chemistry of life
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
why are lipids hydrophobic?
contain a hydrocarbon tail of CH2S which is nonpolar.
difference between saturated and unsaturated
saturated - no double bonds
unsaturated- double bonds (less hydrogen molecules per carbon)
what are steroids?
lipids made of four carbon rings.
difference between catabolic and anabolic
catabolic - breaking down through hydrolysis (add water)
anabolic - building through dehydration synthesis (remove water)
describe the properties of water.
cohesion
adhesion
evaporative cooling
universal solvent
surface tension
why is water a polar molecule?
positive hydrogens at one end and negative oxygen at another
why are hydrogen bonds weaker than other bonds?
intermolecular bond which means its between molecules, which are generally weaker.
what element must be included in an organic compound?
carbon and sometimes hydrogen
describe functional groups
amino - nitrogen based, NH2 or NH3 - amines
carbonyl - hydrophilic and polar - two structures ( C=O )
carboxyl - similar to carbonyl plus hydroxide, acidic
hydroxyl - alcohols, hydrophilic and polar
phosphates - energy sources (atp, adp, gtp) - acidic
sulfhydryl - amino acids (methionine, cytosine)
what are phospholipids?
lipids with glycerol molecule and two fatty acids and phosphate group.
what is unique about phospholipids?
amphipathetic - hydrophobic tail inside and hydrophilic head outside
what are the elements involved in carbs?
Carbon, Hydrogen and Oxgyen. 1:2:1
why is cellulose harder to break down?
cellulose is harder to break down since you can hydrolyze the bonds - no enzymes to break the bonds
state three types of carbs
monosaccharides, disaccharides, polysaccharides
what is sucrose?
disaccharide in plants for energy - fructose and glucose
what is lactose?
disaccharide found in dairy - galactose and glucose
what enzyme breaks lactose down?
lactase
what is the difference glycogen and starch
starch is glucose molecules linked together and is a storage for, for plants
glycogen is found in liver and cells from glucose molecules linked together
both are for energy storage
what are two important polysaccharides?
cellulose and chitin
what are the functions of a protein?
serve as structural components, transport aids, enzymes and cell signals.
what does an amino acid consist of?
carbon center surrounded by an amino and carboxyl group, hydrogen and an R group
describe the structure of proteins
primary
secondary
tertiary
quaternary
what is the process of protein synthesis?
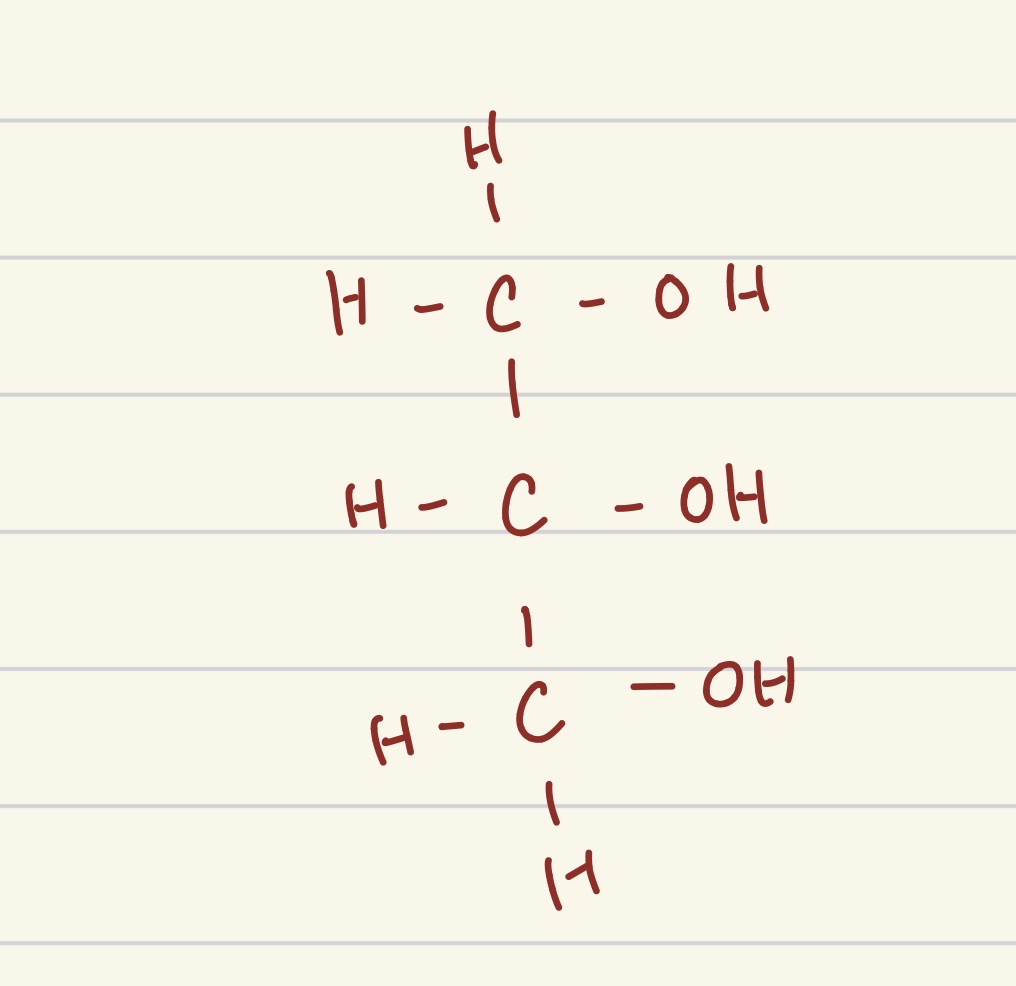
which part of a lipid is this?
glycerol
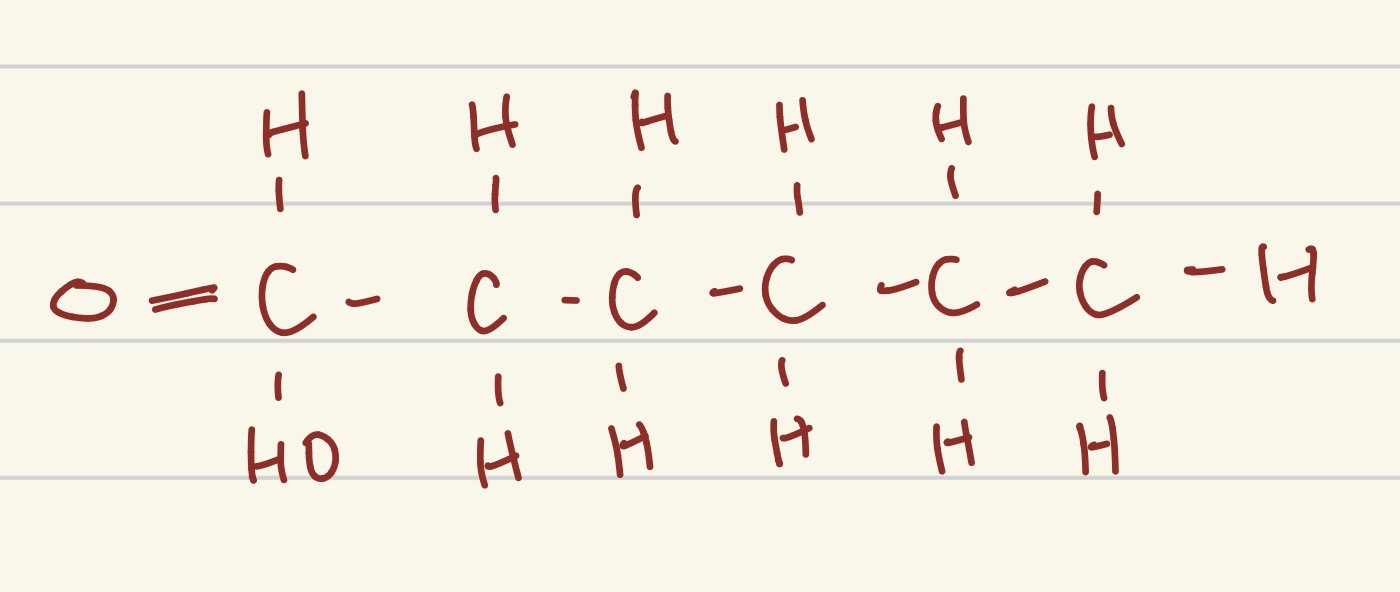
which part of a lipid is this?
