michniak transdermal and topical drug delivery I
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms
human skin cross section
- stratum corneum
- dermatoepidermal junction
- when looking down on skin surface, cells are peeling off
- cross section of human stratum corneum looks like the inside of a croissant
epidermis cell type
- keratinocytes
layers of epidermis
- stratum corneum
- granular layer
- spinous layer
- basal layer
dermis major cell type
- fibroblasts in dermal matrix
additional components of the dermis
- langerhans cells
- melanocytes
- hair follicles
- eccrine sweat glands
- sebaceous glands
- adipocytes
- blood supply
- nerves
interfacial boundaries
- surface
- stratum corneum
- appendages
- viable epidermis
- dermis
- circulation
surface penetration routes
- drug dissolves, diffuses, and releases from vehicle
some treatments of surface
- camouflage
- protective layer
- insect repellent
- antimicrobial/antifungal
stratum corneum penetration routes
- trans epidermal
- partition/diffusion, stratum corneum
stratum corneum treatments
- emolliency
- keratosis [exfolients]
appendages penetration route
- transappendageal 2 routes
- pilosebaceous unit
- eccrine gland
appendages treatment
- antiperspirant
- exfolient
- antibiotic/antifungal
- depilatory
viable epidermis penetration route
- partition/diffusion
viable epidermis treatment
- antiinflammatory
- anesthetic
- antipruritic
- antihistamine
dermis penetration route
- partition/diffusion
dermis treatment
- same as viable epidermis
- antiinflammatory
- anesthetic
- antipruritic
- antihistamine
