character tables & vibrational spectroscopy
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
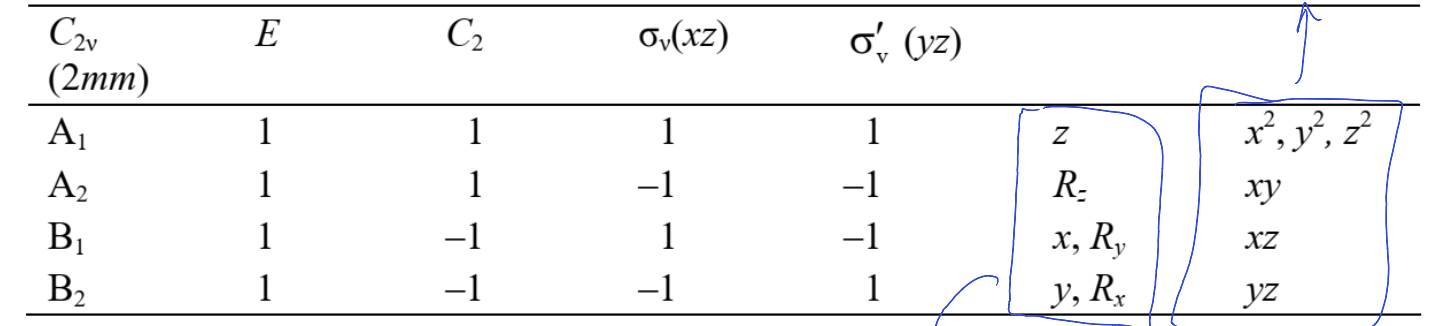
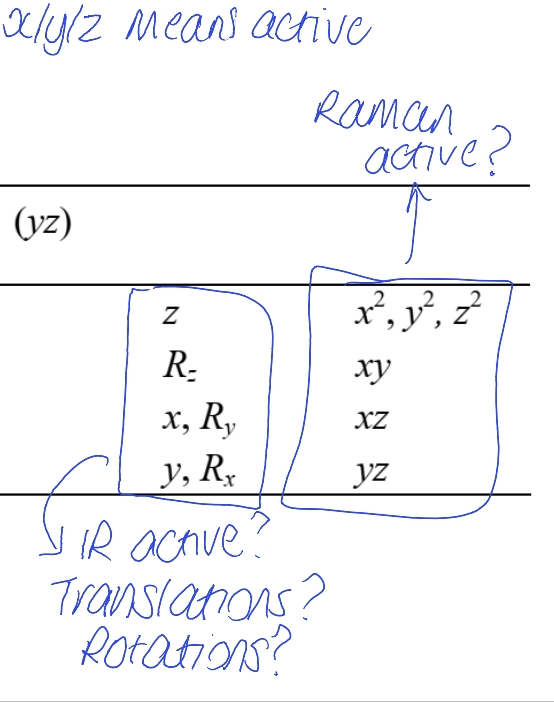
In a character table, what does the final two columns mean?
what does A mean?
a singly degenerate state which is symmetric with respect to rotation about the principal Cn axis
what does B mean?
a singly degenerate state which is antisymmetric with respect to rotation about the principal Cn axis
In Mulliken symbols - what does E mean?
what does T(F) mean?
a doubly degenerate state
triply degenerate - G,H etc continue this sequence of degeneracy
In Mulliken symbols - what is Xg?
symmetric with respect to inversion through the centre of the molecule
In Mulliken symbols - what is Xu?
antisymmetric with respect to inversion through the centre of the molecule
In Mulliken symbols - what is X1?
symmetric with respect to a vertical mirror plane perpendicular to the principal axis
In Mulliken symbols - what is X2?
anti symmetric with respect to a vertical mirror plane perpendicular to the principal axis
In Mulliken symbols - what is X’?
symmetric with respect to a horizontal mirror plane
In Mulliken symbols - what is X’’?
anti symmetric with respect to a horizontal mirror plane
how many degrees of freedom does a molecular system with N nuclei and n electrons have?
how can these be treated and therefore how many nuclear degrees of freedom are there?
3(N+n) degrees of freedom
can be treated separately due to Born Oppenheimer approx.
consider 3N nuclear degrees of freedom
N is number of atoms
in linear molecules, what are the degrees of freedom?
3 DOF are translation in x,y,z
2 DOF are rotational (rotating around central principal axis of molecule causes no change in energy)
3N-5 = vibrations
in non linear molecules, what are the degrees of freedom?
3 DOF are translation in x,y,z
3 DOF are rotational (rotating around x,y,z - all require energy)
3N-6 = vibrations
is antisymmetric higher/lower/the same freq as symmetric? why?
intensity?
higher
takes more energy to distort the molecule
higher intensity as distorts dipole moment a lot
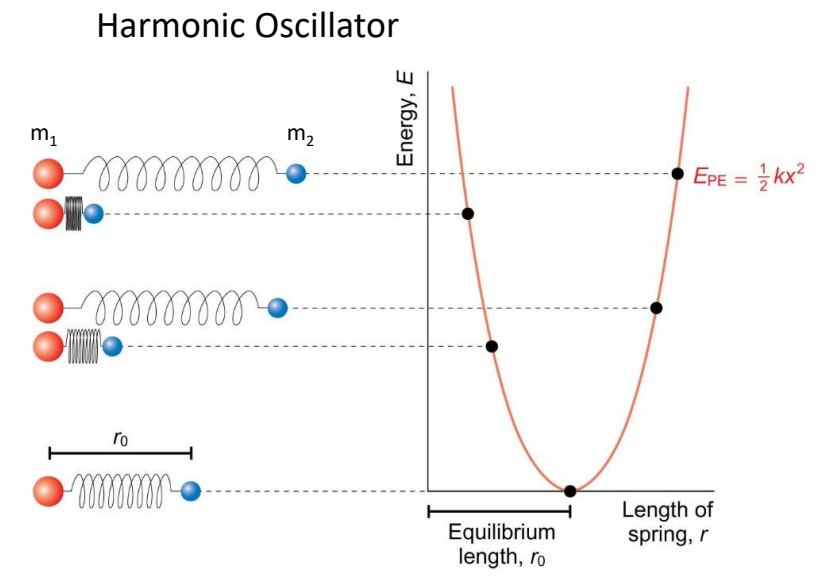
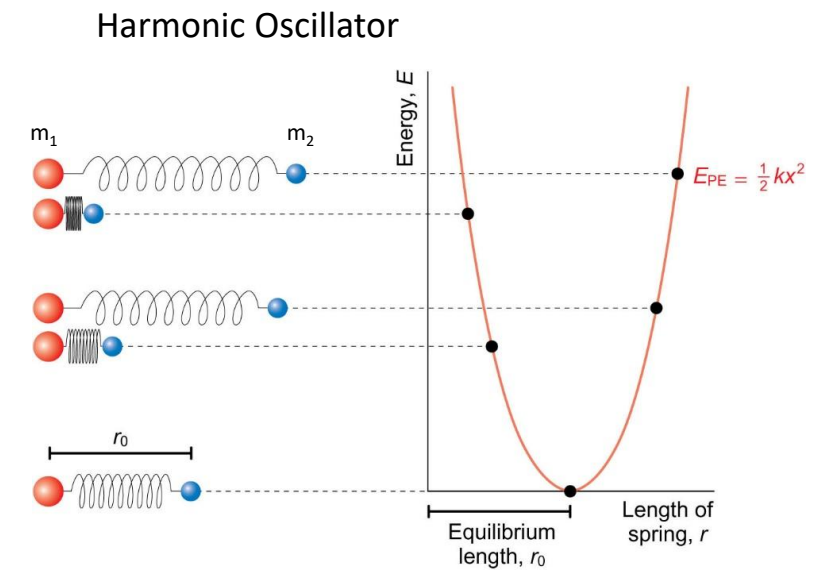
show harmonic oscillator graph
show different springs and their position on the graph
what is on axes?
what is the E equation?
in a harmonic oscillator, how is force related to potential energy (EPE)?
