4. psychopharmacology (for exam 2)
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
agonist
facilitates NT effects
antagonist
inhibits NT effects
precursor
building blocks of NT
which class of drugs usually increase precursors?
agonists
which class of drugs stimulate autoreceptors and how?
antagonists
autoreceptors located on the pre-synaptic terminal buttons — if NT docks, it creates a negative feedback loop that closes the calcium ion channels which create NT release
which class of drugs blocks autoreceptors?
agonists
4 criteria for NT
must be found in the neuron
with an AP, must be released from terminal buttons
must make EPSP or IPSP on target tissue
must have a mechanism to “turn it off”
2 ways which NTs can be “turned off”
enzyme degrades it in the synapse
reuptake into presynaptic neuron
who discovered the first NT? what is this NT? how did he do it?
Otto Loewi, ACh
stimulated vagus nerve of frog and saw that it lowered heart rate (parasympathetic, craniosacral vagus nerve)
then absorbed the NT fluid and put it on another frog’s heart without a vagus nerve, whose HR also declined
acetylcholine (primary NT secreted by what cells, % of CNS neurons it takes up, 2 places where it’s found in the periphery, 2 places where it’s found in the brain)
primary NT secreted by efferent PNS cells
10-15% of all CNS neurons
in the periphery, neurons found in
autonomic ganglia (e.g. the heart)
the neuromuscular junction (activation of muscle mvmt)
in brain, neurons are found in
nucleus basalis (mvmt)
hippocampus (memory)
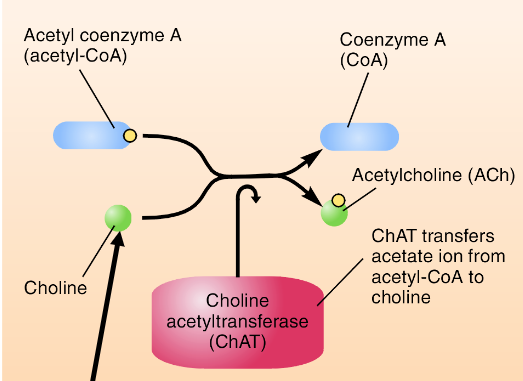
ACh synthesis pathway
note: choline is derived from lipid breakdown
2 natural drugs that release ACh
black widow spider venom, botulinum toxin
black widow spider venom (what does it do, where does it work)
stimulates release of ACh (agonist) to make muscles contract (looks like paralysis)
usually not lethal; just works at neuromuscular junction
botulinum toxin (what it causes and what it’s caused by, class of drug, what it does)
botulinism, caused by improperly canned food
ACh antagonist
extremely toxic → paralysis, death
hemicholinium
prevents reabsorption of ACh, ACh antagonist
acetylcholinesterase + process it’s involved in
destroys ACh into choline and acetate in the synapse, and ~50% of choline is recycled into new NTs
termination of ACh
neostigmine + what disease it helps
interferes w AChE activity → increases ACh and therefore restores muscle strength for those affected by myasthenia gravis
myasthenia gravis
autoimmune disorder that attacks peripheral ACh receptors
what else inhibits AChE?
insecticides
2 types of receptors (excitatory or inhibitory, where it acts, metabotropic or ionotropic) + give examples of agonists and antagonists
nicotinic - excitatory (either Na+ or Ca2+), in skeletal muscle, ionotropic (ligand-gated channels)
agonist: nicotine
antagonist: curare
muscarinic - inhibitory (mostly K+ channels), acts on heart and other smooth muscle, metabotropic (GPCRs, slower than nicotinic)
agonist: muscarine (found in poison mushrooms)
antagonist: atropine (blocks receptors; “belladonna”) (prevents parasympathetic arousal)
2 things ppl w alzheimer’s have less of + why?