circulation penetration routes
- removal via circulation
circulation treatments
- transdermal
order of skin layers that a drug must go through
- surface --> epidermis --> dermis --> subdermal tissue
gradient of skin
- pH gradient since top of skin at pH about 5.2
- water gradient
- calcium gradient
maximizing transdermal drug delivery
- vehicle-drug interactions
- vesicles and particles
- horny layer modified
- horny layer bypassed or removed
- electrically driven procedures
vehicle-drug interactions
- drug and prodrug selection
- thermodynamic activity
- ion pairs and coacervates
- eutectic systems
vesicles and particles
- liposomes and analogues
- high velocity particles
horny layer modified
- hydration
- chemical enhancers
horny layer bypassed or removed
- microneedle array
- stratum corneum removed
- follicular delivery
electrically driven procedures
- ultrasound
- iontophoresis
- electroporation
- magnetophoresis
therapeutic window
- peaking and valley between toxic and minimum effective levels
- the ideal transdermal dose is in the middle of the toxic vs min therapeutic level
advantages of TDDS
- bypass first pass metabolism and GI incompatibility with pH, food, enzymes
- reduce side effects [better plasma concentration time profiles]
- predictable and extended duration of activity
- greater patient compliance
- enhances therapeutic efficacy
- reduction in frequency of dosing
- reversibility of drug delivery which allows removal of drug source
- minimizing inter- and intra-patient variability as compared with oral
- self-administration
- good in patients with nausea, etc
