Chemistry Extended response
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What did John Dalton to advance the atomic model and when
Converted atomic philosophy of the greeks into a scientific theory between 1803 and 1808
What was John dalton’s atomic model
· All matter is made of tiny, indivisible particles called atoms.
· Atoms of the same element are identical in mass and properties.
· Atoms cannot be created or destroyed in chemical reactions.
· Compounds form when atoms combine in simple whole‑number ratios.
· Chemical reactions involve the rearrangement of atoms, not their creation or destruction.
What was John Dalton’s reasoning for his model of the atomic model
· Careful mass measurements of reactants and products in many reactions showed fixed composition (definite proportions) and simple whole‑number mass ratios in different compounds of the same elements (multiple proportions)
· Gas‑mixture experiments → led to Dalton’s law of partial pressures, explained by independent, moving particles (atoms or molecules).
Putting these together, Dalton argued that the best explanation is that matter is made of indivisible atoms that rearrange in simple integer ratios during chemical reactions, without being created or destroyed
How do you draw Dalton’s model
Draw a small solid circle:
Draw a filled circle.
Label it: “Solid, indivisible sphere (atom).”
You can add the element symbol inside (e.g. “O” or “Na”) and label: “All atoms of this element are identical in mass and properties.”
There are no internal parts (no protons, neutrons or electrons) in Dalton’s model.
When and what did JJ thompson do to advance the atomic model
1890
Thomson discovered the electron using cathode rays and showed that atoms contain small, negatively charged particles embedded in a positive background
What did JJ Thompson actually deduce and what from?
He carried out experiments using cathode rays produced in a discharge tube and found that the rays were attracted by positively charged metal plates but repelled by negatively charged ones. From this, he deduced the rays must be negatively charged.
