Chemistry Week 3
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
Periodic Table Group
Vertical columns on the periodic table
All elements have same number of valance electrons and core charge
Elements inside a group have different properties but due to their identical number of valance electrons react similarly
Core charge
Calculated by Z value - core electrons
Periodic Table Period
Horizontal rows
All elements have the same number of core electrons and shells
If shell empty the shell disappears
Octet Rule
Elements react in a way that gives them 8 valance electrons
8 valance electrons is identical to a noble gas
This rule only applies to C, N, O, and F
All the rest of the element try to match the valance electrons of the nearest noble gas
Ionic Compounds
Type of bionic compound
Always involves a Cation and Anion
Always between a metal and non-metal
Electrons are transferred from 1 element to the other
Metals tend to loose their electrons
Nonmetals tend to gain their electrons
Electrostatic Force
Part of ionic bonding
Attractive or repulsive force between two electrical charged objects
Lattice Energy
Part of ionic bonding
Energy contained in a crystal lattice
Coulomb’s Law
Used to determine attraction between two ions
If formula results in negative number the reaction will happen spontaneously
r = size of the atom, found by looking at how many electron shells it has
q1 = Charge of the first ion in the bond
q2 = Charge of the second ion in the bond
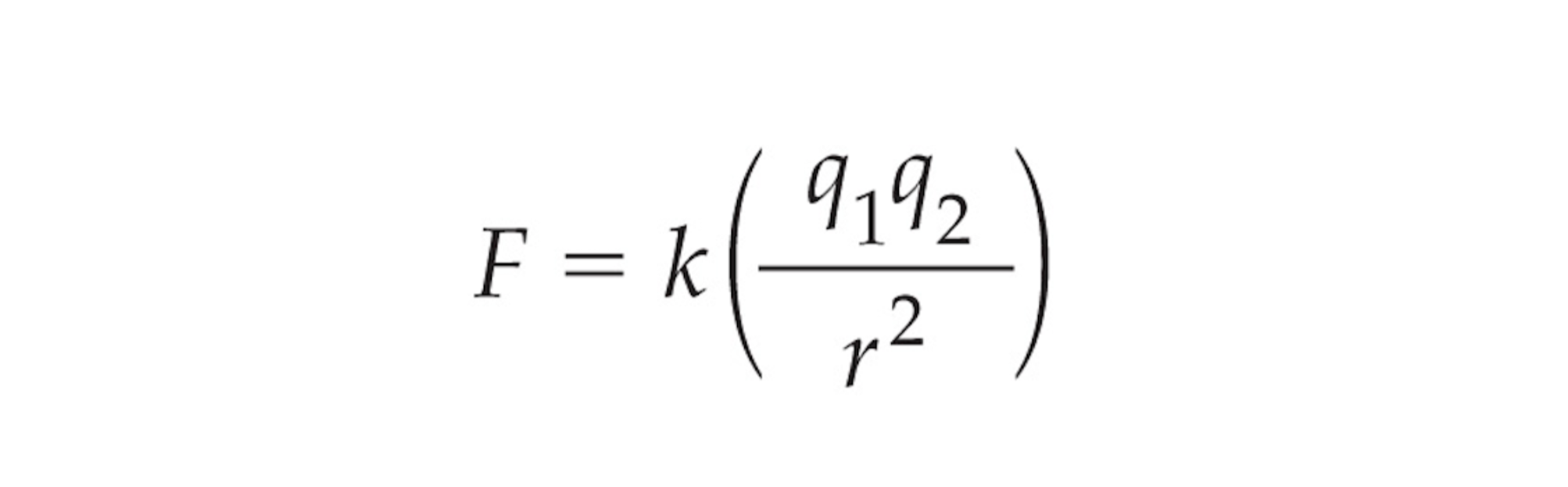
Molecular Substance
Ex: Individual ice molecules being able to be identified in a lattice of ice
Covalent Bonding
Type of binary compound
Always between two non-metals and or metalloids
A chemical bond involving sharing electrons between atoms
However many electrons an atom wants to reach it’s nearest noble gas is how many bonds it will make with other atoms
- Ratios for pairing can not be scaled due to physical limitations with the molecular shape
Water
Common chemical bond
(H2O)
Ammonia
Common chemical bond
(NH3)
Calomel
Common chemical bond
(Hg2Cl2)
Binary Compounds
Compound consisting of two elements
Subdivided into Binary ionic compounds and Binary covalent compounds
Binary Ionic compounds
Compounds consist of a metal and nonmetal
Electron is donated
Binary covalent compounds
Compounds consisting of nonmetals and or metalloids
Shair electrons
mono-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 1
di-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 2
tri-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 3
Word association: Tricycle
tetra-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 4
penta-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 5
Word association: Pentagon
hexa-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 6
Word association: Hexagon
hepta-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 7
octa-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 8
Word association: Octagon
nona-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 9
deca-
Greek prefix
Used when naming binary covalent compounds to show how many atoms of each type there are
Means 10
Word association: Decade
Naming Ionic Compounds
Name consists of two ion names, the metal one always lister first
Metal ion: Based off of element name (doesn’t change from original element name)
Nonmetal ion: Element name with the suffix of “-ide” added
The metal has same ionic charge in all compounds: Name of the metal is just the original name of the metal
Ex: Lithium chloride (LiCl)
The metal does not have the same ionic charge in all compounds (systematic names): Indicate the charge of the metal with a roman numeral in parentheses following the metal name