criteria for drug selection
- correct physicochemical properties
- crystalline drugs have low permeability compared to amorphous
- need potent drugs
- no long lag times
- no dermatitis
- clinical need
correct physicochemical properties
- low molecular weight/size
- melting point
- want low water solubility and high partitioning
why do crystalline drugs have low permeability
- they have high melting points
- slow dissolution
dermatitis
- irritant to skin
clinical need
- prolonged administration
- patient compliance
- reduction in dosage
- adverse reaction to non-target tissues avoided
release kinetics of TDDS
- not all are zero order although this is most preferable
- estraderm
- nitrodisc
- transderm-scop
estraderm (17-beta estradiol) order
- zero
nitrodisc (nitroglycerin) order
- first
transderm-scop (scopolamine) order
- mixed
classes of TDDS systems
- membrane permeation-controlled (reservoir) TDDS
- adhesive dispersion-type TDDS
- matric diffusion controlled TDDS
- microreservoir dissolution controlled TDDS
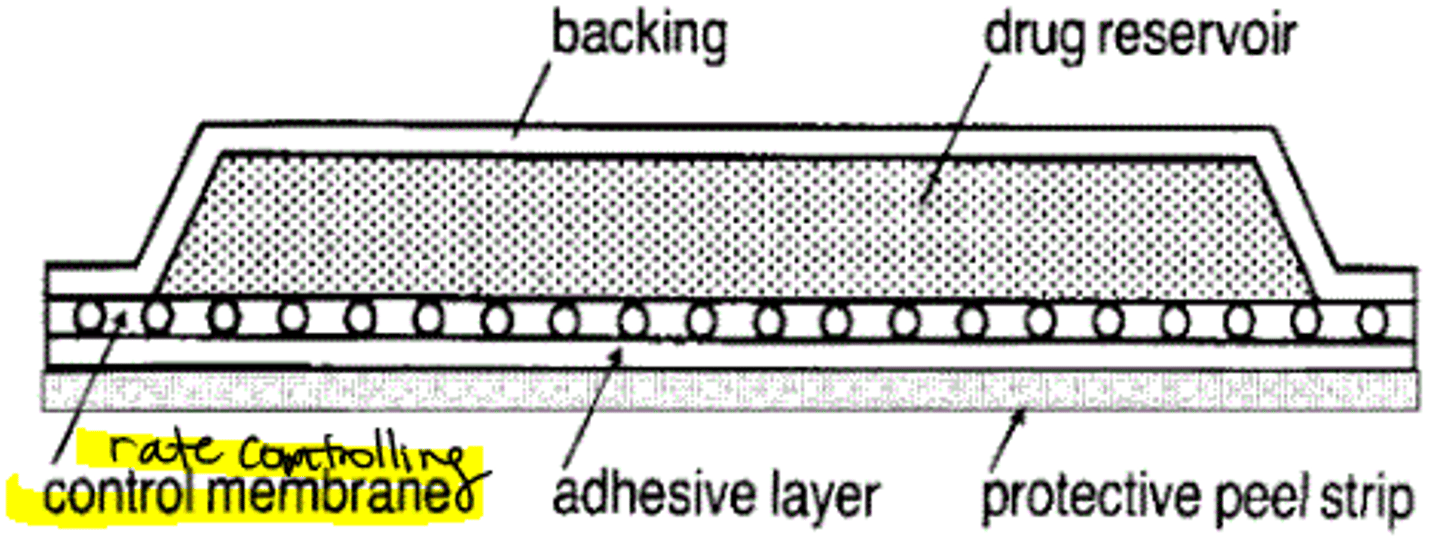
membrane permeation-controlled (reservoir) TDDS
- transderm-scop
- transderm-nitro
- clonidine-TTS
- estraderm
- androderm
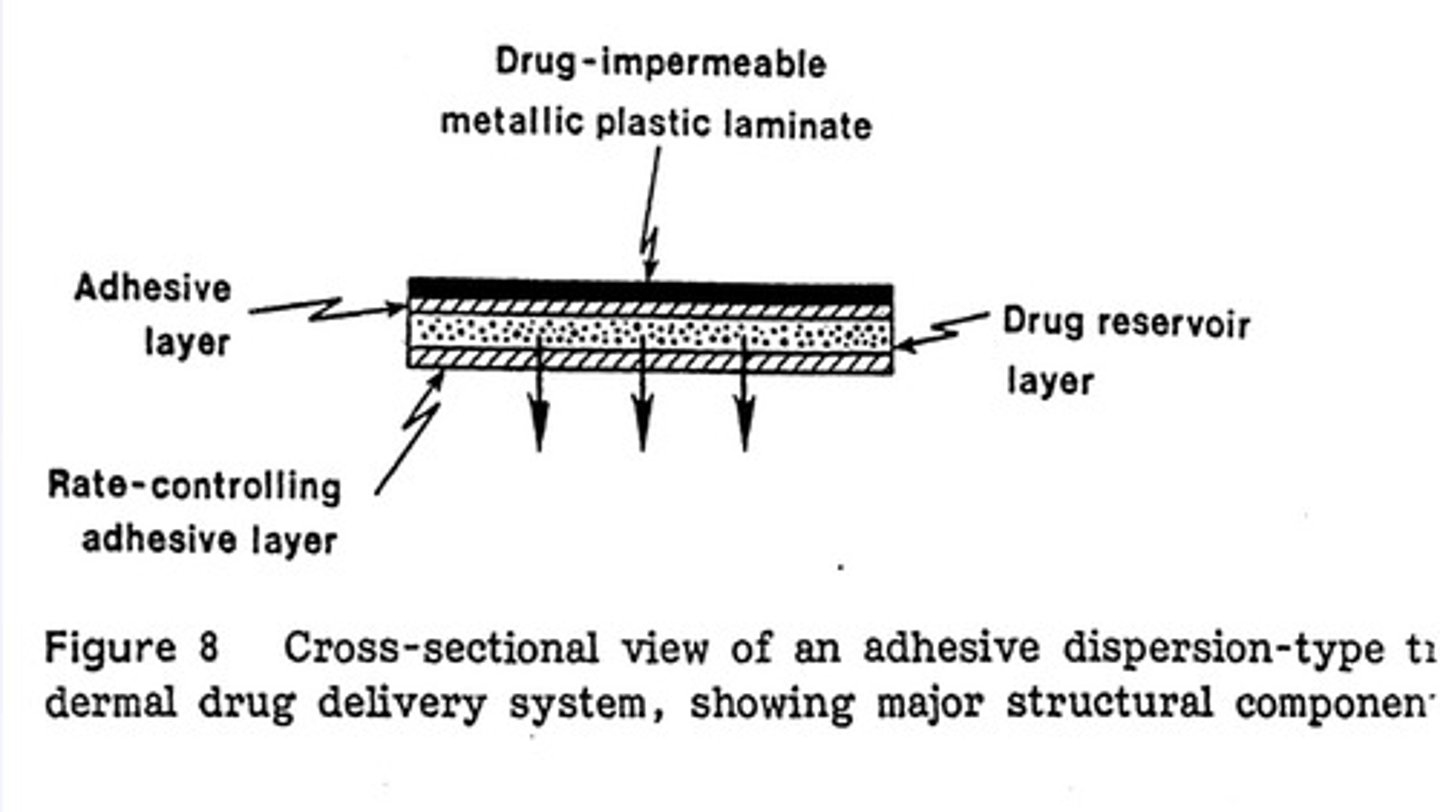
adhesive dispersion-type TDDS
- deponit
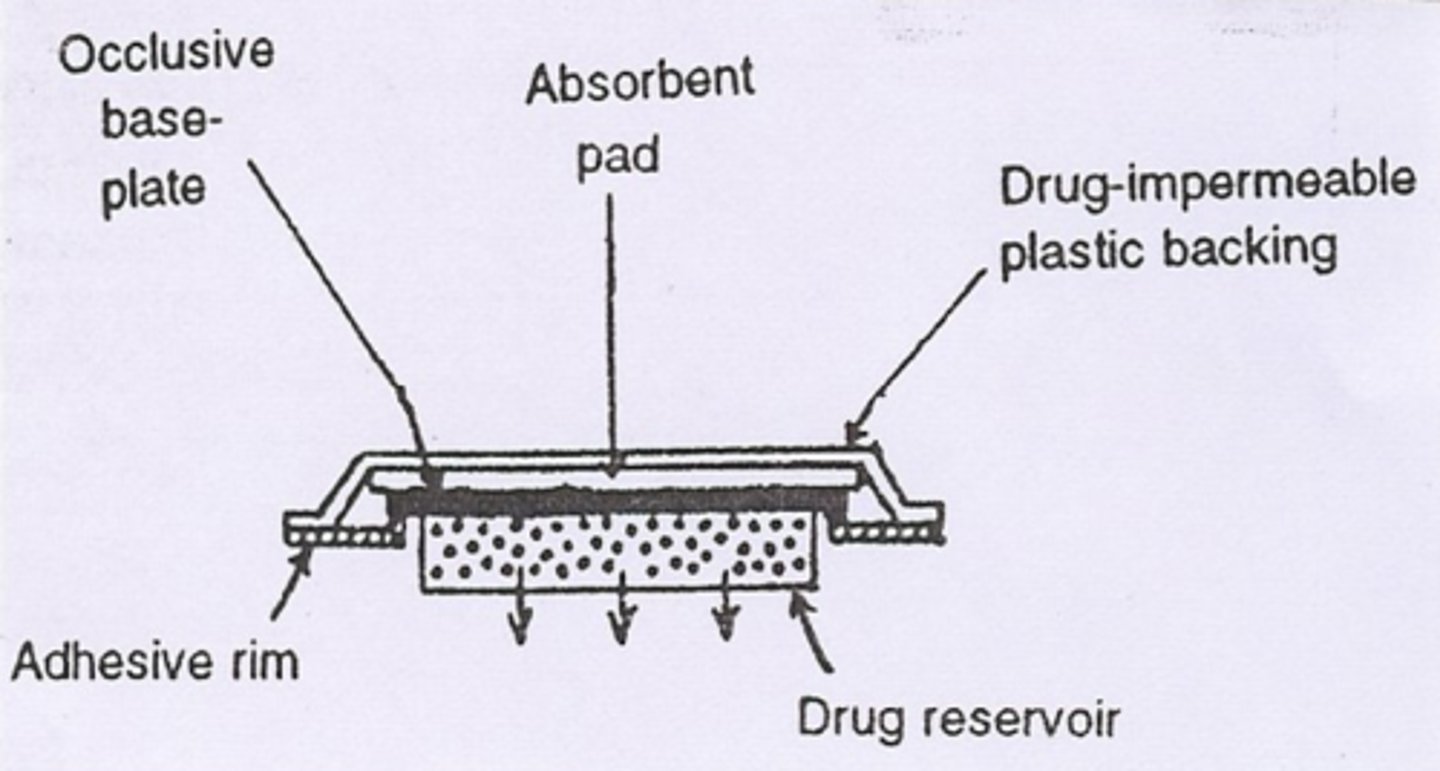
matrix diffusion controlled TDDS
- NitroDur
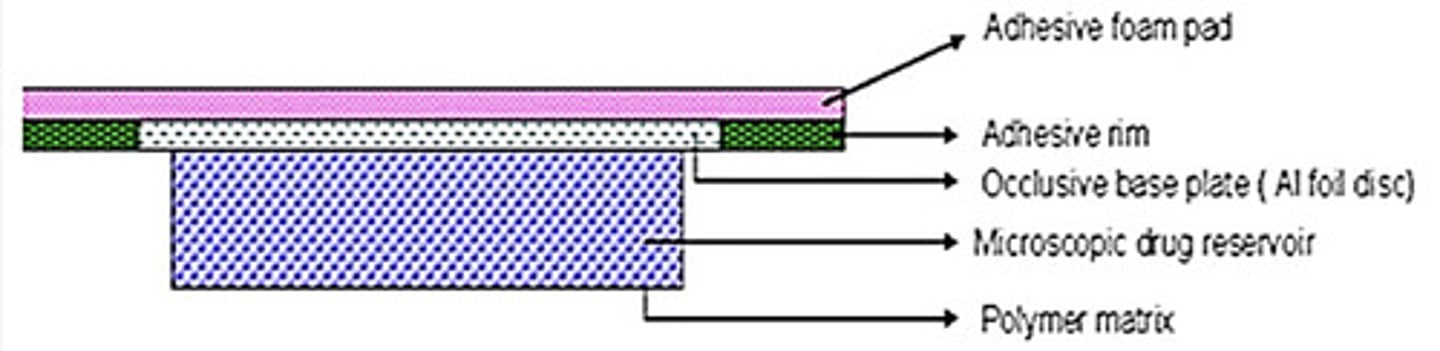
microreservoir dissolution controlled TDDS
- Nitrodisc
marketed systems
- fentanyl-duragesic for chronic pain
- nitroglycerin-many... for angina
- scopolamine-Transderm-Scop- FIST EVER TDDS EVER MARKETED for motion sickness