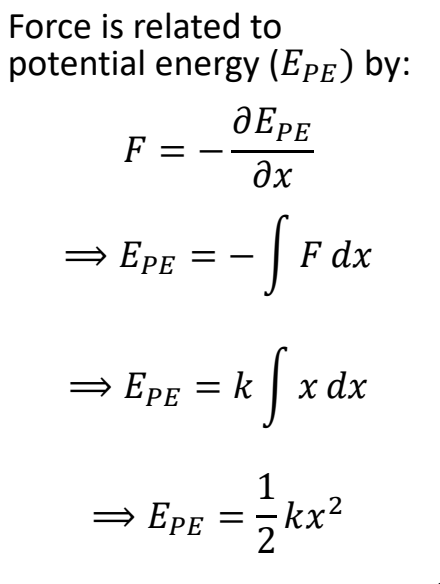
show potential energy vs displacement (x) graph and show how it changes with increasing kf
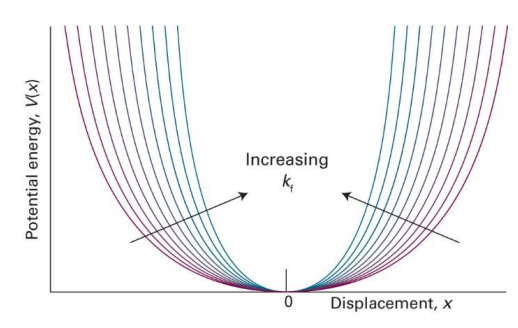
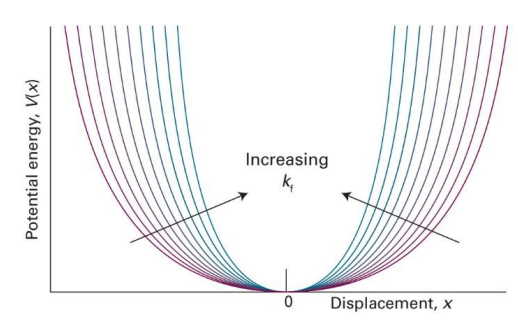
what is k in this graph? (equation)
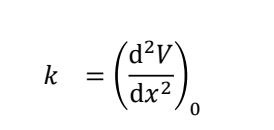
show rigid body rotor (rotational)
energy levels (what symbol?)
what are the gaps between ELs
what are the E values?
show energy vs percentage of radiation transmitted
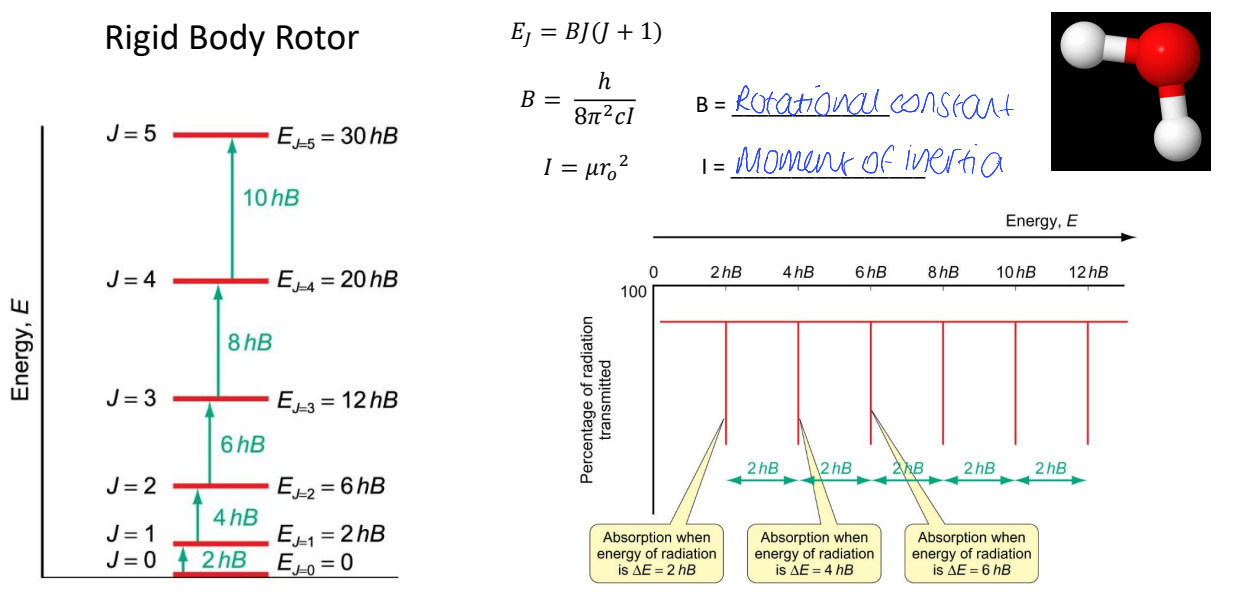
show radiation and then absorption
what is energy of photon (general)?
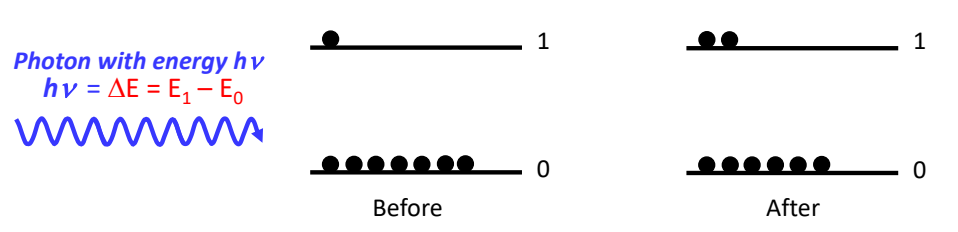
show emission
what is energy of photon?