- Thomson found that the charge would deflect away from the neg coil. He stated that his charge was thus negative
- He found the charge was 1000 times lighter than an atom fo hydrogen
- He then said that this negative charge must be inside an atom
- Called such charge a corpuscle
- Was the electron
How did JJ Thompson’s experiment work
Cathode ray method:
1 nearly empty tube with air sucked out
2 an electric charge is passed thru the tube. Travels from cathode to anode
3 charge is invisible so to see it fluorescent screen is placed at the back of the tube. where the beam hits a dot appears on the screen also can use a fluorescent gas and the whole tube would light up
4 the beam always travels straight if no interference
5 the deflection coils each have a specific charge one is pos and one is neg
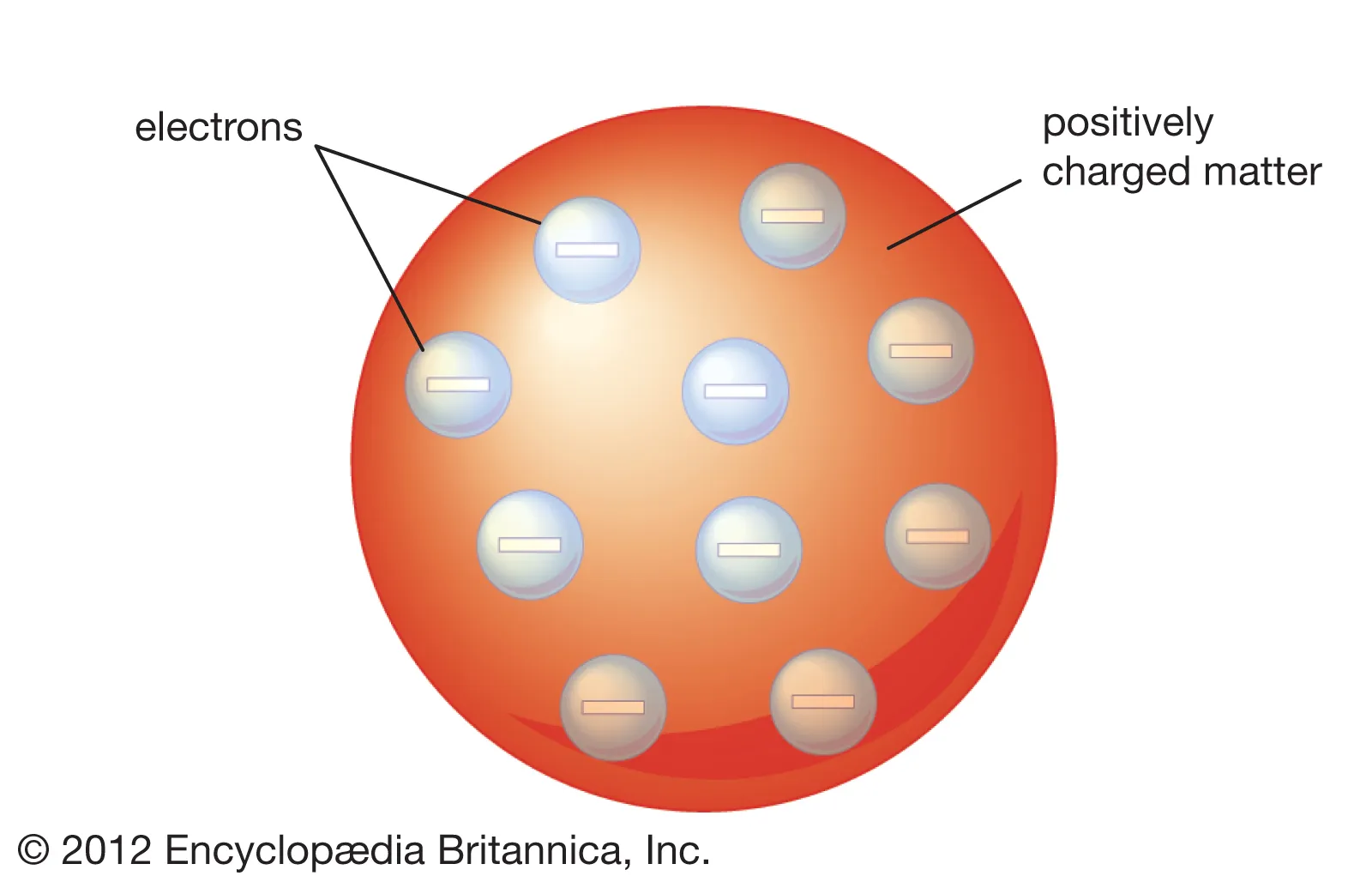
What did JJ Thompson’s model look like ?
An atom is filled with positive ‘fluid’ This resembles the jam in the pudding
Corpuscles are the neg charged particles ‘suspended’ in such fluid Resembles plums in the pudding
He did not predict the movement
When and what did Ernest Rutherford do to advance the atomic model
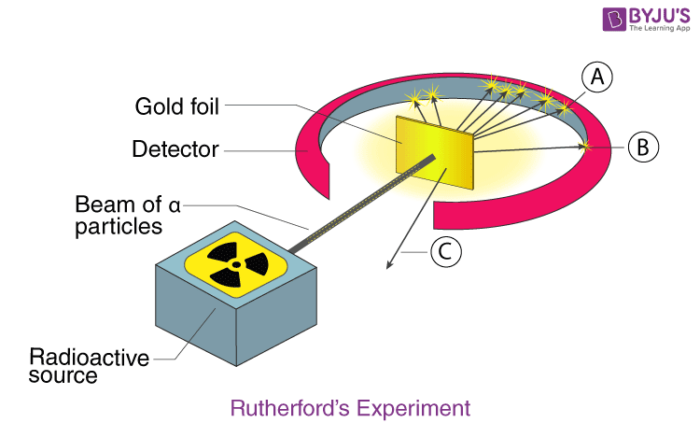
1910 the gold foil experiment
How did Ernest Rutherford’s experiment work and what did he deduce from it
Gold foil experiment
Alpha particles at gold sheet
Measured the deflection as the particles came out the other side
Most went thru and did not deflect whatsoever
hwever about 1 in 8000 particles bounced off the gold foil at very large angles.
In contrast, the particles that were highly deflected must have experienced a tremendously powerful force within the atom He concluded that all of the positive charge and the majority of the mass of the atom must be concentrated in a very small space in the atom's interior, which he called thenucleus. The nucleus is the tiny, dense, central core of the atom and is composed of protons and neutrons.
