Lecture 23 - Therapy for prostate cancer
1/56
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
What is the prostate and where is it located?
Accessory male sex organ
Secretes fluid which is a major constituent of the ejaculatory fluid
Located below the bladder
What are the three zones of the prostate?
Transition
Surrounds urethra
Central
Surrounds ejaculatory ducts
Peripheral
Encompasses transmission and central zones
Which zone do most prostate cancers arise from?
Most prostate cancers arise in the peripheral zone
What is the role of androgens for the prostate/prostate cancer?
Development and growth are dependent on androgens
Often prostate cancers are dependent on androgens for their growth
What is the lifetime risk of prostate cancer in men?
~12%
What is the lifetime risk of death by prostate cancer in men?
~2.5%
What are examples of established risk factors of prostate cancer?
Age
Ethnic origin
Country of residence
Family history
Diet
How does age play a large role in prostate cancer?
Disease of elderly men
50% of men diagnosed are over the age of 70
Generally, very slow growing cancers
10-15 years
Where is the highest incidence rate of prostate cancer globally?
Highest incidence rate, standardised to population, occurs in Western Europe and North America
Are most prostate cancers hereditary or sporadic?
Sporadic
What is a common cause of early-onset prostate cancer?
I.e. in younger men
Hereditary factors for prostate cancer due to polymorphisms/mutations
What are common polymorphism targets in hereditary prostate cancers?
Androgen receptor (AR)
Vitamin D receptor
5-alpha reductase
BRCA1/2
What are examples of protective dietary factors against prostate cancer?
Soya beans (phytoestrogens)
Vitamin E
Green tea
Lycopenes
Selenium (NO?)
What are examples of negative dietary factors for prostate cancer?
Red meats
Obesity
How can the gut microbiota impact prostate cancer risk?
Butyrate-generating Ruminococcaceae over-represented in patients with high tumour burden and aggressiveness
Butyrate increases development and differentiation of intestinal regulatory T-cells, migrate to tumour site and cause local immunosuppression
How can the gut microbiota lessen the impact of androgen deprivation therapy?
Therapy tries to remove androgens from the body
However, it has been shown that bacteria in the gut are capable of using steroids and turning them into androgens
This can then help keep the cancer growing by supplying androgens
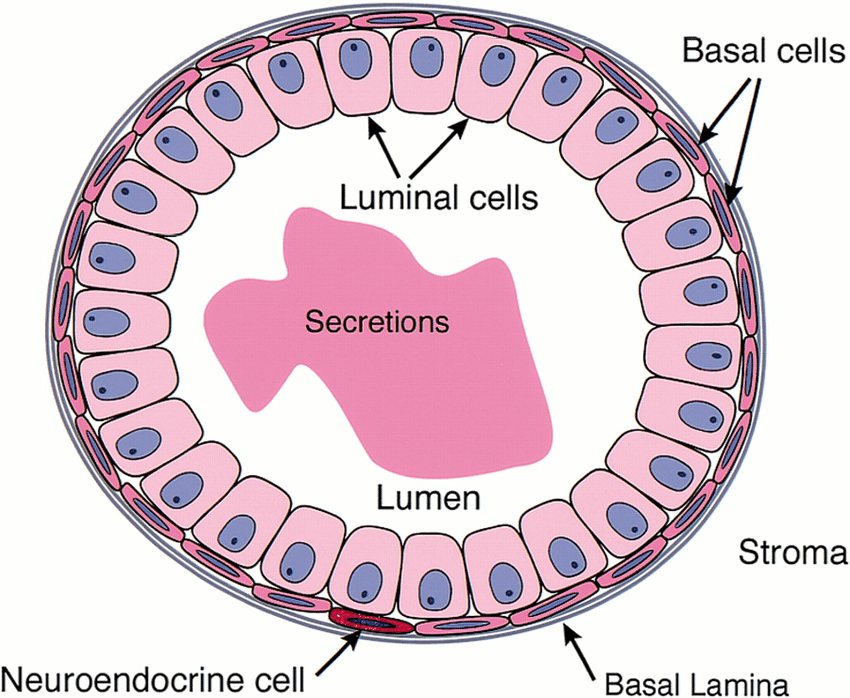
What is the structure of the prostatic duct?