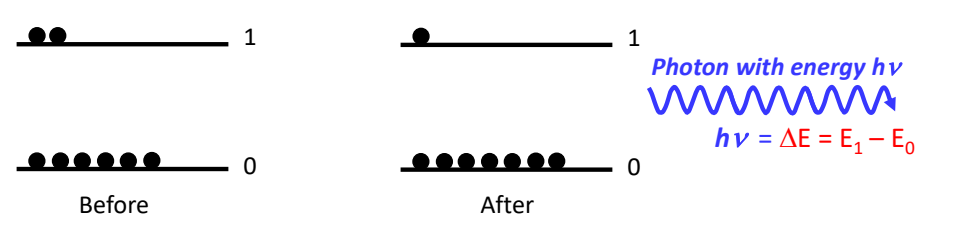
what is a key parameter to consider when thinking about absorption/emission?
excited state lifetime
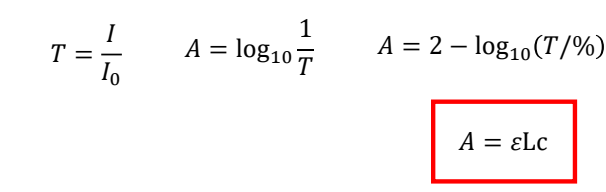
show transmission vs frequency (Beer-Lambert_Bouguer Law) graph
explain about width
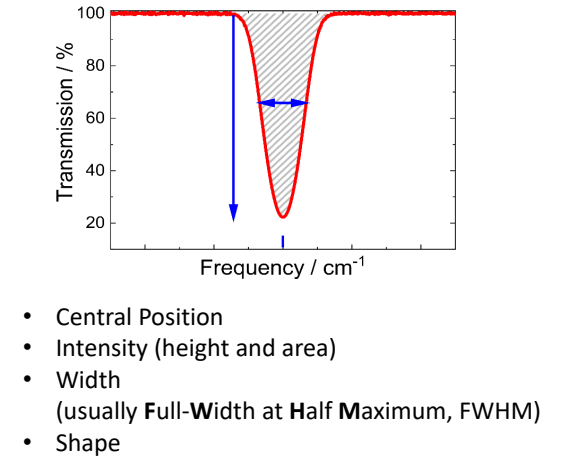
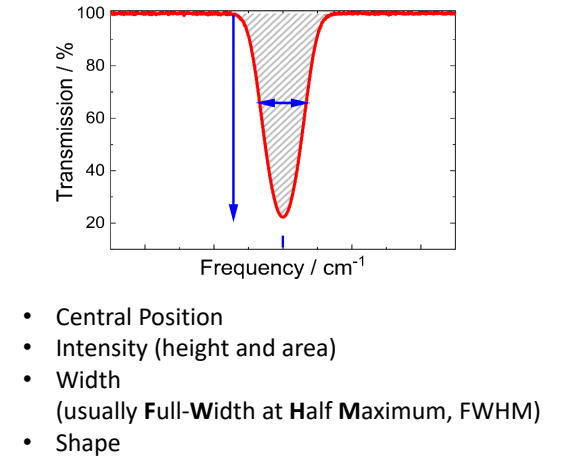
when does this apply?2
measurement of gas where reference is empty gas cell
measurement of KBr pellets
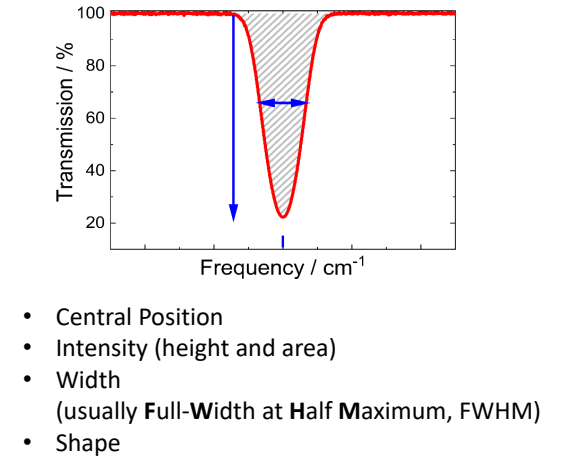
what is usually calculated?
integrated intensity = area under the peak
used rather than peak height
what is a gross selection rule? (general - how does this then apply to vibrational and rotational transitions?)
something about the molecule that decides if a transition is allowed
vibrational = must be a change in dipole during vibration
rotational = must be a permanent dipole
what is a specific selection rule (i.e. where does it come from)
how does this apply to Harmonic oscillator and to Diatomic Rigid Rotor
comes from quantum mechanical understanding of ELs and tells which transitions are allowed
harmonic oscillator - Δv = ±1
diatomic rigid rotor - ΔJ = ±1
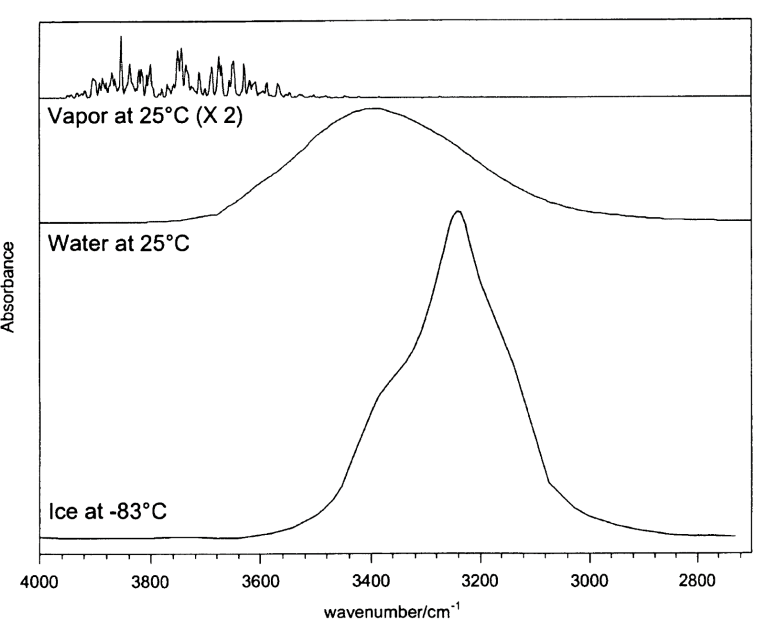
how does concentration change over these three plots?
why are the spectra different? 4
same amount of water so concentration is the same in each spectra
central frequencies move, number of modes change, peak widths change, change in spectral intensity

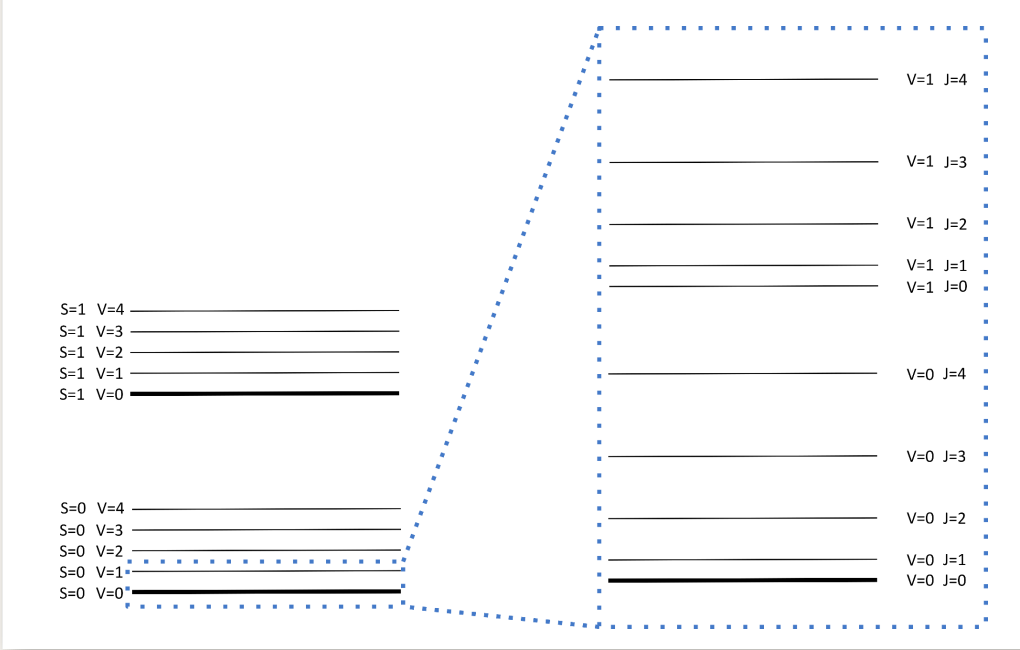
draw S=0 and S=1 energy levels
show up to V=4 for each
where are the J levels?
