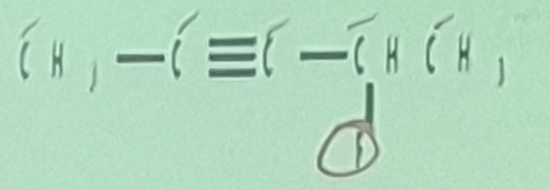Chapter 6 Unsaturated Hydrocarbons
1/72
Earn XP
Description and Tags
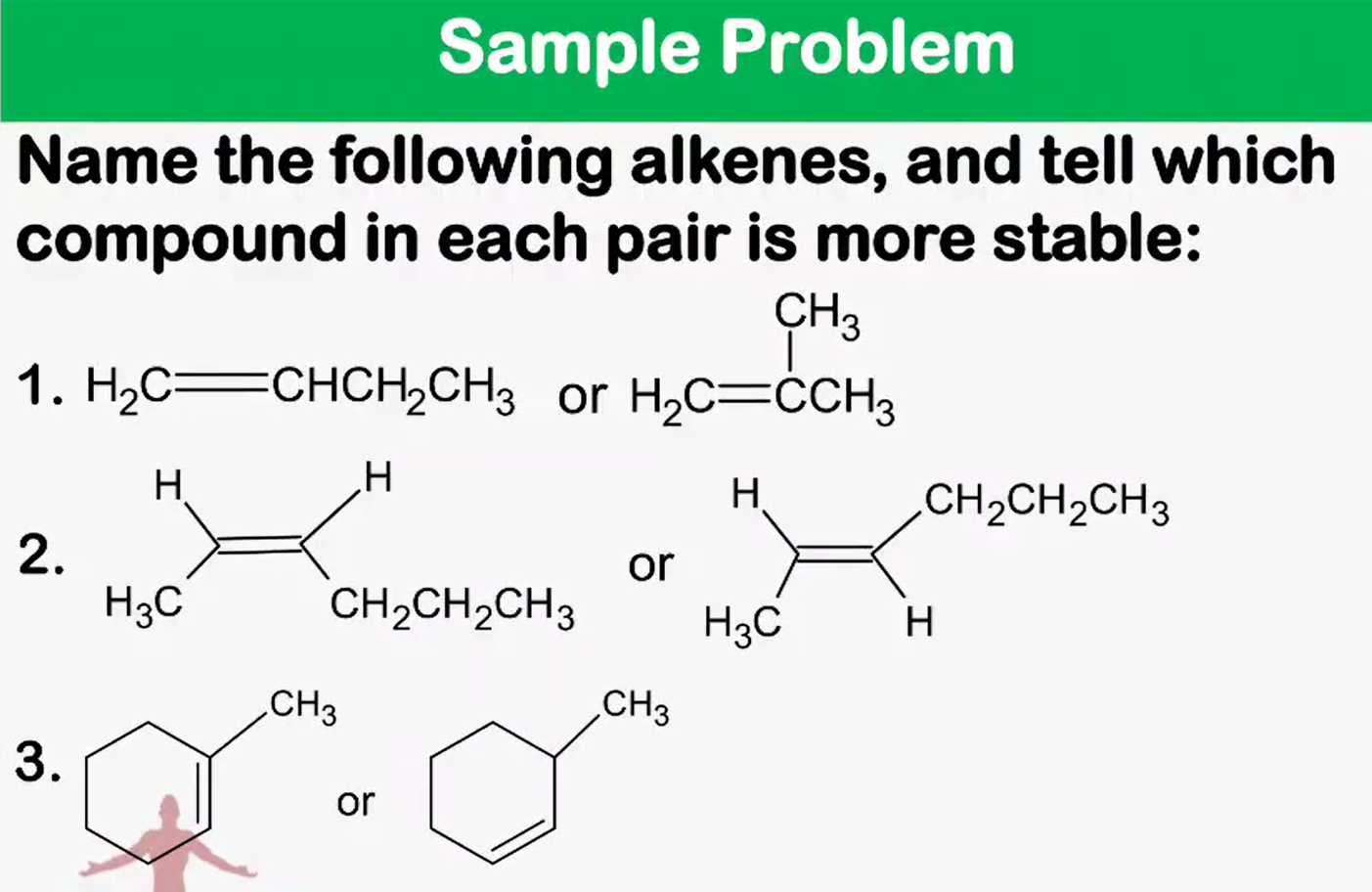
Naming of alkene, diene, cycloalkene, and cyclodiene