ACh
ACh synthesizing enzyme, choline acetyltransferase
prolly due to the degeneration of ACh neurons in the hippocampus and inferior frontal lobes
aricept
AChE inhibitor to keep ACh in synapse of still-living receptors
2 types of monoamine transmitters + examples
catecholamines - dopamine (DA), norepinephrine (NE), epinephrine (EPI)
indolamines - serotonin (5-HT)
process of making catecholamines
tyrosine → L-DOPA (hydroxyl group was gained) → dopamine (carboxyl group was lost) → norepinephrine (hydroxyl group was gained) → epinephrine
nigrostriatal system (location + function)
projects from the substantia nigra to the mid and hindbrain regions (basal ganglia, substantia nigra)
for motor processing
mesolimbic system (location + function)
projects from ventral tegmental area to the limbic system (tegmentum, nucleus accumbens, amygdala, hippocampus)
involved w emotion
mesocortical system (location + function)
projects from the ventral tegmental area to the prefrontal cortex
involved in thinking, planning
nucleus accumbens
associated w reward/pleasure
what 2 things block DA reuptake?
amphetamines
cocaine
so these physiologically addictive drugs INCREASE DA release, and so they feel more rewarding
3 disorders due to catecholamines
Parkinson’s
depression
schizophrenia
Parkinson’s
DA neuron degeneration within the basal ganglia, or btwn substantia nigra and caudate nucleus
depression
decreased catecholamines within, perhaps, all systems
schizophrenia
increased DA within the mesolimbic and mesocortical systems
2 treatments to Parkinson’s
L-DOPA
deep-brain stimulation to basal ganglia — often done in later stage Parkinson’s, has IMMEDIATE effects on motor symptoms
reserpine
an antagonist that prevents the storage of dopamine within vesicles by blocking monoamine transporters within the neuron
Monoamine oxidase (MAO-B)
destroys excess DA in terminal button
Deprenyl
blocks MAO-B to increase catecholamines, agonist
ways motor, emotion, and cognition are affected by depression
motor - increased isolation, decreased activity
emotion - self-critical thoughts, relative inability to experience pleasure
cognition - reduced ability to concentrate
foot-shock avoidance in rats experiment
dopamine antagonists decrease “escape” tendency while dopamine agonists increase it
antagonist rats: 10% escape
placebo: 50%
agonist: 90%
related to learned helplessness model of depression
describe CA (catecholamine) levels in depression and mania
aka catecholamine hypothesis of depression
depression associated w decreased CA lvls within the brain
mania results from increased CA lvls
abt 15% of humans who take __ for hypertension (to decrease NOR and adrenaline) develop clinical depression
reserpine
tricylic antidepressants (function + 3 examples)
treatments for depression that serve as reuptake blockers; affect primarily both NOR and SER
amitriptyline (Elavil)
desipramine (Norpramin)
imipramine (Tofranil)
side effects of tricylics
rapid heartbeat and dizziness upon standing
prone to overdose (cardiac complications)
occasionally “anticholinergic effects”
dry mouth/eyes
bright light sensitivity (dilation of pupils)
blurry vision
types of symptoms of schizophrenia + examples
positive symptoms (most recognizable)
hallucinations/delusions
disorganized thoughts, speech, and behavior
stereotyped behavior - repetitive behavior without significant meaning
negative symptoms
decreased affect
alogia - decreased verbal output
avolition - difficulty initiating goal-directed behavior
how did we find the cause of schizophrenia to be increased DA lvls?
found out bc amphetamine blocks DA reuptake → amphetamine psychosis (similar to paranoid schizophrenia)
typical/classic antipsychotics/neuroleptics, what they affect
have more effect on reducing “positive” relative to “negative” symptoms
chlorpromazine (Thorazine)
haloperidol (Haldol)
mesoridazine (Serentil)
thioridazine (Mellaril)
side effects of antipsychotics
decreased DA → Parkinson’s disease
acute dystonia
oculomotor crisis
akathisia
tardive dyskinesia
acute dystonia
uncontrolled mvmt of face, neck, and tongue
oculomotor crisis
uncontrollable eye mvmts
akathisia
restlessness and agitation
tardive dyskinesia
“late appearing mvmt disorder” (starts w neck/tongue and, eventually, can involve the whole body)
atypical antipsychotics + examples + what it’s better at treating
attempt to have more specific effects on psychosis w fewer extrapyramidal side effects (basal ganglia) by targeting receptors as opposed to DA
Zyprexa (D2), Clozapine (D4), Risperdal (D2)
negative symptoms
the __ within __ gives rise to NE (norepinephrine) fiber systems to areas responsible for __ (frontal systems). what system is it a part of?
locus coeruleus, pons, vigilance/attentiveness
sympathetic nervous system
are noradrenergic receptors metabotropic or ionotropic?