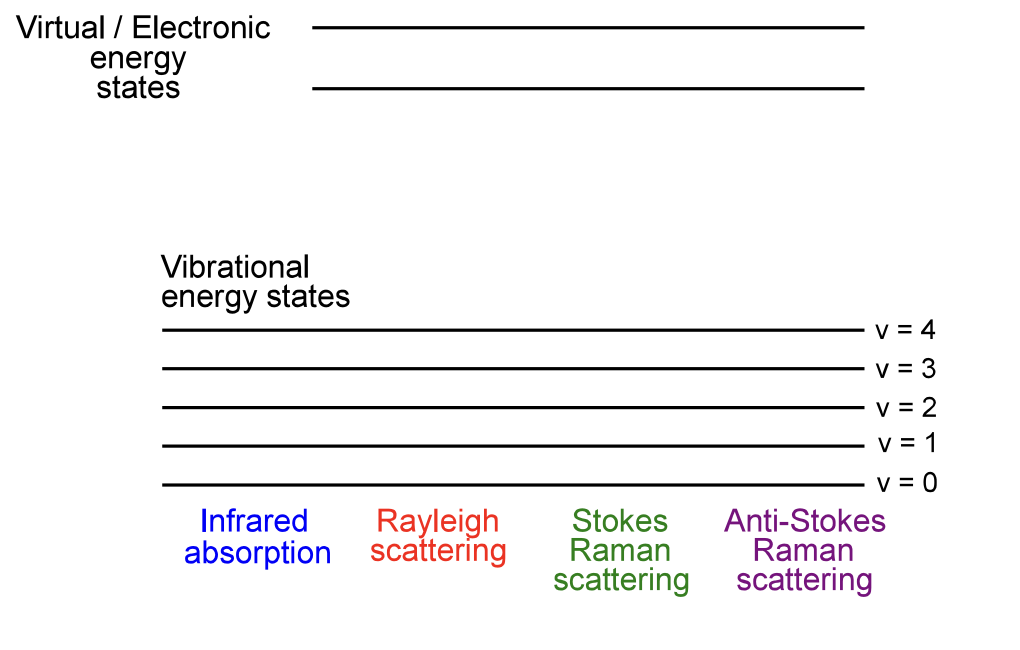
show these different transitions
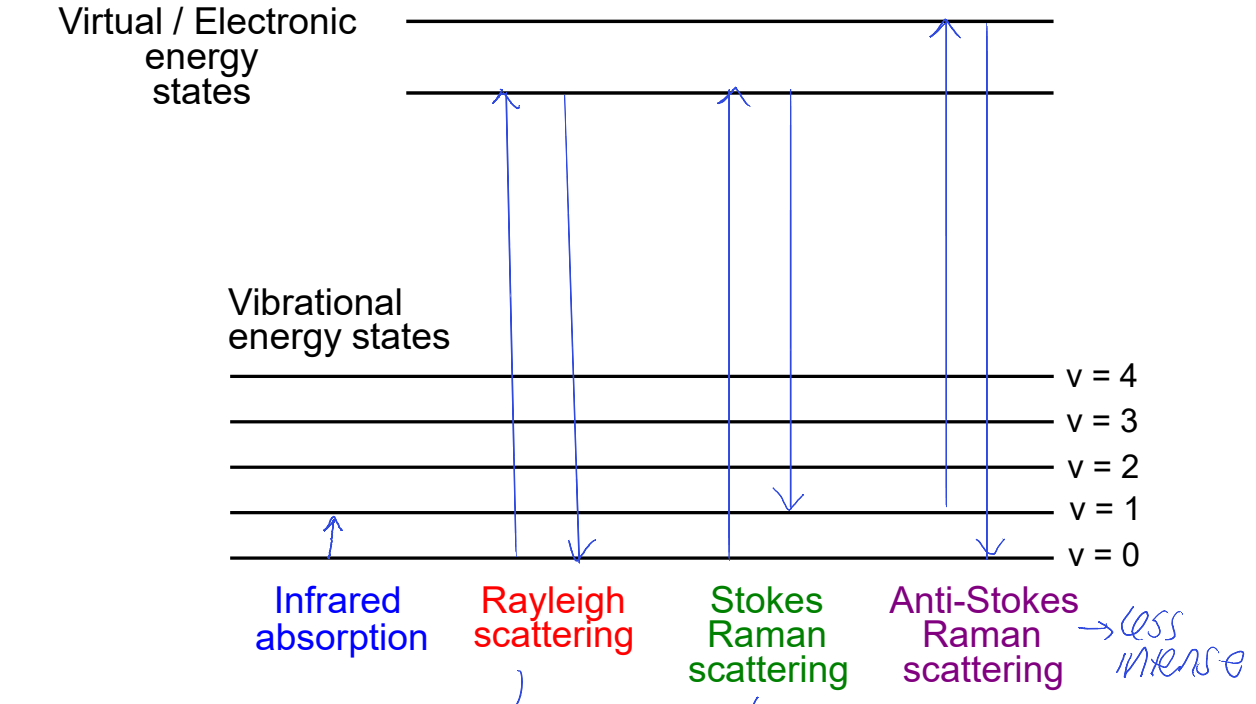
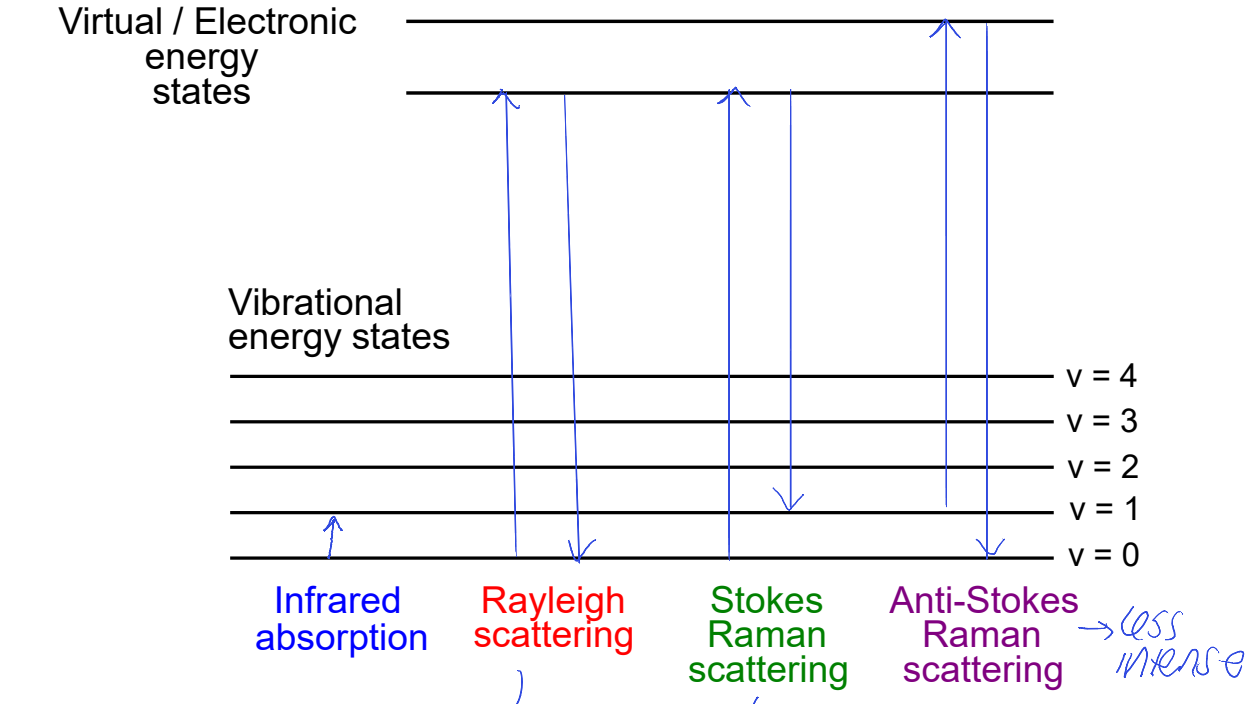
what is Rayleigh scattering? why does Stokes Raman scattering not come all the way back to ground state?
