Chapter 18 Chemistry: Metabolic Pathways and ATP Production
1/85
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
86 Terms
Metabolism
involves
all chemical reactions that provide energy and substances needed for growth
Catabolic reactions
anabolic reaction
Catabolic reactions
break down large, complex molecules to provide energy and smaller molecules
Anabolic reactions
Use ATO energy to build up larger molecules
Stages of Metabolism
Catabolic reactions are organized in stages.
Stage 1: Digestion and hydrolysis
Stage 2: Degradation
Stage 3: Oxidation
Stage 1: Digestion and hydrolysis
break down large molecules to smaller ones that enter the blood stream
Stage 2: Degradation
breaks down molecules to two- and three-carbon compounds
Stage 3: Oxidation
oxidation of small molecules in the citric acid cycle and electron transport provides ATP energy
Cell membrane
Separates the contents of a cell from the external environment and contains structures that communicate with other cells
Cytoplasm
Consists of the cellular contents between the cell membrane and nucleus
Cytosol
Fluid part of the cytoplasm that contains enzymes for many of the cell’s chemical reactions
Mitochondrion
Contains the structures for the synthesis of ATP from energy-releasing reactions
Nucleus
Contains genetic information for the replication of DNA and the synthesis of protein
Ribosome
Site of protein synthesis using mRNA templates
How is energy stored in the body?
as adenosine triphosphate (ATP)
When does ATP hydrolysis happen?
Every time we contract muscles, move substances across cellular membranes, send nerve signals, or synthesize an enzyme, we use the energy from it.
Hydrolysis of ATP
The hydrolysis of ATP to ADP (adenosine diphosphate) releases 7.3 kilocalories per mole of ATP.
ATP —> ADP + Pi + 7.3 kcal/mole
Hydrolysis of ADP
ADP can also hydrolyze to form adenosine monophosphate (AMP) and an inorganic phosphate i (P)
ADP —> AMP + Pi + 7.3 kcal/mole
ATP
the energy-storage molecule, links energy-producing reactions with energy-requiring reactions in the cells
Digestion of carbohydrates in mouth
Enzymes produced in the salivary glands hydrolyze some of the a -glycosidic bonds in amylose and amylopectin, producing smaller polysaccharides such as
maltose
glucose
dextrins (may contain 3-8 glucose units)
What happens after the partially digested starches are swallowed?
starches enter the acidic environment of the stomach, where the low pH stops carbohydrate digestion.
Digestion of carbohydrates in the small intestine
In the small intestine, which has a pH of about 8,
enzymes produced in the pancreas hydrolyze the remaining dextrins to maltose and glucose
enzymes produced in the mucosal cells that line the small intestine hydrolyze maltose as well as lactose and sucrose into monosaccharides
What happens to the monosaccharides after digestion in the small intestine?
Monosaccharides are then absorbed through the intestinal wall to the blood stream and carried to the liver, where any fructose and galactose are converted to glucose.
Digestion
Carbohydrates begin digestion in the mouth, lipids in the small intestine, and proteins in the stomach and small intestine.
Stage 1 of catabolism in carbohydrates
the digestion of carbohydrates begins in the mouth and is completed in the small intestine
Digestion of fats (triacylglycerols)
begins in the small intestine, where bile salts break fat globules into smaller particles called micelles
What happens in the digestion of triacylglycerols
The triacylglycerols are hydrolyzed in the small intestine and re-formed in the intestinal lining, where they bind to proteins for transport through the lymphatic system and bloodstream to the cells.