Basal cells and luminal cells surround the prostatic ducts
Basal lamina membrane surrounds the cells
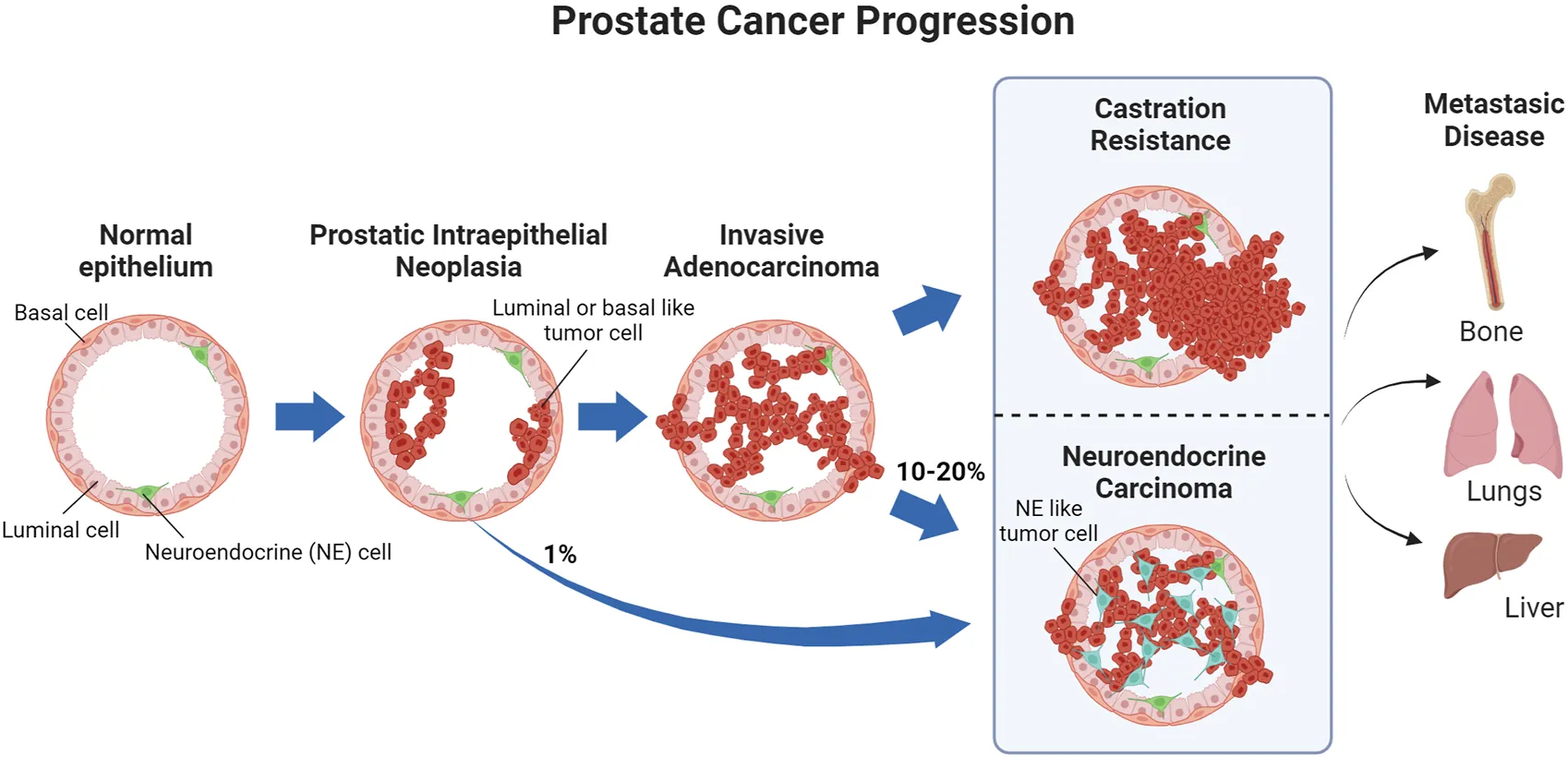
What is PIN in the context of prostate cancer?
