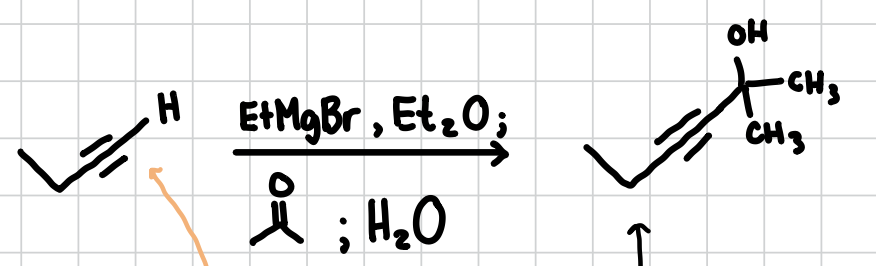ORGO 2 General Concepts + Reactions FINAL
1/131
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
132 Terms
Carbonyl + Primary Amine
Fuses a carbonyl group (C=O) and a amine (-NH2) in the primary location together. They will fuse at the carbonyl oxygen and both of the hydrogens will leave. Water will be a byproduct
Reagents: -R-C=O + NH2-R, acid (catalytic)
Carbonyl + Secondary Amine
Has two separate iterations, one where there is a proton next to the carbonyl group and one where there isn’t. In the no-proton reaction, there are no reagents; the two will just react at the carbonyl site as expected. If there is a proton present, the two will still fuse, but the double bond will move down to where the proton was just removed.
Reagents: Acid
Wolf-Kishner Reduction
This reduction will reduce a carboxyl (C=O) group all the way down to two separate hydrogens.
Reagents: 1) H2NNH2, EtOH 2) KOH, 180°C plus weird oxygen chain mouth
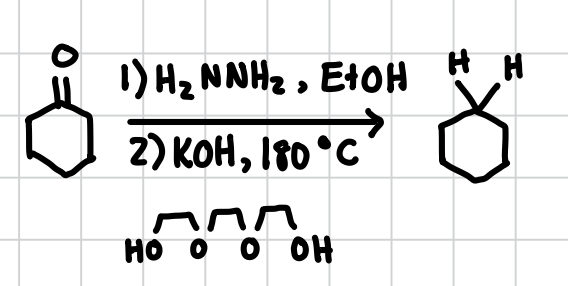
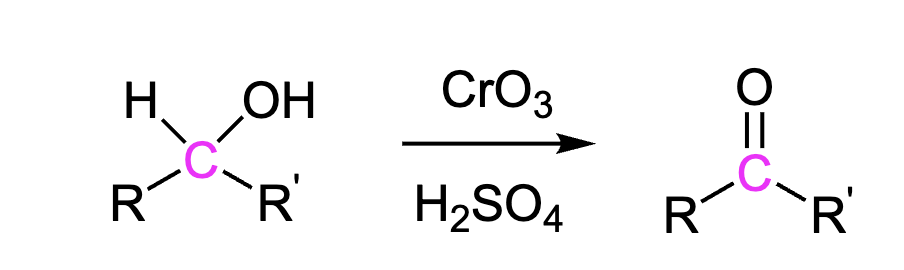
Secondary Alcohols to Ketone
This transforms secondary alcohols (-OH) into ketones (C=O)
Reagents: CrO3, H2SO4
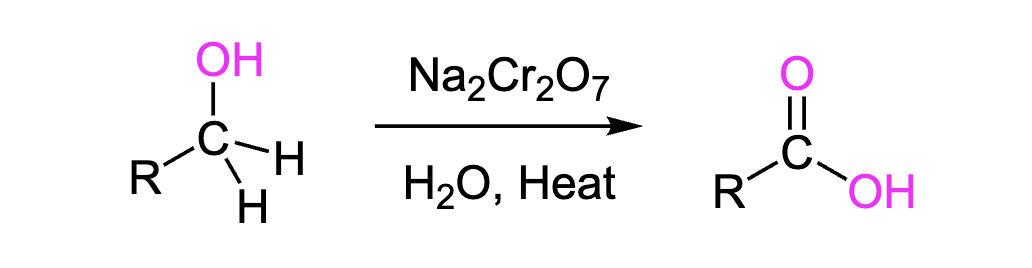
Primary Alcohols to Carboxylic Acids
Transforms primary alcohols (-OH) into carboxylic acids (O=C-OH)
Reagents: Na2Cr2O7, H2O, Heat
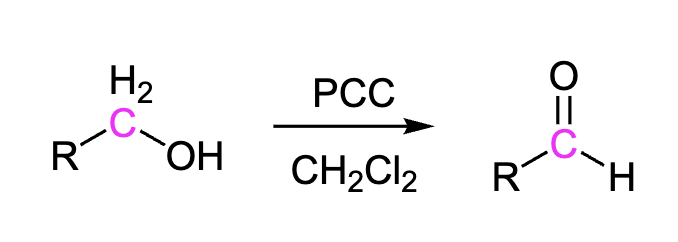
Primary Alcohols to Aldehydes
Using PCC we can turn primary alcohols (-OH) into aldehydes (C=O). Also could be done with DMP
Reagents: PCC, CH2Cl2
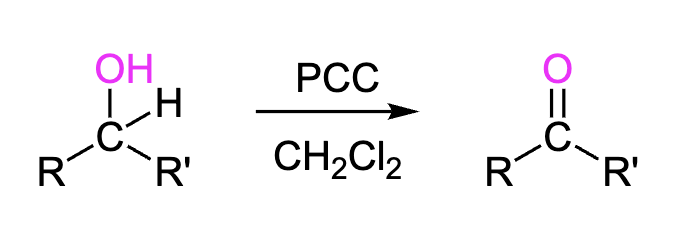
Secondary Alcohols to Ketones
Using PCC we can turn secondary alcohols (-OH) into ketones (O=C-R). Also could be done with DMP.
Reagents: PCC, CH2Cl2
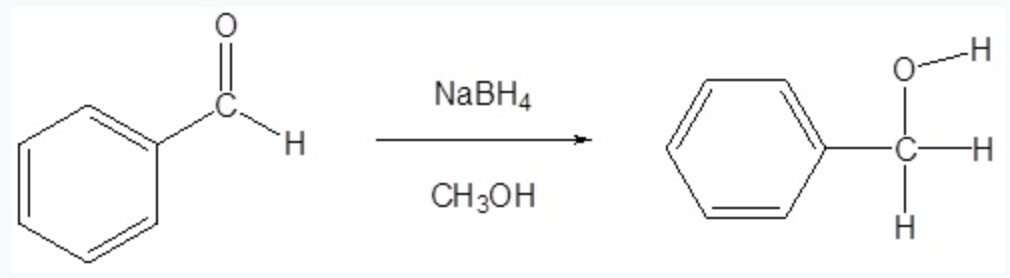
Carbonyl Reduction
Using NaBH4, we can reduce the carbonyl group of an aldehyde or ketone. Will remove the double bond and make the oxygen an alcohol
Reagents: NaBH4, CH3OH ( OR ) LiAlH4, H2O
Nucleophilic Addition
Typically the nucleophile will attack beneath the carbonyl group and reduce it to an OH. If the Nucleophile has an extra hydrogen attached, it will remove a water molecule afterwards and replace the carbonyl double bond.
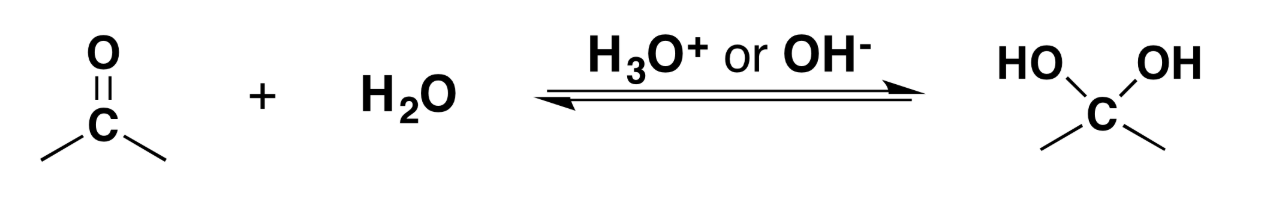
Hydration of Ketones and Aldehydes
When mixing a carbonyl group with water and an acid or base, the carbonyl will turn into a acetal (2 -OH groups where the =O once was).
Reagents: H2O, H3O+ or OH-
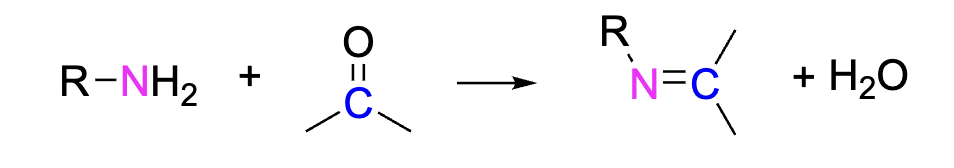
Formation of Imines
Imines can be formed when an aldehyde or ketone is reacted with a primary amine (-NH2). The Nitrogen attaches to the oxygen in the carbonyl group and the hydrogens leave.
Reagents: Primary amine + Carboxyl group
Formation of Enamines
Enamines can be formed when an aldehyde or ketone is reacted with a secondary amine. The Nitrogen attaches to the oxygen in the carbonyl group, and the hydrogen leaves. The double bond is also shifted down. This reaction requires an acid.
Reagents: Secondary amine + Carboxyl group, H3O+
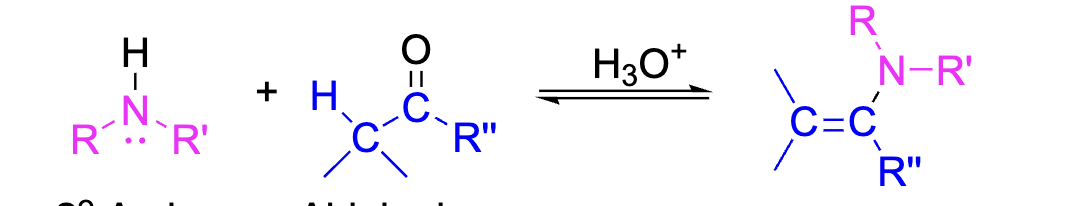
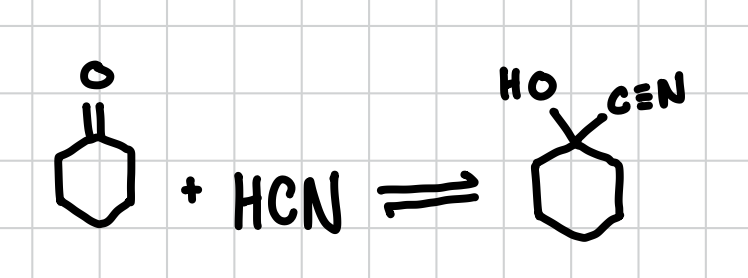
Cyanohydrins
Reduces a carboxyl group (C=O) to an alcohol and a CN triple bond.
Reagents: HCN ( OR ) NaCN, HCl
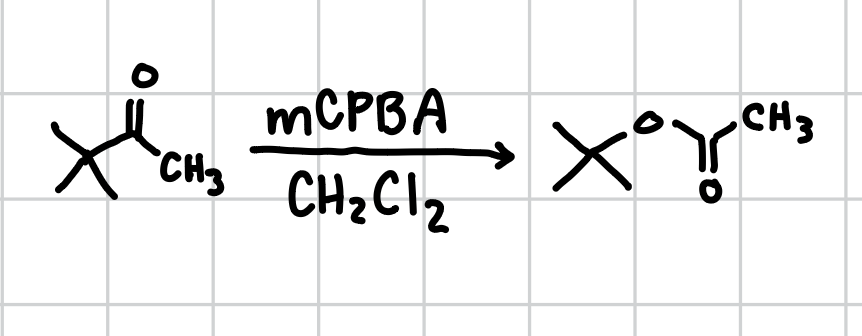
Baeyer-Villiger Oxidation
Adds an oxygen to right before the carboxyl group (C=O). Be careful when using mCPBA as if theres extra alkenes it will form an epoxide instead of extra oxygen.
Reagents: mCPBA, CH2Cl
Reagents if can’t use mCPBA: tBuOOH, KOH, heat
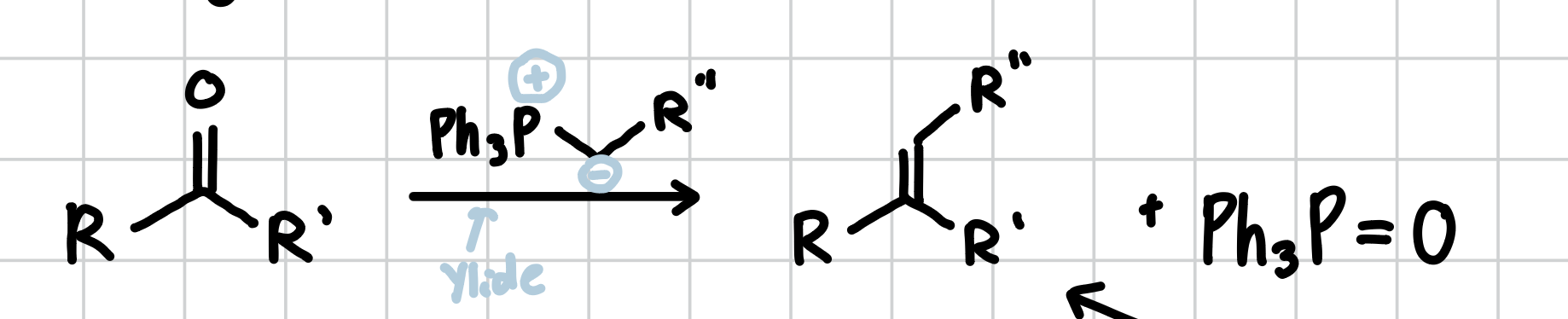
Wittig Olefination
Using an Ylide, you can insert any R group into a carboxyl (C=O) group. The part after the negative sign on the ylide is what gets inserted. Can happen intramolecularly.