Digestion of proteins
begins in the stomach, where HCl at pH 2 denatures proteins and activates enzymes such as pepsin to hydrolyze peptide bonds
moves out of the stomach to the small intestine, where trypsin and chymotrypsin hydrolyze the polypeptides to amino acids
ends as amino acids are absorbed through the intestinal walls and enter the bloodstream for transport to the cells
Where are proteins hydrolyzed?
in the stomach and the small intestine
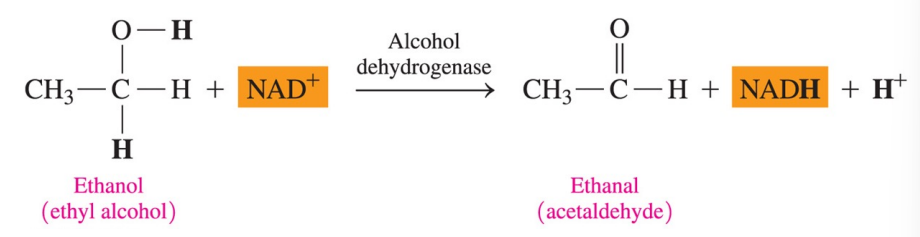
Coenzymes in Metabolic Pathways
The NAD+ coenzyme is required for metabolic reactions that produce carbon-oxygen (C=O) double bonds, such as in the oxidation of alcohols to aldehydes and ketones.
Oxidation reactions
involve
a loss of hydrogen
a loss of electrons
and increase in number of bonds to hydrogen
Reduction reactions
involve
the gain of hydrogen ions and electrons
a decrease in the number of bonds to oxygen
Coenzyme NAD+
NAD+ (nicotinamide adenine dinucleotide) is an important coenzyme in which the B3 vitamin, niacin, provides the nicotinamide group, which is bonded to ADP
What does the coenzyme NAD+ participates in?
the reaction that produce a carbon-oxygen double bonds (C=O)
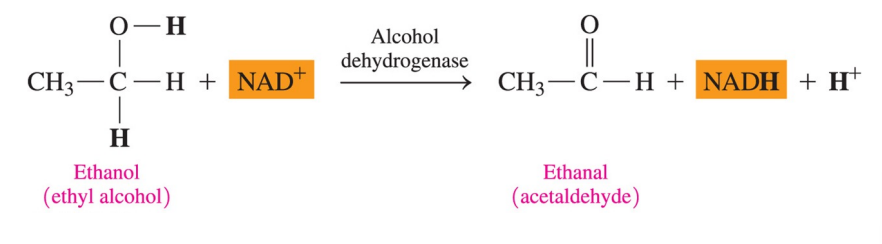
Coenzyme FAD
FAD (flavin adenine dinucleotide) is a coenzyme that
contains ADP and riboflavin (vitamin B2 )
is reduced to FADH2 when flavin accepts 2H+ and 2e-
What does the coenzyme FAD participates in?
in reactions that convert a carbon–carbon single bond to a carbon–carbon double bond (C=C)
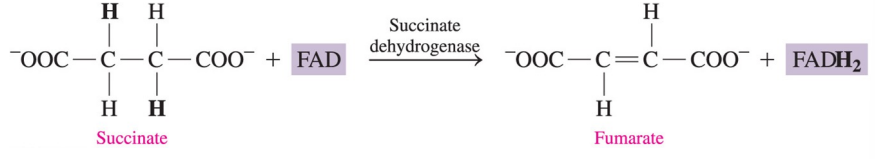
How is FAD used in the citric acid cycle?
in the conversion of the carbon–carbon single bond in succinate to a double bond in fumarate
Structure of Coenzyme A
Coenzyme A (CoA) contains pantothenic acid (vitamin B5 ) phosphorylated ADP, and aminoethanethiol.