fatty acid
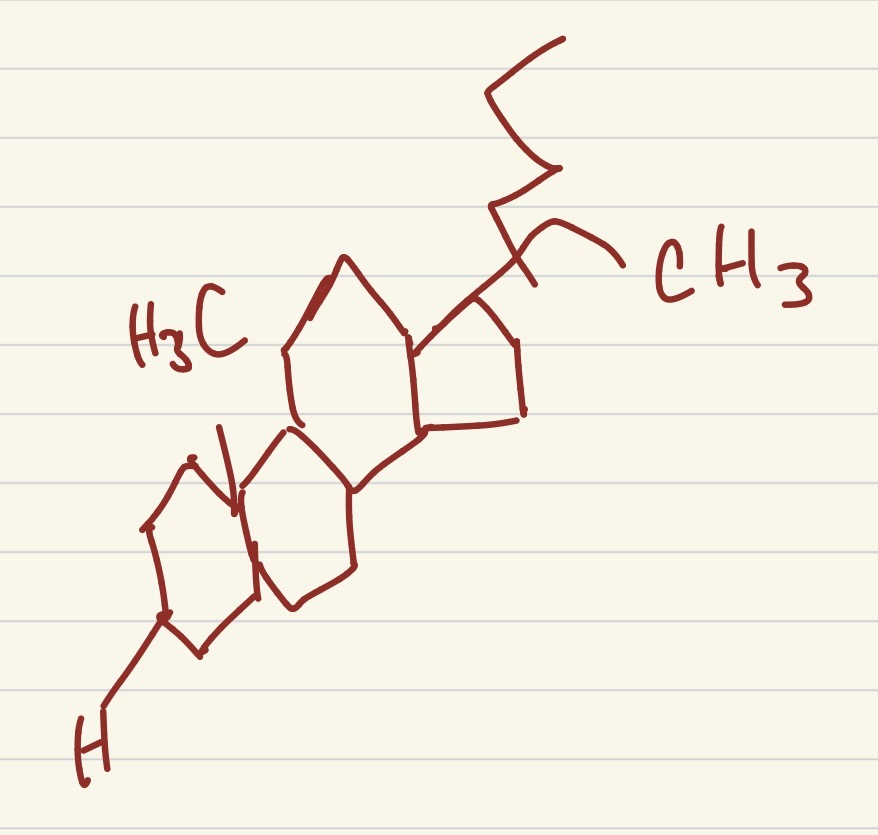
what type of steroid is this?
