ACS Organic Chemistry II Final Examination
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
What is the IUPAC priority for naming?
Carboxylic acid > Ketone > Aldehyde > Alcohol
In IUPAC naming, what is the suffix used for ketone groups?
-one
In IUPAC naming, what is the suffix used for alcohol groups?
-nol
In IUPAC naming, what is the suffix used for esters?
-noate
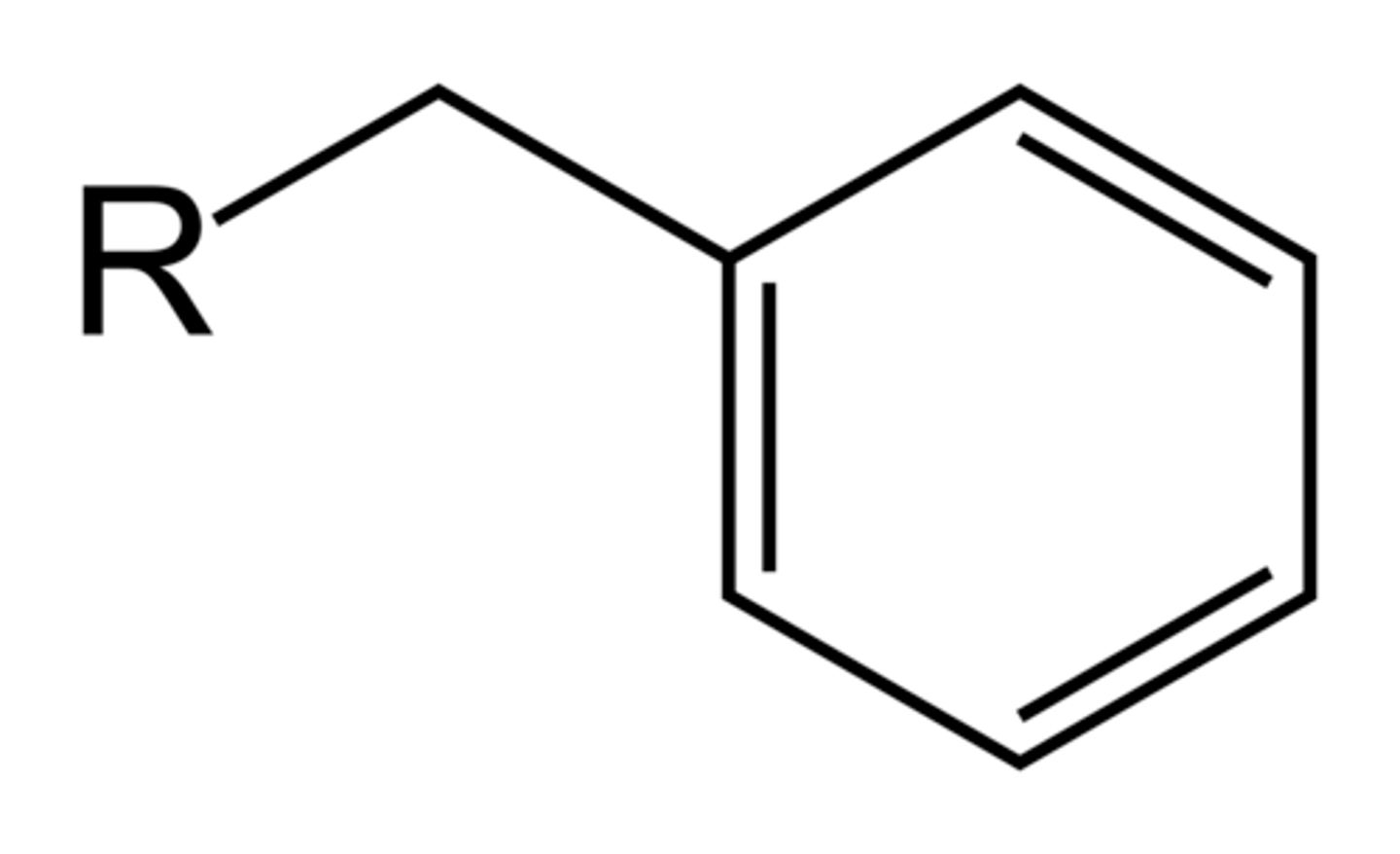
Benzyl group
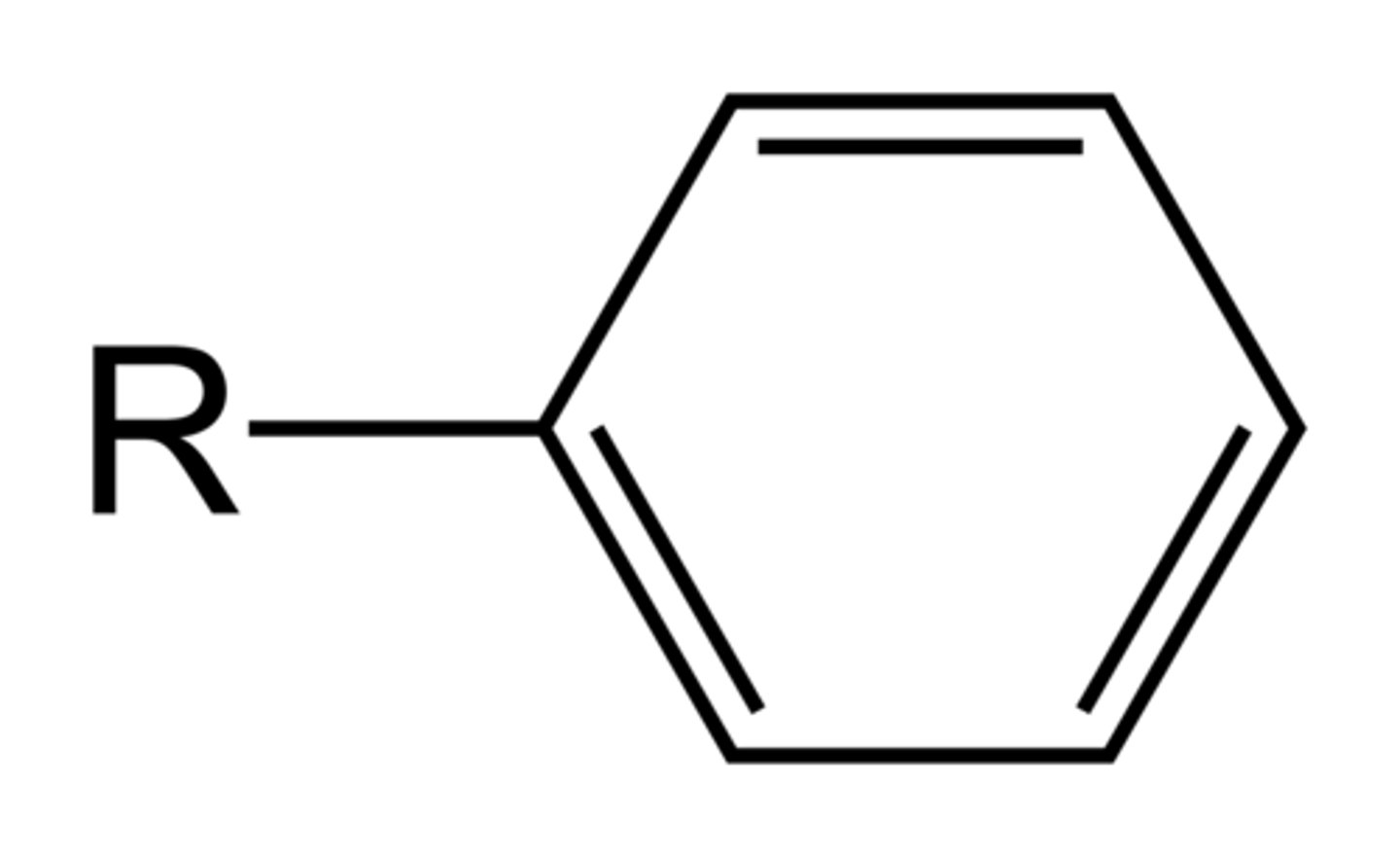
Phenyl group
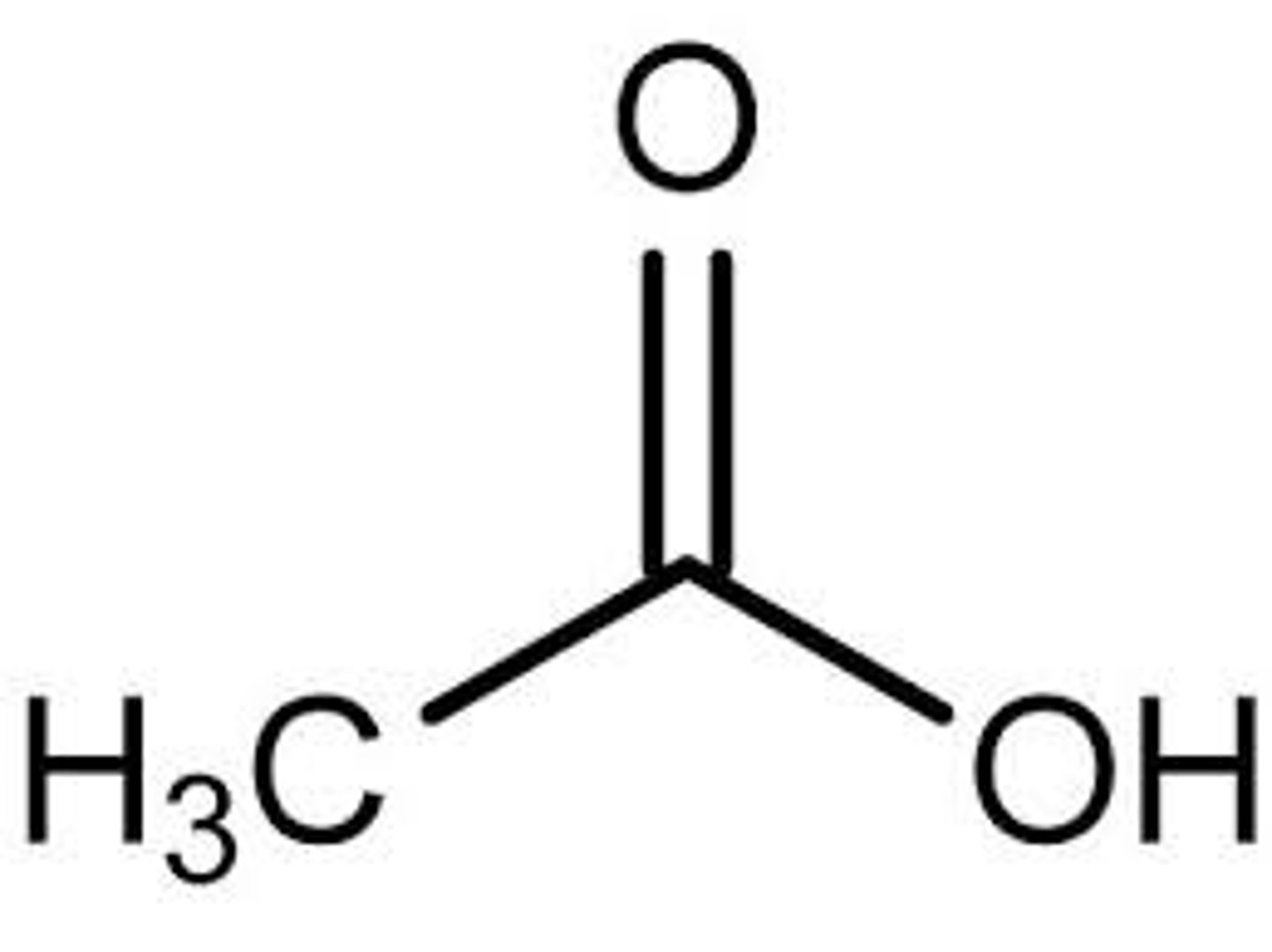
ethanoic acid
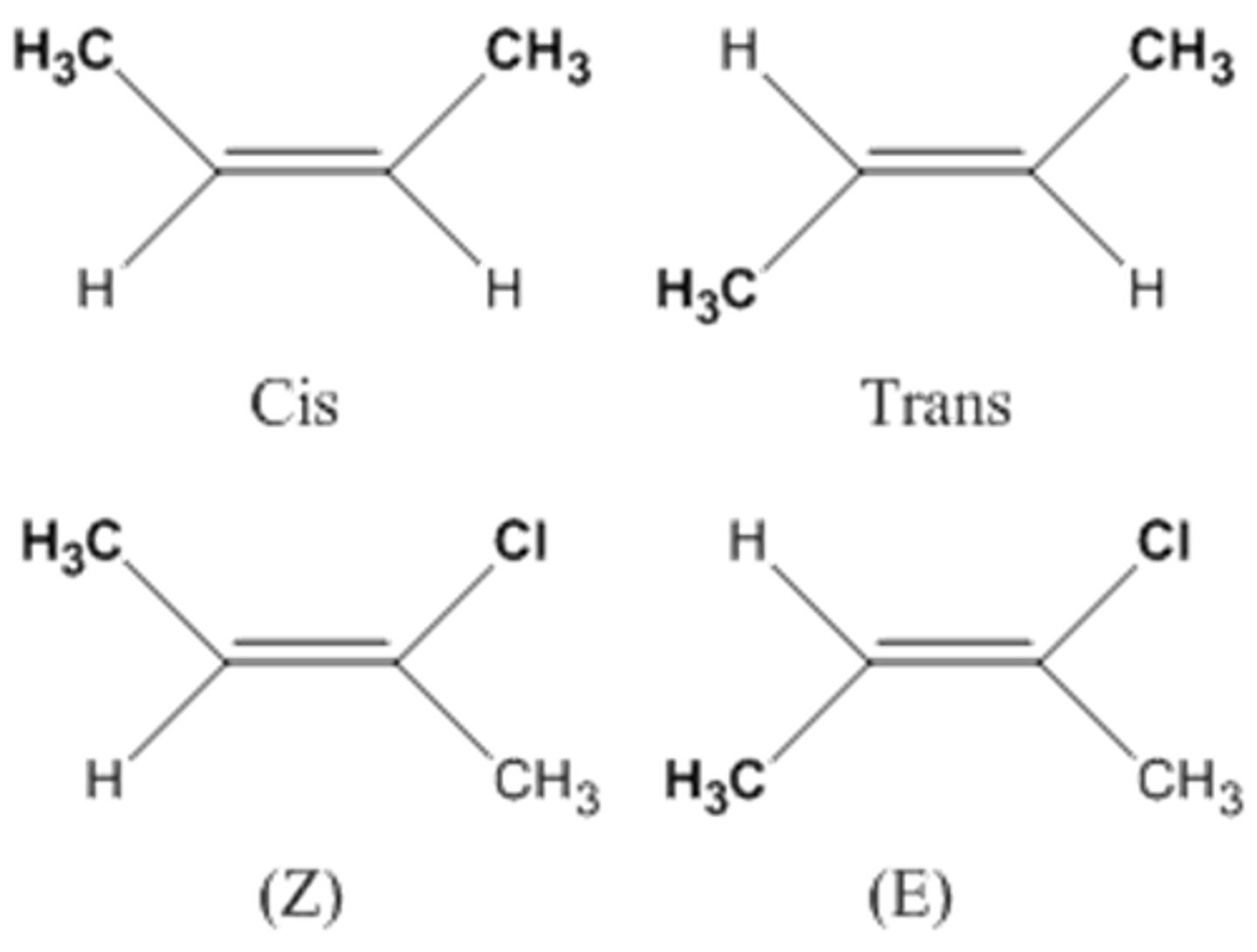
(E) isomers vs. (Z) isomers. Which one is observed when the higher ranked molecules are on the same side? opposite?
highest on same side: Z
highest on opposite sides: E
Electron-withdrawing groups are what type of directors? What is the one and only exception?