cis/trans isomers E/Z isomers
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
Alkenes
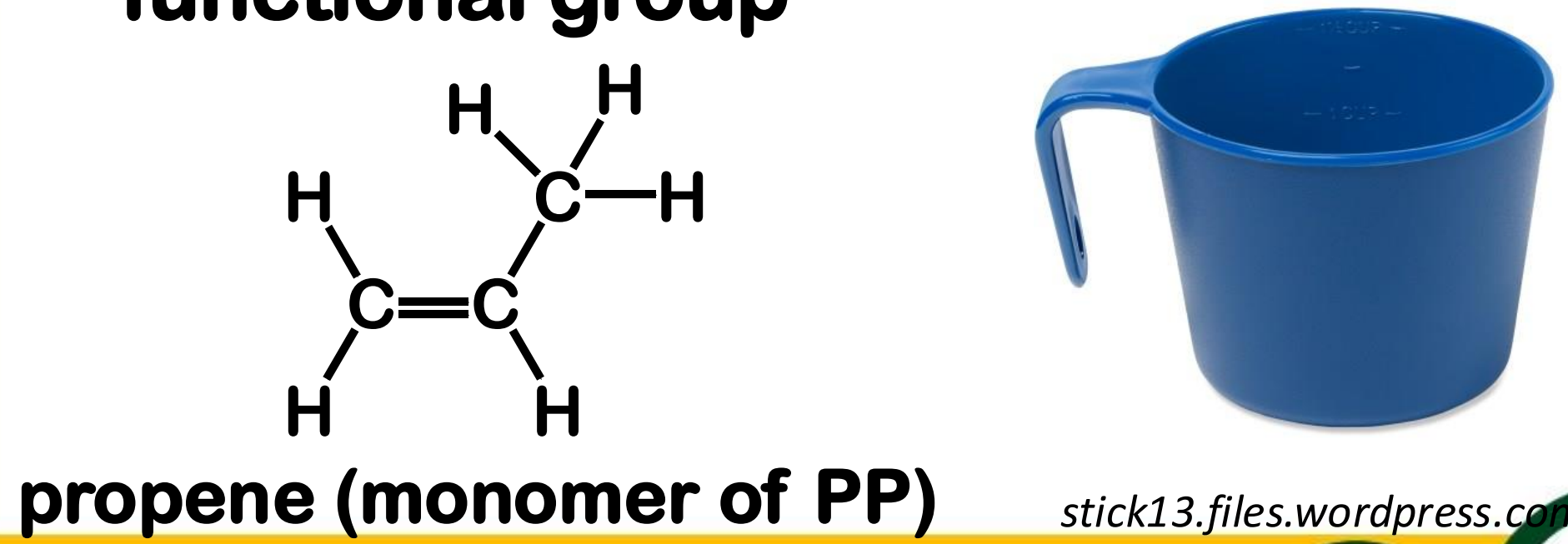
sometimes called olefins
have C-C double bonds as functional group