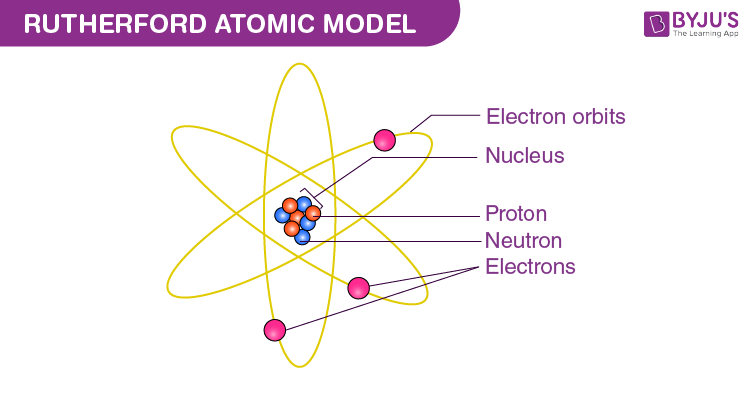
What is rutherford’s model and how do you draw it
Nucleus of atom is a dense mass of pos charged particles
Electrons orbits the nucleus
A problem raised was that why are the negatively charged particles not attracted to the positively charged nucleus
Rutherford stated that this was because the atom was likea mini solar system and the elctrons orbited the nucleus in wide orbit
When and what did Niel Bohr do to advance the atomic model
1910s -Physicist
He agreed with Rutherford but found flaw with the electrons not collapsing
What did Bohr deduce to change the atomic model
Bohr proposed that electrons do not radiate energy as they orbit the nucleus, but exist in states of constant energy that he called stationary states. This means that the electrons orbit at fixed distances from the nucleus. Bohr's work was primarily based on the emission spectra of hydrogen. This is also referred to as the planetary model of the atom. It explained the inner workings of the hydrogen atom.
Classical physics predicted that an orbiting electron should continuously radiate energy and spiral into the nucleus, so Bohr introduced “stationary states” in which electrons do not lose energy.
Hydrogen Emission Spectrum (Balmer Series): Bohr's model perfectly accounted for the discrete wavelengths of light (spectral lines) emitted by excited hydrogen atoms.
Ionization Energy of Hydrogen: Bohr calculated the energy required to remove an electron completely from a hydrogen atom, and this calculated value matched experimental measurements exactly.
X-ray Spectra of Heavier Elements: Bohr expanded his model to explain X-ray emissions from heavier atoms, showing that inner shells behaved in a "hydrogen-like" manner. He predicted X-ray frequencies (Moseley's Law) that were subsequently confirmed by experiments.
What does the rutherford-bohr model look like
Electrons orbit nucleus in orbits that have a set size and energy
The lower the energy of the electron, the lower the orbit
This means that as electrons fill up the orbitals they will fill the lower energy level first
I.e the model we use today
What did James Chadwick do in relation to the atomic model?
Chadwick is best known for his discovery of the neutron in 1932. A neutron is a particle with no electric charge that, along with positively charged protons, makes up an atom’s nucleus
How did James chadwick deduce what he found
Rutherford's model had a proton-electron nucleus, but this failed to explain atomic mass discrepancies—helium's mass was about four times a proton's, yet it had only two protons.
Chadwick was not convinced that alpha particles could produce gamma rays. Hence he performed other experiments and found that this unknown radiation caused nuclei recoil that could only be of a particle of proton like weight, but of no charge, being unaffected by electric or magnetic fields
Theories for the additional particles included additional protons whose charge was shielded by electrons in the nucleus or an unknown neutral particle. In 1932 French physicists Frédéric and Irène Joliot-Curie bombarded beryllium with alpha particles and observed that an unknown radiation was released that in turn ejected protons from the nuclei of various substances. The Joliot-Curies hypothesized that this radiation was gamma-rays. Chadwick was convinced that alpha particles did not have enough energy to produce such powerful gamma-rays.
gamma rays (photons, massless) couldn't transfer enough momentum to eject fast protons, as calculated from proton ranges. Only massive neutral particles could.
Describe the structure and bonding of diamond
Diamond is of a covalent network structure. Each carbon in diamond is bonded to 4 others, filling its octet. The carbons bond in a tetrahedral arrangement forming a rigid 3d lattic. Diamond exhibits covalent bonding
No. of bonds on each carbon atom of diamond
Four
Properties of diamond
- Very hard
- Brittle
- High mp/bp
- Non conductive
Examples of use of diamond
Jewellry
Lustrous and resists scratching hence is durable and has high brilliance due to high refractive index
Cutting and drilling tools
Diamond is used in cutting and drilling tools because it is the hardest natural material, so it can cut through hard substances like stone, concrete, and metal more effectively than most materials. It also has high wear resistance, so the tool stays sharp for longer.