- testosterone-Testoderm, etc for hypogonadism
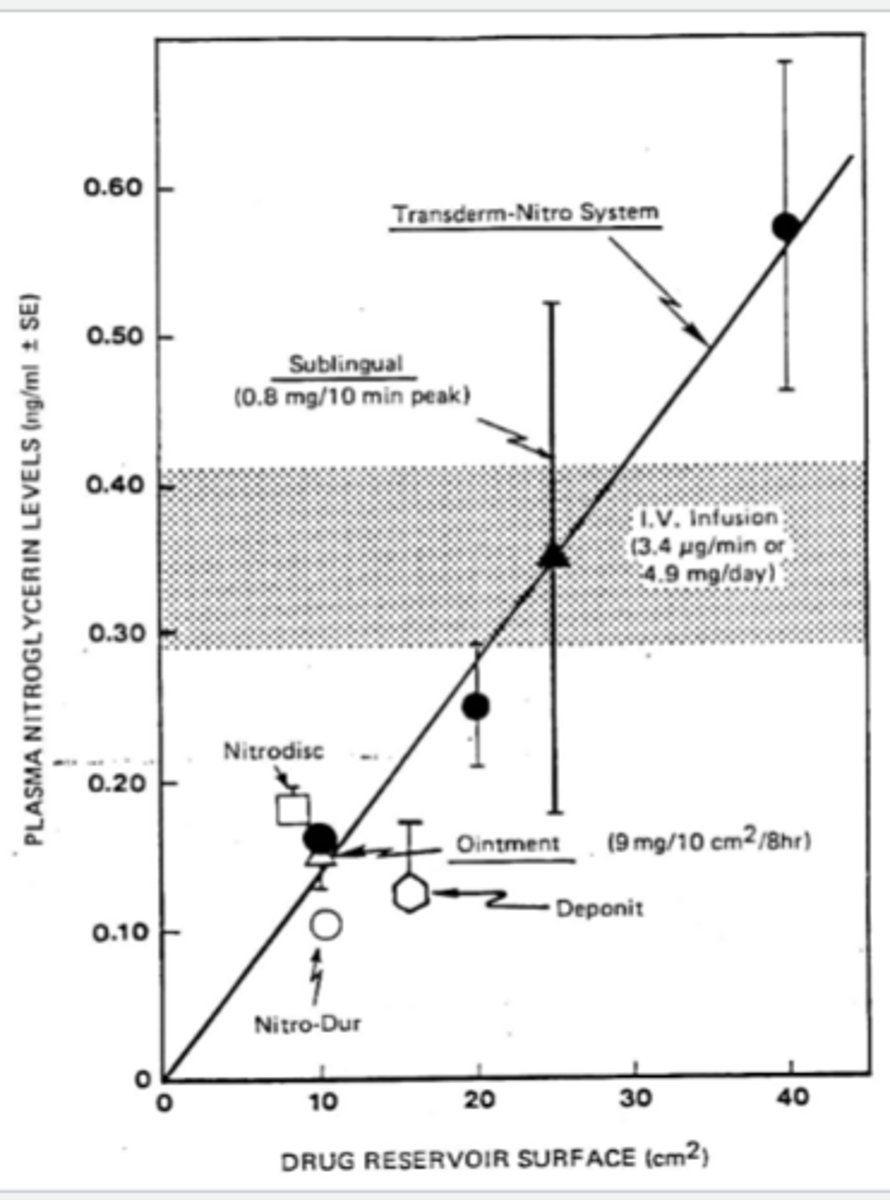
plasma nitroglycerin levels vs drug reservoir surface
- positive linear graph
- transderm-nitro system farthest right
- SL second farthest right
- ointment, Deponit, and ointment is the third most right
- Nitro-Dur is around the same region, but more below the line than the rest of them
release rate
- refers to how much drug is released over a certain time from the patch
permeation rate
- rate of diffusion of active across the skin membrane
in vitro testing of drug permeation
- using human or porcine skin membranes
- placed in glass diffusion cells called Franz cells
- donor compartment has formulation placed in it of known weight/drug concentration
- receptor sampled over time
- flux graph plotted
- skin kinetic parameters recorded
flux
- cumulative amount/unit time
skin kinetic parameters
- lag time
franz diffusion cells components
- donor compartment
- O-ring
- membrane
- receptor compartment with sampling port for analysis
- pinch clamp
- optimal water jacket
pinch clamp connects
- donor compartment
- O-ring
- membrane
- receptor compartment
in vivo testing of TDDS
- use larger animal
- best is weanling pig
- skin similar in thickness and lipid organization of stratum corneum to human
- patches often applied to the back of animal
in vivo technique of TDDS
- gauze pad with taped edges on top of window screen
- window screen on top of foam pad
- foam pad on clipped skin surface
Androderm TDDS
- testosterone for 24 hr application
- non-scrotal application: back, abdomen, thighs