Reagents: Ylide, THF or Et2O
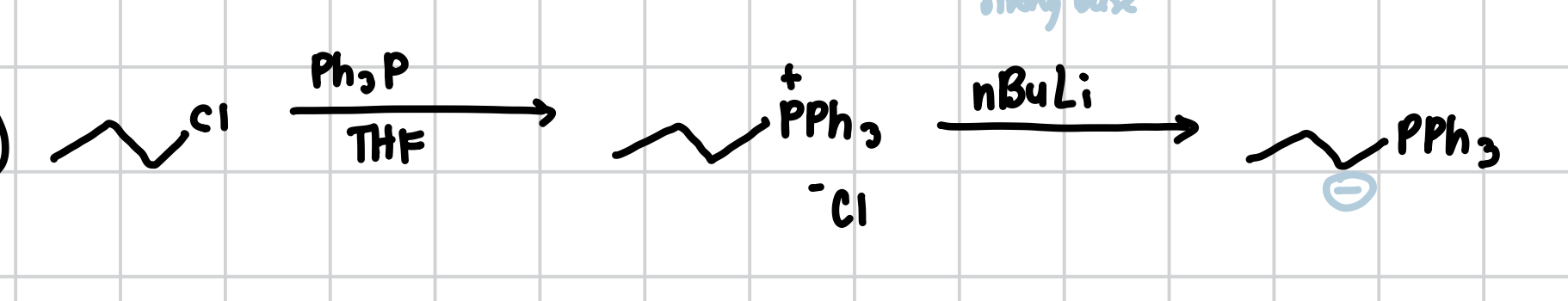
Synthesis of Ylides
Ylides can be formed by reacting Ph3P with a primary halogen. Afterwards, a strong base is necessary to solidify the Ylide.
Reagents: 1) Ph3P, THF 2) nBuLi or KOtBu
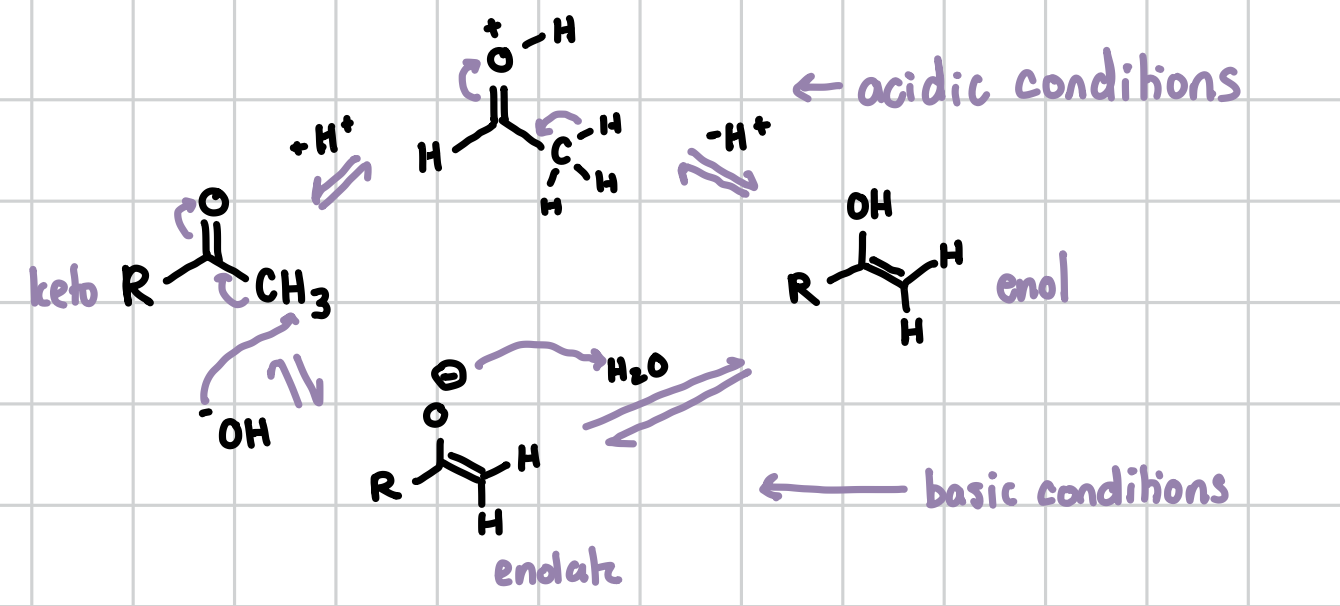
Keto-enol tautomerization
This reaction serves to go from the keto form of a molecule to its enol form. The keto form (electrophile) is a ketone or aldehyde, and the enol form (nucleophile) is an alcohol and alkene. These reactions can take place under acidic conditions or basic conditions. Ketones favor the enol confirmation while aldehydes favor the keto confirmation. Adding an acid and removing it makes the keto into an enol. Adding a base and then adding water takes the enol to a keto.
⍺-hydrogens
Hydrogens located right next to a carbonyl (C=O) group
Adding Deuterium
Deuterium replaces all ⍺-hydrogens in a trans fashion
Reagents: HCl, D2O
⍺-Halogenation
Can occur under basic or acidic conditions. In its acidic form will replace the least sterically hindered ⍺-hydrogen with a halogen. In its basic form will replace the least sterically hindered total amount on one side of the carboxyl of ⍺-hydrogens with halogens.
Acidic Reagents: X2, AcOH, heat
Basic Reagents: X2, KOH

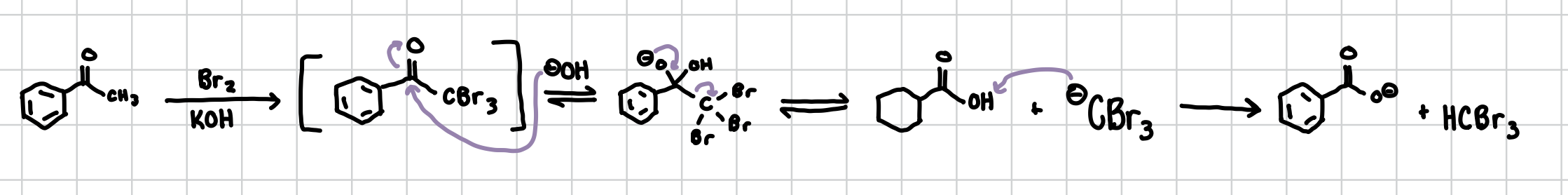
Haloform Reaction
Basically the ⍺-Halogenation but if an extra base is added afterwards turns the free standing CBr3 into an alcohol and then reduced down to an O-
Reagents: 1) X2, KOH, 2) OH-
⍺-Alkylation
Will replace least sterically hindered ⍺-hydrogens with methyl groups. Will also work if the carboxyl is in resonance.
Reagents: CH3I, KOtBu
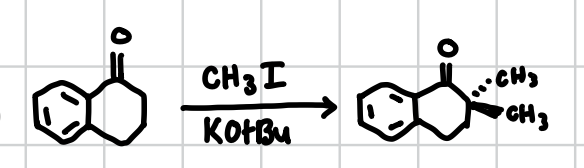
“O” vs “C” confirmation
An ⍺-alkylation with differing amounts of base added. If above 1 equiv, the resonance will form with the least sterically hindered side, which you can then add the Phenol group to. If below 1 equiv, the resonance will form with the most sterically hindered side, which you an then add the phenol group to.
Reagents: 1) LDA (equiv matters), THF if > or RT if < 2) PhCH2Br (add -78°C → RT if LDA was >1)
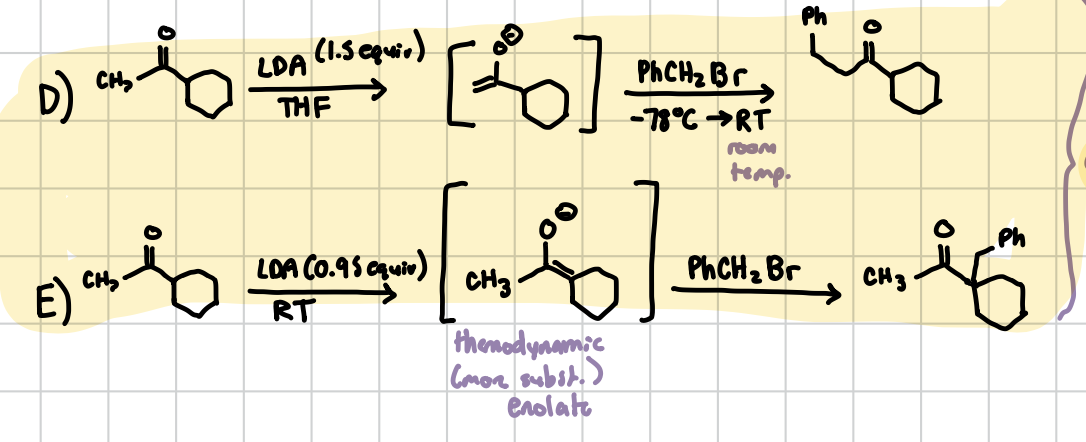
Aldol Condensation w/ heat
In an Aldol Condensation, the carboxyl group is reacting with its own resonance form to create an extra carbon-carbon bond. When heat is added, this reaction goes all the way through and will create an alkene. Can occur intramolecularly
Reagents: NaOH, H2O, HEAT (IMPORTANT)
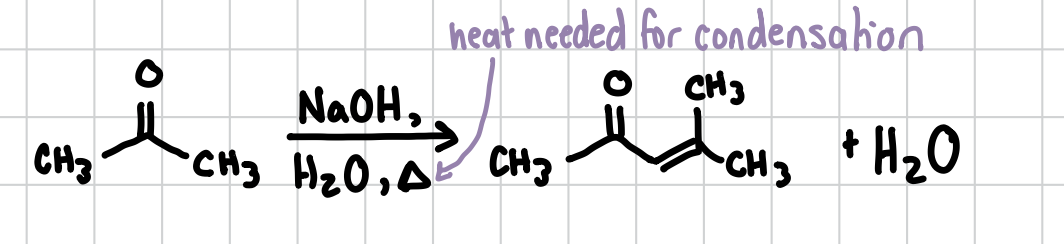
Aldol Reaction (no heat)
Same as a regular Aldol Reaction except it does not go all the way through and does not make an alkene, stops at the alcohol.
Reagents: NaOH, H2O
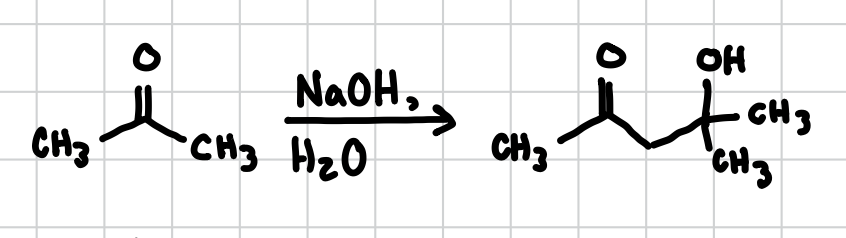
⍺,β-Unsaturated Carbonyls
basically if a copper halide is present in the reagents, you have to go to the other alkene and not the carboxyl group. Otherwise continue as normal.
Micheal Addition
Attaches a primary alkene group by expanding that double bond into an extra single bond, will attach to any carbon attached to ⍺-carbons
Reagents: tBuOK, Et2O, Group you wish to attach with primary alkene
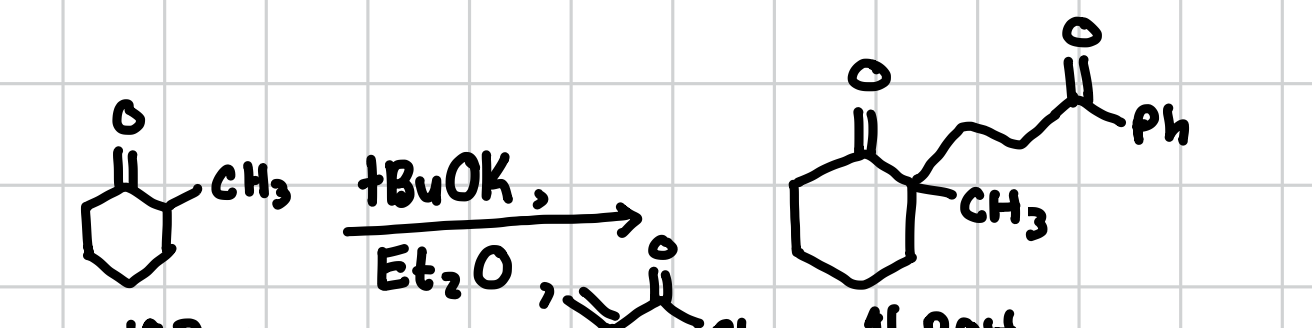
Robinson Annulation
This is basically a Michael addition followed by an aldol intermolecular.
Carboxylic Acid oxidation state
Can be found out by counting all of the N,O,X around a carbon. Should be equal to 3. EXCEPTION: H2C=C=O.
Carboxylic Acid from Alcohol
Can be done with a primary alcohol. Will form the carboxyl group just before the primary acid
Reagents: CrO3, H2O, H2SO4, 0°C (Jane’s Reagent)
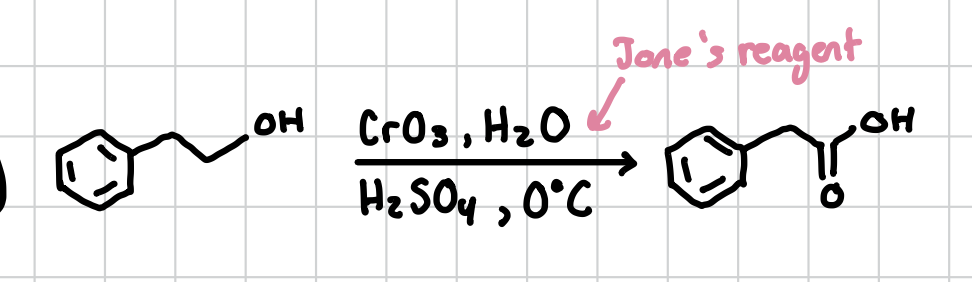
Carboxylic Acid from Aldehyde
Can be done with any detached aldehyde. Will form the carboxyl group just before the primary acid. Can be done with Jane’s but can also be done with
Reagents: tBuOOH, KOH
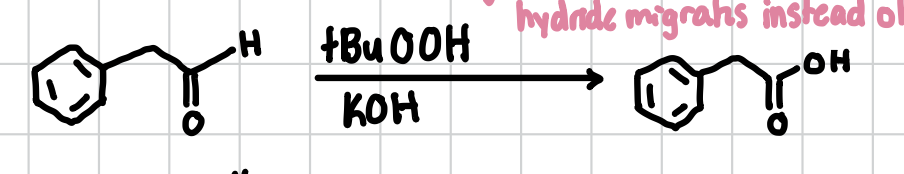
Carboxylic Acid from Halogens
Can be done with any primary halogen, will replace right on the spot of the halogen instead of the spot before.