Meta (deactivators)
Halogens
If the atom directly attached to the aromatic ring has lone pairs what type of director is it? what if it does not have lone pairs?
lone pairs: activating, ortho, para director
no lone pairs: deactivating, meta director
When are the terms ortho, meta, para used?
when there are 2 substituents on the aromatic ring
In IUPAC naming, what is NH2 on a phenyl ring referred to as?
aniline
Analine
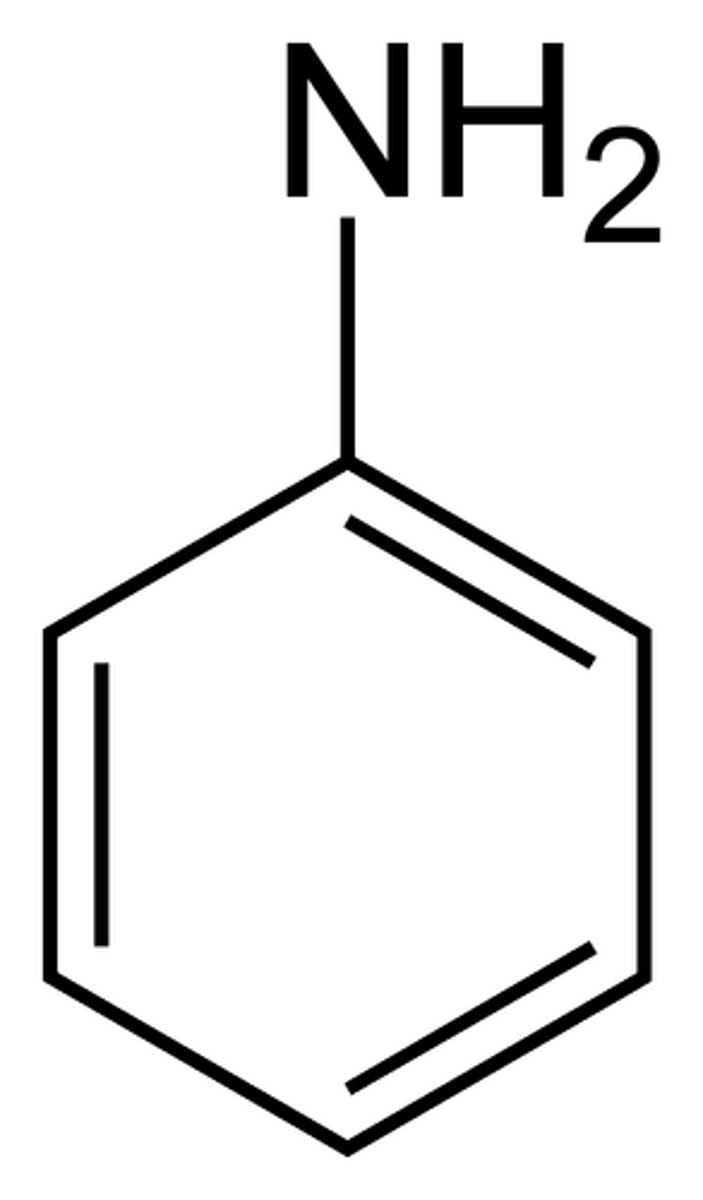
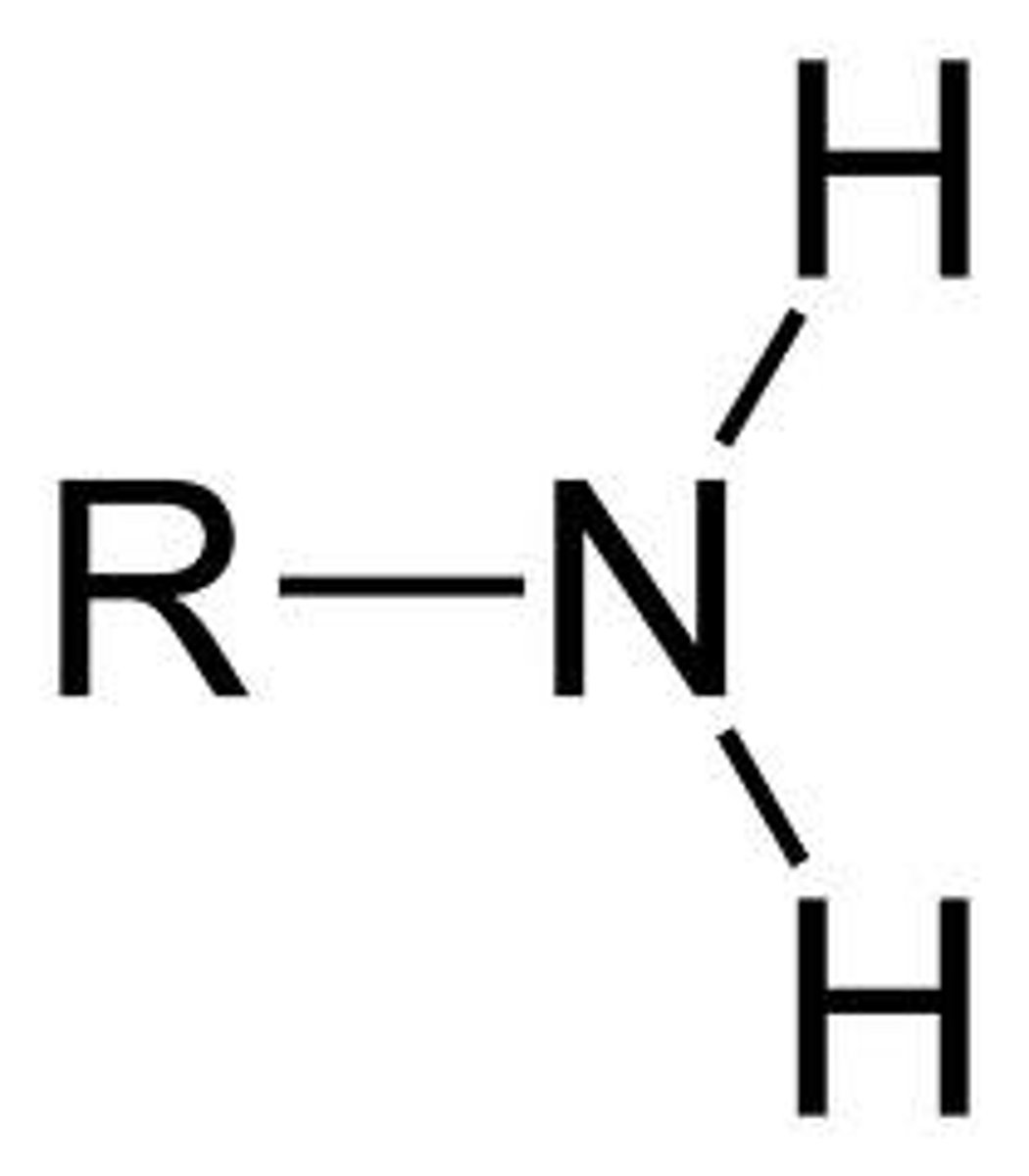
Amine
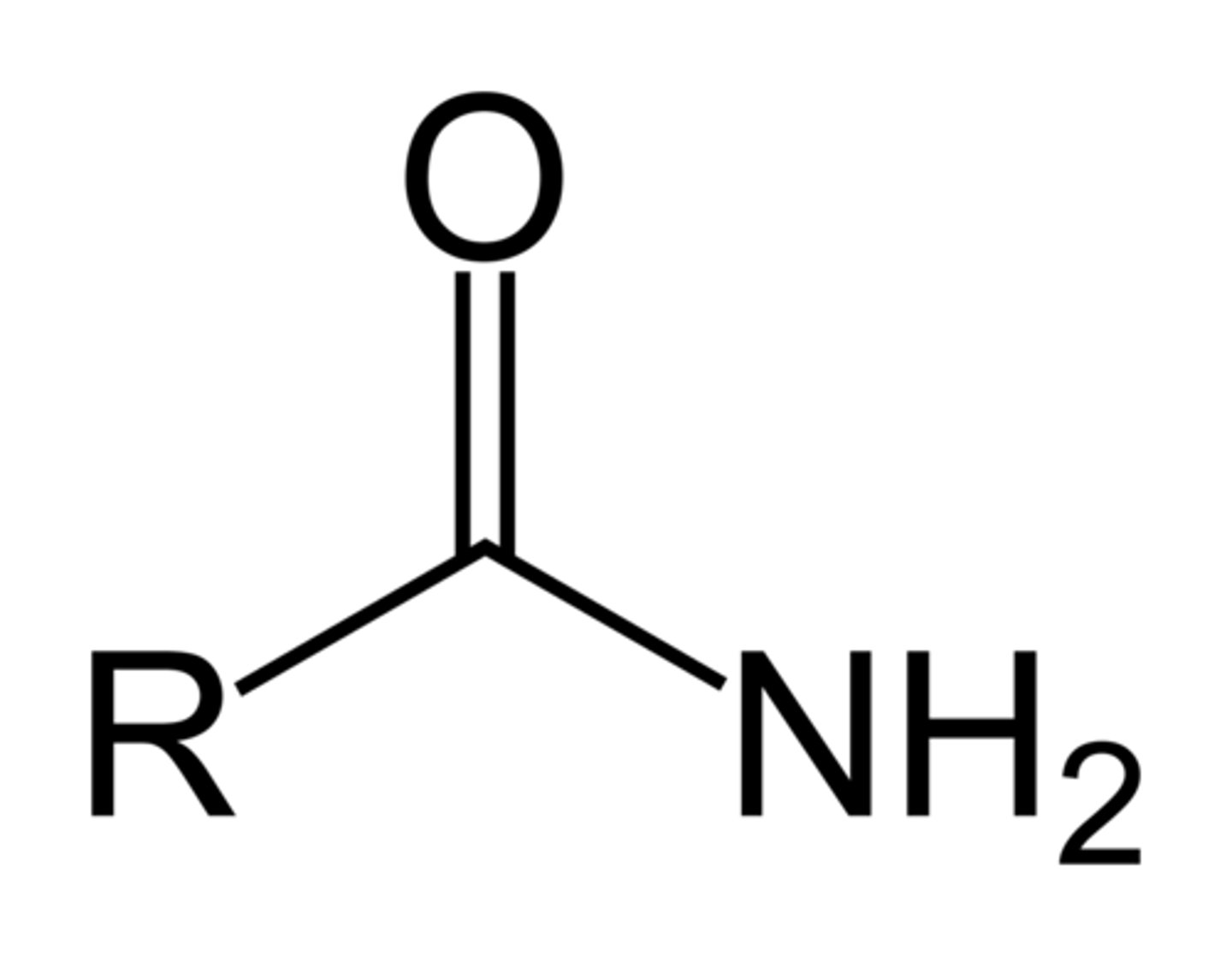
Amide
If there is a NO2 group present, what IUPAC prefix will also be evident?
nitro
What is the predicted reaction? 1º alkyl halide with a strong base? weak base? poor nucleophile?
strong base: Sn2 favored but E2 with strong non-nucleophilic bases
weak base: Sn2
poor nucleophile: no reaction
What is the predicted reaction? 2º alkyl halide with a strong base? weak base? poor nucleophile?
strong base: mostly E2
strong non-nucleophilic bases: mostly Sn2
poor nucleophile: Sn1/E2 (slow) in polar, protic solvents
What is the predicted reaction? 3º alkyl halide with a strong base? weak base? poor nucleophile?
strong base: E2
strong non-nucleophilic bases: Sn1/E1 in polar protic solvents
poor nucleophile: Sn1/E1 in polar, protic solvents
What is the configuration of the product in the base-catalyzed hydrolysis of (R)-1-choloro-1-deuteriobutane?