- 2.5 or 5 mg
- drug dissolved in alcohol based gel: 12.2 mg for 2.5 mg TDDS
- alcohol, glycerin, glycerol mono-oleate, methyl laurate gelled with acrylic acid copolymer
- reservoir TDDS
Testoderm TDDS types of systems
- non-scrotal application
- scrotum with adhesive
- scrotum without adhesive
nonscrotal application Testoderm
- Testoderm TTS 5mg/day
- reservoir type TDDS
scrotum without adhesive Testoderm
- Testoderm 5 mg or 4 mg/day
- membrane-controlled TDDS
scrotum with adhesive Testoderm
- Testoderm wit adhesive 6 mg/day
- adhesive type TDDS
Fentanyl TDDS
- duragesic 25, 50, 75, and 100 mcg/hr
- only for chronic pain and NOT acute, so there are lag time issues
- opioid analgesic
- not to be used for postoperative analgesia
- used for 72 hr
- contains 0.1 mL alcohol USP per 10 cm^2
- time to attain Tmax is about 30-38 hr
- membrane controlled type reservoir TDDS OLD
- matrix type patch NOW
Ortho-Evra TDDS
- Ortho-McNeil contraceptive patch
- worn 1 week at a time for 3 consecutive weeks
- contains 6 mg norelgestromin and 0.75 mg ethinyl estradiol, releases 150 mcg and 20 mcg resp/24 hrs
- approved by FDA 11/20/01
- 20 cm^2 patch
- middle adhesive layer contains drugs embedded in matrix and a non-woven polyester fabric
amount of transdermal patch products approved in US over past 2 decades
- more than 35
transdermal product prescriptions
- have been used by about 12 million people worldwide for ailments ranging from bladder control to heart disease
marketed products
- Transderm Scop® (scopolamine)
- Duragesic® (fentanyl)
- Transderm-Nitro® (nitroglycerin)