metabotropic - slow-acting, diffuse effects
what is NE synthesized from and where?
dopamine within synaptic vesicles
what leads to NE breakdown? what 2 drugs inhibit NE breakdown and how? what are the effects?
excess NE destroyed by MAO-A
moclobemide inhibits MAO-A (increases NE, agonist)
increases attentiveness, alertness, vigilance
Ritalin - an NE agonist used for ADHD, stimulant, DA/NE reuptake blocker
3 antagonists/agonists for NE based off its relation to DA
L-DOPA - agonist
AMPT - antagonist
reserpine - antagonist
epinephrine (aka…, synthesized from what, synthesizing enzyme, where it’s released from, what it does to the body)
adrenaline
synthesized from norepinephrine/noradrenaline
PNMT (phenylethanolamine-N-methyltransferase)
released from adrenal medulla
preps body for action under stress… usually as a sympathetic hormone
serotonin location + involved in + synthesized from
serotonin cell bodies are located in brainstem raphe (RAS) and project to cortex
involved in emotion, arousal [sleep-wake cycles], visual system
tryptophan
substances responsible for 5-HT/serotonin release and termination
serotonin release
8-OHDPAT is an autoreceptor stimulator that reduces 5-HT release → antagonist
serotonin termination
reuptake is blocked by fluoxetine, which elevates 5-HT → agonist
degradation: MAO-A converts serotonin to 5-HIAA
relationship btwn serotonin and aggression in animals
serotonin suppresses aggression
Higley studied wild rhesus monkeys. 5-HIAA (metabolite) in CSF was inversely associated w aggression
low 5-HIAA → increased attacks on dominant monkeys + longer leaps from tree to tree (so more risk-taking behavior) → often leading to death
5-HT higher in dominant rather than submissive male vervet monkeys
removing dominant monkey increases 5-HT in 2nd dominant male
restoring dominant male lowers 5-HT once again
relationship btwn 5-HT and aggression in humans
in humans, aggression is rare
reduced 5-HT and 5-HIAA in brains of suicide completers relative to those severely depressed
reduced 5-HIAA and 5=HT in those completing suicide via VIOLENT relative to non-violent means
reduced 5-HIAA is associated w the “urge to act out hostility” subscale of the Hostility and Direction of Hostility Questionnaire
neuromodulators (what are they, how far do they travel, how much is secreted, 2 important types, function)
chains of amino acids
travel farther than NTs (not “private” communication)
secreted in larger amounts than NTs
glutamate (produces EPSPs), GABA (produces IPSPs)
well-suited for setting lvls of central neural activity underlying behavioral states
alertness, arousal, anxiety by modulating the excitability of the postsynaptic cell
relaxation
how does glutamate and GABA produce EPSPs and IPSPs?
glutamate increases likelihood of neuronal firing → Na+ or Ca2+ channels open → EPSPs
GABA decreases likelihood of firing → K+ or Cl- channels open → IPSPs
4 receptor types glutamate interacts w
NMDA receptor - controls a Ca2+ channel, closely associated w learning
AMPA receptor - controls sodium channels
kainate receptor - controls sodium channels
metabotropic glutamate receptor
2 receptors GABA acts on + examples of drugs that affect them
GABAsubA - ionotropic receptor (controls a chloride channel) - benzodiazepines (e.g., valium) bind here → reduce anxiety and seizures; promote sleep and muscle relaxation
GABAsubB - metabotropic receptor (controls a K+ channel)
Baclofen (major muscle-relaxant) acts as a direct agonist
what receptors does alcohol affect, and how?
direct effect on GABAsubA receptors
GABA agonist by increasing the amt of time that Cl- channels are open
relationship btwn seizures and GABA + drug that may help
seizures (uncontrollable neuronal firing) appear associated w decreased GABA
tiagabine reduces GABA reuptake
tetrahydrocannibol (THC) (what is it, what does it stimulate, associate w 4 things)
a lipid
stimulates cannibinoid receptors
associated w
analgesic effects
increased appetite
reduced nausea
distorted perception of time
soluble gases + example
can diffuse somewhat and affect more distant cells
nitric oxide (NO) exerts effects within intestinal muscles, dilates brain blood vessels