Reagents: 1) Mg°, H2O 2) CO2 ; H3O+ ( OR ) 1) NaCN, DMF 2) H2O, H2SO4, heat
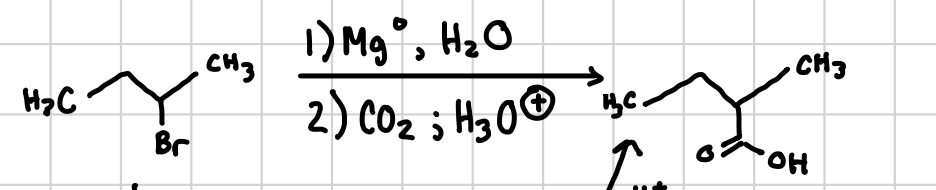
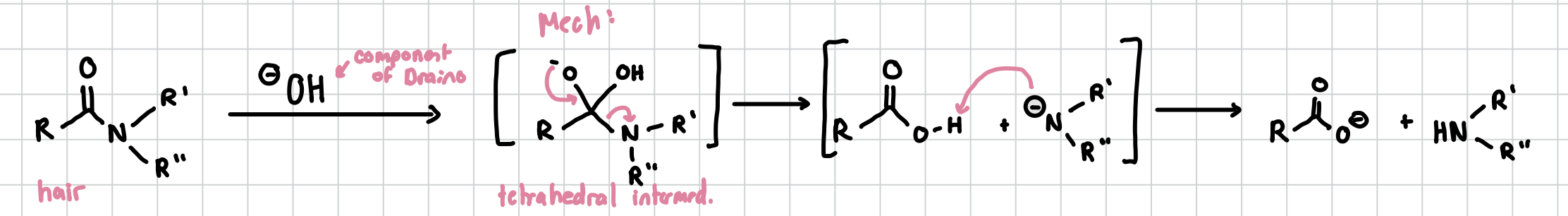
Amide-Bond Hydrolysis
Basically separates an amide group from a carboxyl group
Reagents: OH-
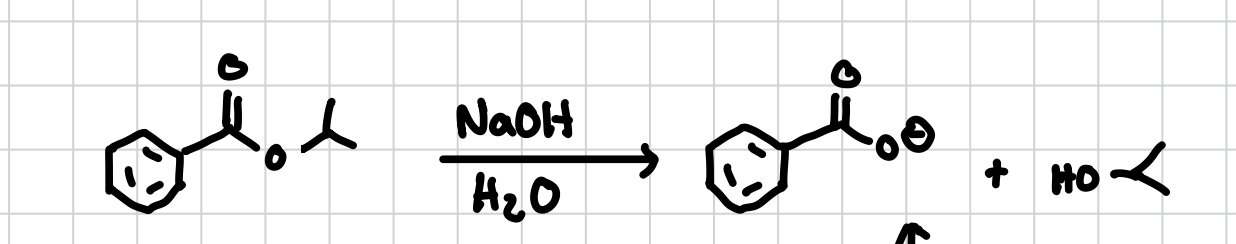
Ester-Bond Hydrolysis: Basic Conditions
Separates the other side of the ester connected to a carboxyl group, leaving the carboxyl group with an O- and the separated group with an Alcohol. better if you dont want a ring closure
Reagents: NaOH, H2O
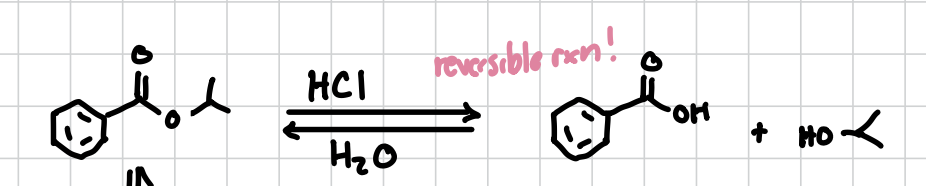
Ester-Bond: Acidic Conditions
Will separate a carboxyl group and an ester leaving both sides as alcohols, better if you want to react something with it afterwards.
Reagents: HCl
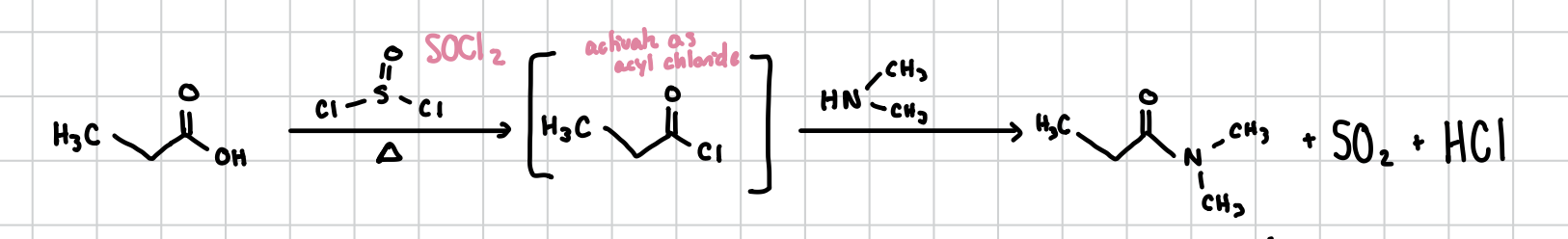
Amide Formation
Amides can be forms from a carboxylic acid when reacted with SOCl2 and then the amide itself.
Reagents: 1) SOCl2, heat 2) The amide
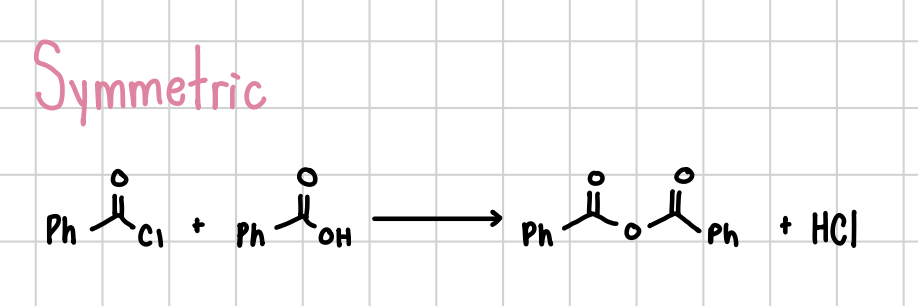
Anhydride Formation
Merges two carboxyl groups if one has an alcohol group and the other one has a halogen. Will proceed without reagents if the molecules are symmetrical. Otherwise, it will need reagents. Can occur intermolecularly
Unsymmetrical Reagents: Heat, removal of HX
Intramolecular Reagents: Removal of HX
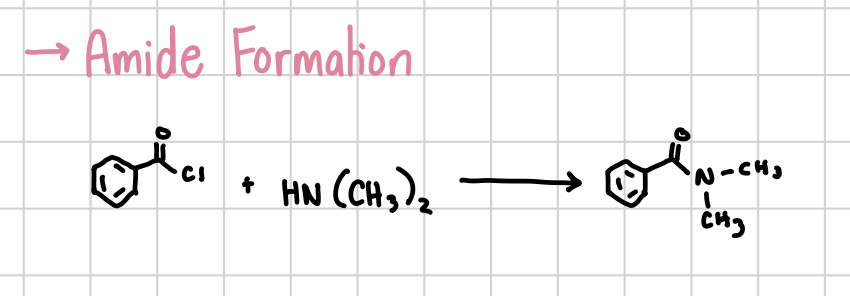
Amide Formation w/ Acyl Chloride
Reacting an acyl chloride with an HN(CH3)2 will form the amide
Reagents: R-Acyl Chloride + HN(CH3)2
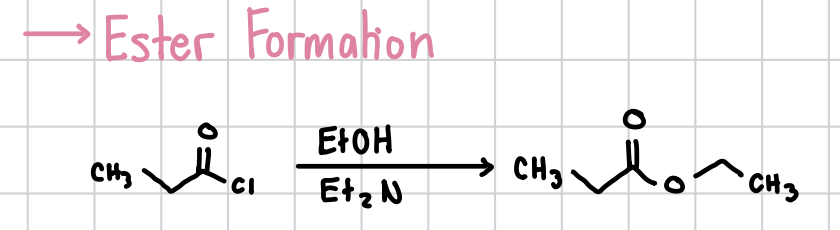
Ester Formation w/ Acyl Chloride
An ester can be formed through an acyl chloride. Attached to the oxygen will be an ethyl methyl (-CH2CH3).
Reagents: Acyl chloride + EtOH, Et2N
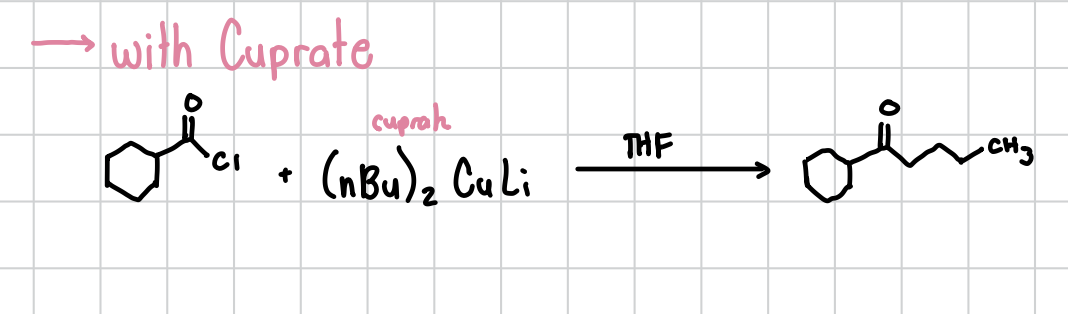
Acyl Chloride reaction with Cuprate
When an acyl chloride group is reacted with the cuprate, a CH2CH2CH3 group will replace the chlorine in the chloride.
Reagents: Acyl chloride + (nBu)2CuLi (cuprate), THF
Acyl Chloride reaction with LiAl(OtBu)3H
Will reduce an acyl chloride to a carboxyl attached to a hydrogen.
Reagents: Acyl chloride + LiAl(OtBu)3H
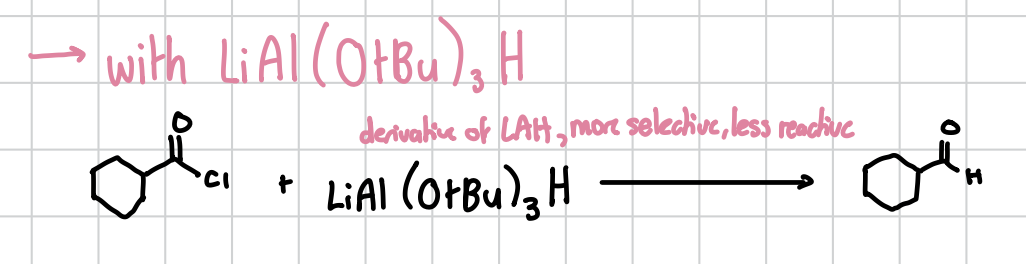
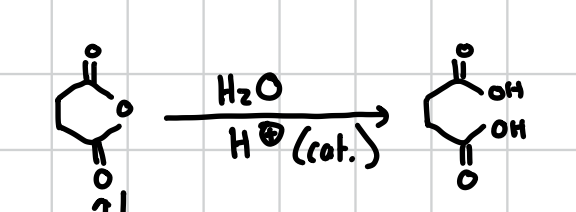
Opening a Cyclic Anhydride
This basically removes the oxygen from the center of a cyclic molecule and ends the two sides in alcohol groups
Reagents: H2O, H+ (cat.)
If EtOH is used instead of H2O one of the alcohol groups will instead be OEt
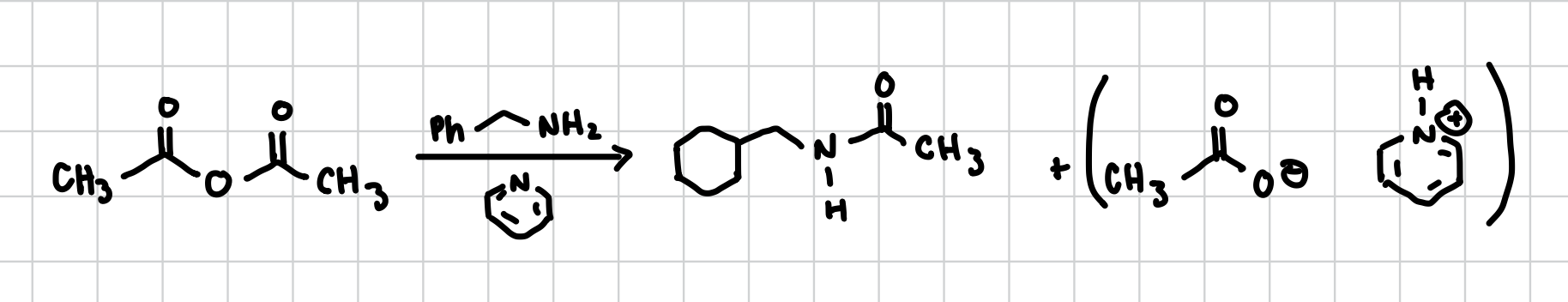
Anhydride into an Amine
An anhydride is the W looking thing with oxygens this reaction turns that into an amine. Attaches the NH group to where the oxygen was and replaces everything with what it had attached.