cholesterol
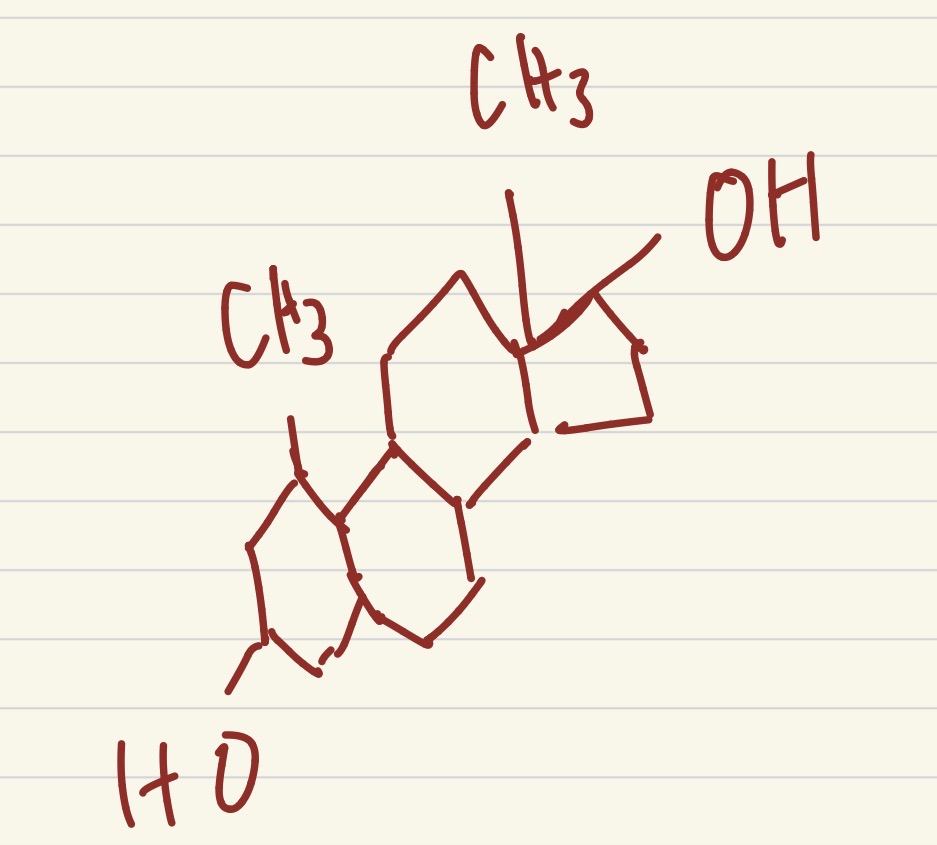
which steroid is this?
testosterone

what is this structure
