MB2 - oncogenes
1/103
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
104 Terms
What is the Knudson Two-Hit Hypothesis?
Hypothesis suggests that two mutations (or "hits") are required to inactivate tumor suppressor genes and lead to cancer development. The first hit is inherited, while the second hit occurs somatically.
Why is the first mutation in the Knudson Two-Hit Hypothesis typically inherited?
The first mutation is inherited because it occurs in a germline cell, meaning it is present in all cells of the body. This makes the person more susceptible to a second mutation in the same gene, which can then lead to cancer.
How does the Two-Hit Hypothesis relate to the development of retinoblastoma?
In retinoblastoma, children inherit one defective allele of the RB1 gene. The second "hit" (mutation) occurs later, in a retinal cell, causing the development of cancer. This explains why retinoblastoma often affects children, and why both hits are required for cancer development.
What is the role of proto-oncogenes in normal cells?
Proto-oncogenes normally stimulate cell growth, division, and help cells move through cell cycle checkpoints to ensure they are inspected and remain normal.
What happens when a proto-oncogene mutates?
When a proto-oncogene mutates, it becomes an oncogene, which no longer responds to normal cell cycle checkpoints. As a result, the cell continues dividing uncontrollably, potentially leading to cancer.
Why do oncogenes cause cancer?
Oncogenes cause cancer because they promote continuous cell division without the normal checks and balances. They override the usual signals that stop abnormal cells from dividing, allowing uncontrolled cell proliferation.
What is the Two-Hit Hypothesis in cancer development?
The Two-Hit Hypothesis states that two mutations in a tumor suppressor gene are required for cancer development. The first mutation is usually inherited (making the person a susceptible carrier), and the second mutation or loss of the second allele leads to loss of function and cancer
How does the first mutation in the Two-Hit Hypothesis contribute to cancer risk?
The first mutation in a tumor suppressor gene is usually inherited, making the individual a susceptible carrier who has one defective copy of the gene. This increases the risk of a second mutation in the other allele, which can cause cancer.
What happens when the second mutation or loss of a tumor suppressor gene occurs?
The second mutation or loss of the remaining normal allele leads to the inactivation of the tumor suppressor gene, which normally regulates cell growth and DNA repair. Without these functions, uncontrolled cell division occurs, leading to cancer.
What role do tumor suppressor genes play in regulating cell growth?
Tumor suppressor genes act to stop excessive cell growth, ensuring that cell division is controlled. They also play a role in DNA repair to fix errors in the genome, helping maintain genetic stability.
What happens when a proto-oncogene becomes an oncogene?
When a proto-oncogene mutates, it becomes an oncogene. This active oncogene no longer responds to normal growth control signals, leading to uncontrolled cell division. It essentially removes the "brakes" on cell growth, promoting cancer.
Why are DNA repair genes important in preventing cancer?
DNA repair genes are crucial because they fix errors that occur during DNA replication. If these genes are mutated or dysfunctional, errors accumulate in the genome, contributing to genomic instability and increasing the risk of cancer.
What is a proto-oncogene?
A proto-oncogene is a group of normal genes that regulate cell growth and division. When mutated, they can cause normal cells to become cancerous.
ARE MUTATIONS IN PROTO ONCOGENES DOMINANT OR RECESSIVE?
How do mutations in proto-oncogenes typically affect their function?
Mutations in proto-oncogenes are typically dominant, meaning only one mutated copy is needed to cause the gene to become an oncogene, leading to abnormal cell growth.
What are the different ways a proto-oncogene can become an oncogene?
