PChem II Final
1/90
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
91 Terms
Combustion reactions are examples of ______ processes
exothermic
The decomposition of stable compounds tends to be an ______ process
endothermic
Does it take energy to break chemical bonds?
Yes
Is energy released when a chemical bond is broken?
No
Does it take energy to form a chemical bond between two atoms?
No
Is energy released when a chemical bond is formed?
Yes
Where does bond formation lie on the potential energy graph
the minimum
Potential Energy is transformed into _____ energy when a bond is formed
kinetic
Are A-A bonds or A-B bonds stronger?
A-B bonds
How do you calculate ΔHrxn
Bonds broken-bonds formed
The more oxygen required during combustion of burning fuel the ____ energy will be released
more
predicting whether a chemical reaction will be product-favored or reactant-favored is
reaction directionality
Reaction directionality is affected by
potential energy and number of configurations
The most favored state will have
Lower potential energy and higher number of configurations
Potential energy is a _____ factor
energetic
Number of configurations is an ______ factor
entropic
A molecule is more energetically stable if it has a _____ potential energy
lower
Lower potential energy is correlated with _____ distances
shorter
Lower potential energy is correlated with _____ attractions
stronger
Compounds with strong bonds are ____ energetically stable
more
longer bonds are _____ than shorter bonds
weaker
______ center’s are negative with high electron density
nucleophilic
______ center’s are positive or have a low electron density
electrophillic
species that have a pair of electrons that can be shared to form a new chemical bond
nucleophiles
species that are capable of accepting the pair of electrons from the nucleophile
electrophiles
If a species is negative it is a
nucleophile
If a species is positive it is an
electrophile
The _____ moves closer and attacks the _____
nucleophile, electrophile
Negative charges, lone pairs, and pi bonds are all features of
nucleophiles
carbocation, electronegative atoms, and carbonyls are all features of
electrophiles
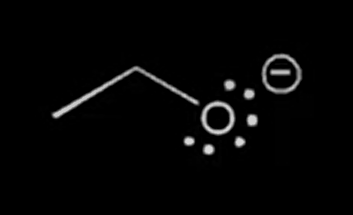
nucleophile
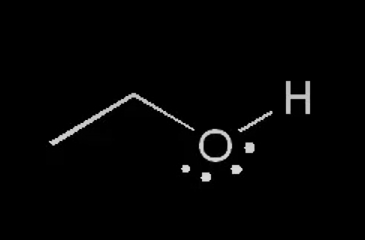
nucleophile
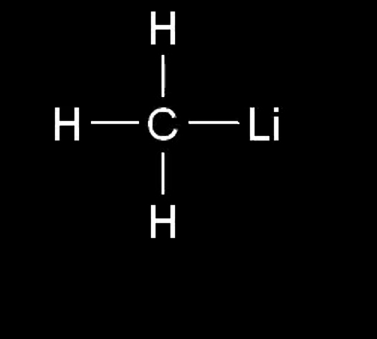
what is the nucleophilic center
carbon
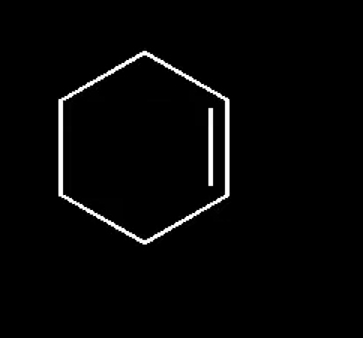
where is the nucleophilic center
pi bond
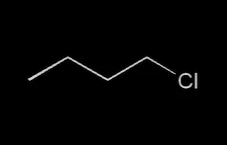
where is the electrophilic center
carbon attached to Cl
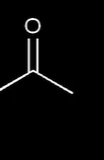
where is the electrophilic center
carbon attachted to O
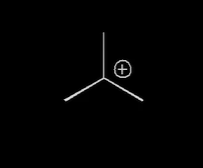
where is the electrophilic center
center carbon
an arrow origination on a lone pair is a
bond forming reaction
an arrow originating from a bond is a
bond breaking reaction
compounds with the same formula but different structures
isomers
carbons with four different groups attached to them are
chiral centers
all amino acids except _____ are chiral
glycine
equal mixtures of enantiomers
racemic mixture
in a racemic mixture often only one enantiomer is _____; the other one can be ______
clinically active, inert
an example of harmful racemic mixtures where one enantiomer is dangerous would be
thalidomide
a molecule wit hthe same molecular formula but different connectivity is a
consituational isomer
C4H10 and CH3CH2CH2CH3 are examples of
constitutional isomers
the _____ the number of carbons the ____ the number of isomers
greater,greater
a _____ temperature is required to boil the less branches molecules
higher
Cis isomers will have a _____ boiling point than trans isomers
greater
fats without a bend
trans
fats with a bend
cis
double bonds are stiffer so they are
flat
reaction where we are adding something to a molecule
addition reaction
a reaction where we are swapping out a functional group from a molecule
substitution reaction
in an alkene addition reaction, X and Y have _____ electronegativity
different
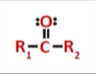
ketone
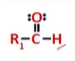
aldehyde
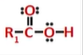
carboxylic acid
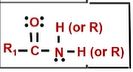
amide
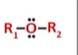
ether
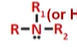
amine
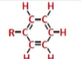
phenyl

alcohol
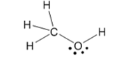
alcohol
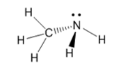
amine
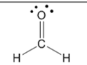
aldehyde
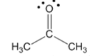
ketone
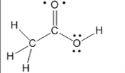
carboxylic acid
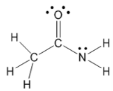
amide
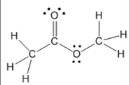
ester
the less stable the _____ reactive the compound will be
more
the most likely location to add an electronegative ion will be the more _____ carbon
subsituted
the double bond usually _____ during an addition reaction
breaks
the atom within a molecule bonded to the highest number of other carbon atoms rather than hydrogens
most substituted carbon
a two-step subsitution reaction where a leaving group departs before the nucleophile attacks, forming a carbocation intermediate
SN1 reaction
a one step subsitution reaction where a nucleophile attacks a substrate and a leaving group departs
SN2
the bulkier the electrophile the _____ for the nucleophile to attack
harder
if there are fewer R groups there are on the carbon that you need to bond to, the reaction will be
SN2
if there are more R groups that are in the way of the carbon, the reaction will be
SN1
the chirality of an electrophile is inverted in a ____ reaction
SN2
both the left handed and right handed product are created in ____ reactions
SN1
rate law for SN2 reactions
k[Nu][R-LG]
rate law for SN1 reactions
k[R-LG]
SN2 reactions are ___ order
2nd
SN1 is ___ order
1st
a sample containing equal amounts of left handed and right handed ciral molecules
racemic mixture
which reaction results in racemization?
SN1
the slowing or prevention of reactions caused by the physical bulk of atoms or groups within a molecule
steric hinderance
in the context of SN2 reactions, _____ reactions will go quicker because of steric hinderance
primary
in the context of SN1 reactions, ______ reactions will go quicker
tertiary