Rayleigh = elastic scattering, energy doesn’t change
Stokes Raman loses some E corresponding to vibration
what are the Raman gross selection rules? 2(vibrational and rotational)
vibrational = must be a change in polarisability during vibration
rotational = polarisability must be anisotropic (depends on orientation of molecule)
what are the Raman specific selection rules? 2
harmonic oscillator Δv=±1
diatomic rigid rotor ΔJ=±2
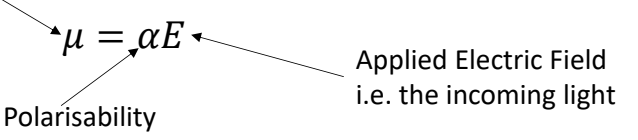
what is equation for induced dipole
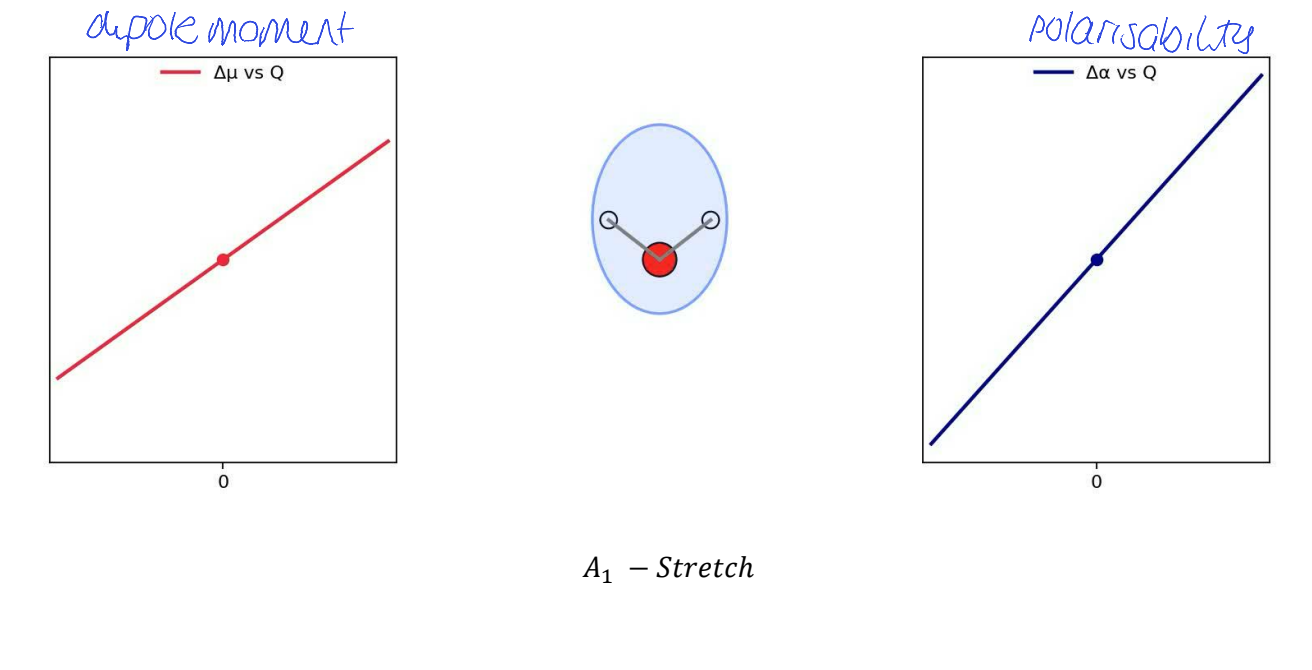
show Δμ vs Q (dipole moment) and Δɑ vs Q (polarisability) for A1 stretch