A proto-oncogene can become an oncogene through:
Increased activity (mutation)
Gene amplification (increased gene levels)
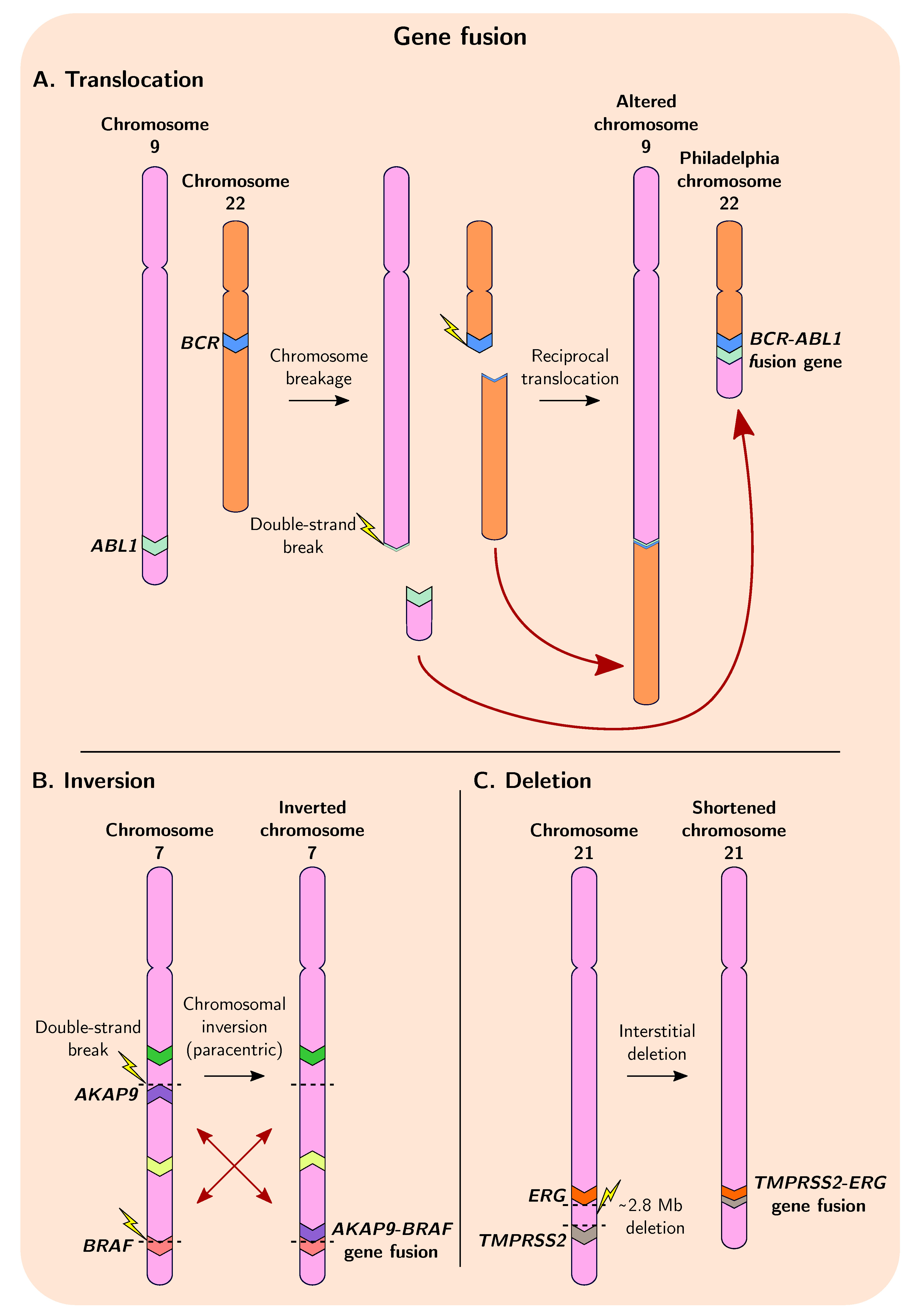
Chromosomal translocation (loss of regulation, gene fusion)
What is the consequence of an oncogene on cell behavior?
An oncogene causes increased activity of the gene, which can result in uncontrolled cell growth, contributing to cancer development.
How does gene amplification affect proto-oncogenes?
Gene amplification results in increased levels of the proto-oncogene product, leading to overactivity and disruption of normal cell growth regulation.
How does chromosomal translocation contribute to cancer?
Chromosomal translocation involves the fusion of a proto-oncogene with another gene, leading to loss of regulation and abnormal cell division.
fusion example BCR -ABL (a segment of abl gene on chromosome 9 breaks off and attaches to bcr on chromosome 22)
at chromosomal level its a swap (two chromosomes exchange segments with each other, both chromosomes are altered).
at the gene level its a fusion, as a result of the swap, created a hybrid gene.