Prostatic intraepithelial neoplasia
Proliferation of cancerous prostate cells while still being contained by the basement membrane
Not yet invasive
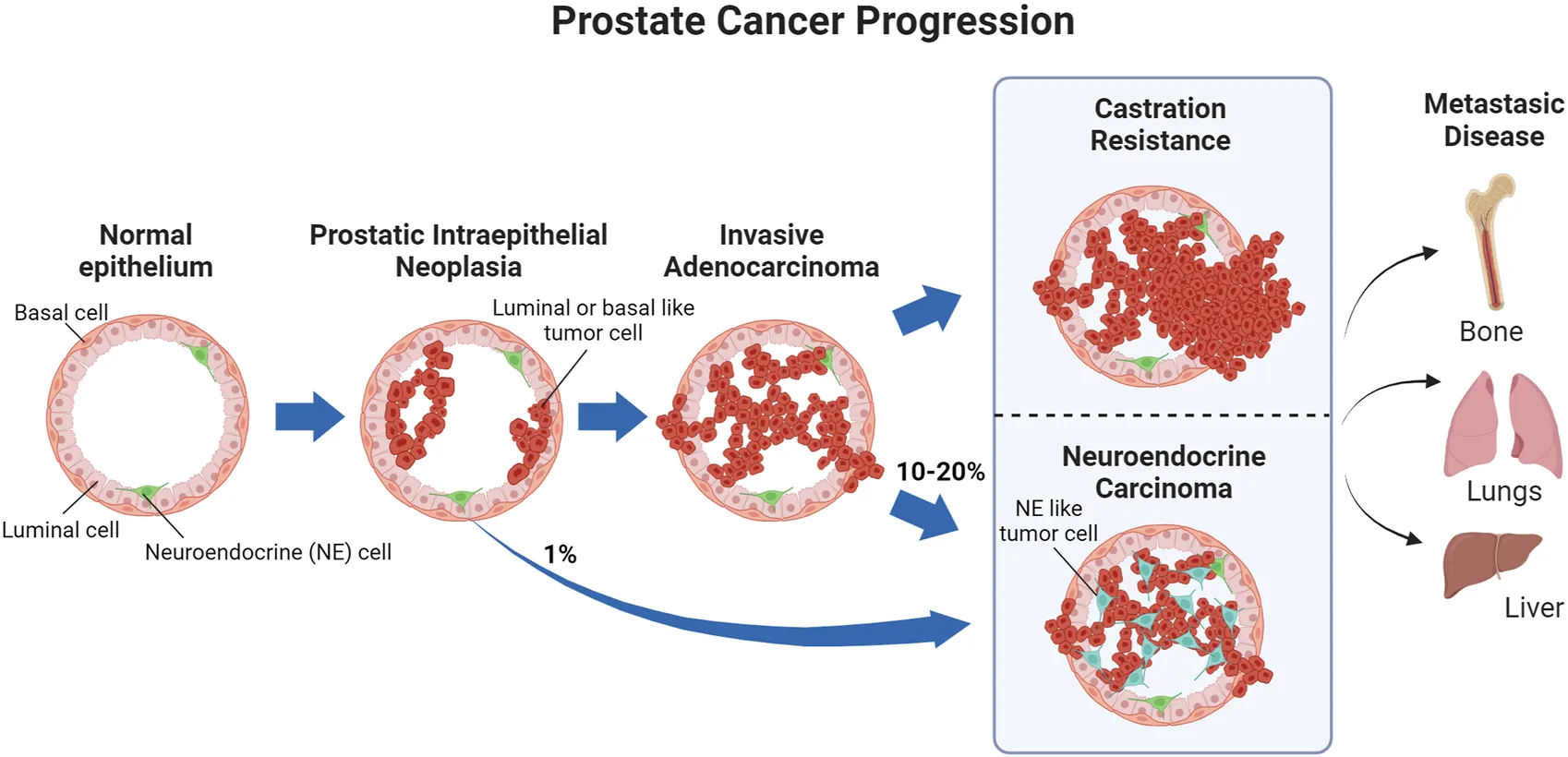
What occurs following PIN development?
