3.3 Cellular Energy
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Energy
the ability to do work, which is any change in the state or motion of matter
all living systems require an input of energy to survive and function
to sustain life, energy input must exceed energy output
significant loss of order or energy flow can result in death
cells need ways to extract energy and turn it into usable forms(ATP), metabolic pathways(ex. glycolysis and oxidative phosphorylation) to do this are conserved across all domains(supports common ancestry of life)
cells receive energy in many forms(light,organic molecules like glucose, etc.), but can rarely use it as-is, so it must be converted/transformed before it can be used ex. kinetic energy converted to potential(chemical energy) and vice versa
Kinetic energy
energy associated with motion
Potential energy
stored energy
chemical energy is potential energy available for release in a chemical reaction
Laws of thermodynamics
the study of energy transformations in matter is called thermodynamics
the laws apply to the universe as a whole
life requires a highly ordered system that does not violate the first and second law of thermodynamics
1st law: energy cannot be created or destroyed
energy can be transferred or transformed
ex. chemical(potential) energy in a nut will be transformed into kinetic energy for a squirrel to climb a tree
2nd law: energy transformation increases the entropy (measure of disorder) of the universe. The entropy/disorder of organisms can decrease as long as the entropy/disorder of the entire universe is still always increasing
when energy is converted from one form to another, some energy is lost as heat
heat=less usable energy=disorganized
organized/ordered is energy we can use and disorganized/disordered is energy that is less usable
the organization seen in organisms(like living cells, tissues, and organs)represents a local(not universal) decrease in entropy, which requires a constant input of energy. Some of this energy is lost to heat to the organism’s surroundings, which increases the total amount of entropy/disorder in the universe, which makes sure the 2nd law of thermodynamics is not violated
Metabolism
the sum of all chemical reactions in a cell that transform energy and matter
Metabolic pathways
series of linked chemical reactions that either break down complex molecules(catabolic pathways) or build complex molecules(anabolic pathways)
paths are sequential, meaning the product of a reaction is the reactant in the next step
substrate→intermediate→intermediate→product
ex. glycolysis and oxidate phosphorylation
metabolic pathways are conserved across all domains( archea, bacteria, eukarya), which supports the theory of common ancestry
the specific chemical reactions happening within metabolic pathways can be classified as either endergonic or exergonic
reactions that are endergonic are often coupled with those that are exergonic through the use of ATP
Catabolic pathways
pathways that breakdown complex molecules
releases energy

Anabolic pathways
pathways that build complex molecules from simpler compounds
require energy

Endergonic
chemical reaction that needs energy
Exergonic
chemical reaction that releases energy
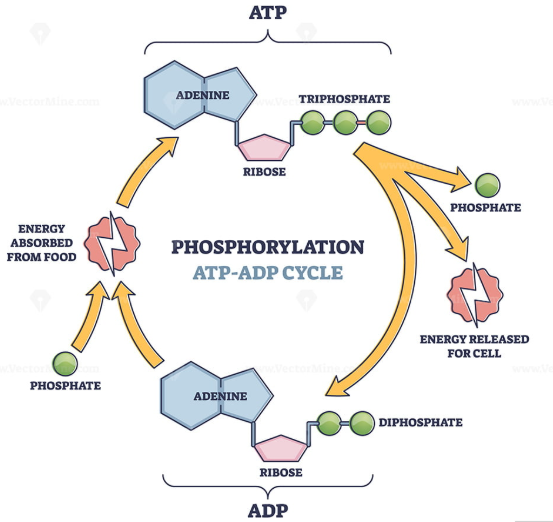
ATP
adenosine triphosphate
molecule that organisms use as a source of energy to perform work
ADP can be regenerated to ATP via the ATP cycle
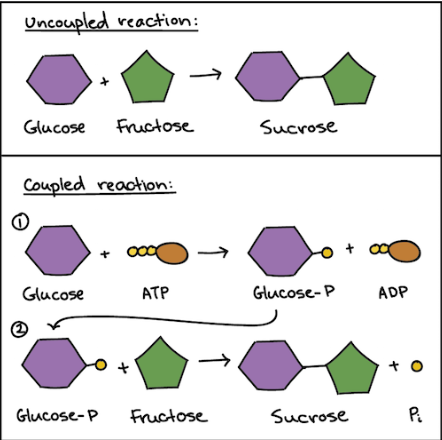
Coupling reactions
cells carry out energy-needing reactions(anabolic) by coupling them to ATP hydrolysis (catabolic)
ATP + H2O→ADP + P(free phosphate)+energy- 1. releases energy(the catabolic reaction,ATP hydrolysis)
the free phosphate that was released moves to another molecule and phosphorylates it(intermediate)- 2. transfers the energy(intermediate)
phosphorylation makes the molecule more reactive, which drives the energy needing reaction- 3. energy needing reaction occurs(anabolic)