Heat sinks and electronics
Diamond is used in heat sinks and some electronic applications because it has very high thermal conductivity, meaning it transfers heat away quickly. It is also an electrical insulator, so it can remove heat without conducting electricity.
Diamond has an exceptionally high thermal conductivity (ranging from 1000–2200 W/m·K) despite being an electrical insulator because its heat conduction is mediated by phonons (lattice vibrations) rather than free electrons. The rigid, highly ordered 3D tetrahedral structure allows these vibrational waves to propagate rapidly through the material with minimal scattering.
Describe the structure and bonding of graphite
Graphite consists of sheets of graphene that are sheets of carbon bonded to 3 other carbons, each having a delocalised electron associated with each carbon
Graphite exhibits covalent and intermolecular bonding. The inside the the graphene, carbons bonded covalently but the graphene sheets are bonded intermolecularly to form graphite
characterized by a layered, hexagonal, crystalline structure.
No. of bonds on each carbon atom of garphite
three
Properties of graphite
Graphite is a soft, black, and opaque allotrope of carbon,
excellent conductor of electricity and heat, chemically inert, and highly stable at high temperatures, with natural lubricating properties due to weak forces between its layers
High MP due to having to break apart graphene
Uses of graphite
Writing (pencil)
Weak IMF between layers allow them to come apart easily
Lubricants
The layers slide over each other, so graphite reduces friction.
Electrodes
Because graphite is conductive
Descripton of structure and bonding of graphene
Carbon bonded to three others with a free electron
Hexagonal structure
single layer of carbon atoms arranged in a hexagonal lattice
Properties
Very strong: graphene has exceptional tensile strength because of the strong covalent bonds between carbon atoms in its 2D lattice.
Very thin: it is one atom thick, making it the thinnest known material.
Lightweight: because it is just a single atomic layer, it has very low mass.
Excellent electrical conductor: electrons move very easily through its structure.
Excellent thermal conductor: it transfers heat very efficiently.
Flexible and elastic: it can bend and stretch without easily breaking.
Transparent: despite being a solid material, it lets most visible light pass through.
Large surface area: its 2D nature gives it an extremely high surface area for its mass.
Nearly impermeable: perfect graphene does not easily let gases through.
Uses of graphene
Microchips and transistors
Graphene has high conductivity so it can help devices switch and carry signals very quickly.
Batteries and supercapacitors
Large surface area and conductivity means that it can store more charge and allows fastor ion movement
Solar panels and transparent conductors
It can carry charge whilst being transparent
Descripton of structure and bonding of buckminsterfullerene
Buckminsterfullerene is C60 and is a ball composed of 12 pentagons and 20 hexagons
The presence of pentagons is what makes the sheet curve and close up into a sphere; if it stayed flat like graphite, the carbon atoms would have a different arrangement.
No. of bonds on each carbon atom
tres
Properties of buckminsterfullerne
- Spherical
- Molecular bonding between balls
- Soft solid because of that
- Low solubility in water (insoluble)
- Soluble in some organic solvents
- Stable due to closed cage structure
- Semi conductor
- Reactive in addition reactions
it behaves a bit like an electron-deficient alkene and can undergo hydrogenation and halogenation.
Uses of buckminsterfullerene
Drug delivery
Its hollow spherical cage can trap or carry other molecules, so it can transport drugs to target sites
Lubricant
Fullerene molecules can improve antiwear and anti-friction properties, helping surfaces slide more smoothly.
Catalysts / chemical applications
Its reactive surface and ability to take on added groups make it useful in chemical and industrial processes.
Many of these properties come from the fact that C60 is a small molecule with weak forces between molecules, unlike the giant covalent networks in diamond or graphite. Its delocalised electron system and curved cage shape also give it unusual chemical and electronic behavior.
It suits these uses because its hollow cage structure, large surface area, electron-accepting ability, and ability to be modified
Define allotrope
- Allotropes are two or more different forms of an element in the same physical state, having different atomic arrangements that produce different properties