Reagents: R-NH2, plus aromatic Nitryl ring
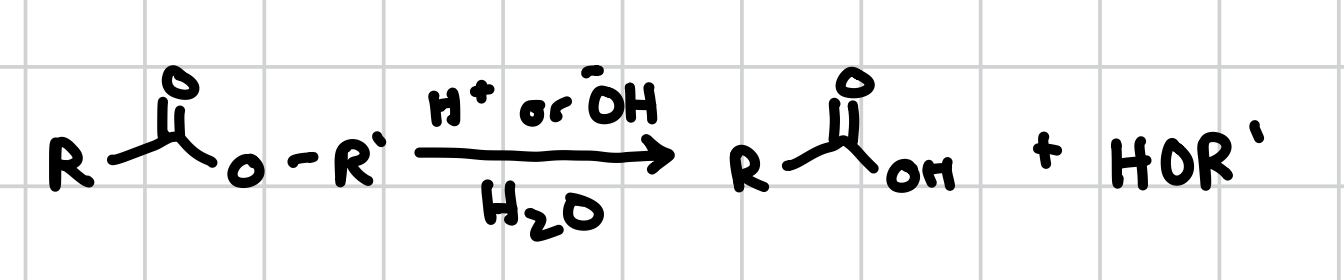
Ester Hydrolysis
Will turn the oxygen on an ester group into an alcohol
Reagents: H+ or OH-, H2O
Trans-esterification
Will attach any -ROH to the oxygen of an ester.
Reagents H2SO4 + ROH
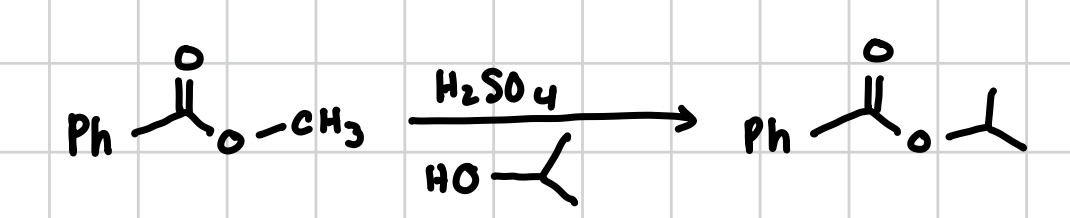
Addition of Grignards to Carboxylic Acid Derivatives
Ketone: Will add to the keytone and then split the carbonyl into an alcohol and whatever R group was attached
Carboxylic Acid: Will not Grignard
Ester: Will do so twice both at the carbonyl and leaves at as a OH
Reduction of Carboxylic Acids
Will remove the cabonyl group and make it into two hydrogens
Reagents: 1) LAH, THF, heat 2) H30+
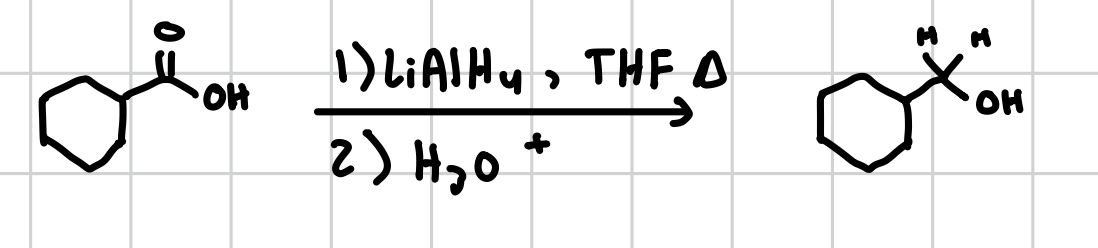
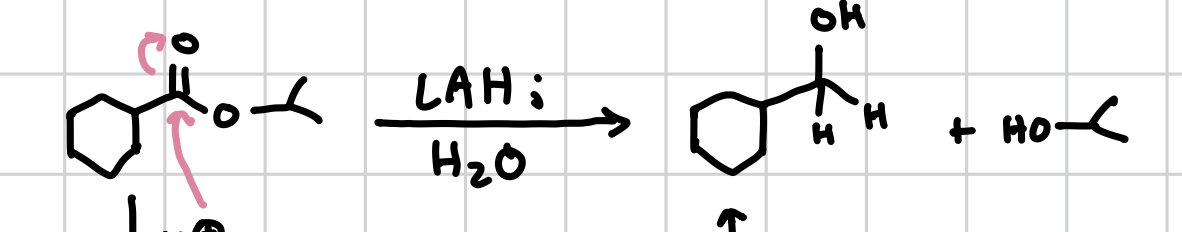
Reduction of Esters
Will remove the oxygen group and turn the carbonyl into an alcohol.
Reagents: LAH; H2O
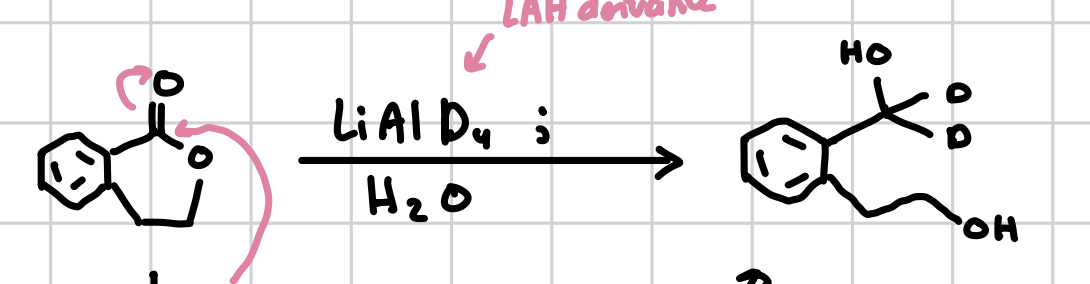
Reduction of Cyclic Esters
Will open the ring adding a carbon to the side that did not have the ether and making it an alcohol and then turning the carbonyl into an alcohol and adding a D grouo
Reagents: LiAlD4;H2O
e-alkylation of an ester
LDA, THF will make the enolate and then you can react the enolate with any electrophile. However if using a phenol connected to an aldehyde, will do the weird stereo chem stuff
Reagents: LDA, THF, electrophile, water (if using phenol aldehyde)
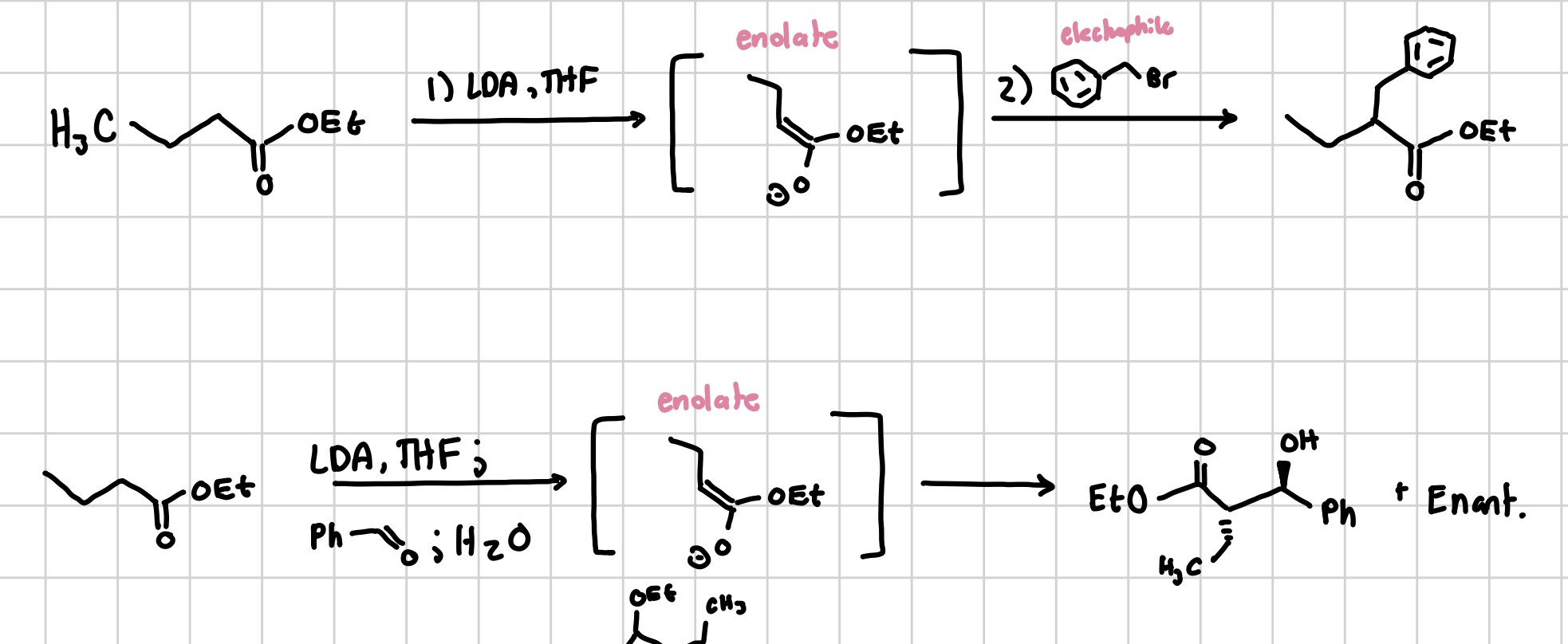
Adding two things to a cyclic ester with an alkene
This will add things in a trans fashion across the alkene in a cyclic ester with an alkene
Reagents: (CH3)2 CuLi, THF; CH3I
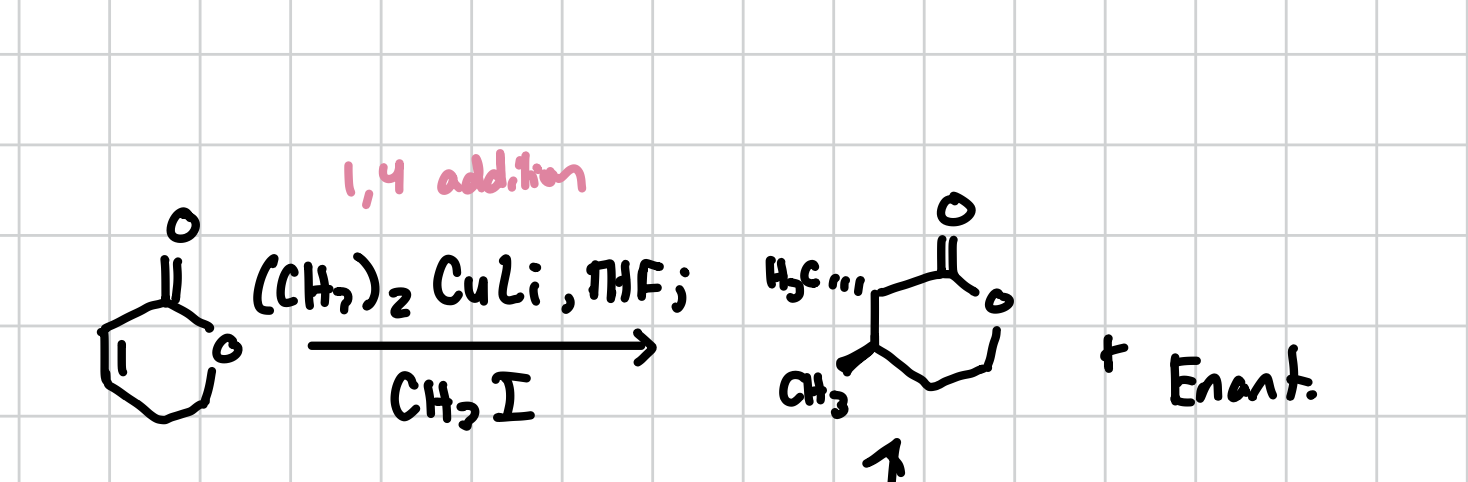
Amide Reduction to Amine
Self-Explanatory
Reagents: LAH, THF; H2O
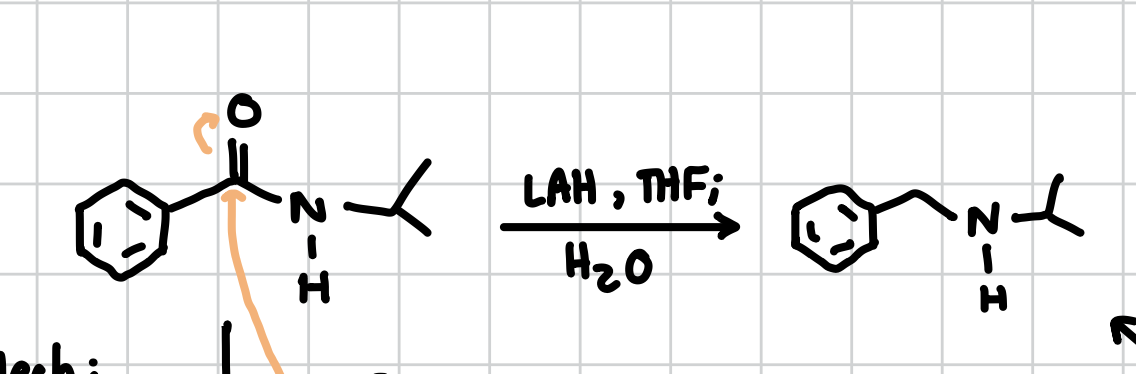
Addition to Amide
If there is a proton on the N:
Add directly to the N
Reagents: LDA, THF; PhCH2Br
If there is no proton on the N:
Add to the other side of the enolate at the end of its point
