General Chemistry II Formulas
1/30
Earn XP
Description and Tags
just kill me now
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
when solving a heating/cooling curve problem, what formulas do you use? (the q ones)
q = mcΔT for the original equation
q = nΔH for the combined states
m = mass
c = specific heat capacity (water = 4.184)
ΔT = change in temperature (in KELVINS)
n = moles
ΔH = change in enthalpy
cell density formula (think perovskite)
P = Z(MM)/(6.022E23)(a³)
P = cell density
Z = atomic number
MM = molar mass
a = side length
how many atoms are in a face centered cubic unit cell?
4 (there are atoms present in the corners and faces of the cube → 8(1/8) + 6(1/2) = 4)
how many atoms are in a body centered cubic unit cell?
2 (there are atoms present in the corners and center of the cube → 8(1/8) + 1 = 2)
how many atoms are in a simple cubic unit cell?
1 (there are atoms only in the corners of the cube → 8(1/8) = 1)
What do you use when you have to calculate how much a gas dissolves in a liquid (hint: Henry’s Law)?
C = kP
C = concentration
k = solubility constant
P = partial pressure of the gas
what is the formula for morality?
M = n/L
n = moles
L = volume (in LITERS)
what is the formula for molality?
m = nsolute/kgsolvent
what is the formula for mole fraction (X)?
X = nsolute/nsolution
what is the formula for ppm?
ppm = (masssolute/masssolution) x 1,000,000
1,000,000 = 106
what is the formula for ppb?
ppb = (masssolute/masssolution) x 1,000,000,000
1,000,000,000 = 109
What equations do you use when dealing with vapor pressure and mole fraction? (hint: Raoult’s Law)
Vapor Pressure: Psolvent = (Xsolvent)(P°solvent)
Change in Vapor Pressure: ΔP = Psolvent - Psolvent = (Xsolute)(P°solution)
What equation do you use for measuring boiling point elevation?
ΔT = iKbm
i = van’t Hoff factor (ex: NaCl i = 2 and CaCl2 i = 3)
Kb = constant of the solvent
m = molality
What equation do you use for measuring freezing point depression?
ΔT = iKfm
i = van’t Hoff factor (ex: NaCl i = 2 and CaCl2 i = 3)
Kf = constant of the solvent
m = molality
What formula do you use to find osmotic pressure?
π = iMRT
π = osmotic pressure (in ATM)
i = van’t Hoff factor (ex: NaCl i = 2 and CaCl2 i = 3)
M = molarity
R = 0.08206
T = temperature (in KELVIN)
What formula do you use to find the enthalpy of sublimation (ΔHsub)?
ΔHsub = ΔHfus + ΔHvap
What is the half-life formula for the zero order rate law?
t = [Ao]/2k
What is the half-life formula for the first order rate law?
t = 0.693/k
What is the half-life formula for the second order rate law?
t = 1/k[Ao]
What equation do you use when you’re given the Kp and Kc?
Kp = Kc(RT)^Δn
What are all of the pH equations?
pH = -log[H+] and pOH = -log[OH-]
[H+] = 10-pH and [OH-] = 10-pOH
pH + pOH = 14
Kw = KaKb = [H3O+][OH-]
When looking at buffer capacity and titrations (before eq. pt.) what equation should you use? (hint: Henderson Hasselbach)
pH = pKa + log(base/acid)
How do you calculate Ka, Kb, and Ksp?
Ka = [H+]2 / [HA]
Kb = [OH-]2 / [A-]
Ksp = [products]/[reactants]
How do you calculate Ka and Kb during salt hydrolysis?
producing H3O+: Ka = [A-][H3O+]/[HA]
producing OH-: Kb = [BH+][OH-]/[B]
What formulas do you use for Gibbs Free Energy?
ΔG = ΔH - TΔS
ΔG = ΔG° + RTln(Q)
ΔG° = -RTln(k)
What is the formula to find cell potential (Ecell)?
Ecell = cathode - anode
What are the Nernst equations related to Ecell?
Ecell = E°cell - (RT/nF)ln(Q)
Ecell = E°cell - (0.0592/n)log(Q)
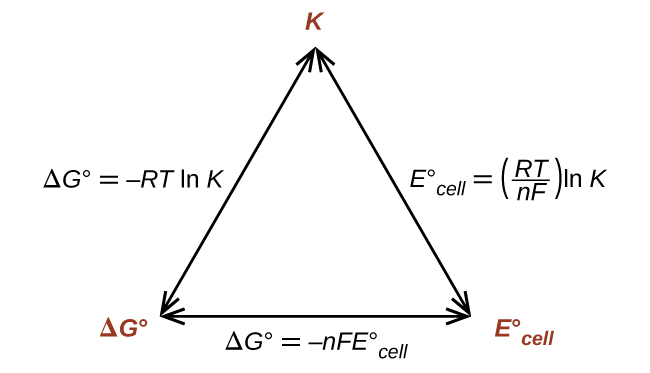
What are the Nernst equations related to ΔG, Keq, and Ecell?
F = 96485 c
What is the galvanic cell notation?
anode | anode solution || cathode solution | cathode