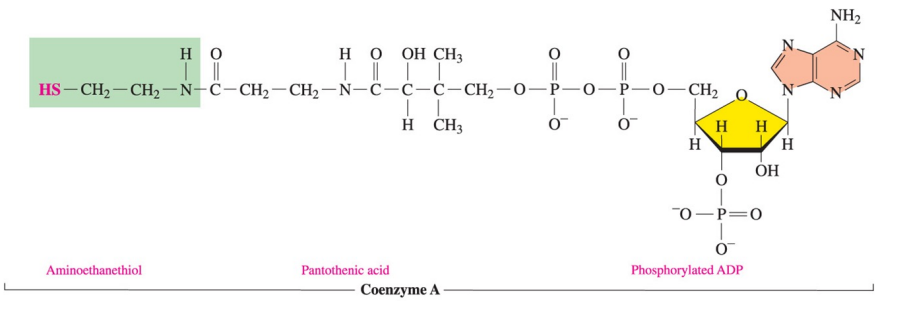
Function of coenzyme A
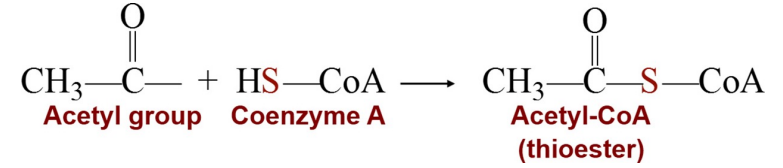
is to prepare small acyl groups (represented by the letter A in the name), such as acetyl, for reactions with enzymes.
The thiol group ( SH) -- bonds to a two-carbon acetyl group to produce the energy-rich thioester acetyl-CoA
Glycolysis
is a metabolic pathway that uses glucose, a digestion product from carbohydrates
degrades six-carbon glucose molecules to three-carbon pyruvate molecules
Where does glycolysis take place?
in the cytoplasm of the cell
What kind of process is glycolysis?
is an anaerobic process: no oxygen is required
In reaction 1 to 5 of glycolysis,
energy is required to add phosphate groups to glucose
glucose is converted to two three-carbon molecules
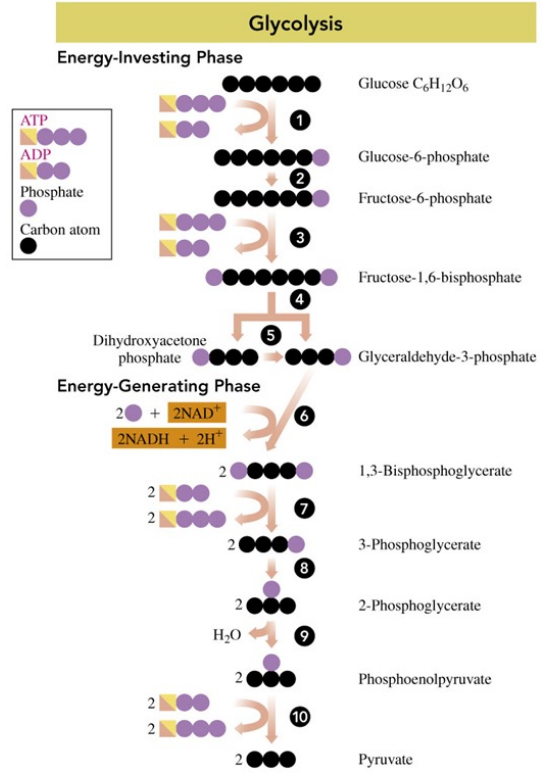
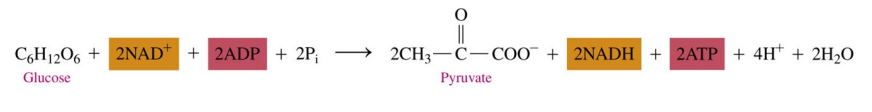
In glycolysis,
two ATP add phosphate to glucose and fructose-1,6- bisphosphate
four ATPs are produced during phosphate transfers
there is a net gain of two ATP and two NADH when glucose is converted to two pyruvate
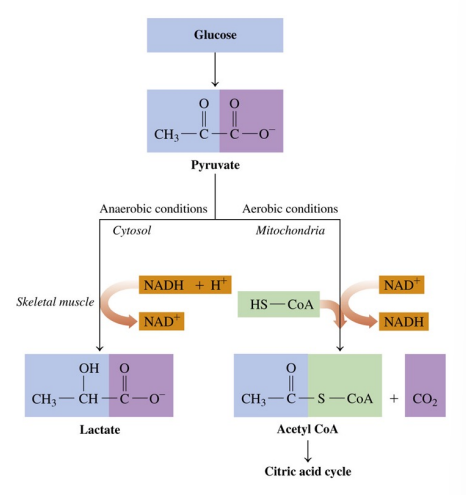
Pyruvate Pathways: Aerobic and Anaerobic
Pyruvate is converted to acetyl CoA under aerobic conditions and to lactate under anaerobic conditions