higher; polarizable
Physical Properties
alkenes have slightly _________ dipole moments compared to alkanes due to the loosely held pi electrons (easily ____________)
physical properties of alkenes are very similar to those of alkanes
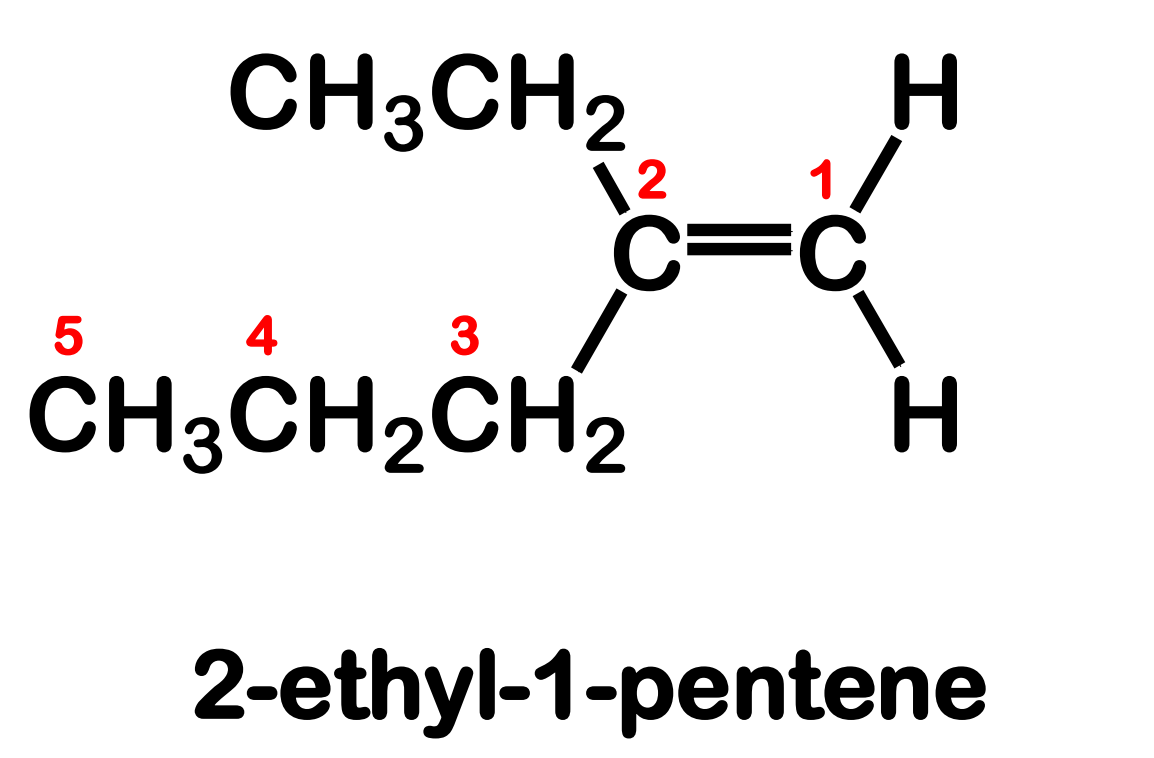
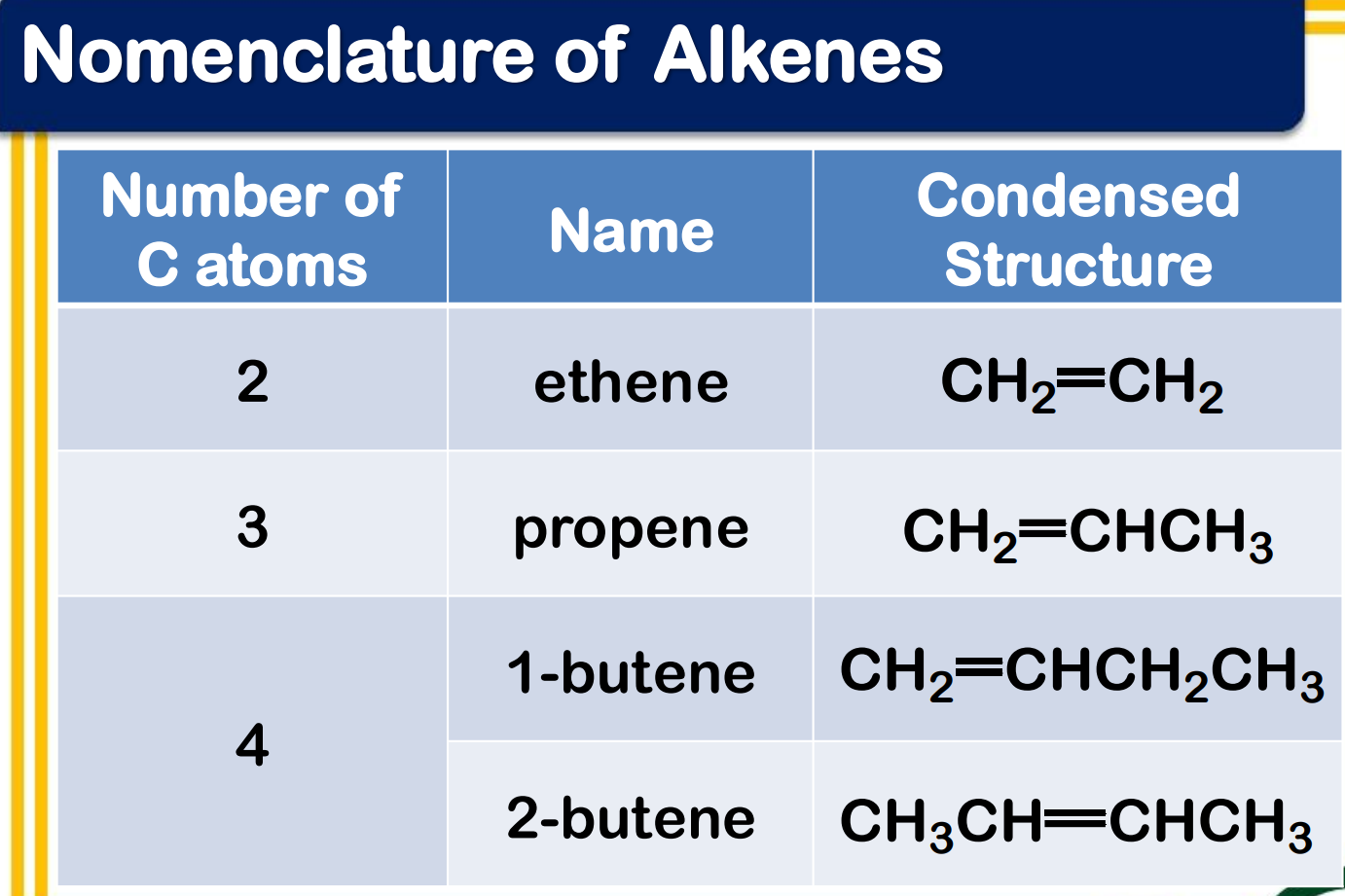
Systematic Names of Linear Alkenes
names end in –ene