phospholipid
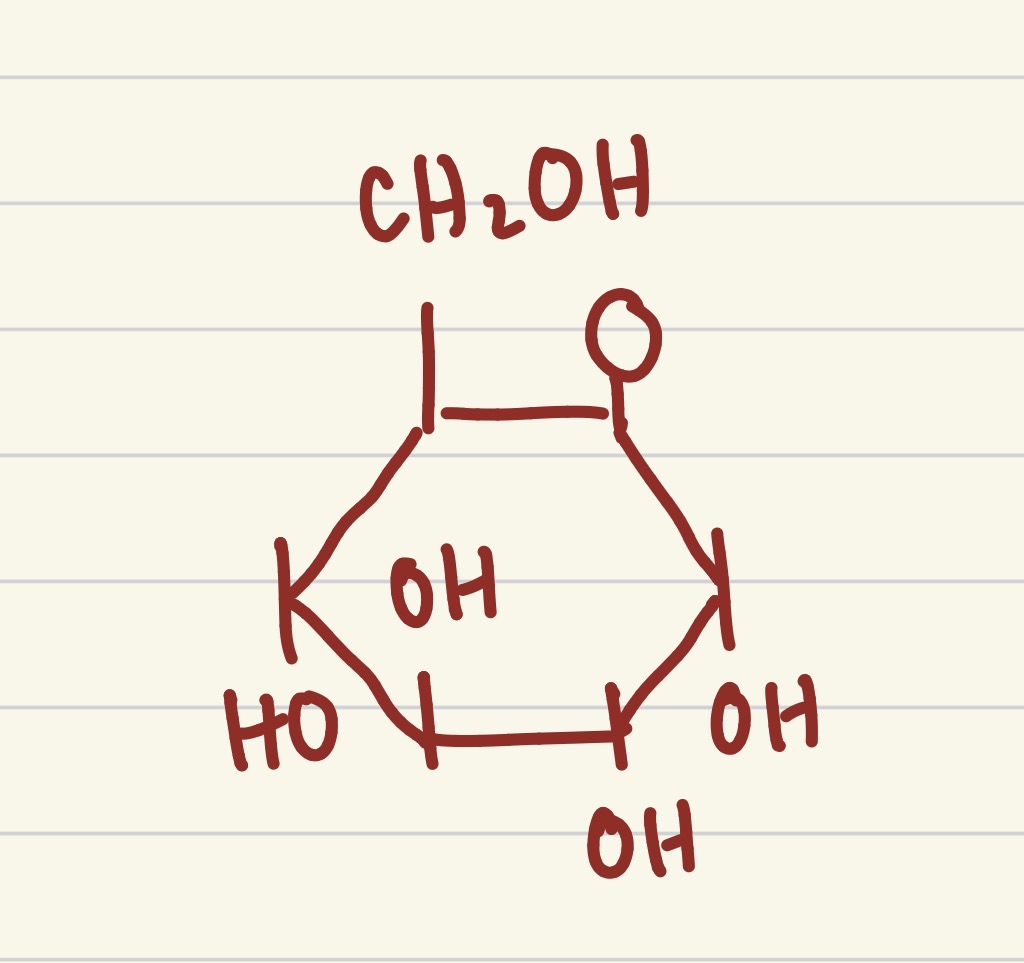
what is this structure
glucose
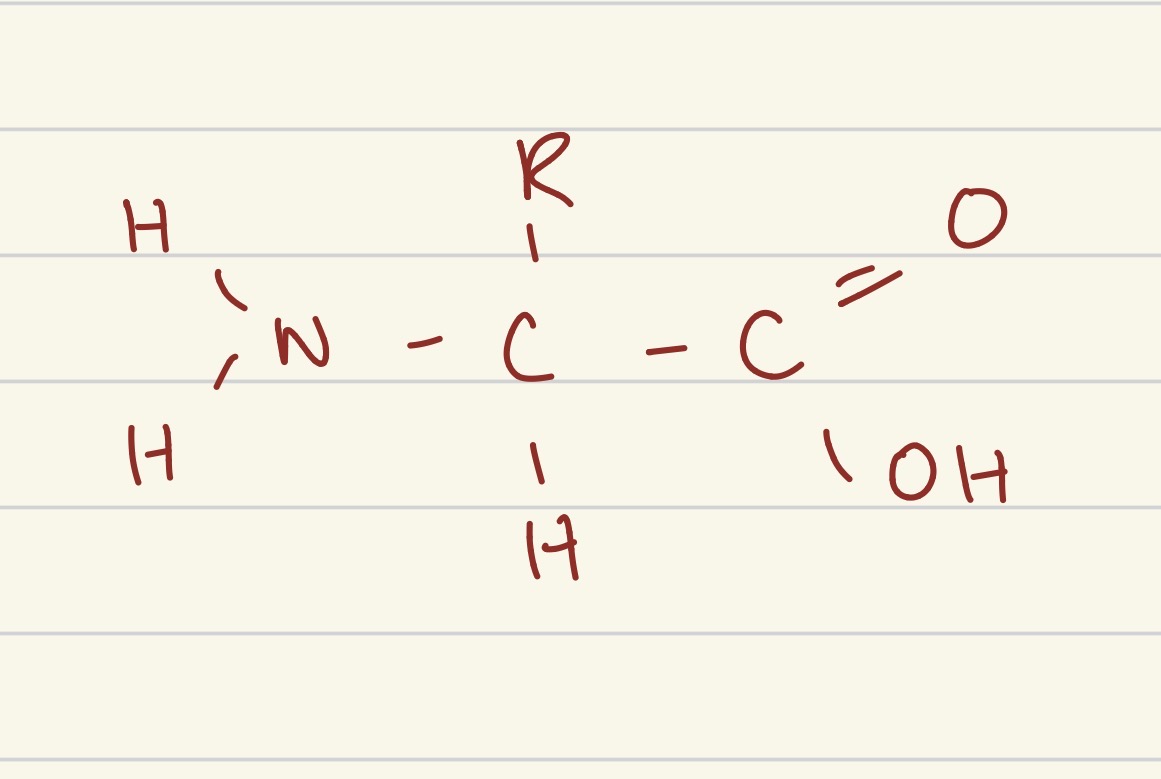
