Synthesis of trans-9-(2-phenylethenyl) Anthracene: A Wittig Reaction
1/26
Earn XP
Description and Tags
OCHEM II Lab Final
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Chemical reactions involving organic molecules can be classified into three categories?
molecular rearrangement
elimination
addition
Alkenes are commonly formed through?
elimination reactions
Wittig reaction
preferred as a method of synthesizing alkenes b/c of its high level of regioselectivity
addition reaction
generates an alkene from the reaction of a carbonyl compound with a carbon containing phosphorous reagent (ylide) which is made from a phosphonium halide
Ph3P = good nucleophile + weak base —> so no elimination reaction
Regioselectivity
the tendency of a reaction to form predominantly one isomer from a single reactant
The product resulting from the reaction of the phosphonium halide with a strong base is called?
ylide
neural molecule, which among its atoms, has two adjacent atoms that have opposite charges
Controversy lies in whether the oxaphosphentane intermediate is formed by one step concerted process or a two step process. What forms oxaphosphetane?
betaine reacts to form the oxaphosphetane, stable at -78 C
at room temp, it decomposes to yield the alkene and triphenyl-phosphine oxide
What is the driving force for the decomposition of oxaphosphetane?
formation of the strong-phosphorous oxygen bond of the phosphine oxide, a bond strength estimated to be at least 540 kj/mol
Wittig reaction forms?
carbon-carbon double bond between the carbonyl carbon and the carbon adjacent to the P atom in the ylide
What are we doing in this experiment?
9-anthraldehyde + ylide ( from triphenylbenzylphosphonium chloride) → trans -9-(2-phenylethenyl)anthracene
Reactions
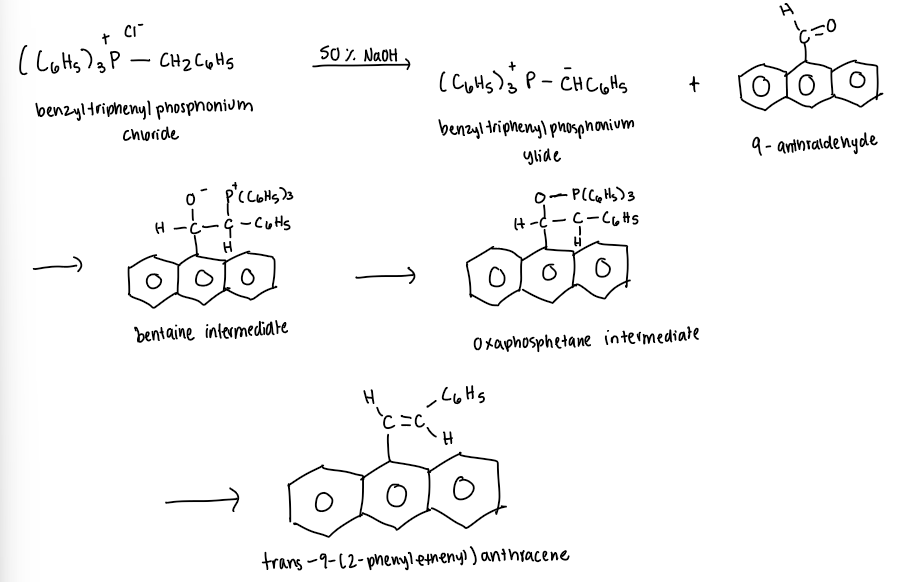
Mechanism
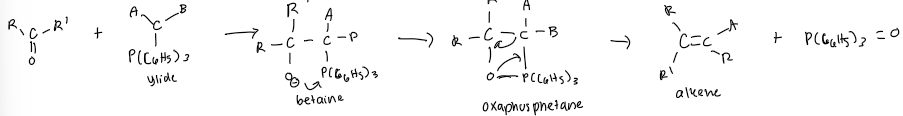
Formation of ylide
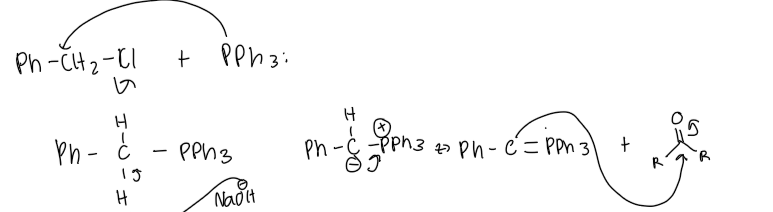
What safety precautions must be observed when using dichloromethane?