How many oncogenes have been identified so far?
Over 100 oncogenes have been identified, with Ras genes accounting for about 25% of them.
What is the relationship between oncogenes and tumor suppressor genes?
Oncogenes promote uncontrolled cell growth, while tumor suppressor genes normally work to stop cell division and repair DNA. Loss of regulation in tumor suppressor genes can also contribute to cancer development.
Why is it important to study proto-oncogenes and oncogenes in cancer research?
: Understanding proto-oncogenes and oncogenes is crucial because their mutations or overactivity drive uncontrolled cell growth in cancer, making them potential targets for therapies.
What factors control the rate of cell division?
The rate of cell division is controlled by external cues, such as signals from other cells, growth factors, and environmental conditions.
What happens when the mechanisms that regulate cell division lose control?
When the mechanisms that regulate cell division lose control, it can lead to cancer, as cells begin to divide uncontrollably.
How does the frequency of cell division vary across different cell types?
The frequency of cell division varies by cell type. Some cells divide frequently (e.g., skin cells), while others divide rarely or only when needed (e.g., liver cells).
What is the cell cycle time like in embryo cells?
Embryo cells have a very fast cell cycle that is less than 20 minutes, allowing rapid cell division during early development.
How often do skin cells divide throughout life?
Skin cells divide frequently, with a cell cycle time of 12-24 hours to replace lost or damaged cells.
How do liver cells regulate their division?
Liver cells retain the ability to divide but do so very infrequently, usually only once every year or two as needed for tissue repair or regeneration.
How is cancer typically described in terms of cell type?
Cancer is typically specific to an individual cell type, and the description of the cancer correlates with the nature of the involved cell type.
Can oncogenic drivers vary across tissues?
Yes, oncogenic drivers can be tissue-specific, meaning that the mutations or genes driving cancer can differ depending on the tissue type involved.
What did Rous discover about cancer and cell-free filtrates?
Rous used cell-free filtrates to show that chicken sarcomas could be transmitted, demonstrating the role of viral oncogenes in cancer.
What is the v-Src gene and what does it do?
the v-Src gene is an oncogene encoded by the RNA "sarcoma" virus. It mimics the host cell's nonreceptor tyrosine kinase Src, leading to uncontrolled activity and promoting cancer.
meaning virus delivers the oncogene into the host cell. when the virus is inside it makes active tyrosine kinase that always signals for cell growth, leading to uncontrolled proliferation and formation of sarcoma.
What is the only human oncogenic retrovirus known?
The only human oncogenic retrovirus is the Human T-cell lymphotropic virus (HTLV).
What are the three major domains of Src?
Src has three major domains:
SH2 (Src Homology 2)
SH3 (Src Homology 3)
Kinase domain (responsible for its kinase activity)
src is a tyrosine kinase protein, normal job is to regulate cell signalling and growth. a proto oncogene.
How does Src switch from inactive to active?
Src switches from inactive to active through the control of its phosphorylation state, which is regulated by phosphorylation at specific tyrosine residues.
What role does CSK (c-Src kinase) play in Src activity?
CSK (c-Src kinase) regulates the phosphorylation of Src, which controls its transition between active and inactive states.
What is the role of auto-phosphorylation in activating Src?
Auto-phosphorylation at Tyr416 (Y416) displaces the P-Tyr416 from a binding pocket, opening Src into its active form.
: How does Y527 phosphorylation affect Src activity?
Y527 phosphorylation folds Src into a closed, inaccessible bundle, preventing its activation.
not activated, not on, off
y627 is tyrosine at position 527 in protein. normally y527 phosphorylation causes inhibition of src, as src is in closed inactive conformation. and when 7527 is absent, inhibit mechanism is lost, vsrc is constit active, proliferation.
What happens when Y527 dephosphorylation occurs in Src?