Q represents progress of stretch
show Δμ vs Q (dipole moment) and Δɑ vs Q (polarisability) for B2 stretch
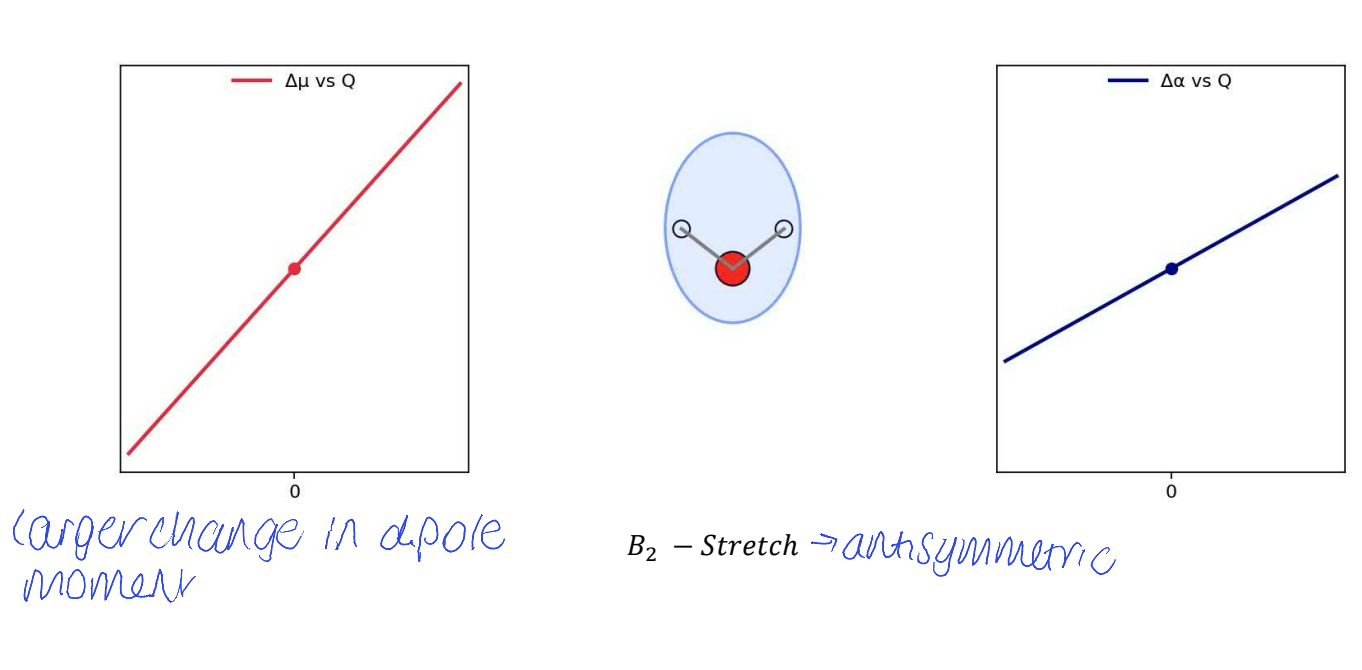
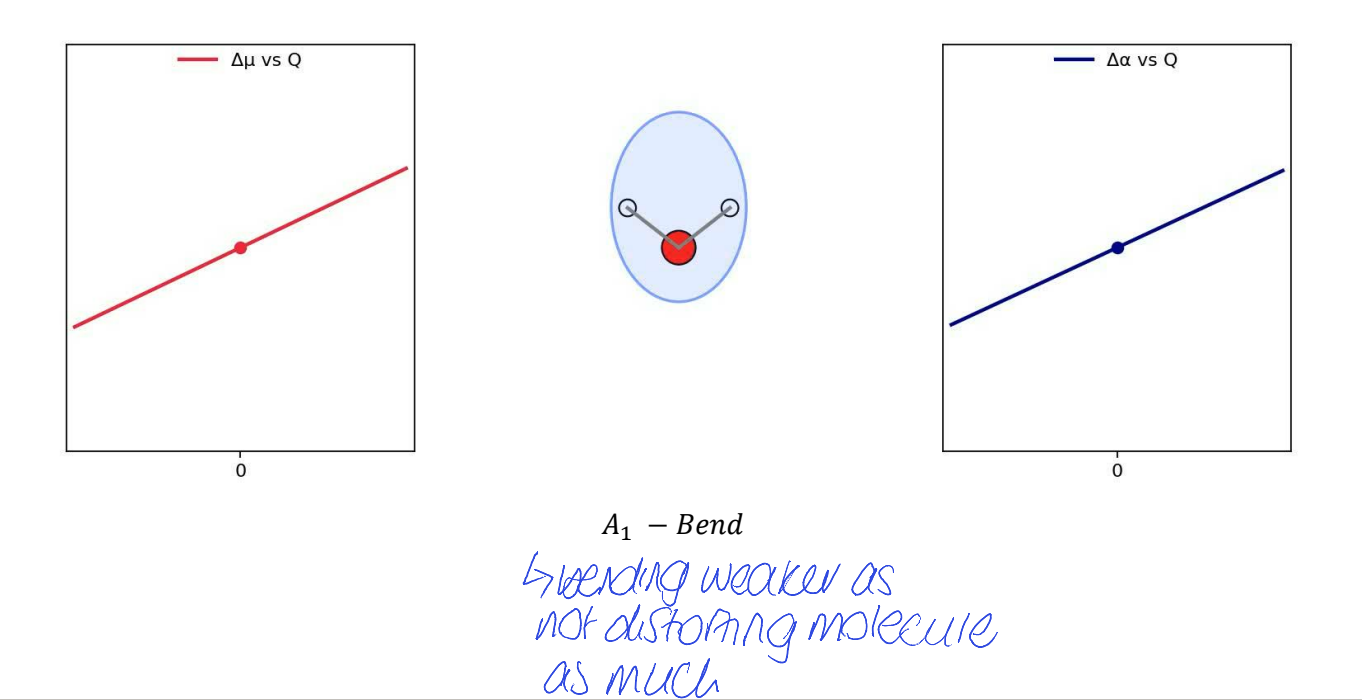
show Δμ vs Q (dipole moment) and Δɑ vs Q (polarisability) for A1 bend
for CO2, which has the biggest peak and why?
(asymmetric/symmetric, bend, stretch)
asymmetric stretch as largest change in dipole
for CO2, show Δμ vs Q (dipole moment) and Δɑ vs Q (polarisability) for asymmetric stretch
explain shape of polarisability graph
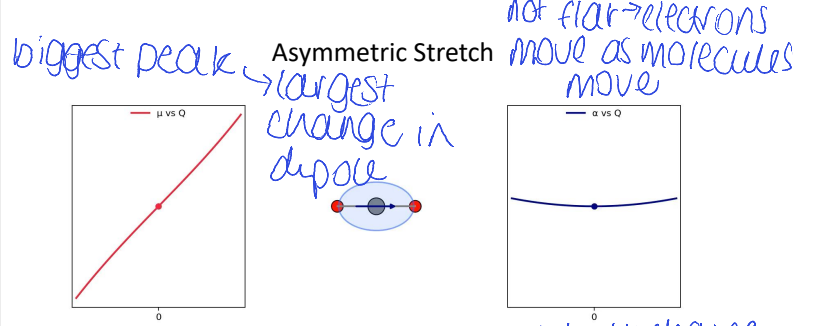
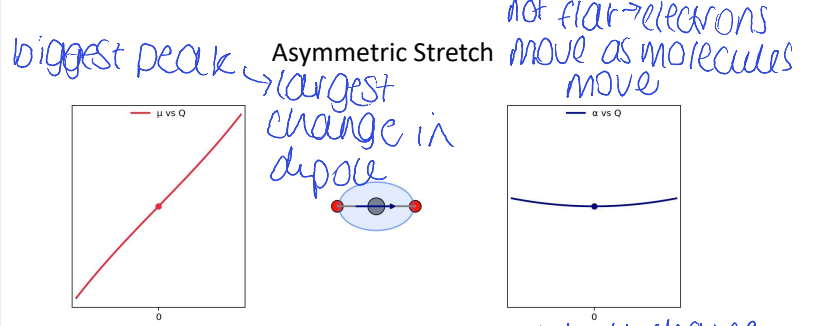
is CO2 Raman active?