Hoffman Rearrangement: Amide to amine
Basically shoves the carbonyl group out of the equation
Reagent: X2, NaOH, H2O
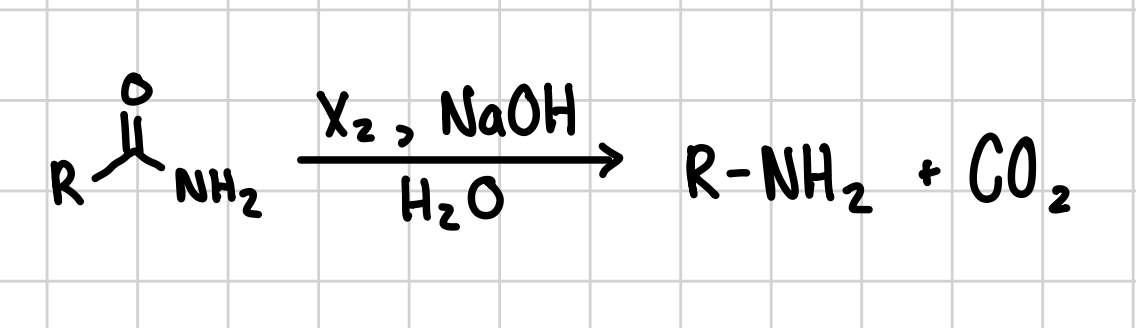
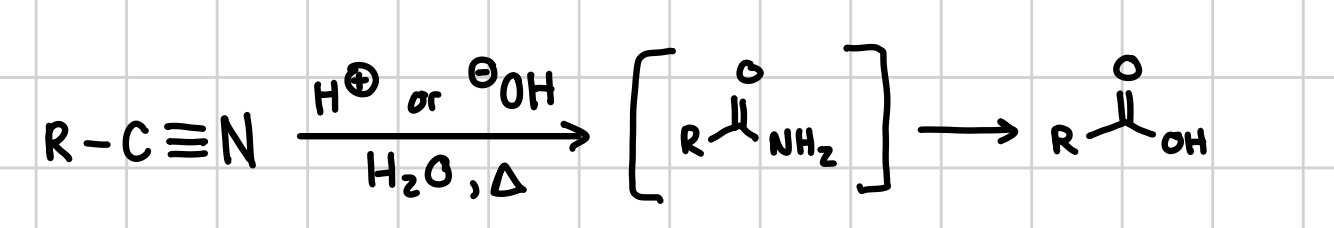
Nitrile to a Carboxylic Acid
To turn a Nitrile into a Carboxylic Acid you need a
Reagents: H+ or OH-, H2O, heat
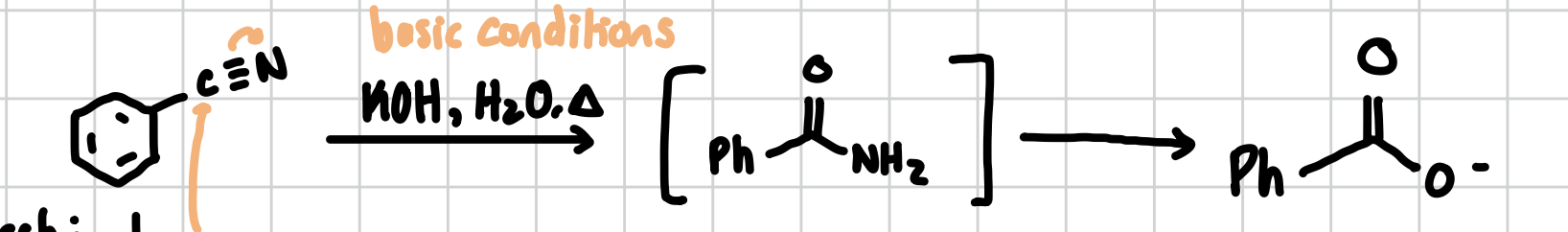
Nitrile Under Basic Conditions
This does the carboxylic acid except instead of a OH it makes a O-
Reagents: KOH, H2O, heat
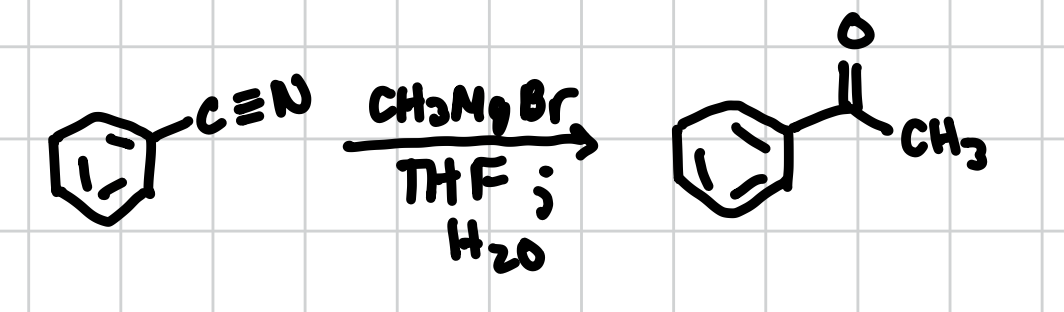
Grignard Addition to Nitrile
Adds a Carbonyl attached to a methyl group where a nitrile is.
Reagents: CH3MgBr, THF; H2O
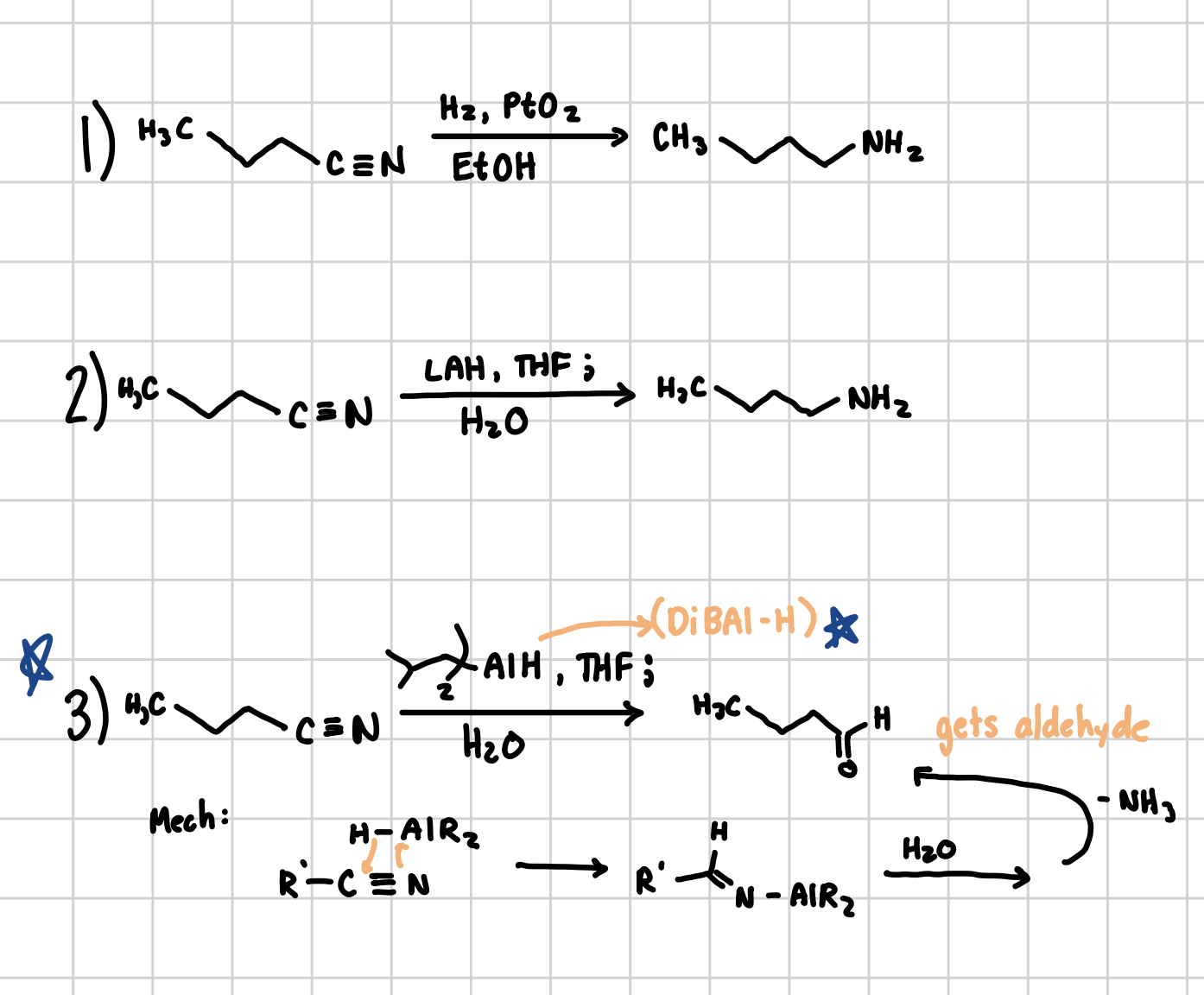
Reduction of Nitriles
Multiple Ways
Adds the NH2 group at the END of the nitrile
Reagents: H2, PtO2, EtOH
Reagents: LAH, THF; H2O
Reagents: DiBAl-H, THF; H2O (This one is special cause instead of NH2 it addes an aldehyde at the BEGINNING of the Nitrile)
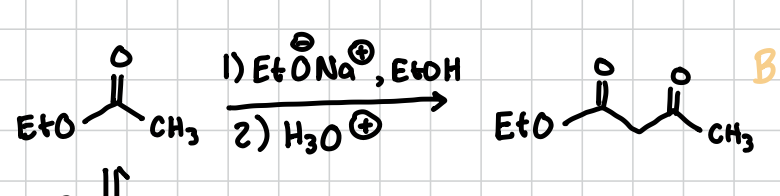
Claisen Condensation
A carbonyl group with an EtO, basically an aldol, but leaves as a carbonyl when done, no alkene
Reagents: 1) NaOEt, EtOH 2) H3O+
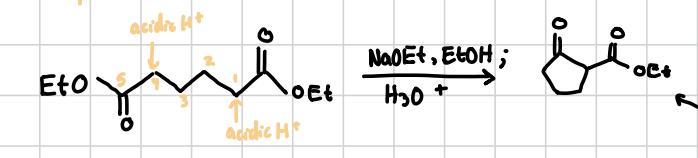
Intramolecular Claisen Condensation (Dieckmann)
Basically Intermolecular Claisen Condensation
Carboxylic Acid
R-COOH. Made of a carbonyl group (C=O) attached to a hydroxyl group (—OH)

Ester
RCOOR’. Made of a carbonyl group (C=O) and a organyl group (—OR’)

Aldehyde
RCHO. Made of a carbonyl group (C=O) and bonded to at least one hydrogen atom.

Alcohol
R-OH. A hydroxyl group (—OH) bonded to a carbon atom

Alkane
Hydrocarbon containing only single carbon-carbon bonds
Alkene
Hydrocarbons containing at least one carbon-carbon double bond
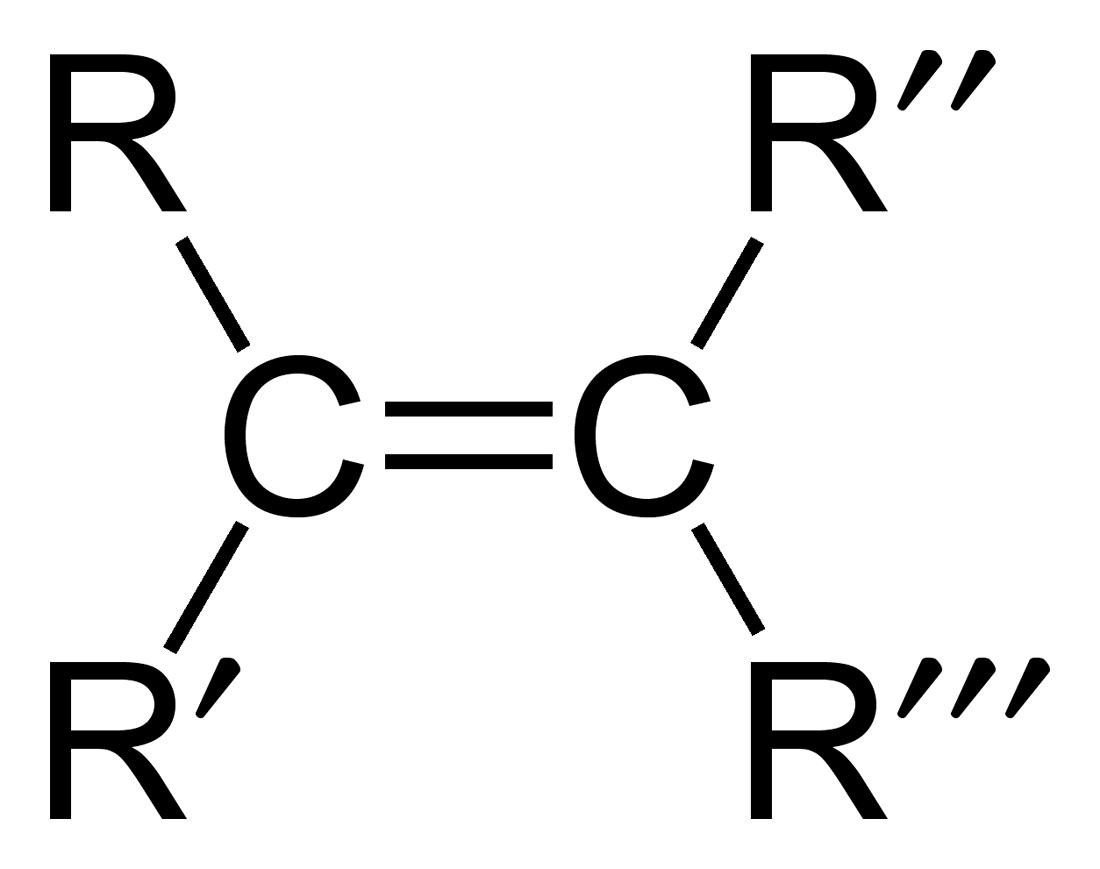
Alkyne
Hydrocarbons containing at least one carbon-carbon triple bond
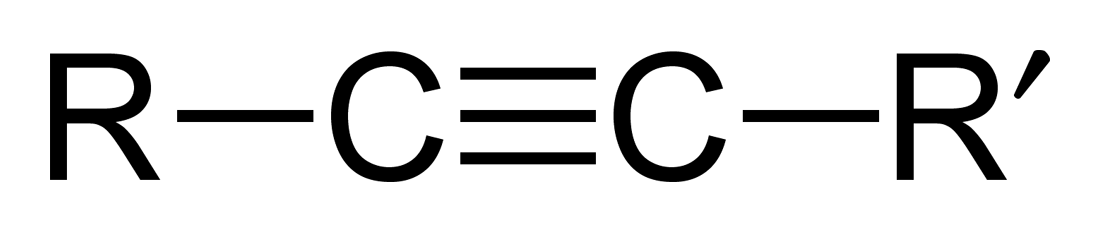
Ether
R-O-R’. Made of an oxygen atom bonded to alkyl or aryl groups

Epoxide
A cyclic ether where the ether forms a three-atom ring.