Dephosphorylation of Y527 leads to Src being in an active state, allowing it to bind with platelet-derived growth factor (PDGF) and focal adhesion kinase (FAK), further promoting its activation.
phosphorylation of src, src in inactive state, y527 present, brake is on, inhibition, no proliferation.
dephosphorylated, src is active, brake is off, proliferation.
What is the relationship between Rous sarcoma virus and avian leucosis virus?
the Rous sarcoma virus evolved from a less harmful virus called avian leucosis virus (which infects birds but doesn't usually cause much trouble).
At some point, this relatively harmless virus picked up the src gene and turned into the cancer-causing Rous sarcoma virus.
What the virus makes:
The virus produces a protein called pp60v-src
"pp60" just means it's a phosphoprotein (has phosphates attached) that weighs 60 kilodaltons (kDa) - that's just the size of the protein
The "v-src" part means it's the viral version of Src
The key problem: This viral Src protein has unregulated activity - meaning it doesn't have the normal OFF switch. Remember how normal Src gets turned off by phosphorylation at Y527? The viral version doesn't respond to those controls. It's constantly active, always pushing cells to grow and divide.
Bottom line: A mostly harmless bird virus stole the src gene, the gene lost its brakes, and now the virus makes a constantly-active growth signal that causes tumors. That's how it went from "benign" to "cancer-causing."
What happened when Src was knocked out in mice?
Knockout of Src produced viable mice, indicating that Src is not essential for survival in mice, but may still be important for certain cellular functions.
What are some members of the kinase family that are related to Src?
The kinase family related to Src includes SRC, LCK, LYN, BLK, HCK, FYN, FGR, YES, and YRK (YRK is found in birds).
What are some Src inhibitors used in cancer treatment?
Some Src inhibitors include dasatinib, saracatinib, and bosutinib.
What percentage of the human genome consists of viral genes?
About 8% of the human genome (around 100,000 genes) consists of viral genes.
What role might viral genes play in the human genome?
These viral genes may help boost host immunity, block viral infections, and help recognize specific invaders, mounting immune responses against entire classes of viruses.
How do retroviruses integrate into the human genome?
When a retrovirus infects a cell, it converts its RNA into DNA, which can then integrate into the host chromosome. This integration can occur in sperm or egg cells, making the virus part of the human genome, and leading to inter-generational transmission.
What is HPV and how common is it?
HPV (Human Papillomavirus) is a non-enveloped, double-stranded, circular DNA virus from the Papillomaviridae family. It is very common, with nearly everyone being infected with one type at some point in their lives.
What are the risks associated with HPV infection?
HPV infection is linked to epithelial lesions and cancers on cutaneous and mucosal surfaces, with major risk factors including:
Sexual activity
Age of first sexual intercourse
Number of sexual partners
Oral contraceptives (over 5 years)
Betel nut chewing, smoking, radiation, and UV light exposure
HPV associated cancers= cervical cancer, vaginal, anal cancer
How does HPV affect the basal stem cells of the skin or mucosa?
HPV infects the basal stem cells of skin and mucosal epithelium, where the viral DNA initially exists as an episome. It then integrates into the host genome at sites prone to chromosomal breakages.
What happens during early HPV infection?
Normal cell cycle: Cells go through phases of growth and division, but they also need to rest. The G0 phase is like a "resting" or "retired" state where cells stop dividing and just do their normal job. Most of your cells are actually in G0 most of the time - they're not constantly dividing.
What HPV does: When HPV (human papillomavirus) infects cells early on, it blocks cells from entering this resting phase. It's like forcing workers to stay on the assembly line and never take a break.
The result:
Dysregulation of the cell cycle = the normal controls that tell cells when to divide and when to rest get messed up
Cells keep cycling through growth and division phases over and over
Neoplastic transformation = the cells start transforming into abnormal, precancerous, or cancerous cells ("neoplastic" just means tumor-like)
Why this matters: By preventing cells from resting, HPV keeps them constantly dividing. This increases the chance of mutations accumulating and gives the virus more opportunities to hijack the cell machinery. Over time, this uncontrolled division can lead to cancer (like cervical cancer, which is caused by certain high-risk HPV strains).
How does HPV affect the cell cycle and cell growth?