except for ethene and propene, locants are used to indicate the position of the carbon to carbon double bond
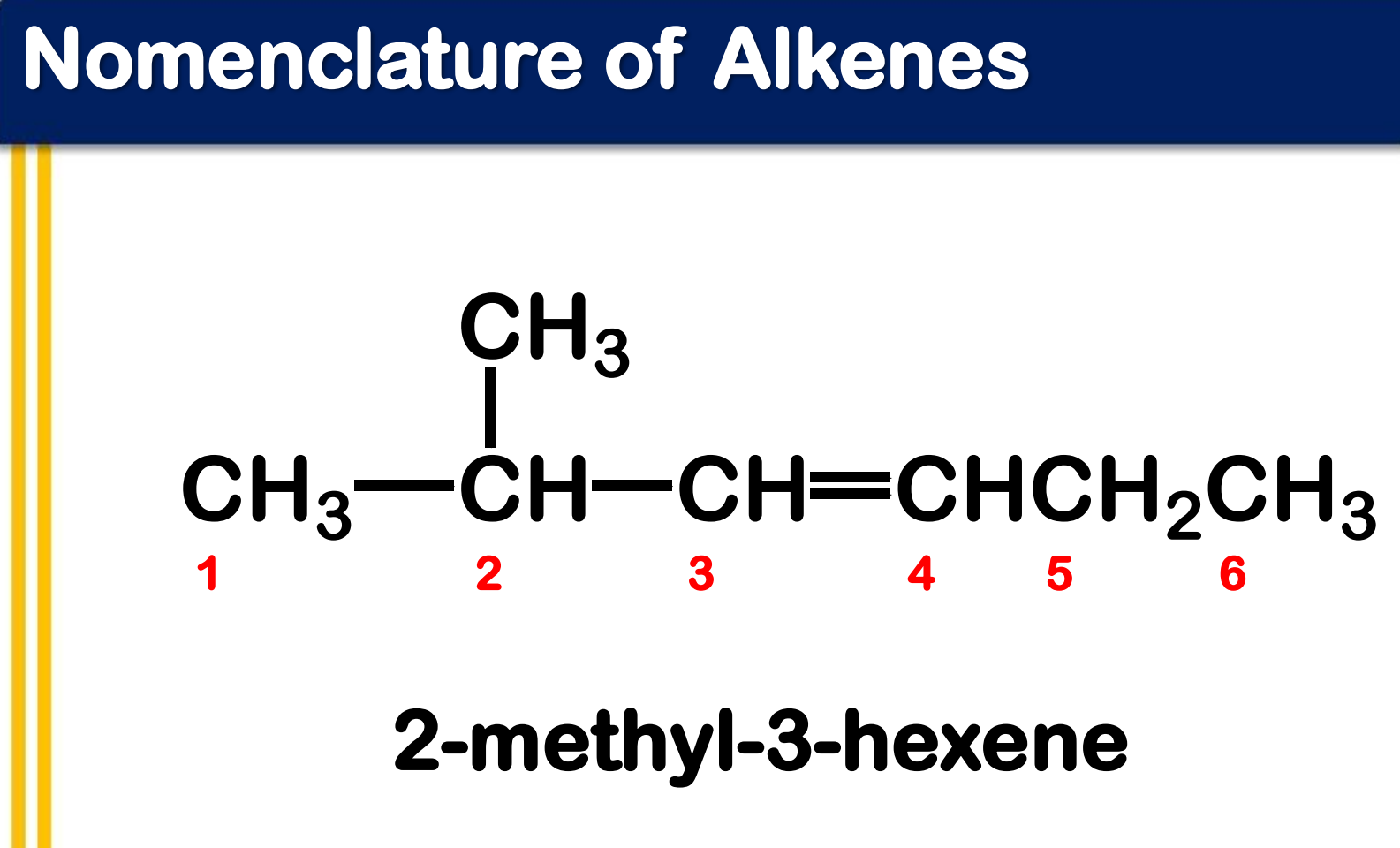
Systematic Names of Branched Alkenes
the longest chain that contains both carbon