- NicoDerm® CQ® (nicotine)
- Catapres-TTS® (clonidine)
- Testoderm® (testosterone)
- Estraderm® (estradiol)
- Oxytrol® (oxybutynin)
in 2006
- transdermal selegiline Emsam
- somerset pharmaceuticals, Inc.
- for parkinsonism and demetia
2007
- Transdermal Rotigotine (Neupro ®) - Schwartz Pharma for Parkinsonism and Restless Leg Syndrome
- Transdermal Rivastigmine (Exelon ®) - Novartis Pharmaceuticals Corp. For dementia.
- Transdermal Methylphenidate (Daytrana ®) - Shire Pharmaceuticals ADHD-attention deficit disorder
current needs
- patients prefer smaller discrete patches
- patches like to have longer lasting patches
- prefer not to have them visible
- need low irritation from polymer adhesive blends
- need to have no crystallization in patch over time [add polyvinyl pyrrolidone PVP)
the future; examples of recent advances
- novel adhesive polymers: improved wear Evra, a 7-day contraceptive patch
- electronics, miniaturization and skin permeation with electronically assisted GlucoWatch, a glucose monitor for diabetes control
- devices- Bioject and device/patch combinations
DOT Matrix Patch
- Noven Pharmaceuticals
- the acrylic serves only one purpose and it is to hold a lot of drug
- loaded with such high drug concentrations that it would never stick on its own
- it's then added with a silicone adhesive whose only job is to make the patch stick
- marketed small patches such as Vivelle Dot-convenient and effective therapeutically
challenge with skin delivery of larger MW agents
- over 500Da need active not passive delivery approaches
- electrically assisted