no because the gradient change of polarisability is 0
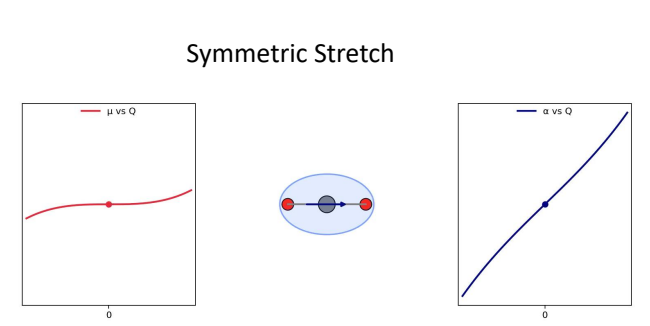
for CO2, show Δμ vs Q (dipole moment) and Δɑ vs Q (polarisability) for symmetric stretch
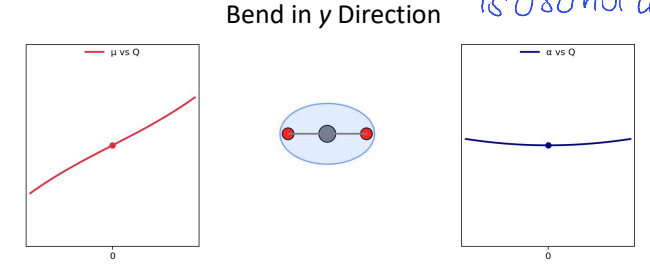
for CO2, show Δμ vs Q (dipole moment) and Δɑ vs Q (polarisability) for bend in y direction
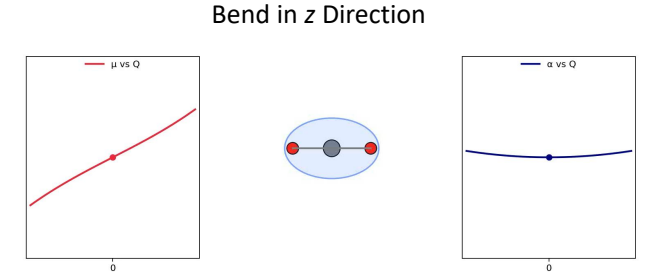
for CO2, show Δμ vs Q (dipole moment) and Δɑ vs Q (polarisability) for bend in z direction
for harmonic oscillator, how does equilibrium bond length change as more E is put in?
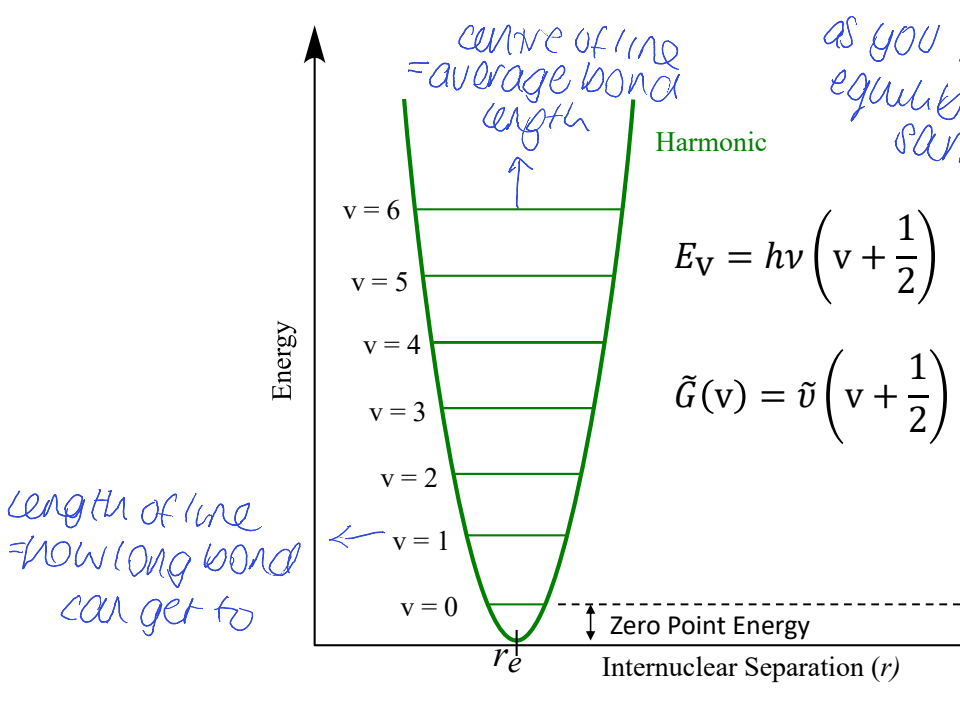
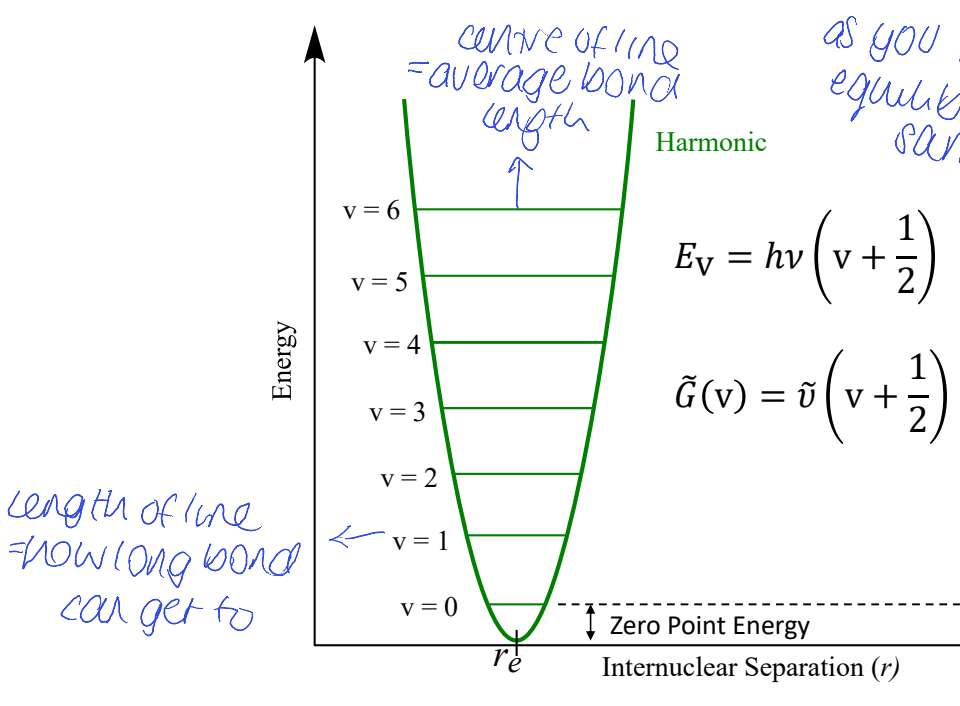
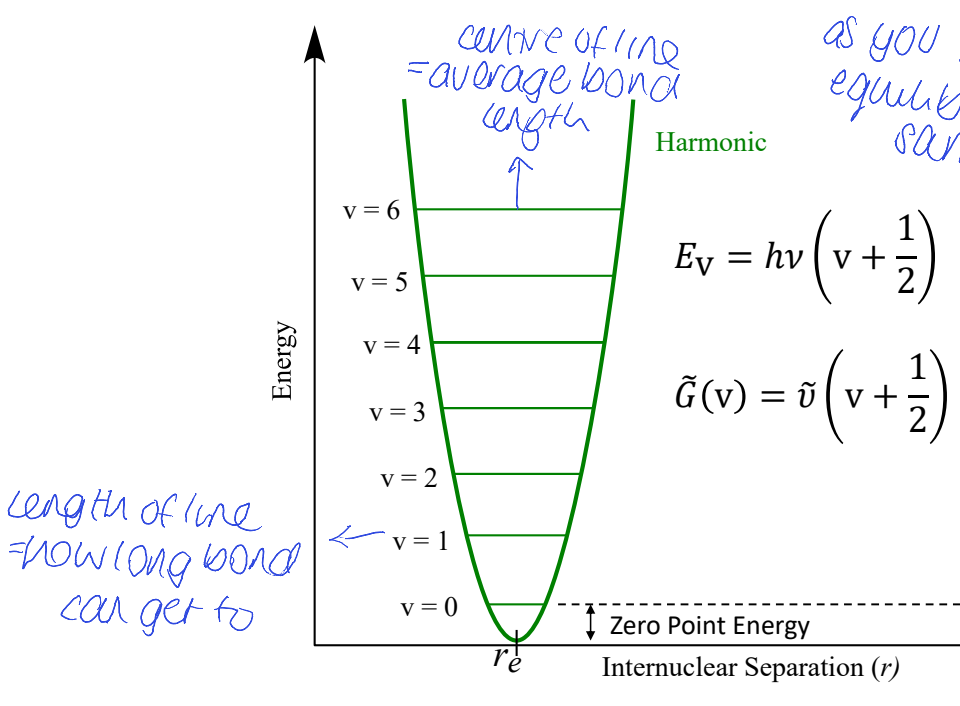
show graph of E vs internuclear separation
where is average bond length
what does length of each line mean
ZPE?