what is this structure
amino acid
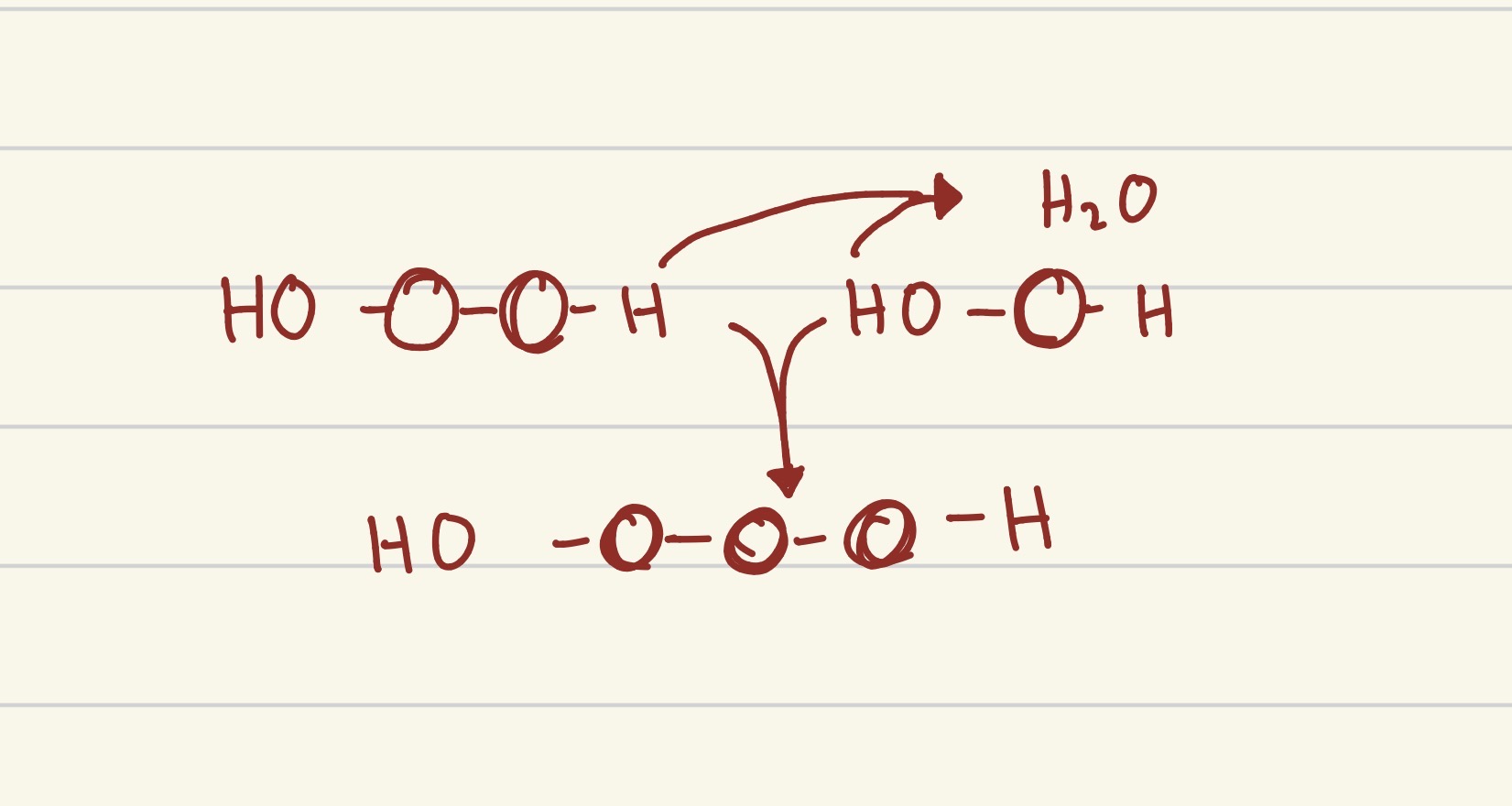
what process is this?