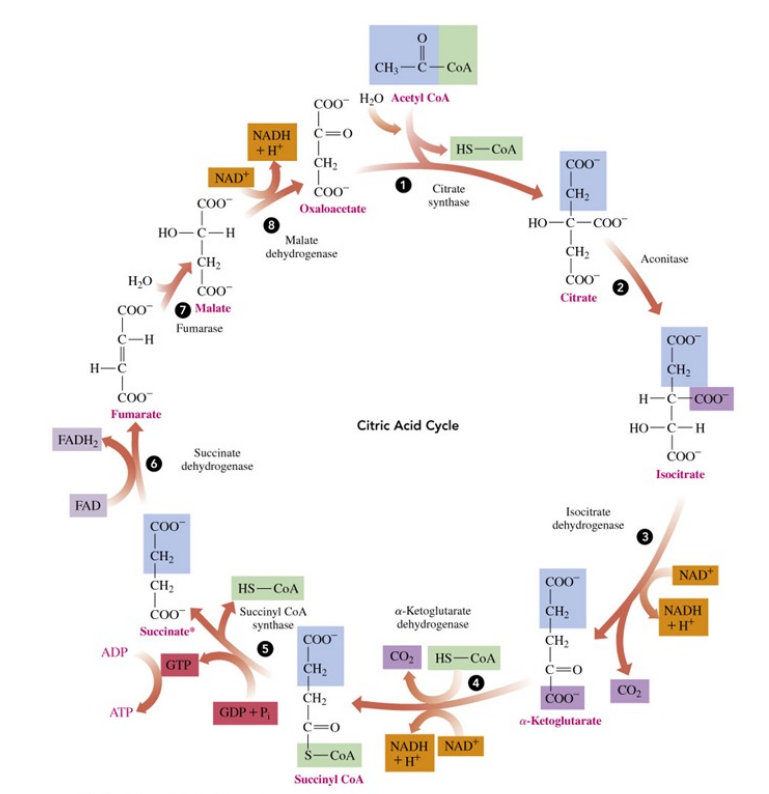
The Citric Acid Cycle
is a series of reactions that connects the intermediate acetyl-CoA from the metabolic pathways in stages 1 and 2 with electron transport and the synthesis of ATP in stage 3.
citric acid cycle components
uses the two-carbon acetyl group in acetyl CoA to produce CO2 , NADH+ H+ , and FADH2
What is the Citric Acid Cycle named after?
the citrate ion from citric acid (CHO , 68 7 ) a tricarboxylic acid, which forms in the first reaction
is also known as the tricarboxylic acid (TCA) cycle or the Krebs cycle
Citric Acid Cycle process
an acetyl group (2C) in acetyl CoA bonds to oxaloacetate (4C) to form citrate (6C)
two decarboxylation reactions remove carbon atoms as C O2 molecules to give succinyl-CoA (4C)
a series of reactions converts four-carbon succinyl-CoA to oxaloacetate, which combines with another acetyl-CoA, and the citric cycle starts all over
Citric Acid cycle: overall reaction
one complete citric acid cycle
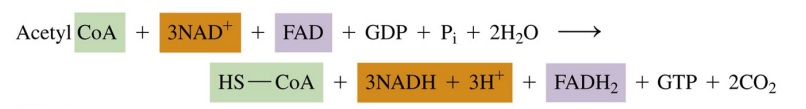
Electron transport chain
hydrogen ions and electrons from NADH and FADH2 are passed from one electron carrier to the next and combine with oxygen to make H O.