(S)-1-deuterio-1-butanol (Sn2)
Where is the largest molecule on the periodic table?
Bottom lefthand corner
Nucleophilic strength increase with increasing what?
atomic size
Which alkyl valid would you expect to undergo Sn1 hydrolysis most rapidly?
I, Br,Cl, or F?
I
In which reaction is a racemic mixture obtained?
In which reaction is an inversion of chirality obtained?
inversion: Sn2
racemic mix: Sn1
Cleavage of an ether usually requires a strong acid that has a conjugate base which is a strong nucleophile, such as...? Depending on the type of alkyl group, the acid reaction with the halide ion produces what?
HBr
HI
alkyl halide and an alcohol
When chirality rotates clockwise the chirality is...
R
When chirality rotates counterclockwise the chirality is...
S
A solution containing equal amounts of both enantiomers is called a..?
racemic mixture
A meso compound contains multiple __________ centers but is nevertheless ________ because it possesses reflection symmetry
chirality
achiral
For each chirality center Fischer projection - the horizontal lines are considered to be coming _______ of the page and the vertical lines are considered to be going ______ the page _
horizontal - coming out of the page (towards you)
vertical - going out of the page (away from you)
A racemic mixture of enantiomers is optically _________.
inactive
Based off chirality, how can you tell if two molecules are enantiomers?
If they contain the same molecules but one is S and the other is R
How do you convert a fischer projection to a wedge and dash diagram?
1. place lowest priority on dash
2. place highest priority up
The _____________ a molecule is, the better/stronger nucleophile it is
larger
Which type of reaction involves a carbocation intermediate?
Sn1
Which type of reaction involves a transition state
Sn2
What is the rate determining step in an Sn1 reaction?
the loss of the leaving group to form the intermediate carbocation.
What is the rate determining step in an Sn2 reaction?
the concentration of substrate and the nucleophile
which reaction will occur if the substrate is 1º?
Sn2
which reaction will occur if the substrate is 3º?
Sn2
Nucleophiles can behave as bronsted-lowry ________ because of their ability to pick up a H+
bases
Nitrile
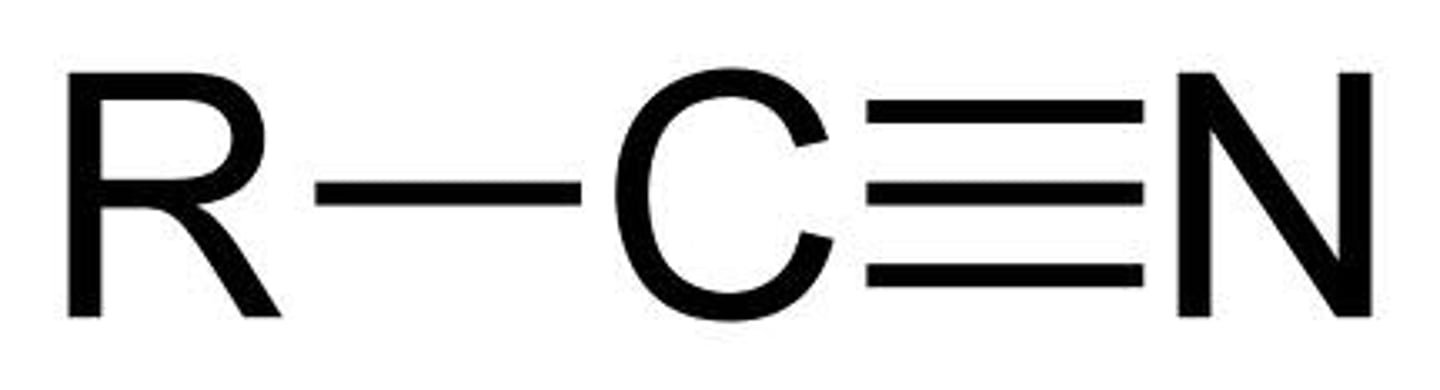
Thiol

E2 reactions occur in ________ step(s)
E1 reactions occur in ________ step(s)
E2: 1 step
E1: 2 steps
Which elimination reaction has a unimolecular rate law determination?
E1
What does the rate of an E1 reaction depend upon?
the concentration of the substrate only (unimolecular)
Which elimination reaction has a bimolecular rate law determination?
E2
What does the rate of an E2 reaction depend upon?
the concentration of the substrate and the base (bimolecular)
What is the rate determining step in an E1 reaction?
the formation of the carbocation
What is the rate determining step in an E2 reaction?
concerted (bimolecular) happens all at once
Which elimination reaction requires a strong base?
E2
Which elimination reaction requires the stereochemistry of the hydrogen to be removed to be anti to the leaving group
E2
The more substituted product is called the..?
Zaitsev product
The less substituted product is called the..?
Hoffmann product
Which isomer is more stable than the other: E or Z?
E
List common base only reagents
NaH
DBN or DBU
tertbutyl (KO+)
triethyl amine (Et3N)
How do all base only reagents reactions proceed (1º, 2º, 3º)
E2
HOCH3
weak nu
weak base
List the two ways 2 water molecules can be added across an alkene in a syn addition manner
1)KMnO4 2)H2O or 1)OsO4 2) H2O2
List the reagents necessary to add two water molecules across an alkene in an anti fashion
MCPBA, H3O+
The majority of E2 eliminations require a transition state conformation in which the B-hydrogen atom and the leaving group are _____ to each other
anti
T/F conjugated alkenes are more stable and favored in E2 reactions
True
NaCC(R)
Strong nucleophile/strong base
Why would concentrated hydrobromic acid be an inappropriate catalyst for the dehydration of alcohols
Because the conjugate base (Br-) is a good nucleophile and it would attack the carbocation to form an alkyl bromide
To double the volume of the solvent, you would multiply the reaction rate by a factor of what?