As the tumour progresses, the basement membrane begins to break down and the proliferating cells are able to break out the duct
Can metastasise to the lymph nodes before eventually metastasising to the bones
What is the Gleason Grading System?
Five distinct patterns of decreasing tissue differentiation
Gleason score is calculated by summing the two most common grades seen under the microscope
E.g. most common cell grade is a 4 but some grade 3 cells are also seen
4+3 = Gleason score of 7
Does the order of the grades added to create the Gleason score matter?
Example: 3+4 and 4+3 both equal a score of 7
YES
In the case of a Gleason score of 7, the order of the scores matters
A 3+4 is generally less-aggressive whereas a 4+3 is more aggressive
What does a high Gleason score suggest?
High score/poorly differentiated tumours have worst prognosis and increased prostate cancer-specific mortality
What is PSA?
Prostate Specific Antigen
Androgen-regulated serine protease which is produced by luminal cells in the prostate
Why is PSA a useful prostate cancer biomarker?
PSA is prostate but not prostate cancer, specific
Disruption of the basal cell layer/basement membrane enables PSA to enter peripheral circulation where it’s level can be measured
Allows detection, staging and monitoring of prostate cancer
What are other causes of high PSA other than prostate cancer?
Benign Prostatic Hyperplasia (BPH)
Prostatitis
Urinary retention
Sexual activity
In a normal prostate gland, what role does androgen have?
Blood brings androgens to the prostate
Androgens act on the androgen receptors in stromal cells
This then causes release of survival factors and growth factors
These have paracrine effects on basal cells and luminal cells
In a prostate cancer, what role does androgen have?
The basement membrane breaks down
Androgen no longer acts on only stromal cells
Androgen can move across into the epithelium and activate luminal cells directly resulting in promotion of proliferation
What does the hypothalamus produce to initiate androgen production?
Hypothalamus produces Gonadotropin-releasing hormone (GnRH) which acts on pituitary cells
What does GnRH act on and what does it cause?
GnRH acts on pituitary cells
The pituitary cells then secrete luteinizing hormone (LH) which travels into the blood
LH then acts on the testes
Specifically, the leydig cells
What is the role of Leydig cells and what can act on them?
The pituitary cells then secrete LH which travels into the blood
LH then acts on the testes
Specifically, the leydig cells
Leydig cells will produce testosterone in response to LH
What androgens are produced by the adrenal glands?
Dehydroepiandrosterone and androstenedione
OR DHEA and A4 for simplicity
In the blood, most androgen is found in what form?
Most androgen are bound to serum proteins
E.g. sex hormone binding globulin (SHBG)
5-alpha reductase is found where in the cell?
5-alpha reductase is a membrane bound protein which converts testosterone to dihydrotestosterone (DHT)
What is the role of 5-alpha reductase?
5-alpha reductase is a membrane bound protein which converts testosterone to dihydrotestosterone (DHT)
Once testosterone is converted to dihydrotestosterone (DHT), what happens?
DHT can the bind to androgen receptor in the cytoplasm
When DHT binds, the receptor releases and undergoes conformational changes
Receptor then localises to nucleus
Receptor binds to androgen-regulated genes and turns on expression
When no androgen ligand is present, how is the androgen receptor found?