What is the energy released during electron transport used for?
to synthesize ATP from ADP and Pi , a process called oxidative phosphorylation.
A mitochondrion contains
an outer membrane, an intermembrane space, and a highly folded inner membrane that surrounds the matrix
enzymes and electron carriers along the inner membrane required for electron transport
four distinct protein complexes—complex I, II, III, and IV — located within these membranes
Two electron carriers, coenzyme Q and cytochrome c,
are firmly attached to the membrane
function as mobile carriers shuttling electrons between the protein complexes that are bound to the inner membrane
Electron carriers
Coenzymes NADH and FADH2 are oxidized in enzyme complexes providing electrons and hydrogen ions for ATP synthesis
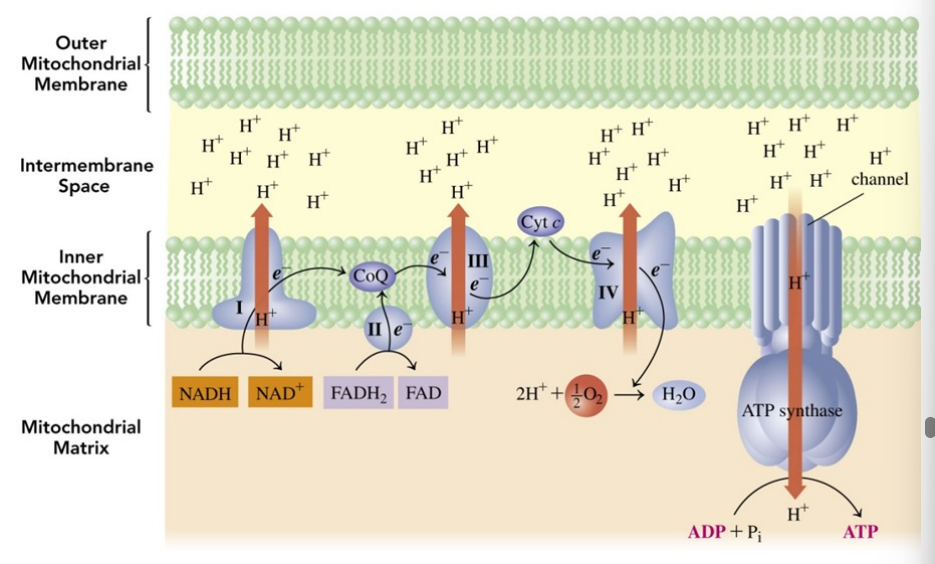
Oxidation phosphorylation in the chemiosmotic model
each complex acts as a proton pump by pushing H+ ions from the oxidation of NADH and FADH2 out of the matrix and into the intermembrane space
the increase in H+ concentration lowers the pH and creates an H+ or electrochemical gradient
What is done to equalize the pH and charge in oxidative phosphorylation?
to equalize the pH and charge between the intermembrane space and the matrix, the + H ions return to the matrix by passing through a protein complex called ATP synthase
The process of oxidative phosphorylation
couples the energy from electron transport to the synthesis of ATP from ADP.
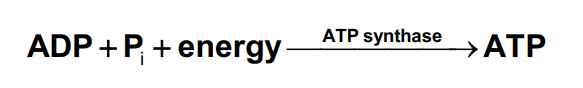
What happens to NADH during ATP synthase?
When NADH enters electron transport at complex I, the energy released from its oxidation is used to synthesize 2.5 ATP

What happens to FADH2 during ATP synthesis?
