Telomerase and Senescence
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
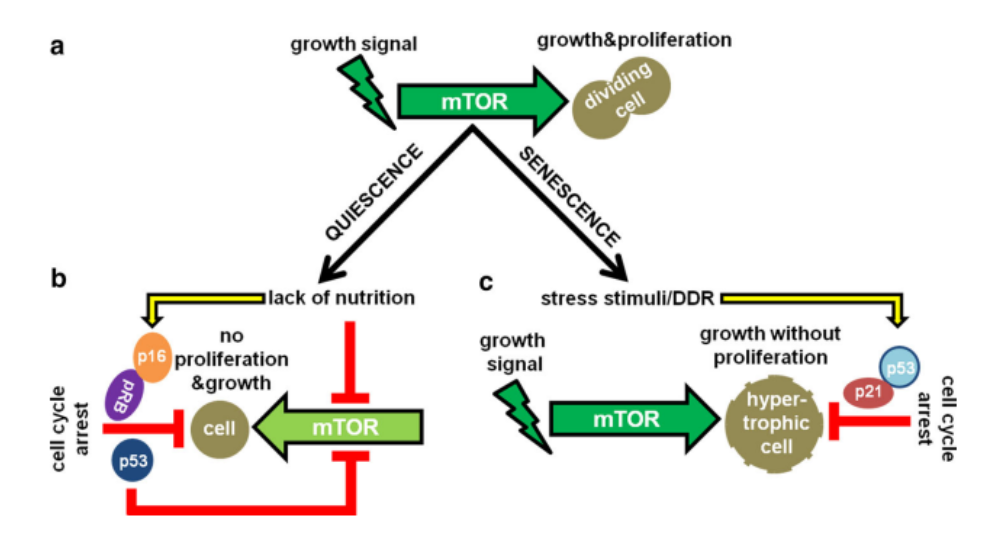
Quiescent Cells is a state of cellular division in which cells
“leave” the cell cycle to remain in a semi-permanent state of no longer actively dividing
cells are still _ active
metabolically
Reversible
Cells can enter and exit from G0 depending on various factors/stimuli
Primary Stimulation
lack of nutrition and growth factors
EX of Quiescence cells
Adult stem cells
capable of regenerating tissues when needed, but otherwise inactive
Hepatocytes
metabolically active liver parenchymal cells that do not divide,
but can enter the cell cycle to regenerate liver tissue when needed
Fibroblasts
active connective tissue cells that become mitotically active
during injury/inflammation to mediate repair/regeneration
Senescent Cells
State of cellular division in which cells permanently arrest somewhere within the cell cycle. Can occur in G1, S, or G2
Cells can remain _ active
metabolically
Once cells undergo senescence, they are
permanently arrested
Primary Stimulation
aging, major DNA damage
EX of Senescence
Replicative senescence
limited replicative potential of normal cells due to telomere attrition or dysfunction
Stress-induced senescence
premature senescence (before telomeres shorten) due to various stressors (e.g., oxidative damage, DNA damage, oncogene activation, etc.)
Postmitotic cellular senescence
permanent growth arrest of terminally differentiated cell types (e.g., neurons, myocytes, adipocytes, etc.)
Difference?
Quiescence is a temporary, reversible resting state where cells can re-enter the cell cycle, while senescence is an irreversible, permanent cell cycle arrest often triggered by stress.
Molecular Players
Telomere
Non-coding, repetitive sequences (TTAGGG) at the ends of linear chromosomes
Shorten with each
DNA replication due to
inability of lagging strand to synthesize (no place to put a
primer to start new Ozakai fragment)
Associated with replicative senescence
when telomeres become sufficiently shortened, stimulates
irreversible cell cycle arrest
Molecular Structure
Shelterin
protein complex bound to telomeric repeats
Telomere loops
ensure that telomere ends are not exposed and are protected from premature degradation
Telomere Shortening: shorten by _ bp with each cell division
50-200
Results of
incomplete synthesis of lagging strand
Also prone to shortening due to
oxidative destruction (despite loops and shelterin complexes)
Mitotic Clock
once reach a threshold length, stimulates senescence. Cells can only divide a finite number of times
Implications for
issue culture of non-transformed cells and aging process
It is not clear what exactly constitutes the
“critically short” telomere length
TFR2
telomere binding factor 2
POT1
protection of telomeres 1
Immortalization
Cell lines that are capable of unlimited replicative potential. Term is usually applied to cultured cells rather than in vivo cells
Process
Repeated cell division of cells in a tissue culture environment
2. Induction of senescence due largely to shortened telomeres
3. A subset may continue dividing despite senescent stimulation crisis phase
4. Majority of cells that continue dividing while in crisis will undergo apoptosis
5. A small subset may survive crisis and become immortalized due to activation of telomerase
Stabilizing Telomeres
Telomerase
reverse transcriptase enzyme that can elongate chromosomes
Two main components
Telomerase reverse transcriptase (TERT)
DNA polymerase that uses internal RNA as a primer
Telomerase RNA component (TERC)
– acts as the RNA template for synthesis of new telomere DNA
Which cells express telomerase
Developmental tissues:
All/most embryonic tissues up to 20 weeks gestation
2. Variable expression in fetal tissues after 20 weeks
Adult tissues:
Lymphocytes in bone marrow and peripheral blood
2. Some epithelial cells (epithelial regenerating cells) in skin, hair, GI, uterus
3. Germ cells in testis
4. Other discreet adult stem cell populations
Majority of cancers (%)
85-95
Hallmarks - Repression of telomerase expression
and phenomenon of telomere shortening are
natural, built-in mechanisms that help prevent transformation
and oncogenesis
Transcriptional Control of TERT: where is it located
5p; complex TF regulation
Upstream promoter element is rich in
CpGs
Normal somatic cells
unmethylated
Malignant Cells
hypermethylated
Overexpression of pro-proliferative TF can activate
promoter (c-myc)
Promoter mutations observed in large percentage of certain cancers
C228T, C250T
Some oncogenic viruses express
transcriptional cofactors that promote expression
Gene amplification also observed in some
cancers