equilibrium bond length stays the same
what does ZPE show?
vibration E is never zero
still oscillates around central position at absolute zero
what is ZPE for Ev and G~(v)?
G~(v) is vibrational
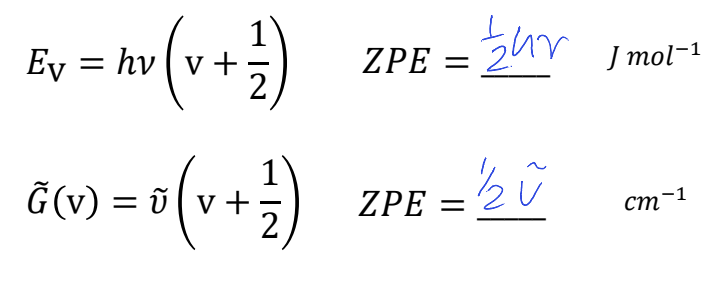
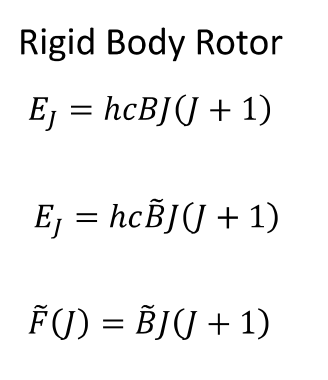
what is the difference between the first two equations?
what is F~(J)?
first has c in ms-1, second has it in cms-1
F~(J) is rotational
for harmonic oscillator - how are energy levels space? is there dissociation? vibrational coupling? how does it change w temp?
all ELs are evenly spaced
peak position doesn’t change with temperature
bonds are not allowed to break = no dissociation
all vibrations are independent meaning no vibrational coupling
show harmonic oscillator with morse potential - where is dissociation E?
where is De and D0?
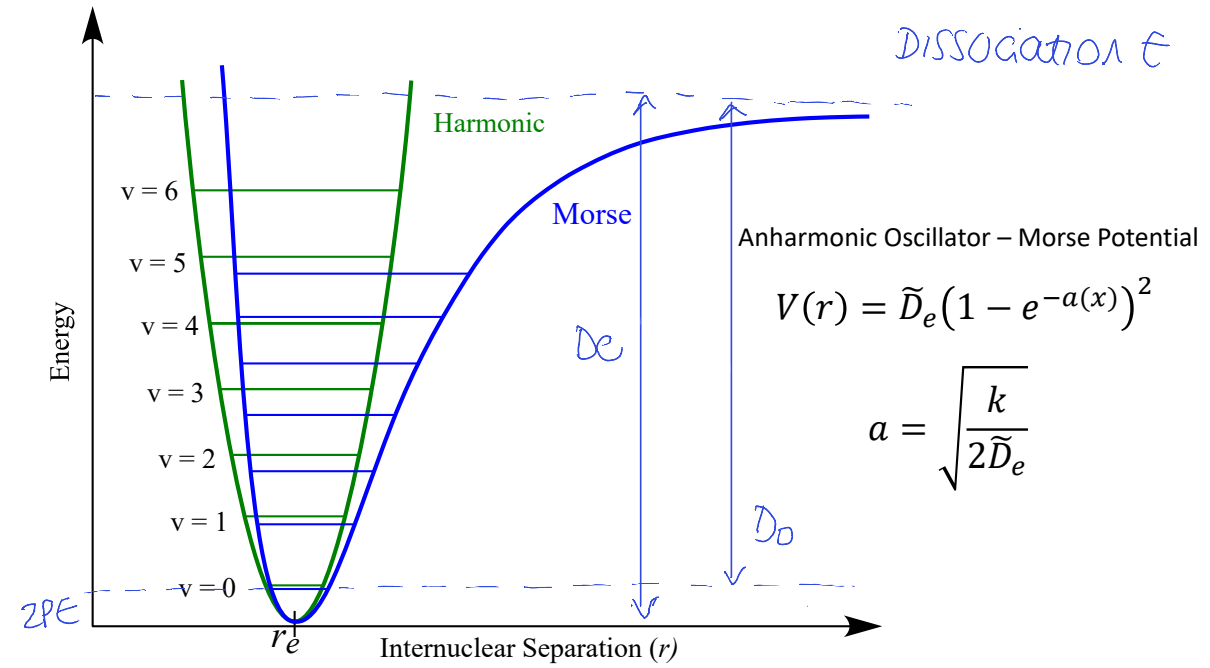
how does anharmonic oscillator differ from harmonic?5
energy levels are lower than corresponding harmonic levels
ELs converge
allows bond dissociation
peak position changes with temp
vibrations can couple
how do gases in the atmosphere heat the earth by the greenhouse effect?
photons are absorbed after bouncing off the earth and remitted back down and heat the earth
what is integrated intensity(area under peak) of a vibrational peak proportional to?
proportional to change in dipole moment/polarisability as a function of vibration
specific selection rules for harmonic vs anharmonic oscillator
Δv = ±1 (harmonic)
Δv = 0,±1,±2 …
why can nuclei and electrons be treated separately?
nuclei are an order of magnitude heavier than electrons
when a nucleus moves, the electrons move instantaneously
when is anharmonicity important?
bond dissociation
temp and pressure induced phase transitions
overtones/combination bands
fermi resonance
thermal expansion
TST
thermal conductivity
mode coupling