dehydration synthesis
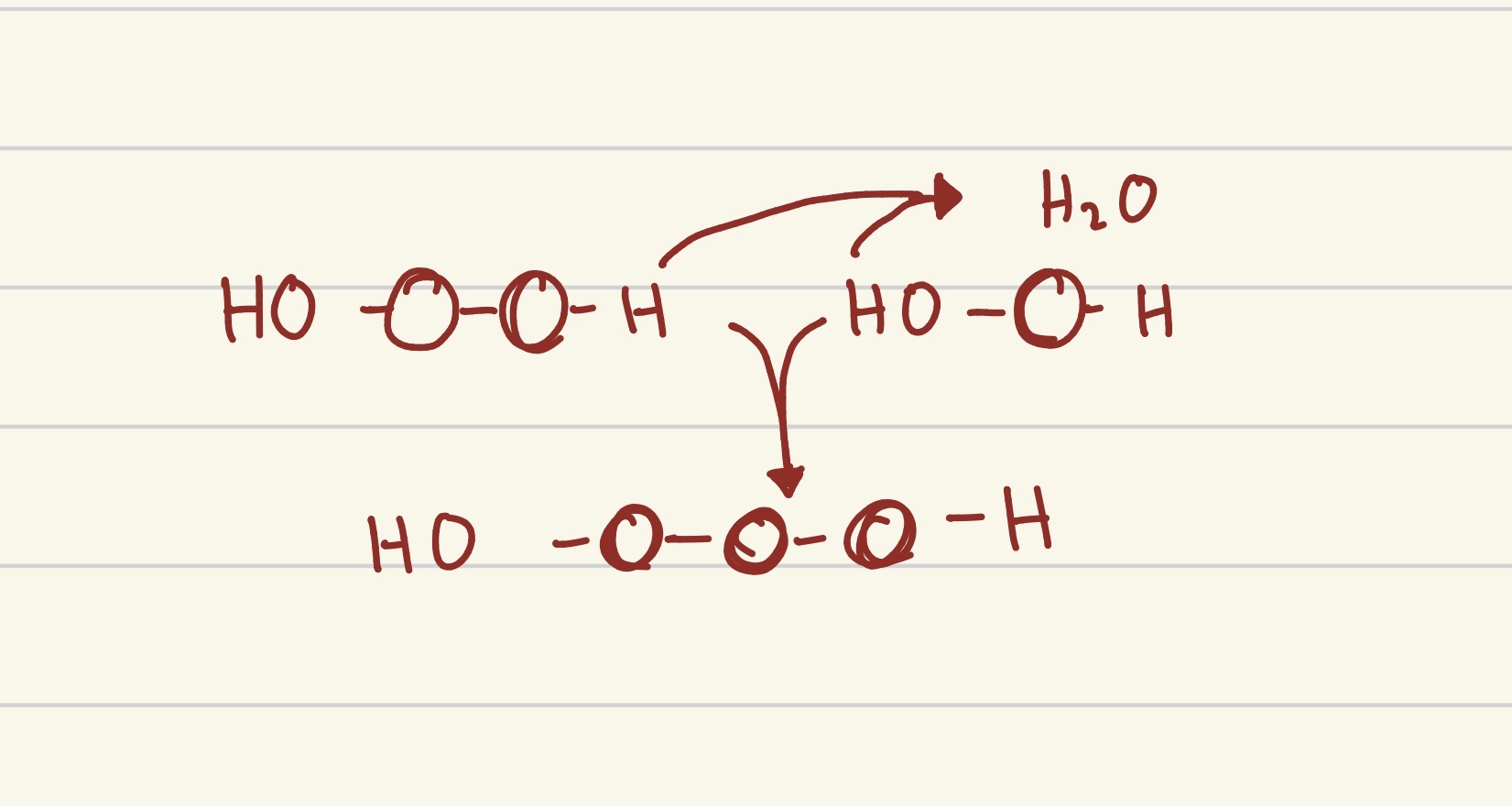
what process is this?
