Unit 2 Matter and Energy
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Matter
anything that has mass and takes up space
gas
has the most energy but the least attraction between particles, no definite shape, and no definite volume
liquid
less energy and attraction between particles than a gas but more than a solid, no definite shape, and has a definite volume
solid
has the least energy but the most attraction between particles, and has both a definite shape and a definite volume
melting
solid to liquid
freezing
liquid to solid
condensing
gas to liquid
evaportaion/boiling
liquid to gas
subliming
solid to gas
depositing
gas to solid
pure substance
made of one species that cannot be separated by a physical means
elements
one type of atom
diatomic elements
H2, O2, N2, Cl2, Br2, I2, F2
compounds
two or more types of atoms that are chemically bonded together
mixtures
made of two or more pure substances that are not chemically bonded
homogeneous mixtures
the same throughout
heterogeneous mixtures
not the same throughout, phases are visable
ways that chemical properties can identify substances
flammable, explosive, reacts with water, oxidizes, and corrosive
chemical change
whenever a substance undergoes a chemical rxn
energy
the ability to do work
kinetic energy
energy due to motion
potential energy
energy due to position, composition, and condition
thermal energy
heat flows from a hot object to a cool one
specific heat
the energy required to raise the temperature of exactly 1g of substance 1C
equation for specific heat
q=mC(delta)T
equation for calorimetry
-(mC(delta)T)=(mC(delta)T)
endothermic reactions
heat flows into the reactants
exothermic reactions
heat flows out of the reactants
how does energy move in odd numbers in a heating curve
kinetic increases while potential stays constant
how does energy move in even numbers in a heating curve
kinetic stays constant while potential increases
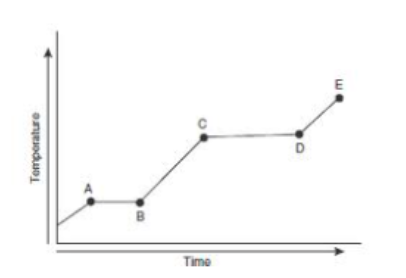
where is the melting point in this heating curve
point B
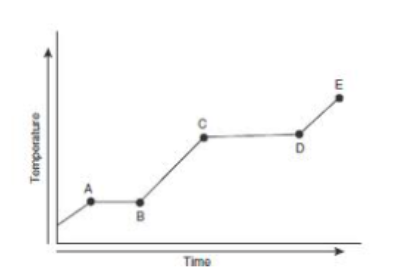
where is the boiling point in this heating curve
point D
triple point
where all three phases exist in equilibrium
critical point
where the liquid and gas exists as a supercritical fluid
supercritical fluid
any point past the critical point
melting line
the line between solid and liquid
boiling line
the line between liquid and gas
what is an alloy
a mixture of two metals
what is smoke
a colloid
extensive
dependent on matter
intensive
independent of matter
solutions
homogeneous