Ex: Iron(III) chloride (FeCl2)
The metal does not have the same ionic charge in all compounds (older system names): When an Ion appears in two charges -ous is used to show the one of low charge and -ic is used to show the one of a higher charge
Ex: Ferrous chloride (FeCl2) or Ferric chloride (FeCl3)
Naming Covalent compounds
Name consists of two ion names, the less electronegative element first
The second ion takes on the suffix -ide
A prefix is used to show the subscript of both elements
If the first element’s prefix is “mono-” is is left off
Ex. Sulfur hexafluoride
Naming Polyatomic Compounds
Same as ionic compounds
The second name is just the name of the polyatomic ion
Ex: Sodium nitrite (NaNO2)
Naming Acids
Follow two different naming rules
Rule 1: When solid named same as binary or polyatomic ions compounds
Rule 2: When dissolved in water follow below rules
Substance does not contain oxygen: Add prefix “hydro-” and the suffix “-ic acid” to the name of the non-hydrogen part or polyatomic ion
Ex: HF Hydrofluoric acid
Substance does contain oxygen (all that contain oxygen are polyatomic compounds) and the anion ends with the suffix “-ite”: Replace “-ite” with “-ous”
Ex: Sulfurous acid (H2SO3) “Sulfite” is being turned to “Sulfurous”
Substance does contain oxygen (all that contain oxygen are polyatomic compounds) and the anion ends with the suffix “-ate”:
Ex: Sulfuric acid (H2SO4) “Sulfate” is being changed to “Sulfuric”
Acid
Simplified def. for purpose of this class: An acid is any substance that dissolves in water to yield H+ ion.
(aq)
Notation meaning that the result is aqueous
(S)
Notation meaning that the result is a solid
(L)
Notation meaning that the result is a liquid
(G)
Notation meaning that the result is gas
Hydrogen
Periodic Element
Atomic Number: 1
Symbol: H
Helium
Periodic Element
Atomic Number: 2
Symbol: He
Lithium
Periodic Element
Atomic Number: 3
Symbol: Li
Beryllium
Periodic Element
Atomic Number: 4
Symbol: Be
Boron
Periodic Element
Atomic Number: 5
Symbol: B
Carbon
Periodic Element
Atomic Number: 6
Symbol: C
Nitrogen
Periodic Element
Atomic Number: 7
Symbol: N
Oxygen
Periodic Element
Atomic Number: 8
Symbol: O
Fluorine
Periodic Element
Atomic Number: 9
Symbol: F
Neon
Periodic Element
Atomic Number: 10
Symbol: Ne
Sodium
Periodic Element
Atomic Number: 11
Symbol: Na
Magnesium
Periodic Element
Atomic Number: 12
Symbol: Mg
Aluminum
Periodic Element
Atomic Number: 13
Symbol: Al
Silicon
Periodic Element
Atomic Number: 14
Symbol: Si
Phosphorus
Periodic Element
Atomic Number: 15
Symbol: P
Sulfur
Periodic Element
Atomic Number: 16
Symbol: S
Chlorine
Periodic Element
Atomic Number: 17
Symbol: Cl
Argon
Periodic Element
Atomic Number: 18
Symbol: Ar
Potassium
Periodic Element
Atomic Number: 19
Symbol: K
Calcium
Periodic Element
Atomic Number: 20
Symbol: Ca
Scandium
Periodic Element
Atomic Number: 21
Symbol: Sc
Titanium
Periodic Element
Atomic Number: 22
Symbol: Ti
Vanadium
Periodic Element
Atomic Number: 23
Symbol: V
Chromium
Periodic Element
Atomic Number: 24
Symbol: Cr
Manganese
Periodic Element
Atomic Number: 25
Symbol: Mn
Iron
Periodic Element
Atomic Number: 26
Symbol: Fe
Cobalt
Periodic Element
Atomic Number: 27
Symbol: Co
Nickle
Periodic Element
Atomic Number: 28
Symbol: Ni
Copper
Periodic Element
Atomic Number: 29
Symbol: Cu
Zinc
Periodic Element
Atomic Number: 30
Symbol: Zn
Gallium
Periodic Element
Atomic Number: 31
Symbol: Ga
Germanium
Periodic Element
Atomic Number: 32
Symbol: Ge
Arsenic
Periodic Element
Atomic Number: 33
Symbol: As
Selenium
Periodic Element
Atomic Number: 34
Symbol: Se
Bromine
Periodic Element
Atomic Number: 35
Symbol: Br
Krypton
Periodic Element
Atomic Number: 36
Symbol: Kr