hydrolysis
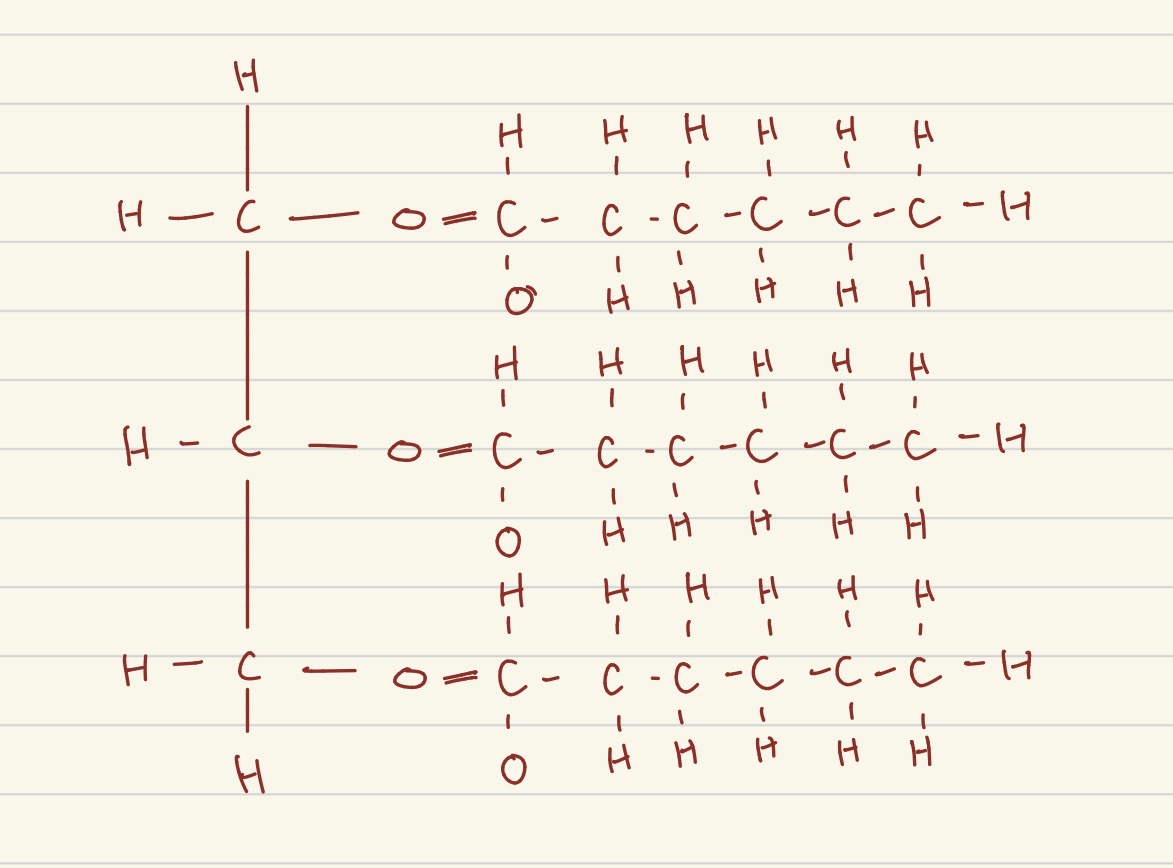
what structure is this?
triglyceride
What is the role of DNA and what is it made of?
genetic information- made of four nitrogenous bases (atcg)
A=T - two h bonds
C=G three h bonds
what is adenine and guanine?
purine with a double ring structure.
what is thymine and cytosine?
pyrimidine - single ring structure.
what is the structure of DNA?
double helix.
what is DNA composed of?
two strands of nucleotides held together by hydrogen bonds. consists of sugar phosphate backbone that keeps nucleotides connected.
How is DNA arranged?
5’ end and 3’ end with the other strand in the opposite direction
What is RNA and what is it made of?
ribonucleic acid - sugar phosphate backbone
and is single stranded (aucg)
three types of RNA and their roles
mRNA ( messenger)
tRNA (transfer)
rRNA (ribosomal)
what are factors that affect the effectiveness of an enzyme?
temperature- PMOM
pH - optimal pH
substrate concentration - more subsrate = more collisions.
what is the difference between endergonic and exergonic reactions?
endo - requires input of energy
exer - gives off energy
what is an enzyme?
catalytic protein that helps break down reactions by lowering activation energy.