- mechanical methods
- miscellaneous
electrically assisted MW agents
- iontophoresis
- electroporation
mechanical MW methods
- microneedles
- abrasion
- needle-less injections
miscellaneous MW agents
- ultrasound, radiofrequency ablation, temperature, laser radiation and photomechanical waves, magnetophoresis
active transdermal delivery
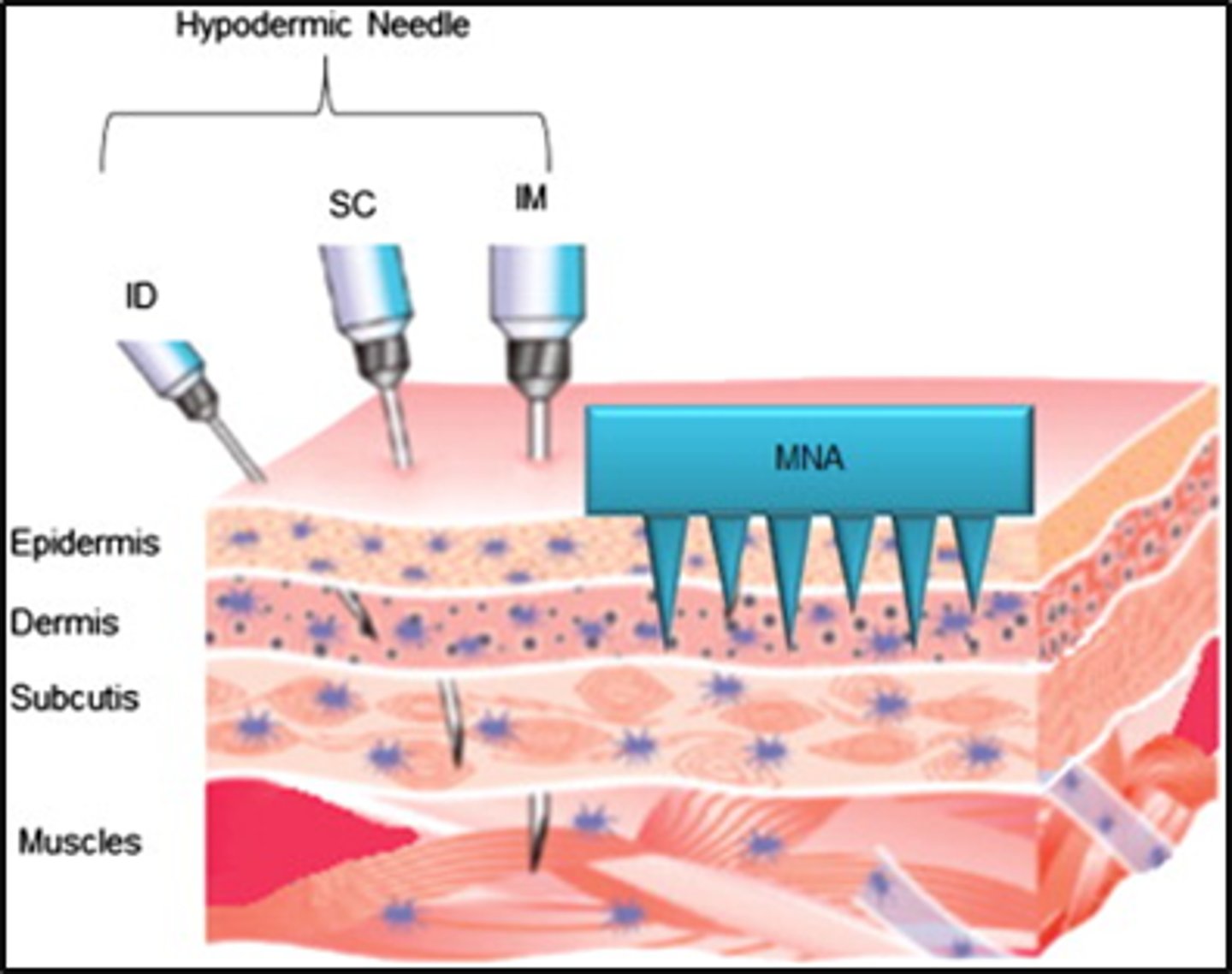
different types of microneedles
- solid
- coated
- dissolving
- hollow
solid microneedles
- silicon
- metal
- polymer
microneedle patches
- currently being explored as mechanisms to deliver vaccines and larger macromolecules
transdermal vaccine technology
- patch delivers vaccine antigen and adjuvant into the stratum corneum
iontophoresis
- a current passed between the active electrode and indifferent electrode repelling drug away from the active electrode and into the skin
electroporation
- also based on the application of a voltage to the skin
- in contrast to iontophoresis where a low voltage os applied, this requires a large voltage treatment for a short period of 10 mcseconds to 100 ms
- produces transient hydrophilic pores across skin barrier, which allow passage of macromolecules via a combination of diffusion, electrophoresis and electroosmosis
principles of electroporation
- short pulses of high voltage current are applied to the skin
- produces hydrophilic pores in the intracellular bilayers via momentary realignment of lipids
sonophoresis/ultrasound examples
- Dermisonics
- U-Strip Transdermal Drug delivery system
- Echo Therapeutics, Inc generation 1.5 and 2.0
more advanced technology becomes...