When no ligand is present, the receptor is held in a complex with other proteins
Which chromosome is the androgen receptor found on?
X chromosome
What 4 domains make up the androgen receptor gene?
Transactivation domain
Involved in transcriptional activation
DNA binding domain
Involved in DNA binding
Hinge domain
Ligand binding domain
Involved in binding androgen ligand
What are examples of genes commonly mutated in early prostate cancer?
SPOP
ERG oncogene fusion with androgen-regulated genes
What is the role of SPOP and what does its mutation cause in prostate cancer?
Normally involved in ubiquination of certain proteins such as the androgen receptor
When it’s mutated in prostate cancer, it can impair its ability to degrade the androgen receptor as well as other oncoproteins
What is the role of ERG and what does its fusion cause in prostate cancer?
Normally a transcription factor critical in stem cell self-renewal and vascular development
When it fuses with androgen-regulated genes, it can be over expressed
Plays a role in activating oncogenic signalling, promoting cell invasion and promoting epithelial-to-mesenchymal transition (EMT)
What are examples of mutations commonly seen later in prostate cancer development?
TP53
MYC
Androgen receptor
What is Castrate resistance disease?
Refractory to castration therapy from outset
Cancer acquires resistance to castration therapy and AR antagonist therapy
What are common aberrations in androgen receptor pathway which allows cancers to gain castrate resistance?
Many have amplification of androgen receptor
~20% have alteration of ligand binding site
Some have mutations in proteins involved in androgen receptor signalling rather than the receptor itself
Knockout of co-repressors of the androgen receptor
What is androgen receptor amplification?
Excessive expression of the androgen receptor which can make the cancer cell hypersensitive to androgen ligands.
How can mutations in the androgen receptor cause increased activation of the receptor?
Promiscuous pathway
Mutations in the binding domain can allow it to be activated by numerous other steroids alongside androgens
How can the androgen receptor be activated independent of steroids?
Outlaw pathway
Can allow for activation of other signalling pathways (e.g. growth factor receptor) which causes activation of androgen receptor
PI3K and MAPK can phosphorylate the androgen receptor and keep it activated
No androgen ligand binding needed
Why is castration a common intervention for prostate cancer?
Castrate men to take away the main source of androgens
In the past, surgical castration was performed
More recently, chemical castration is performed
Can block the production of androgens by the testes
How do anti-androgens work?
Can bind to the androgen receptor binding domain which competitively inhibits and blocks activation of the androgen receptor
What is Abiraterone?
Hormone therapy used to treat high-risk metastatic prostate cancer
Binds to the CYP17 enzyme and blocks activity
This enzyme is involved in activation of adrenal gland androgens
Blocks androgen production
What three broad groups are prostate cancer patients staged into?
Localised
Locally advanced
Metastatic
What is the common clinical management of patients characterised as ‘Localised’?
Observation
Observe the tumour every so often to track progress
Curative
Surgery (removal of the prostate)
Radiotherapy
Adjuvant hormone ablation therapy
Why are localised prostate cancers often only observed and not treated?
As many patents are old, they may die before the tumour becomes an issue
Many tumours are slow growing
What is the common clinical management of patients characterised as ‘Locally advanced’?
Surgery + neoadjuvant hormone ablation therapy
Radiotherapy + hormone ablation therapy
Hormone ablation therapy
What is the common clinical management of patients characterised as ‘Metastatic’?
Patients placed on palliative care and given treatments like:
Hormone ablation therapy
Chemotherapy
Radiotherapy
Bisphosphonates
What are Bisphosphonates?
Medications which preferentially bind to bone surfaces undergoing active remodelling
Inhibit osteoclast maturation and suppress osteoclast function
Inhibit osteoclast recruitment
Inhibit tumour cell invasion and adhesion
What are the future research directions for prostate cancer?
Anti-androgens extend survival, but resistance can develop
Chemoprevention
Earlier detection
Better staging
New prognostic indicators
New therapies
Immunotherapy