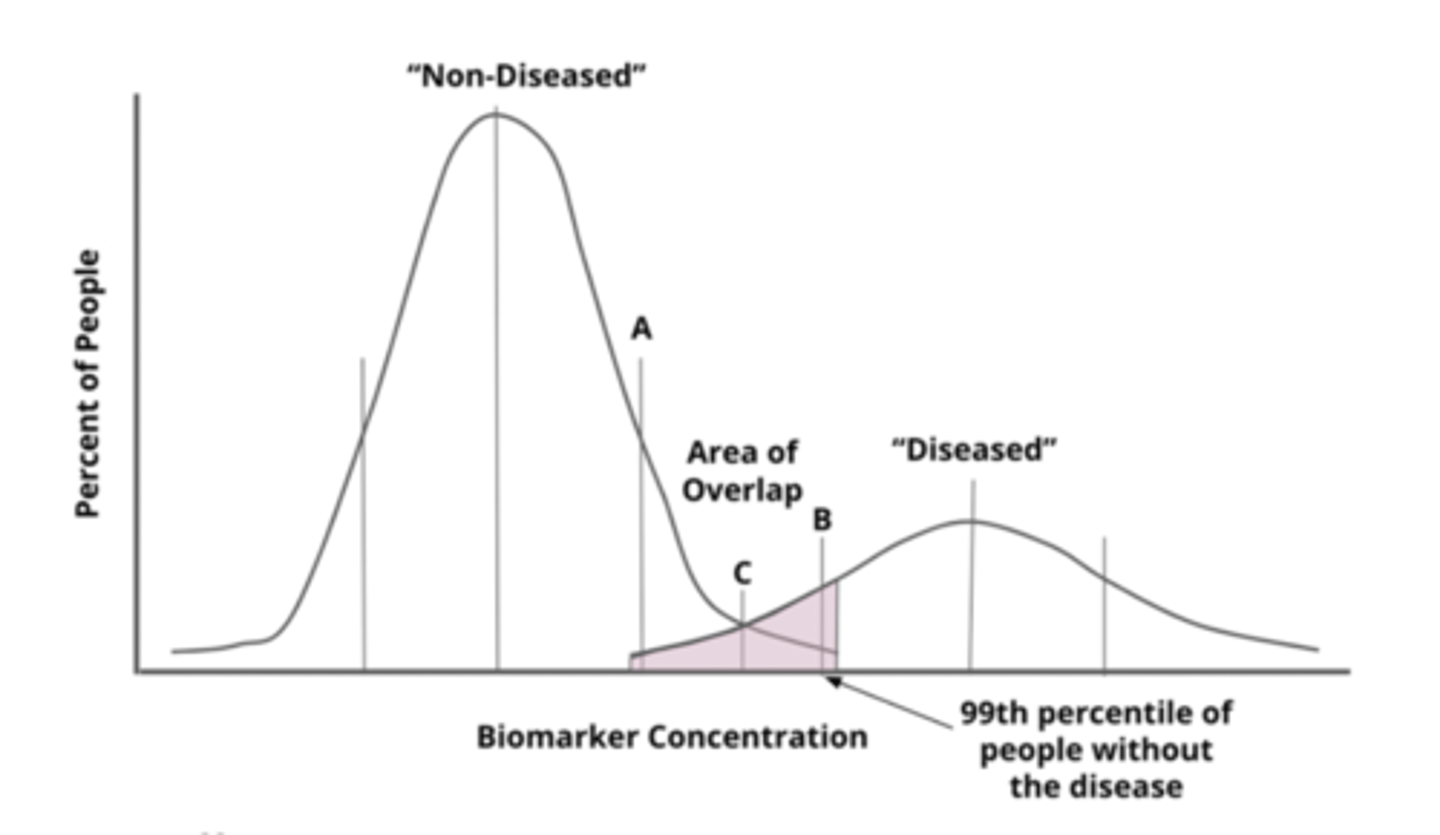
what would a high specificity cut-off be (on a population distribution)
99th percentile of healthy people (only 1% healthy people missed = very low false pos = high SPECificity))
- low sensitivity bc high false neg
- good for confirmatory testing but NOT for screening
what would balancing sensitivity/specificity look like on a population distribution? what does this mean? what would this test be good for?
- cut-off in middle of overlap = similar rates of false neg and false pos = compromise between catching the disease and false alarms
- used when balancing risks is appropriate
5 considerations when setting cut-off concentrations (influencing factors)
1. prevalence and severity of disease in population
2. factors impacted by biologial variation related to disease prevalence
3. environmental factors (temp, altitude) affecting screening/testing
4. dif in way test is performed (methodology)
5. other factors unique to the lab and its equipment
collaborative laboratory integrated reports (CLIR)
- extra risk assessment by comparing NBS results to large, adjusted reference and disease databases
- uses scoring scale and multivariate pattern recognition software to see how closely baby's analyte pattern matches disease profile
= refines cut-offs and reduces false positives
Uses of CLIR (5)
- analyze new screening results in real time
- identify which analytes/ratios are most useful for detecting a disorder
- reduce false positives
- compare results across different labs
- distinguish between similar disorders (better differential diagnosis)