FADH2 enters electron transport at complex II, which is at a lower energy level. Thus, FADH2 provides energy to produce 1.5 ATP

ATP from Glycolysis
Glycolysis yields a total of seven ATP:
five ATP from two NADH
two ATP from direct phosphorylation

ATP from Oxidation of two pyruvates
Under aerobic conditions,
two pyruvate enter the mitochondria and are oxidized to two acetyl-CoA, CO2 , and two NADH
two NADH enter electron transport to provide five ATP
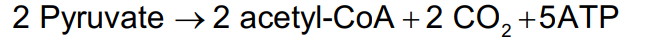
ATP from the Citric Acid Cycle
One turn of the citric acid cycle provides
3 NADH x 2.5 ATP/NADH = 7.5 ATP
1 FADH x 1.5 ATP/FADH2 = 1.5 ATP
1 GTP x 1 ATP/1 GTP = 1 ATP
Total = 10 ATP
Because each glucose provides two acetyl-CoA, two turns of the citric acid cycle produce 20 ATP

The complete oxidation of glucose yield
The complete oxidation of glucose to C2O and H2O yields a total of 32 ATP.
Oxidation of fatty acids
A large amount of energy is obtained when fatty acids undergo oxidation in the mitochondria to yield acetyl-CoA.
Stage 2 of fat metabolism
Fatty acids undergo beta-oxidation (β - oxidation), which removes two-carbon segments, one at a time, from the carboxyl end
Each cycle in oxidation produces acetyl-CoA and a fatty acid that is shorter by two carbons
Fatty acid activation
prepares fatty acids for transport through the inner membrane of mitochondria
combines a fatty acid with coenzyme A to yield fatty acyl-CoA
What does fatty acid activation require?
energy obtained from hydrolysis of ATP to give AMP and 2Pi
Reactions of the β - oxidation cycle
fatty acyl-CoA molecules undergo β oxidation, a cycle of four reactions converting the β - carbon —CH2— to a β -keto
once the β -keto group is formed, a two-carbon acetyl group can be split from the carbon chain, shortening the fatty acyl chain
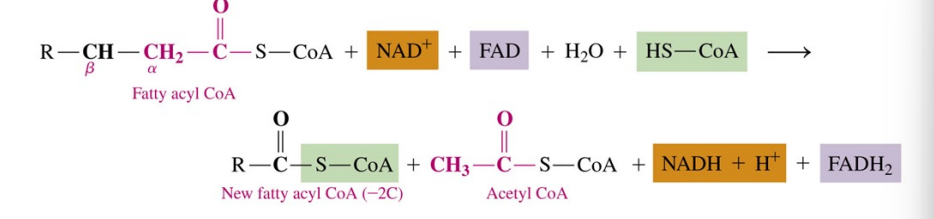
Cycles of β -Oxidation
The number of β -oxidation cycles
depends on the length of a fatty acid
is one less than the number of acetyl- CoA groups formed
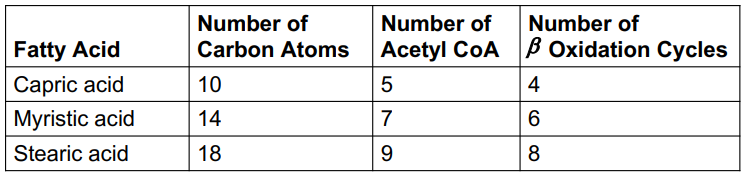
ATP from Fatty Acid Oxidation
In each β -oxidation cycle,
one NADH is produced, generating 2.5 ATP
one FADH2 is produced, generating 1.5 ATP
one acetyl-CoA is produced, generating 10 ATP
What happens if carbohydrates are not available?
the body breaks down to meet energy needs
ketone bodies
How do ketone bodies form?
in a process called ketogenesis
In ketogenesis
acetyl-CoA molecules combine to produce ketone bodies: acetoacetate, β - hydroxybutyrate, and acetone
Formation of Ketone bodies
ketone bodies form
in large amounts of acetyl-CoA accumulate
when two acetyl-CoA molecules form acetoacetyl-CoA
when acetoacetyl-CoA hydrolyzes to acetoacetate
when acetoacetate reduces to β - hydroxybutyrate or loses CO2 to form acetone, both ketone bodies
When does ketosis occur
in diabetes, diets high in fat, and starvation
as ketone bodies accumulate
when acidic ketone bodies lower blood pH below 7.4 (acidosis)
Leptin
hormone that is produced in fat cells
When fat stores are low leptin,
production decreases, which signals the brain to increase food intake
acts on the liver and skeletal muscles, where it stimulates fatty acid oxidation in the mitochondria, which decreases fat storage
When fat cells are full,
high levels of leptin signal the brain to limit the intake of food
What do proteins provide?