E5 activates growth signals (PDGFR) that tell cells to divide.
E6 blocks p53, which is a protein that normally stops damaged cells from dividing or forces them to die.
E7 blocks pRb, another safety checkpoint that prevents cells from entering the division cycle - together these proteins force constant cell division while removing the brakes, leading to cancer.
What is the role of E2 in HPV infection?
E2 regulates the transition from early-phase to late-phase genes in HPV, increasing virion production in HPV-driven lesions, typically manifesting as tissue hypertrophy.
What is the 9-valent HPV vaccine and what does it prevent?
The 9-valent HPV vaccine targets subtypes 6, 11, 16, 18, 31, 33, 45, 52, and 58. It prevents several diseases, including anal cancer, genital warts, cervical intraepithelial neoplasia, vulvar intraepithelial neoplasia, and anal intraepithelial neoplasia
when is the HPV vaccine most effective?
The HPV vaccine is most effective when administered before sexual activity begins, typically at ages 9-12.
Why is the HPV vaccine important in cancer prevention?
he HPV vaccine helps prevent infection with high-risk HPV types that can cause cervical cancer, genital warts, and other cancers, significantly reducing the risk of these diseases.
What are proto-oncogenes?
Proto-oncogenes are normal genes that, when mutated, can cause normal cells to become cancerous by promoting uncontrolled cell growth.
How do proto-oncogenes become oncogenes?
Proto-oncogenes become oncogenes when they undergo mutations that activate them, leading to excessive cell division, inhibition of differentiation, or blockage of cell death pathways.
What proteins do oncogenes encode, and what are their effects on the cell?
Oncogenes encode proteins that:
Stimulate cell division
Inhibit cell differentiation
Block cell death pathways, promoting survival of abnormal cells.
Can oncogenes be endogenous or exogenous?
yes, oncogenes can be endogenous (originating within the body) or exogenous (originating from external sources, such as viruses).
Why are oncogenes (but not proto-oncogenes) a major target for anti-cancer therapies?
Oncogenes are a major target for anti-cancer therapies because they are mutated or overactive in cancer cells, promoting uncontrolled growth, while proto-oncogenes are normal genes that only become problematic when mutated into oncogenes.
How do oncogenes typically differ from tumor suppressor genes?
Oncogenes are typically considered activators of cell growth, while tumor suppressor genes are inhibitors that regulate cell growth and prevent cancer. Both are often required in cancer development.
What are some examples of viruses that can facilitate the development of cancer by activating oncogenes?
Examples of viruses that can facilitate cancer development include:
Human papillomavirus (HPV) → cervical cancer, oropharyngeal squamous cell carcinomas (cancer of squamous epithelial cells lining mouth and throat)
Hepatitis B and C viruses → hepatocellular carcinoma (cancer of cells of liver)
Epstein-Barr virus (EBV) → Burkitt’s lymphoma (cancer of b lymphocytes), gastric, and nasopharyngeal carcinomas
Human T-cell leukemia virus (HTLV) → adult T-cell leukemia/lymphoma
How does HPV contribute to cancer?
HPV can cause oropharyngeal squamous cell carcinomas by encoding oncogenes that disrupt normal cell cycle regulation, promoting uncontrolled cell growth and survival.
What cancers are associated with Epstein-Barr virus (EBV)?
Epstein-Barr virus (EBV) is associated with Burkitt’s lymphoma, as well as gastric and nasopharyngeal carcinomas.
What is a chromosomal translocation?
A chromosomal translocation is when parts of two chromosomes break off and switch places. This can mess up the normal genes and cause cancer.
Why is location important in chromosomal translocations?
The location of the genes on chromosomes matters because if a gene moves to a different spot, it might become overactive or behave abnormally, which can cause cancer.
What is the common translocation in Burkitt lymphoma?
In Burkitt lymphoma, 70% of patients have a translocation t(8;14), where the c-MYC gene on chromosome 8 moves next to the IGH gene on chromosome 14. This causes the MYC gene to become overactive, leading to cancer.
How does the t(2;5) translocation affect patients with anaplastic large-cell lymphoma (ALCL)?
