4.7- Amino acids, Peptides and Proteins
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
What functional groups do amino acids contain?
Amine (NH2) and Carboxyl (COOH)
What is the naturally occurring formula for amino acids?
H2NCH(R)COOH
What does the R represent?
The variable group
What do amino acids have?
A chiral centre
Name H2NCH(C2H5)COOH
2-aminobutanoic acid
Name H2NCH(C3H7)COOH
2-aminopentanoic acid
How would you draw 2-amino-3-phenyl propanoic acid?
C6H5CH2CH(NH2)COOH
What can amino acids form and therefore act as?
Zwitter ions, can act as acids and bases (pH buffer)
At what pH do zwitter ions exist?
Isoelectric point
What may different isoelectric points be because of?
Different variable groups
What structure will HOCH2CH(NH2)COOH have in a low pH?
HOH2CCH(NH3+)COOH
At the isoelectric point?
HOH2CCH(NH3+)COO-
High pH?
HOH2CCH(NH2)COO-
Explain how they act as zwitter ions?
There is an internal transfer of a hydrogen ion from the -COOH group to the -NH2 group to leave an ion with both a positive and negative charge
Does a zwitter ion have an overall electrical charge?
No
How do amino acids have a high melting points and are solids?
Because of the ionic electrostatic forces of attraction between the zwitter ions
How are they more soluble?
Ion-dipole interactions between the zwitter ions and the water molecules
What happens when amino acids react with strong acids such as HCl?
They will form a salt
Eg?
-Cl+H3NCH(R)COOH
What will happen when an amino acid reacts with NaOH?
A salt and water will form
H2NCH(R)COOH + NaOH ->
H2NCH(R)COO-Na+ + H2O
Amino acid + alcohol?
Will form an ester and water
What in the presence of?
Concentrated sulfuric acid
Equation?
H2NCH2COOH + C2H5OH + H+ -> +H3NCH2COOC2H5 +H2O
What are amides the product of?
The reactions of acid chlorides with ammonia and amines
What can two amino acids react with each other to form?
A dipeptide
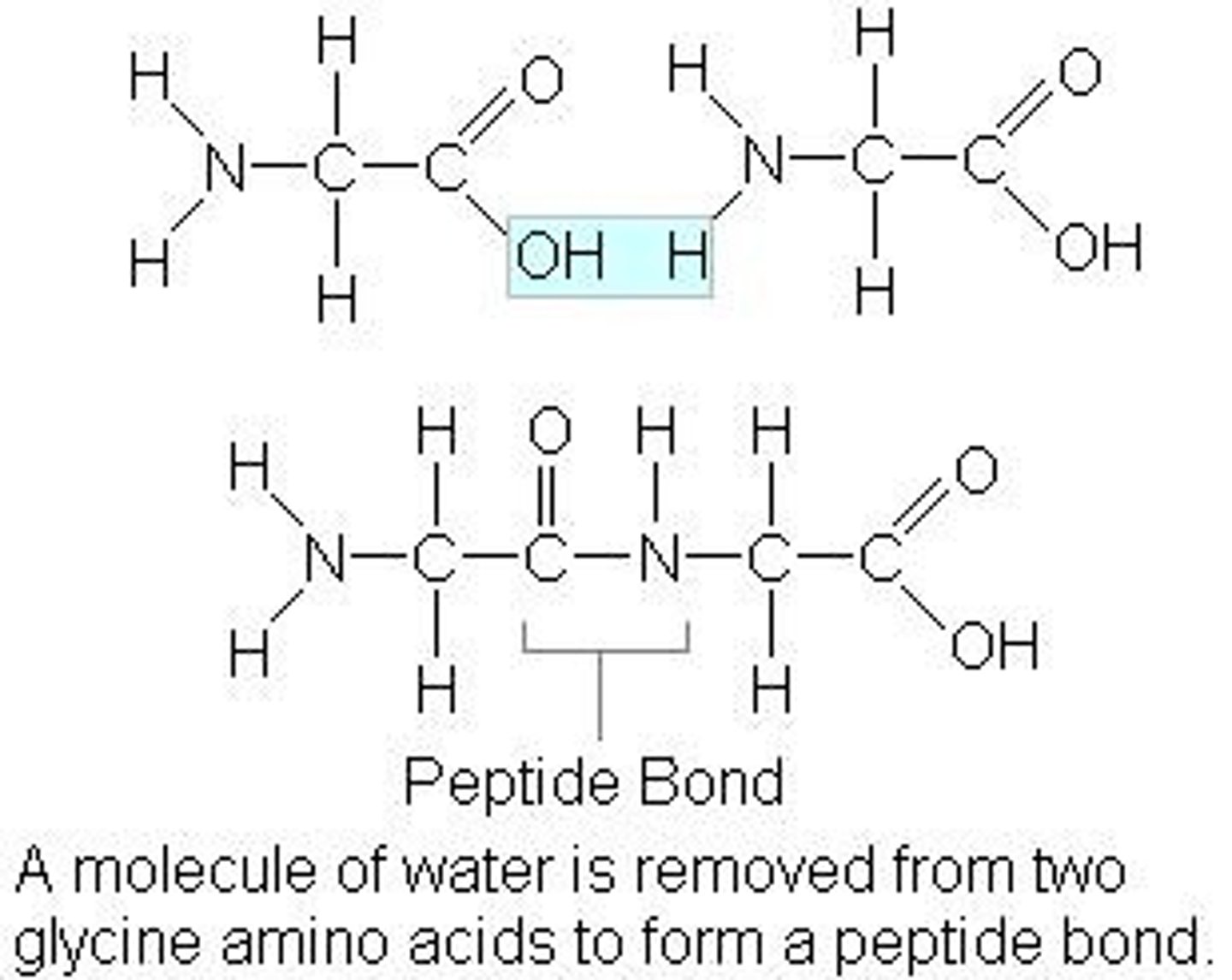
What is eliminated?
A water molecule
What is this type of reaction called?
A condensation reaction
What does the bond look like?
-C=O -N-H -
What can amino acids form?
Polypeptide chain
How to convert drawing of amino acid to polymer?
H2NCH(R)COOH -> [-NH -CH(R) -C=O-]
How can the peptide link be broken?
By heating under reflux in strong acid or strong alkali
H2NCH(R)CONHCH(R)COOH + H2O ->
H2NCH(R)COOH + H2NCH(R)COOH
What would happen in acidic conditions?
+H3NCH(R)COOH + +H3NCH(R)COOH
amino acids are produced in their cationic form
What happens in alkaline hydrolysis?
They are broken down to form carboxylates (eg their sodium salts) by heating with aqueous alkali
Define primary structure
The sequence of amino acids in a polypeptide chain
What bonds are in the primary structure?
Peptide
Define secondary structure
The twisting and folding of a polypeptide chain into an alpha helix or a beta pleated sheet. Held by peptide and hydrogen bonds
What are the hydrogen bonds between?
The N-H hydrogen atom of an amide group and the C=O oxygen atom of another amino acid
What is the beta pleated sheet stabilised by?
Hydrogen bonds between amino acids in different polypeptide chains
Define tertiary structure
further folding and coiling of the secondary structure to give a specific 3D shape. Held in place by ionic, disulphide, hydrogen bonds and hydrophobic interactions.
How are proteins used in living systems?
Biological catalysts eg. amylase catalyses the hydrolysis of starch into sugars
Structural function in living organisms eg. keratin in hair
Hormones eg insulin in blood glucose regulation