1/4
Under basic conditions, nucleophiles will attack the epoxide at the _______ substituted position
least
under acidic conditions, the epoxide becomes __________ and then the nucleophile attacks the more ________ carbon
protonated
substituted
primary alcohol treated with PCC yields?
secondary alcohol treated with PCC yields?
1º = aldehyde
2º = ketone
What two reagents can be used to convert a primary alcohol into a carboxylic acid?
KmNO4 or Na2Cr2O7/H2So4
What reagent can be used to reduce an ether to an alcohol?
1) LiAlH4 2) H2O
What reagent can be used to reduce a carboxylic acid to an alcohol?
1) LiAlH4 2) H2O
What two reagents can be used to reduce aldehydes and ketones to alcohol?
NaBH4 or 1)LiAlH4 2)H2O
What are the two rules for Diels Alder reactions?
1. The diene must always be in the "s-cis" conformation
2. Stereochemistry in the dienophole is always preserved
Under normal Diels Alder conditions, which product dominates?
Endo (everything on wedge or everything on dash)
On the chair conformation, which way does endo point? exo?
endo = down
exo = up
Besides H2O, H30+ what is another way to add OH markovnikov across an alkene?
Hg(OAC)2, H2O followed by NaBH4
What is the stereochemistry for water being added anti-markovnikov?
syn addition
Br2 adds bromines in what fashion across an alkene?
anti addition
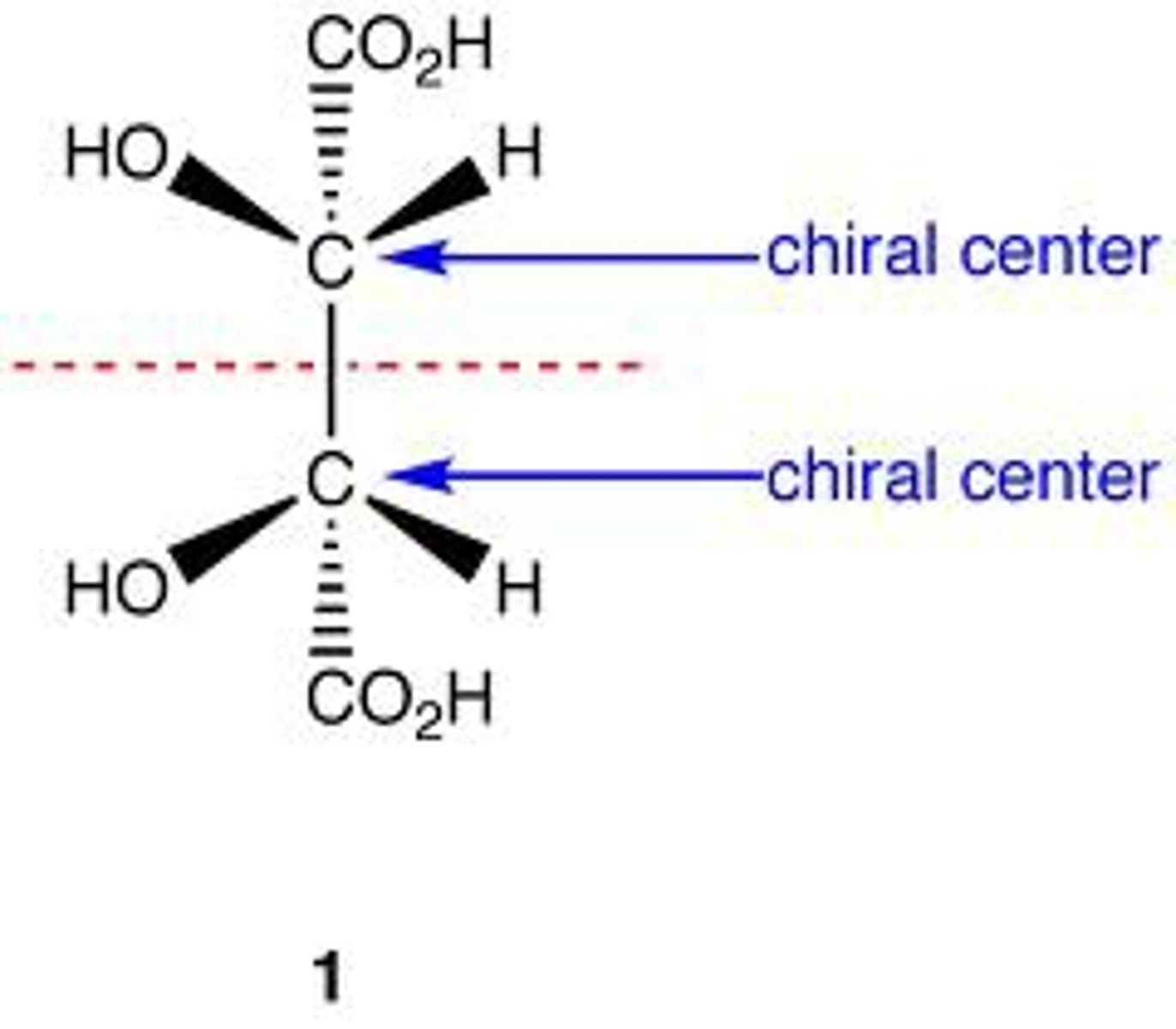
Meso compound
What is formed when an alkene is treated with a peroxyacid? (R'COOOH)
an epoxide branching from the alkene bond
What is the FINAL product when an alkyne is treated with H2O, H2SO4 in the presence of HgSO4
a ketone
What must you keep in mind with addition reactions?