fume hood
What safety precautions must be observed when using 50% aq. sodium hydroxide?
gloves + goggles, prevent skin contact
What safety precautions must be observed when using toluene?
don’t use it near flames or other heat sources
use fume hood
Briefly explain the advantage of a Wittig synthesis over the more common dehydrohalogenation reaction.
high level of regioselectivity
choose precise location of new bond
Limiting reactant
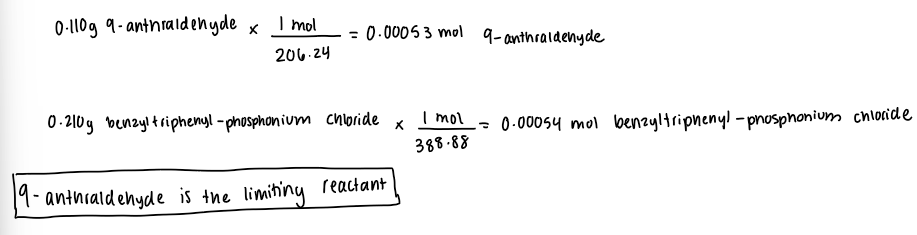
Theoretical Yield
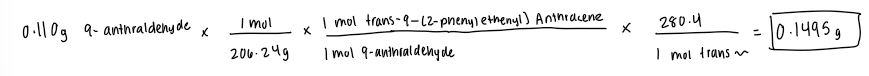
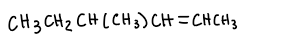
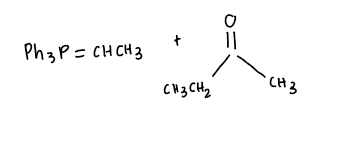
Carbonyl compound + ylide?
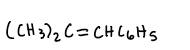
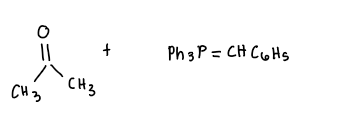
Compare the IR spectra of 9-anthraldehyde + your product.
The IR for 9-anthraldehyde shows a very strong peak at around the 1600-1700 cm-1 mark
This represents an aldehyde peak. However, this peak is not present in the trans-9-(2-phenylethenyl) anthracene product, as there is carbonyl shown.
Additionally, a trans alkene peak is present at the 960 cm-1 mark in the product, which is not shown in the IR for 9-anthraldehyde.
What evidence supports the synthesis of the trans isomer rather than the cis isomer?
A trans isomer showcases a peak at around 960 cm-1, which is present in the IR
a trans isomer has a higher amount of de-shielding protons compared to the cis isomer
What are the steps for this experiment?
benzyltriphenylphosphonium chloride + 9-anthraldehyde added to a test tube
add dichloromethane (DCM) and stir vigorously
add 50% NaOH dropwise slowly while stirring
stopper tube loosely and stir for 30 min
move mixture to centrifuge tube, rinse reaction tube with DCM + H2O
shake + vent centrifuge tube and let layers separate
transfer bottom DCM layer to clean test tube and repeat extraction with DCM
add CaCl2 to DCM layers and let sit for 2 min
decant DCM into RBF and rinse solid with DCM
rotovap
recrystallize w/ 2-propanol
TLC Plate?
9-anthraladehyde has lowest Rf value
trans-9-(2-phenylethenyl)anthracene has higher Rf value
used toluene as eluent + solvent
Why do we use DCM as the solvent?
can dissolve organic molecules and immiscible with water
Why do mix 50% NaOH slowly and stir for 30 minutes?
base deprotonates the phosphonium salt to create ylide
wittig reaction can be exothermic, and adding the base slowly prevents the reaction from overheating
stirring helps the two liquids react
Why do we add water?
water is added to dissolve inorganic salts and pull them away from organic product
cacl2 is the drying agent and absorbs water