Haloalkane
R-X. Derived from alkanes where one or more hydrogen atoms are replaced by a halogen atom.
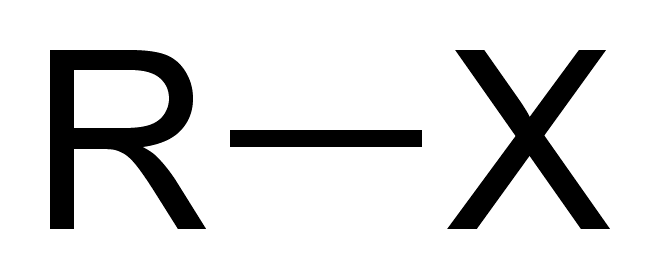
Ketone
R-CO-R’. Made of a carbonyl group (C=O).

Acid Anhydride
(RCO)2O Two acyl groups bonded to an oxygen atom

Amide
R-CO-NR’R’’. Made of a carbonyl group (C=O) bonded to a nitrogen atom.
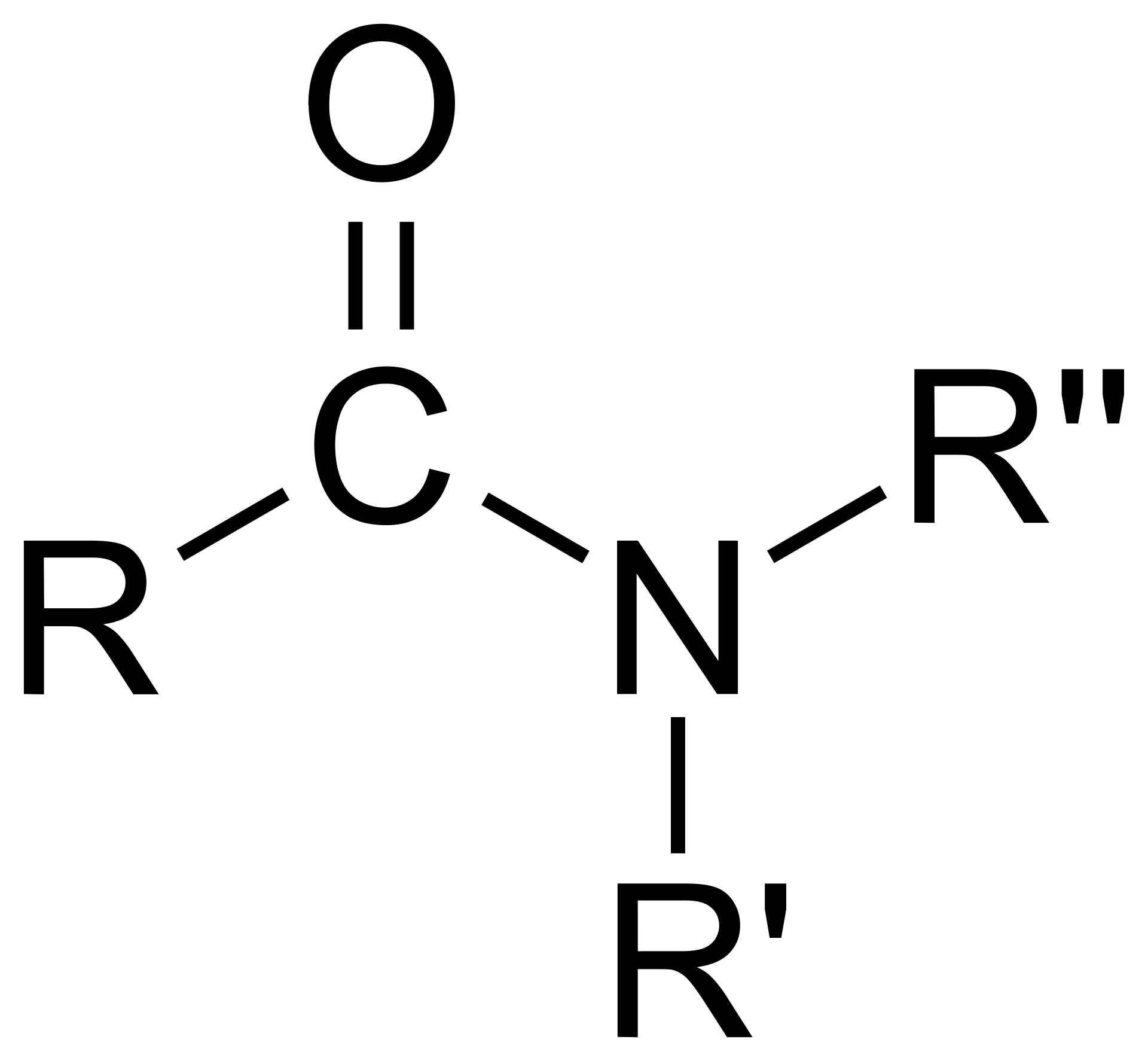
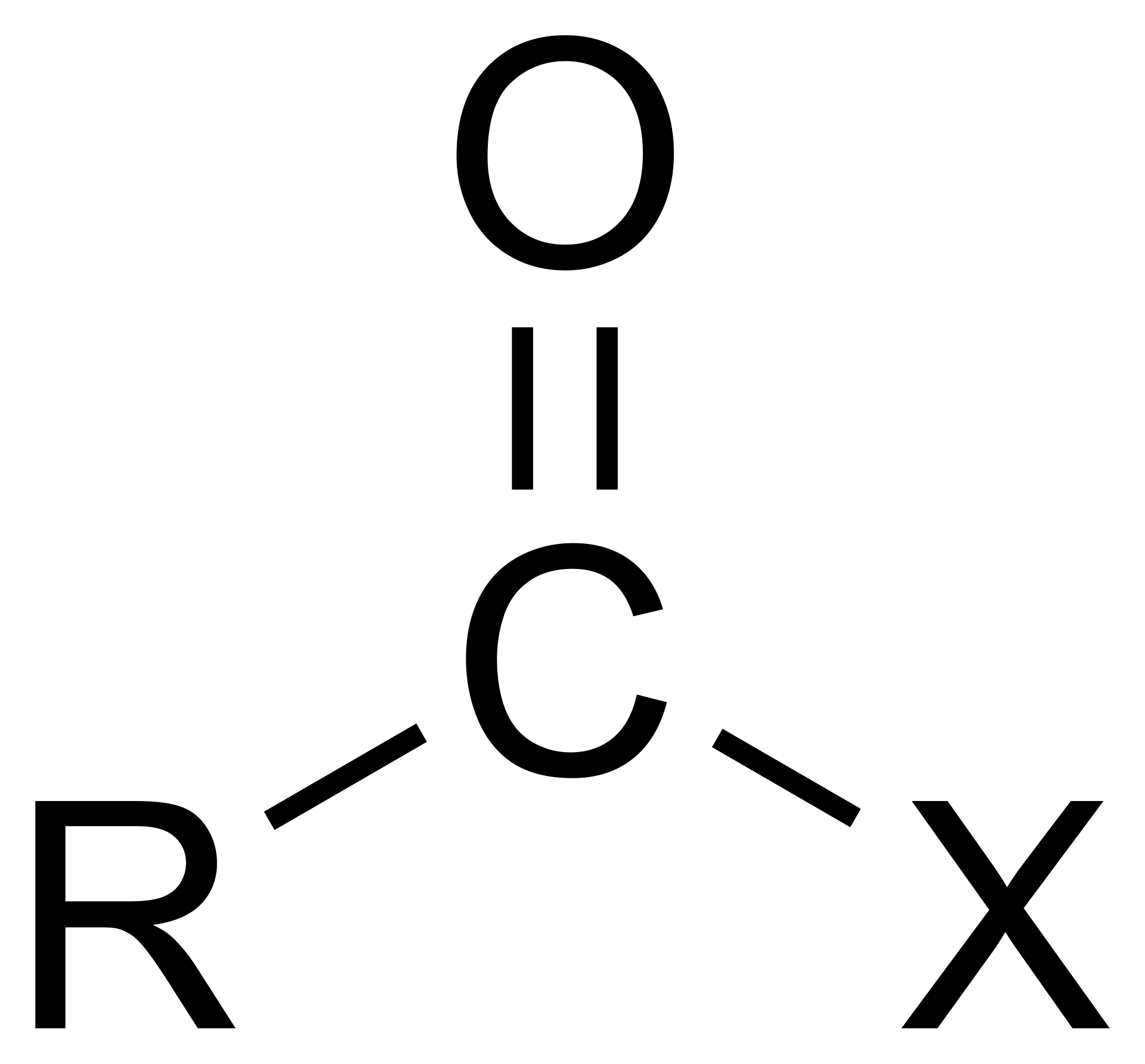
Acyl Halide
RCOX. Made of a carbonyl group (C=O) and a halogen (X)
Amine
Organic compounds that contain a carbon-nitrogen bond.

Nitrile
Compound containing a C(triple bond)N
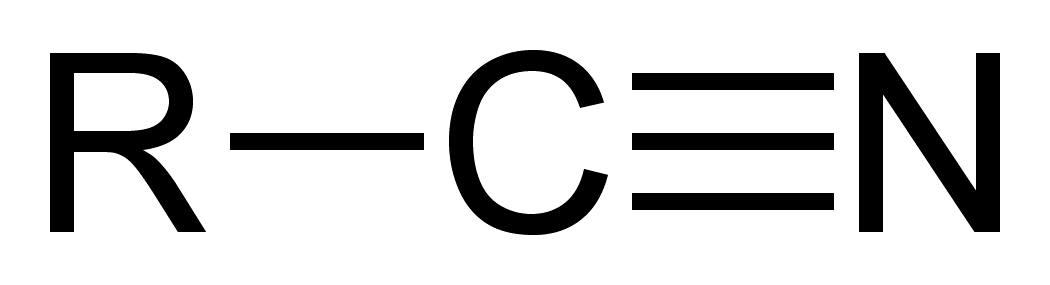
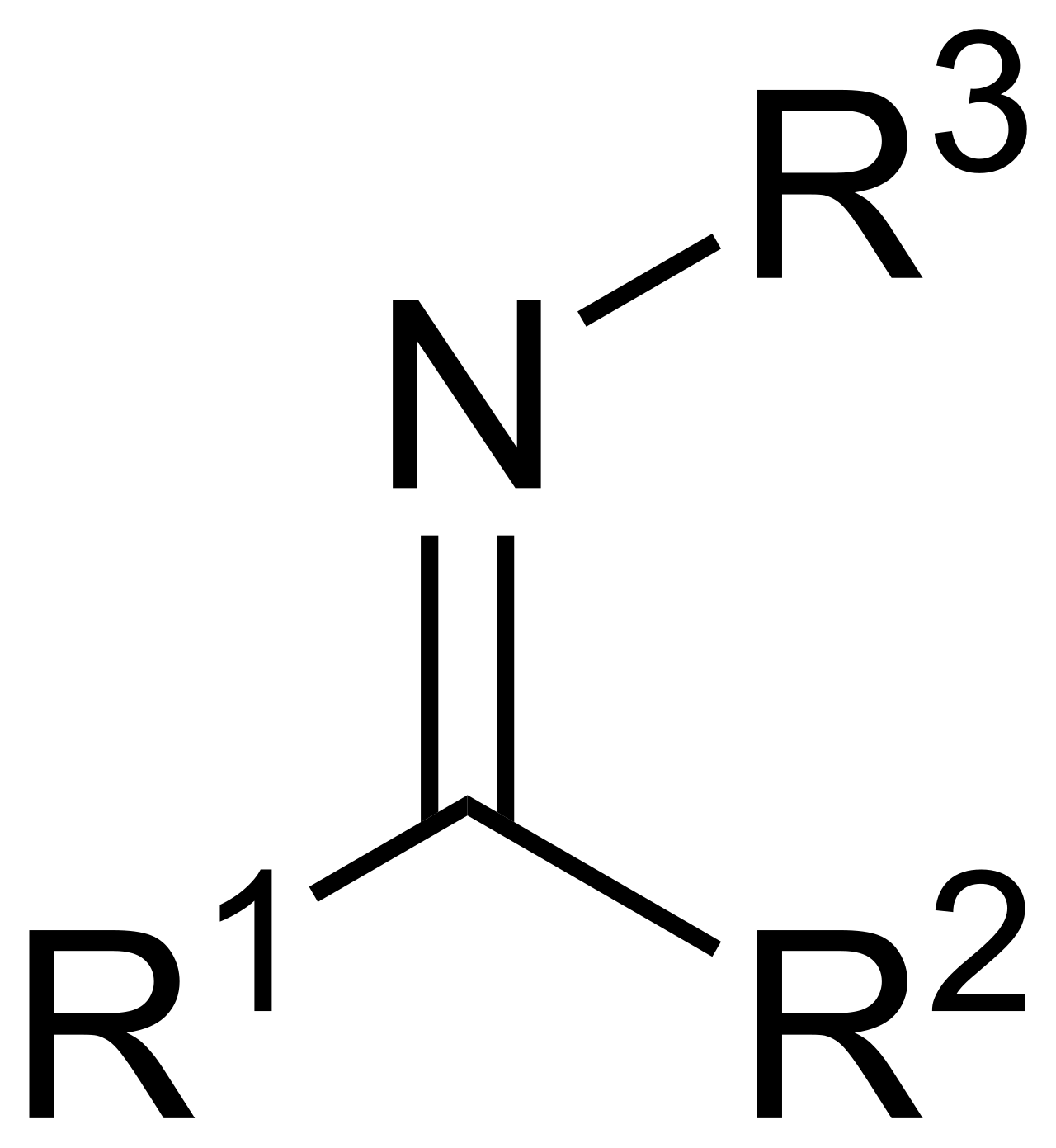
Imine
Compound containing a C=N bond.