- more complicated to explain how to use device to administer a medication
thermal methods
- the use of controlled heat allows drugs to permeate the skin more effectively and efficiently than traditional methods
- controlled heat initiates several physiological responses that facilitate drug penetration through the skin
physiological responses that facilitate drug penetration through skin
- increase in skin permeability
- increase in body fluid circulation
- dilation of blood vessels, thus improving permeability through the blood vessel wall
- an improvement in the solubility of most drugs
- increase in the release rate of the drug, from local skin tissue into systemic circulation
Zars Pharma
- thermal method
- control heat-assisted drug delivery CHADD
CHADD
- consists of a powder-filled pouch laminated between a top cover film with oxygen-regulating holes and a bottom film with a pressure-sensitive adhesive layer
- upon contact with oxygen in ambient air, a chemical reaction occurs in the heat-generating medium
- after an initial rise in temperature, the temp generated by the CHADD unit will reach and remain within the controlled temperature range for a pre-determined period of time
- when the heat generating medium is exhausted, the skin temperature gradually returns to baseline
altea therapeutics PassPort system
- single-use disposable PassPort Patch
- Re-usable handheld Applicator
- a conventional transdermal patch attached to an array of metallic filaments 'porator'
how the PassPort system works
- pressing the activation button of the Applicator releases a single pulse of electrical energy to the porator, where it is converted into thermal energy
- the rapid conduction f this thermal energy into the surface of the skin painlessly ablates the stratum corneum under each filament to reate microchannels
- when the applicator is removed, a simple fold-over design aligns the transdermal patch with the newly formed microchannels
Reverse Iontophoresis and Closed Loop Systems
- Animas Johnson and Johnson
- GlucoWatch G2
Crospon and Hewlett Packard
- technology hybrids and micro systems
- one patch contains about 150 microneedles
- enables the drug's dose and time of delivering to be controlled by a microchip
- the patch also contains 400 cylindrical reservoirs
Chrono Therapeutics
- programmable, passive, multidose transdermal systems
- automated and variable delivery profiles
- drug available when it is needed
- improved patient compliance
example of programmable, passive, multidose transdermal systems
- morning dip
- normal lung function undergoes circadian changes and reaches a low point in the early morning hours
substances with proven chrono pharmacological efficiency
- integrated into a miniaturized, automated, programmable watch-like device
user can pre-program the times and amount of each dosage by
- precisely controlling the amount of drug exposed to the skin during each dosing
- device stops diffusion, consequently dosing
other considerations
- manufacturability
- regulatory paths
- insurance coverage
- marketability
manufacturability
- medical device, pharmaceutical or both
- cost
- easy to make, assemble, QA/QC and package
Regulatory paths
- 510K or NDA 505[b][2]
- FDA Office of Combination Products
Marketability
- it is easy for the patient to understand and use
- is it affordable
conclusions
- transdermals are still a multibillion dollar industry and a viable drug delivery platform technology
- key is to cross the stratum corneum and deliver enough flux across the skin
- traditional passive delivery patches are the mainstay of the industry
- novel techniques are constantly being invented and developed for transdermal drug delivery
- almost all are a combination of active and passive skin permeation technologies
the conclusion in red
- transdermal does not necessarily mean a patch system
- can have transdermal gels or invisible patches, like testosterone or nitroglycerin