energy when carbohydrates and lipids resources are not available
In what processes are the carbon atoms from amino acids used?
in the citric acid cycle
for the synthesis of fatty acids, ketone bodies, and glucose
What are most of the amino groups converted into?
urea
Transamination
amino acids are degraded onto the liver
an amino group is transferred from an amino acid to an ɑ- keto acid, usually ɑ- ketoglutarate
a new amino acid and ɑ- keto acid are formed
when alanine combines with ɑ- ketoglutarate, pyruvate and glutamate are produced
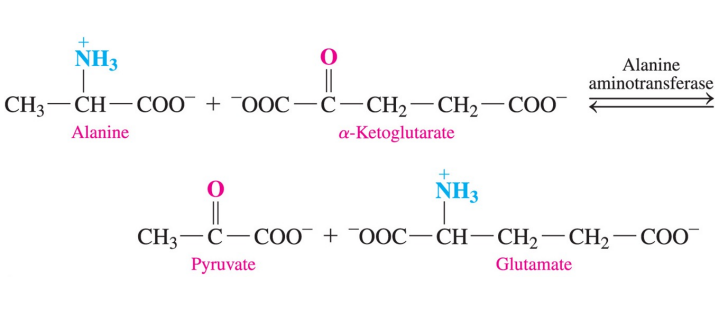
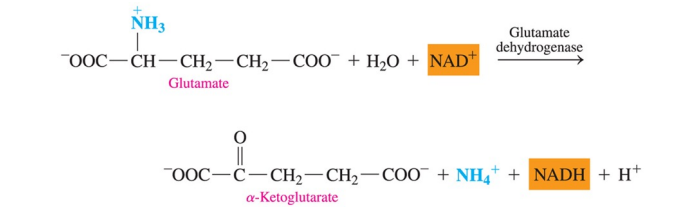
Oxidative deamination
removes the ammonium group ( —NH3+ ) from glutamate as an ammonium ion, NH4+ , and provides hydrogens for the NAD+ coenzyme
regenerates ɑ- ketoglutarate, which can enter transamination with an amino acid
The urea cycle
removes toxic ammonium ions from amino acid degradation
converts ammonium ions to urea in the liver
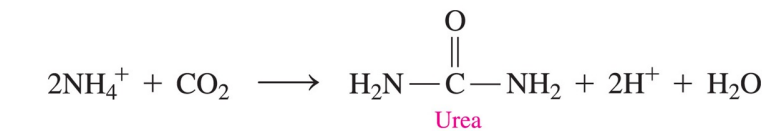
What does the urea cycle produce?
produces 25–30 grams of urea daily for excretion in the urine
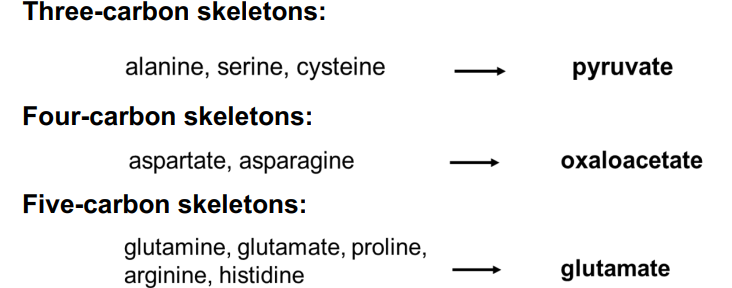
Carbon skeletons of amino acids
form intermediates of the citric acid cycle
produce energy
enter the citric acid cycle at different places depending on the amino acid