Isocyanate
Compound containing the N=C=O group

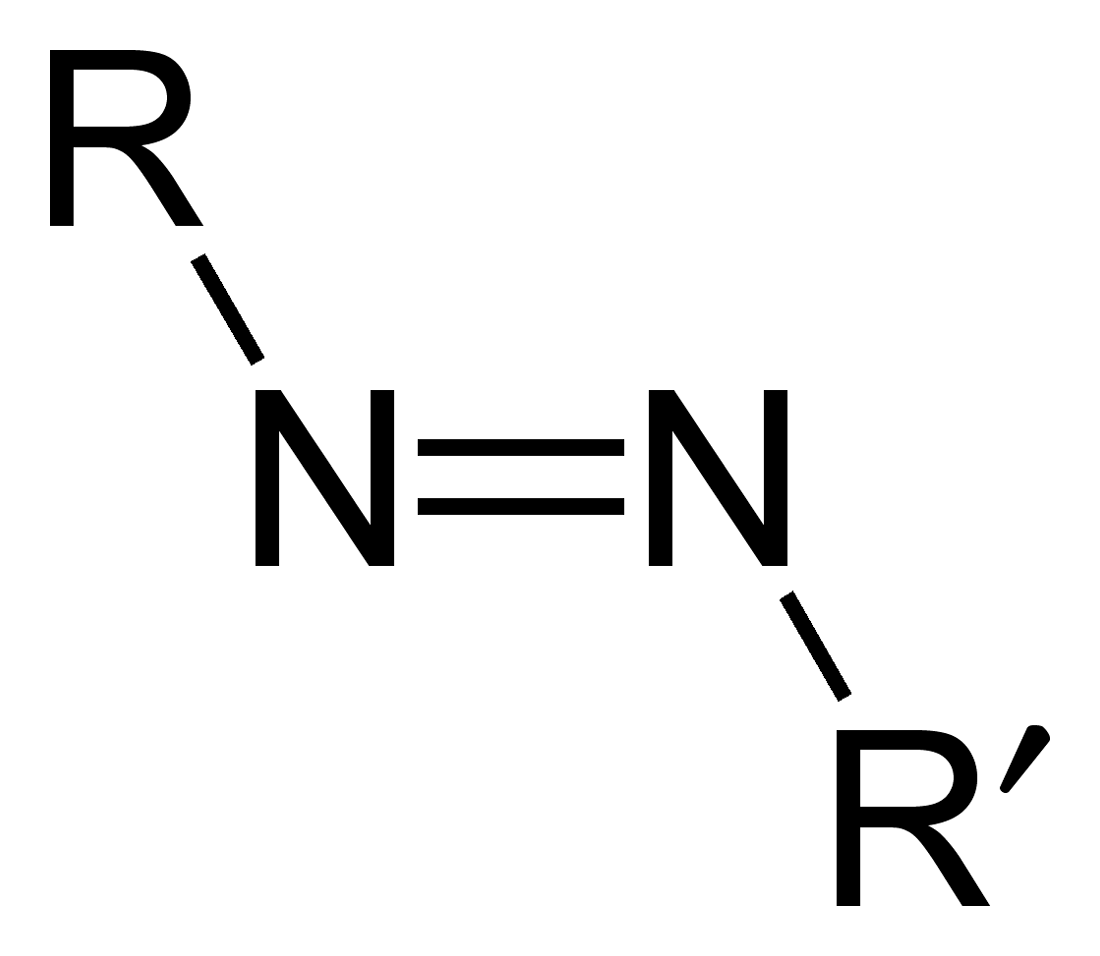
Azo Compound
Compounds containing R-N=N-R’
Thiol
Compounds containing the -SH group

Arene
Compounds containing aromatic groups or are aromatic themselves
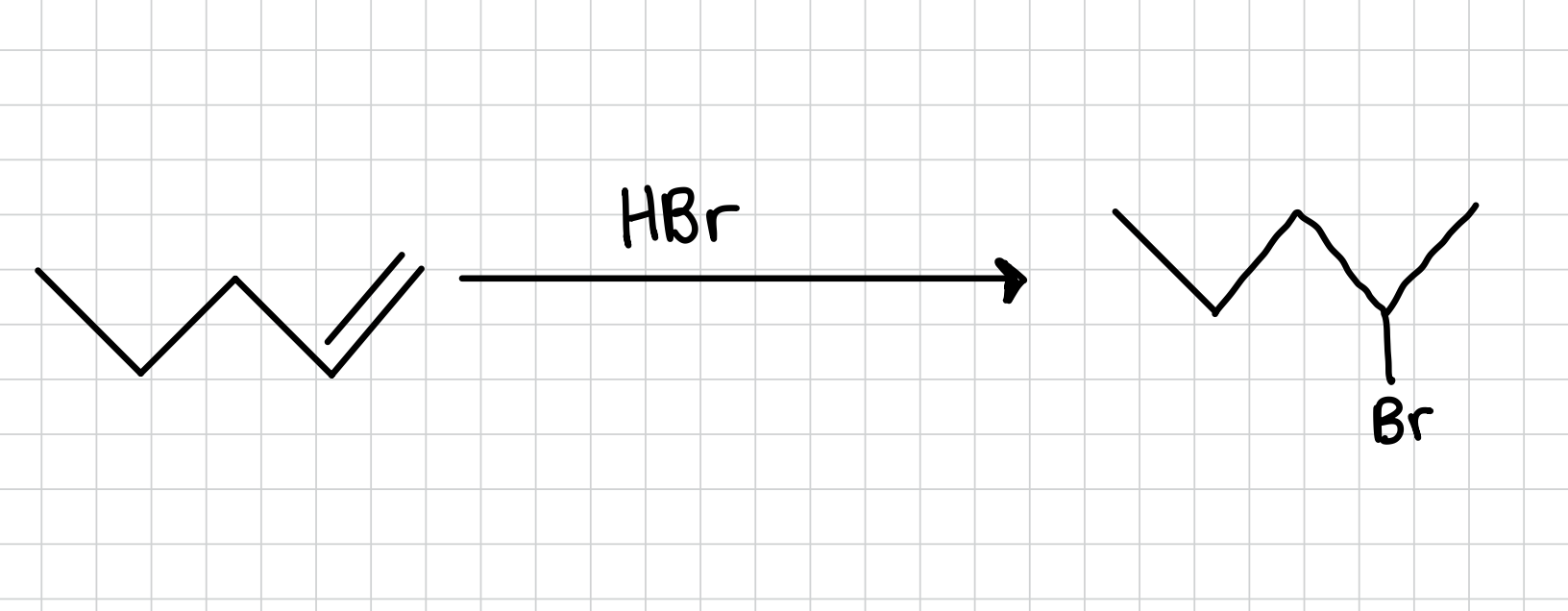
Hydrobromination of an Alkene: Markovnikov
Adds a bromine to the most substituted side of an alkene.
Reagents: HBr
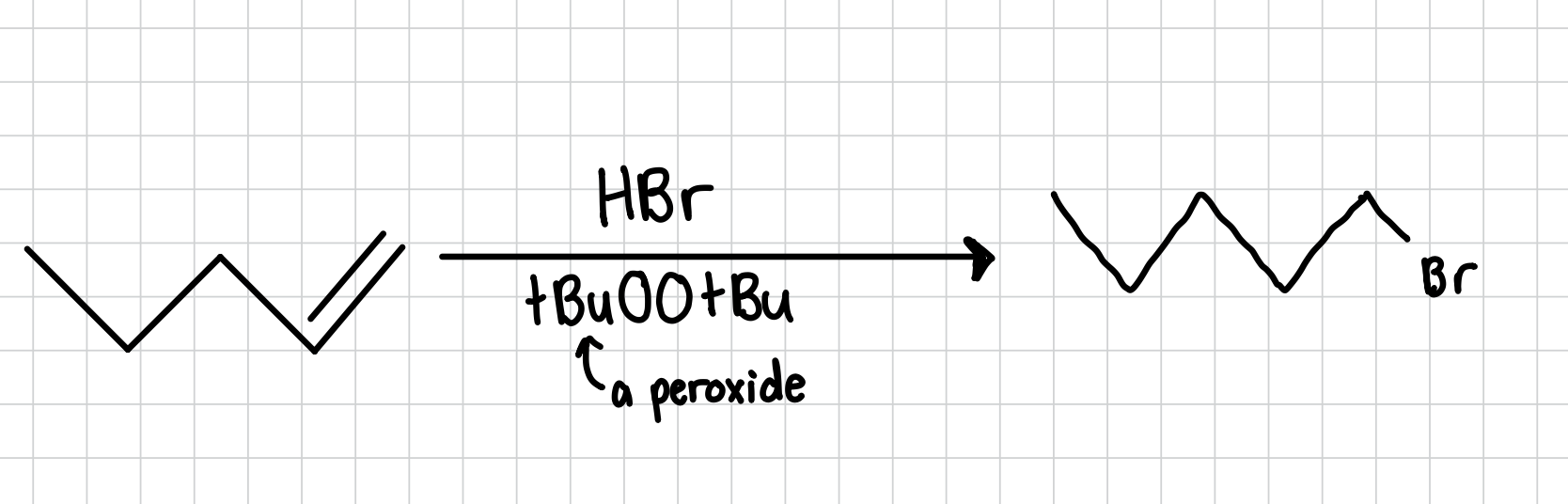
Hydrobromination of an Alkene: Anti-Markovnikov
Adds a bromine to the least substituted side of an alkene
Reagents: HBr, tBuOOtBu (a peroxide)
Acid-Catalyzed Markovnikov Addition of an Alcohol to an Alkene
Adds an methoxy group (-OCH3) to the most substituted side of an alkene.
Reagents: CH3OH, H3PO4 (cat.)
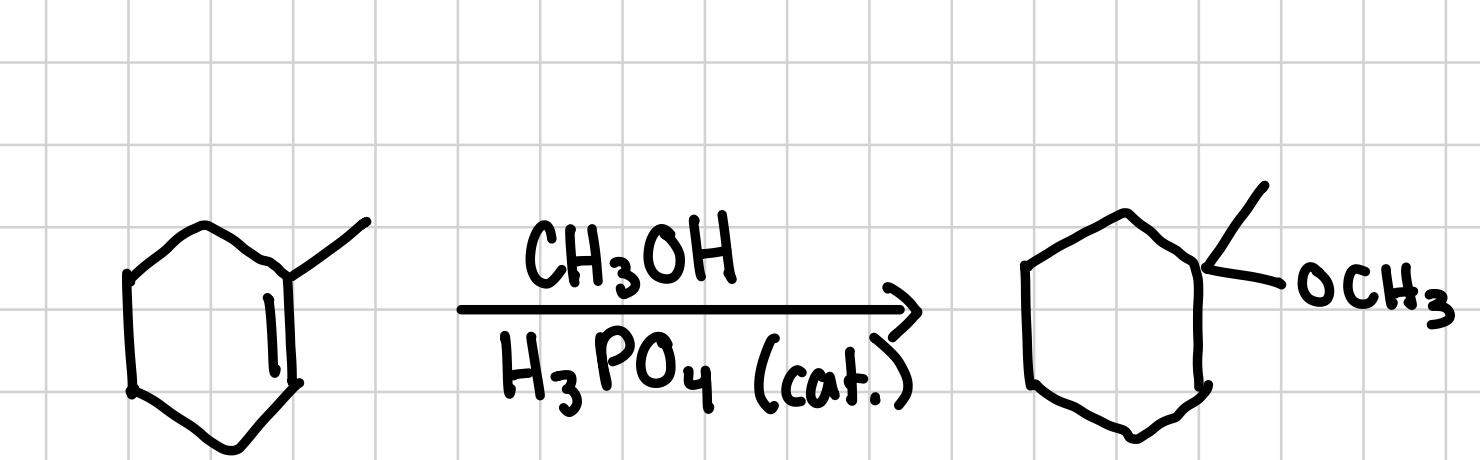
Halogenation of an Alkene
Adds a halogen in a trans fashion to both sides of an alkene. Will also form the enantiomer
Reagents: X2, CCl4
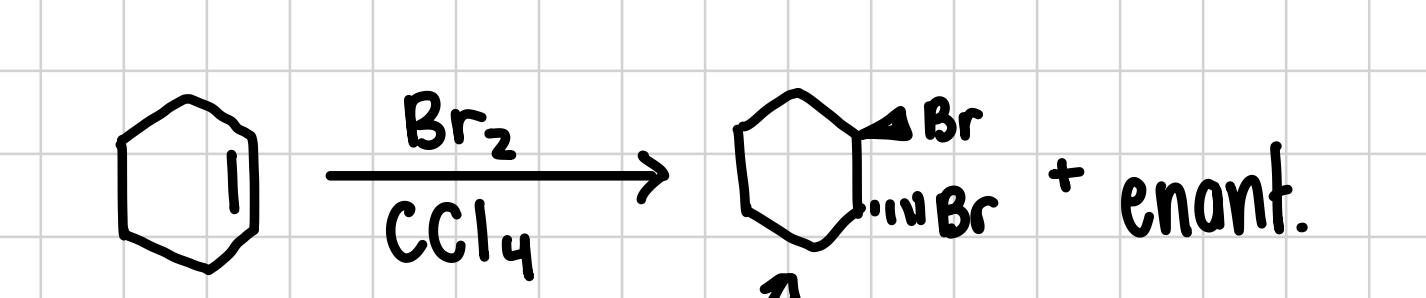
Hydrohalogenation of an Alkene
Adds a halogen in a trans fashion to one side of an alkene and a hydroxyl group to the other side. Will also form the enantiomer.
Reagents: X2, H2O
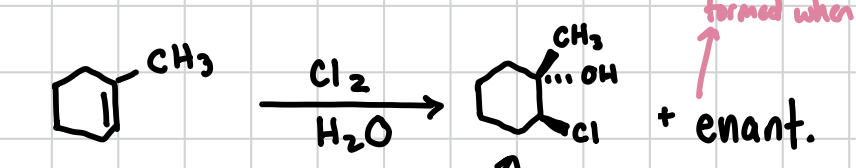
Hydrohalogenation Epoxide of an Alkene
After performing a hydrohalogenation, using a base will form an epoxide.
