AP Chem Flaschards: bond hybridization and molecule geomtry
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
how many sigma bonds does a tetrahedral have
4
(think methane)
bond angle tetrahedral
109.5
trigonal pyrmidial amount of sigma bonds
3 sigma bonds and one unshared pair of electrons
(think ammonia)
bond angle trigonal pyrimdial
107
bond angle for bent molecule
104.5-105 degrees
bent angle sigma bond
2 sigma bonds
2 unshared pairs
(think water)
trigonal planar amoung of sigma bonds (plane since it’s flat)
3 sigma bonds
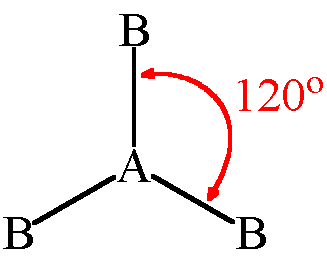
trigonal planar angle
120
angular
2 sigma bonds one unshared pair
bond angle angular
117
linear
2 sigma bond
180 degrees
what are polar molecules
molecules that have one side with more electrons than the rest of the atom (think lopsided)
what makes a molecule have a highe rlattic energy
smaller atomic radii greater charge = greater lattic energy
what do the amount of peaks in a photoelectron spectroscopy tell you
the amount of sublevels
what does the height of a bar in a photoelectron spectorcopy tell u
the amoung of electrons in the shell
what does the last peak in a PES tell u
the last peak shows the one with the lowest ionization energy
how to you calculate ionization energy per electron from PES
multiply ionization energy by a million joules (if ionization energy given in MJ) and divide by avagadros number
also with this the first ionization energy u will lose the peak furthest to the right if u are asked to calculate that