METHYL OR PROTON TRANSFER / CARBOCATION REARRANGEMENTS
Under which set of conditions is a Diels Alder reaction best carried out?
heating in hexanes
Which carbonyl compound would exist to the greatest extent as its hydrate when dissolved in aqueous solution? Why?
An aldehyde would exist at a greater concentration due to electronic and steric effects
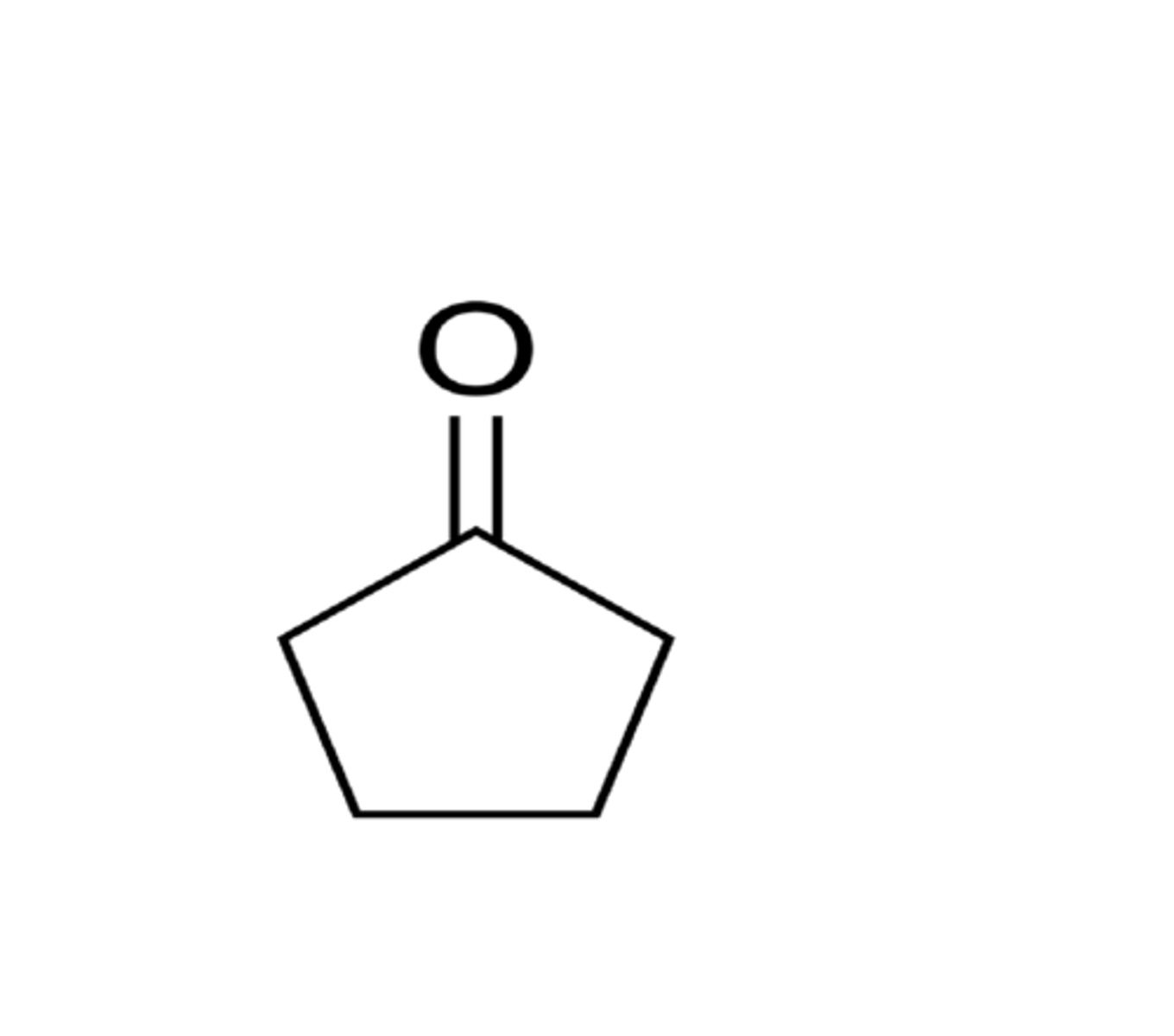
cyclopentanone
when an ether (acetal) is treated with H2O, H3O+ in the presence of heat, what occurs?
ketone
hemiacetal
are the reaction conditions TsOH, C6H6 (heat) considered acidic or basic conditions
acidic
What can/cannot NaBH4 be used to reduce?
ketones or aldehydes not esters or carboxylic acids.
Methyl vinyl ketone is a conjugated ketone that may undergo 1,2 or 1,4 addition. if the nucleophile is strongly basic (LiAlH4, Ch3MgBr, phosphonium ylide) what type of addition occurs? What about if its not as basic (NaCN)? Which type of reagent is more reversible?
strongly basic: 1,2 addition
Not as basic: 1,4 addition
Not as basic: NaCN
List 4 carboxylic acid derivatives starting most reactive to least reactive
acyl chloride
acid anhydride
ester
amide
Best way to convert a carboxylic acid to an ester
SOCl2
Which type of polymer could be readily prepared by condensation polymerization?
Any amine containing compound --> peptide
activator or deactivator: CH3
weak activator: para, oath director
conjugation causes a shift to a lower/higher wavenumber?
lower wavenumber
strong absorption at 1720
ketone
strong band 2500-3300
OH - carboxylic acid
broad with spikes 3300
amine or amide
3300 sharp
CH (sp)
just above 3000
CH (sp2)