Reagents: 1) Hydrohalogenation, 2) Base (NaOH)

Haloetherification of Alkenes
Adds a halogen to the least substituted side of an alkene and a methoxy group (-OCH3) to the most substituted side.
Reagents: X2, CH3OH
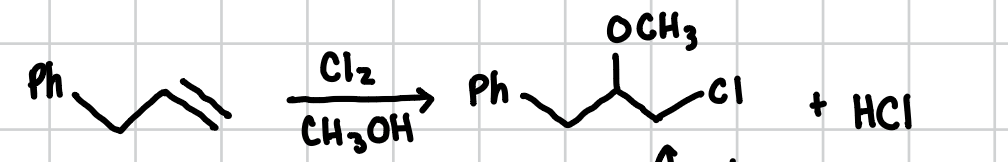
Intermolecular Haloetherification of Alkenes
Can occur when there is a detached alcohol group that can attach one side of an alkene while the halogen attaches to the other in a trans fashion.
Reagents: X2, CCl4
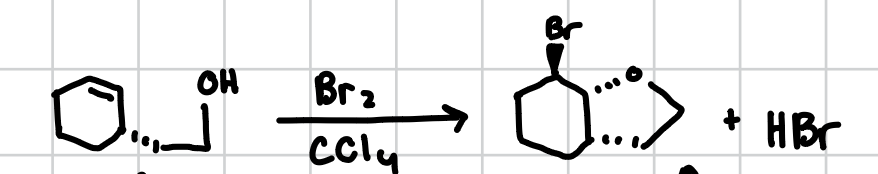
Oxymercuration of Alkenes
Adds an alcohol group to the most substituted side of an alkene
Reagents: 1) Hg(OAc)2, H2O, THF 2) NaBH4, NaOH, H2O
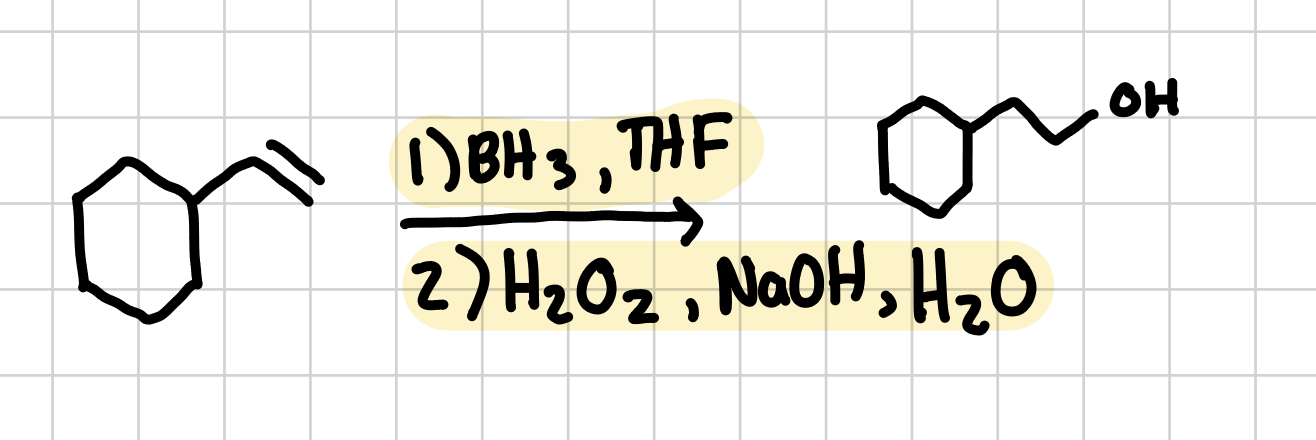
Hydroboration Oxidation
Adds a hydroxyl group to the least substituted side of the alkene
Reagents: 1) BH3, THF 2) H2O2, NaOH, H2O
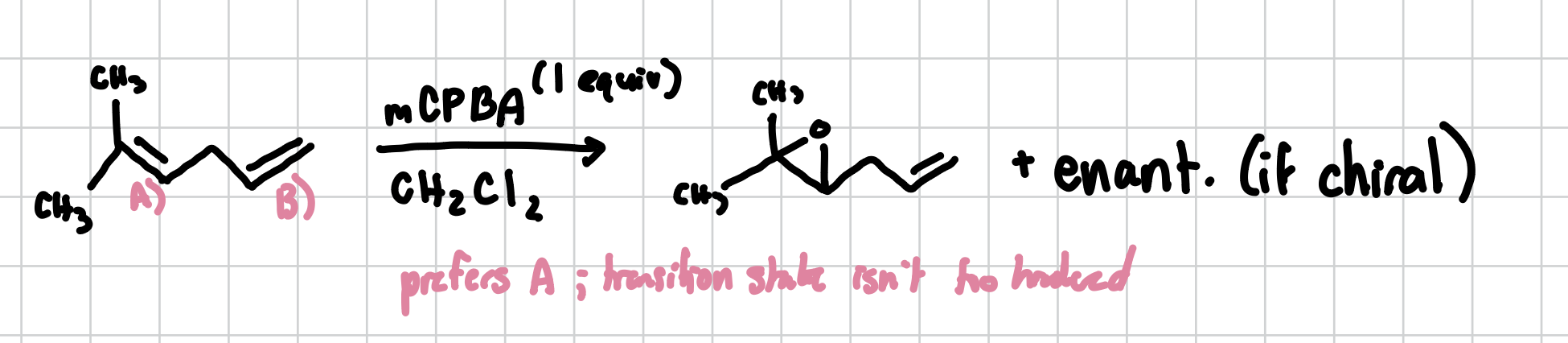
Epoxidation
Will always target the more substituted alkene. Forms an epoxide ring which is basically a oxygen triangle.
Reagents: mCPBA, CH2Cl2
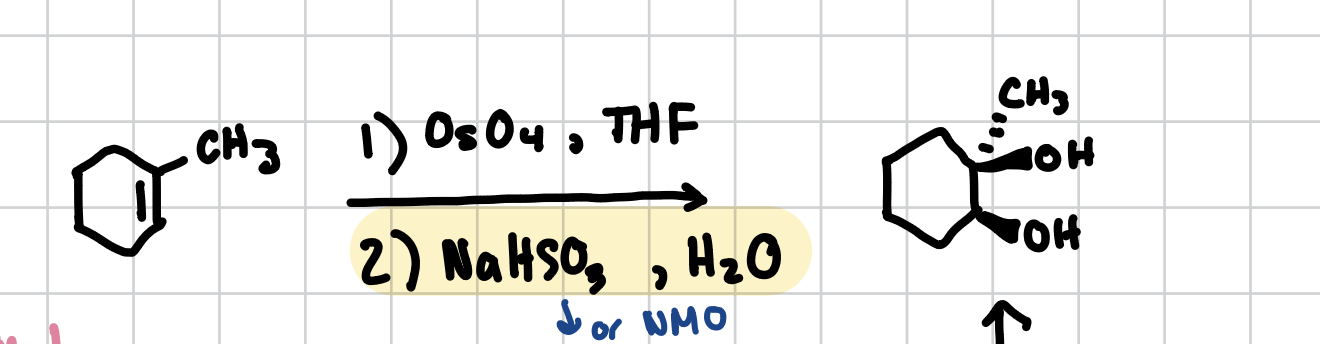
Dihydroxylation
Adds two alcohol groups in a cis fashion across an alkene.
Reagents: 1) OsO4, THF 2)NaHSO3 (or NMO), H2O
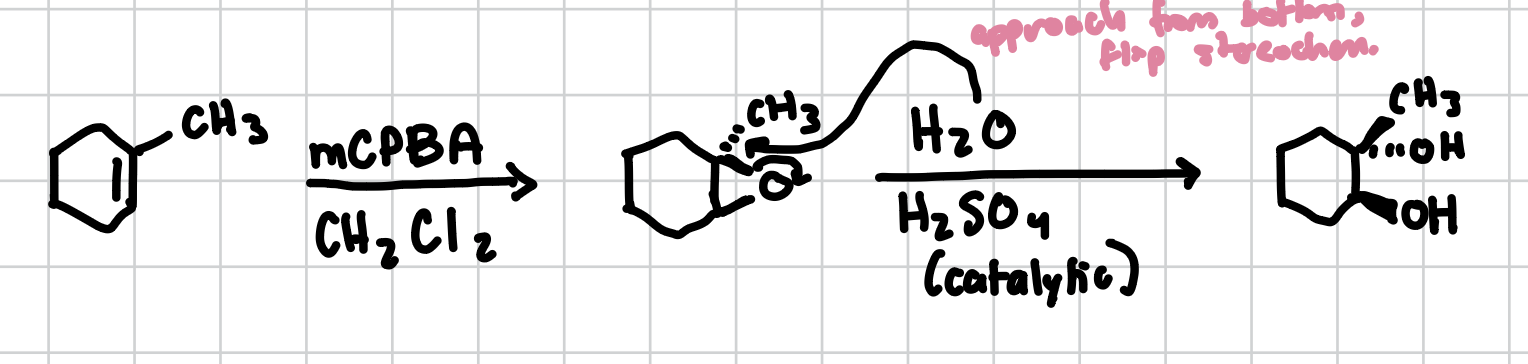
Trans-Dihydroxylation
If an epoxide is present, dihydroxylation can occur in a trans fashion adding alcohol groups trans to each other across an alkene.
Reagents: H2O, H2SO4 (cat.)
Ozonolysis
Splits the alkene in half forming two carbonyl groups (C=O)
Reagents: 1) O3, CH2Cl2 2) (CH3)2S
Cyclopropanations
An addition reaction on a carbene to create those small little triangles across it (No oxygen like in epoxides). Has three forms of carrying it out
1) Reagents: CH2N2, hv or heat. Leaves N2 as an extra product
2) Reagents: CH2I2, Zn°Cu, Et2O
3) Reagents: Connection with a =N, Cu or Rh, Et2O. Will have the cyclopropanated product as well as whatever was attached to the =N
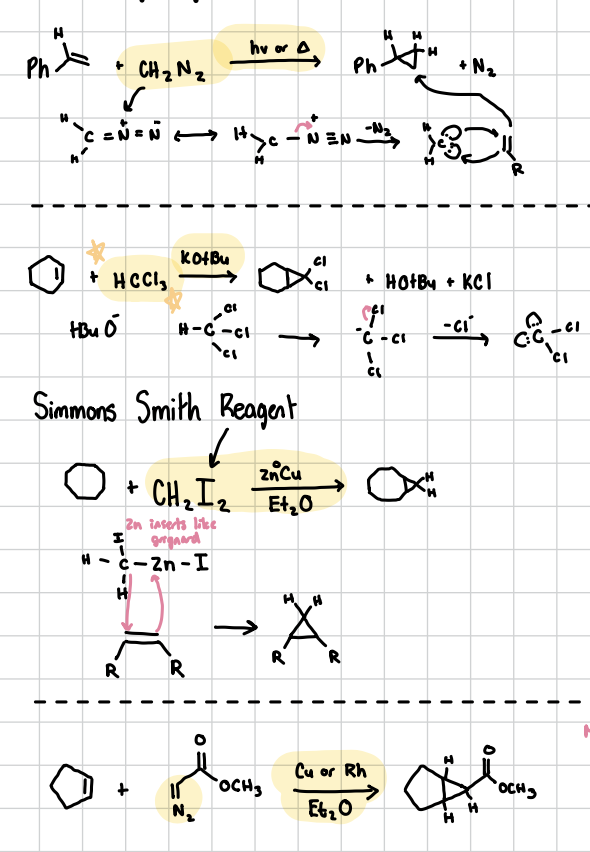
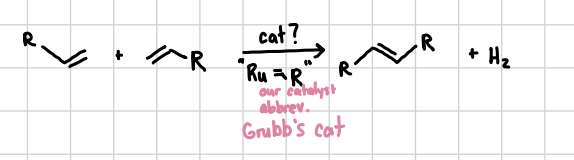
Olefin Metathesis
Will fuse two exposed alkenes together
Reagents: Grubb’s Catalyst
Alkyne Synthesis from Alkenes
Will turn an alkene into an alkyne
Reagents: 1) Br2, CCl4 2) NaNH2,NH3; H2O
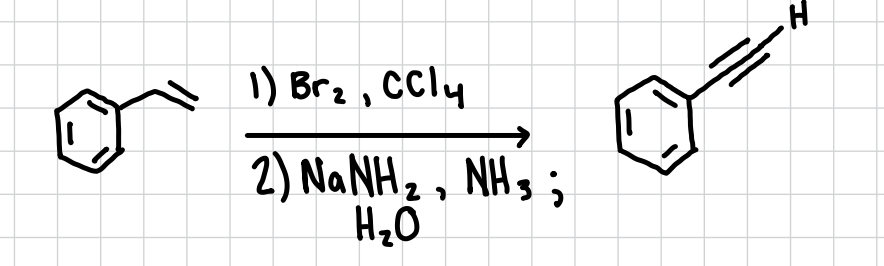
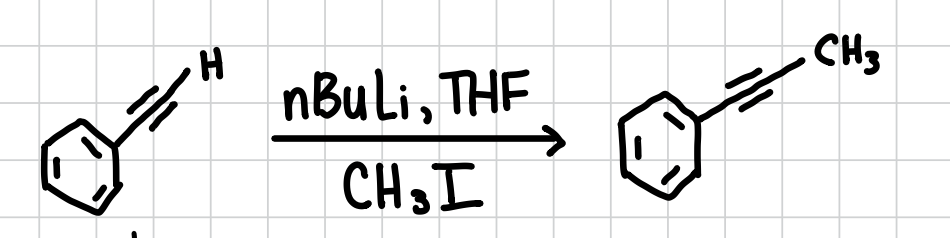
Adding a Methyl group to an Alkyne
Adds a methyl group to an alkyne.
Reagents: nBuLi, THF, CH3I
Adding a ethyl-alcohol group to an Alkyne
Adds an ethyl-alcohol group to an alkyne
Reagents: EtMgBr, Et2O; Acetone; H2O