In ALCL, 60% of patients have a t(2;5) translocation, which moves the ALK gene on chromosome 2 to a new position. This causes the gene to become active and can lead to cancer.
What translocation is common in follicular lymphoma?
In follicular lymphoma, 70-95% of patients have the translocation t(14;18), where the BCL-2 gene on chromosome 18 moves next to the IGH gene on chromosome 14. This causes the BCL-2 protein to be overproduced, helping cancer cells avoid normal cell death.
Why is the c-MYC gene important in Burkitt lymphoma?
The c-MYC gene promotes cell growth. When it’s near the IGH gene because of a translocation, it becomes overactive, leading to uncontrolled growth and cancer.
How does the BCL-2 protein affect cancer cells in follicular lymphoma?
The BCL-2 protein helps cancer cells survive by stopping them from dying when they should. In follicular lymphoma, the BCL-2 gene becomes overactive due to a translocation, allowing the cancer cells to live longer and grow uncontrollably.
What is the mechanism in carcinogenesis where a normal gene regulating cell growth is abnormally replicated?
The mechanism in carcinogenesis involves the abnormal replication of a normal gene, which can lead to the gene becoming an oncogene. This causes uncontrolled cell growth, contributing to cancer development.
carcinogenesis= process by which normal cells become cancer cells
What role do proto-oncogenes play in normal cell growth, and how are they involved in cancer?
Proto-oncogenes regulate normal cell growth and division. However, when mutated or altered, they become oncogenes, which promote uncontrolled cell growth and cancer development.
What happens when a proto-oncogene becomes an oncogene in terms of protein production?
When a proto-oncogene becomes an oncogene, it often leads to the overproduction of a protein, such as a growth-driving kinase or receptor, which can promote excessive cell division.
How do oncogenes contribute to cancer by affecting proteins?
: Oncogenes can lead to the overproduction of proteins that either activate growth pathways or cause excessive amounts of protein to be produced, driving uncontrolled cell division and contributing to cancer.
What is one example of an oncogene, and how does it contribute to cancer?
An example of an oncogene is MYC, which regulates the cell cycle. When mutated, MYC becomes overactive and promotes continuous cell division, a key factor in cancer.
What is HER2/neu, and how does it lead to cancer?
: HER2/neu is a gene that encodes a receptor involved in cell growth. Overexpression of HER2/neu is linked to certain cancers, such as breast cancer, by causing uncontrolled cell division through an overactive receptor.
her2 = oncogene. a receptor tyrosine kinase. When HER2 becomes an oncogene through gene amplification, resulting in overexpression of the HER2 receptor protein on the surface of the cell, driving uncontrolled growth signalling
How can point mutations lead to the activation of an oncogene?
Point mutations in a proto-oncogene can change its DNA sequence, producing an abnormal protein or causing the gene to produce an excessive amount of protein, promoting cancer.
: How does gene amplification contribute to cancer?
: Gene amplification increases the number of copies of a proto-oncogene, leading to overproduction of its protein, which drives uncontrolled cell division and contributes to cancer.
What is a fusion gene, and how does it contribute to cancer?
A fusion gene occurs when two genes fuse together, creating a novel protein. This new protein can have abnormal activity or be produced in excessive amounts, contributing to cancer.
How does a fusion gene differ from other mutations that cause cancer?
Unlike point mutations or amplifications, a fusion gene creates a novel protein A HYBRID by combining parts of two different genes, often leading to abnormal function and excessive activity that drives cancer.
How does a proto-oncogene become an oncogene through mutation?
: A proto-oncogene becomes an oncogene through mutations like point mutations, translocations, gene amplifications, or fusion genes, leading to the production of abnormal or excessive proteins that drive cancer.
What is the significance of oncogene activation in the development of cancer?
Oncogene activation results in the overproduction of growth-promoting proteins or abnormal proteins, causing uncontrolled cell division and contributing to the formation of tumors and cancer.
How do cell proliferation pathways work in cancer?
cell proliferation pathways can independently drive cell division. They often work together, coordinating and affecting each other to promote uncontrolled growth in cancer.
How does activation of a proto-oncogene affect cell growth?
Activation of a proto-oncogene often involves kinase activation and changes in the access of growth-regulating proteins to the nucleus, driving uncontrolled cell division.
What are SNVs (single nucleotide variations), and how do they contribute to cancer?
SNVs are small changes in the DNA sequence, and they are the most common mutations in cancer. These changes can activate genes and lead to uncontrolled cell growth.
SNV is just 1 single base change in the DNA sequence:
normal= ATGCAT
mutated= ACGCAT
How do point mutations affect kinases like KRAS or EGFR in cancer?
Point mutations (e.g., C>T or G>T changes) in genes like KRAS or EGFR can activate kinases, leading to overactive signaling pathways that drive tumor growth in cancer.
a point mutation is a more broader term and can include a snv, but the single nucleotide variation is explicitly when there is a nucelotide change
What is the difference between somatic mutations and germline mutations?
Somatic mutations occur in specific body cells during a person’s lifetime and are acquired,and are not inherited. Germline mutations are inherited and are present in every cell of the body.
How do somatic mutations drive cancer growth?
Somatic mutations occur during a person’s lifetime in specific cells, and they can drive tumor growth by altering important genes in those cells, contributing to cancer.
How does adenocarcinoma develop from benign polyps?
Adenocarcinoma can develop when benign polyps in the colon grow too quickly (called hyperproliferation) and eventually become malignant (cancerous).
adeno= gland or glandular tissue. carcinoma= cancer of epithelial cells. so adenocarcinoma can ocur in any part of body that has glandular epithelium. like, colon, lung, breast, stomach, prostate.
=cancer in epithelial lining.
What is severe dysplasia, and how does it relate to cancer development?
Severe dysplasia is when cells in a polyp become abnormally shaped. at this stage it is pre-cancerous stage, not yet cancer.
can eventually lead to adenocarcinoma (cancer) if the cells continue to grow uncontrollably, and when cells have broken through the basement membrane - this is malignancy
What is intrinsic resistance in cancer?
Intrinsic resistance is when existing mutations in cancer cells already make them resistant to certain treatments, making it harder to treat the cancer.
What is acquired resistance in cancer?
Acquired resistance occurs when cancer cells develop new mutations over time that allow them to resist treatment that initially worked
How do point mutations in EGFR lead to cancer?
Point mutations in the EGFR gene cause the receptor to become constantly active, leading to uncontrolled signaling and cell growth.
How does Src become constitutively active in cancer?
Deletion of C-terminal tyrosine in Src leads to constant activation of its kinase activity, promoting uncontrolled cell growth and cancer
How do kinase signaling pathways contribute to cancer?
Kinase signaling pathways become constantly activated in many cancers, often due to mutations in genes like EGFR or Ras, driving uncontrolled cell division.
What happens when oncogenes are activated by gain-of-function mutations?
Gain-of-function mutations in oncogenes cause constant activation of growth receptors (like EGFR) or signaling molecules (like Ras), leading to uncontrolled cell growth
gain of function, dominant, only 1 mutated copy is sufficient. e.g. bcr-abl, egfr,

Why is no single cancer therapy 100% effective?
No single cancer therapy is 100% effective because cancer cells can develop resistance over time, and the disease is complex and varied.
Why are combination therapies more effective in treating cancer?
Combination therapies are more effective because they target multiple pathways of cancer growth, reducing the chances of resistance and improving the overall chance of success.
What is v-Src and how does it cause cancer?
v-Src is an oncogene carried by the Rous sarcoma virus (an RNA virus). It's a viral copy of the normal cellular gene c-Src. Unlike the tightly regulated c-Src, v-Src is constitutively active (always "on"), causing constant signaling for cell growth and division, leading to uncontrolled proliferation and cancer
What is a tyrosine kinase and what role does it play in the Src story?
A tyrosine kinase is an enzyme that adds phosphate groups to proteins at tyrosine amino acids - a key way cells send signals to control growth and division. Src is a nonreceptor tyrosine kinase. When the viral version (v-Src) becomes constitutively active, it continuously phosphorylates target proteins, driving uncontrolled cell growth