atoms of the double bond is assigned as parent chain
number the parent chain to give the double bond the lower number
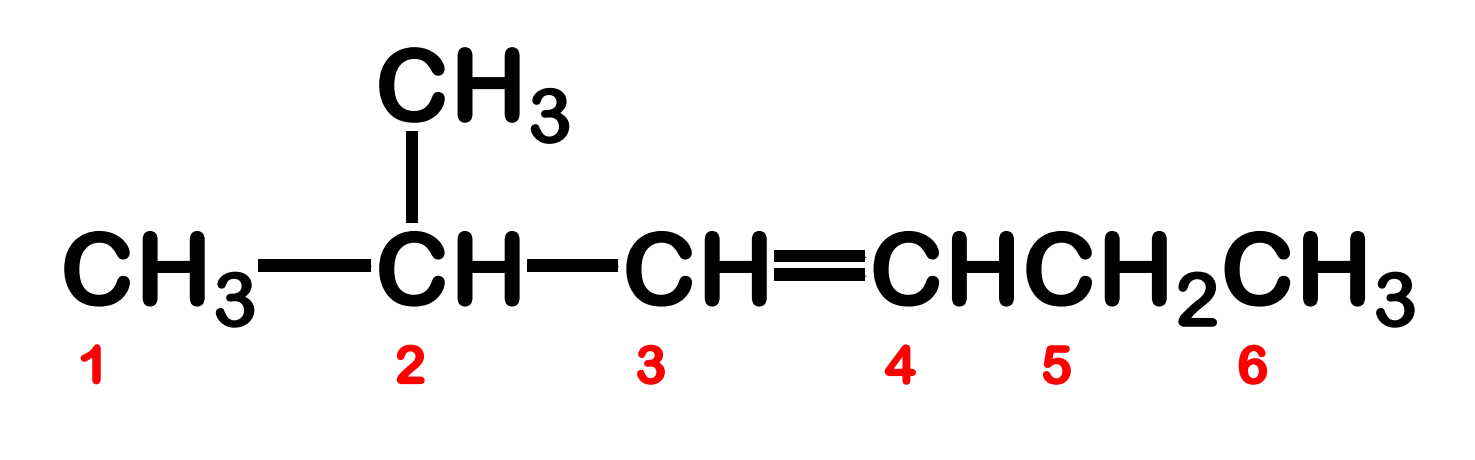
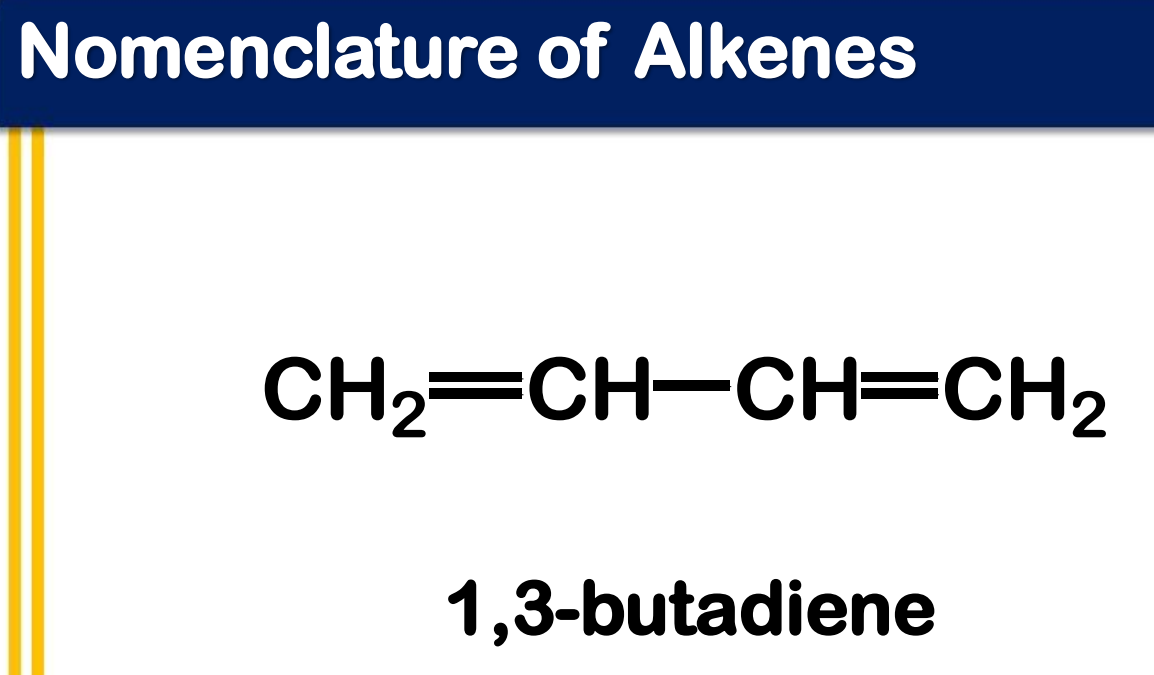
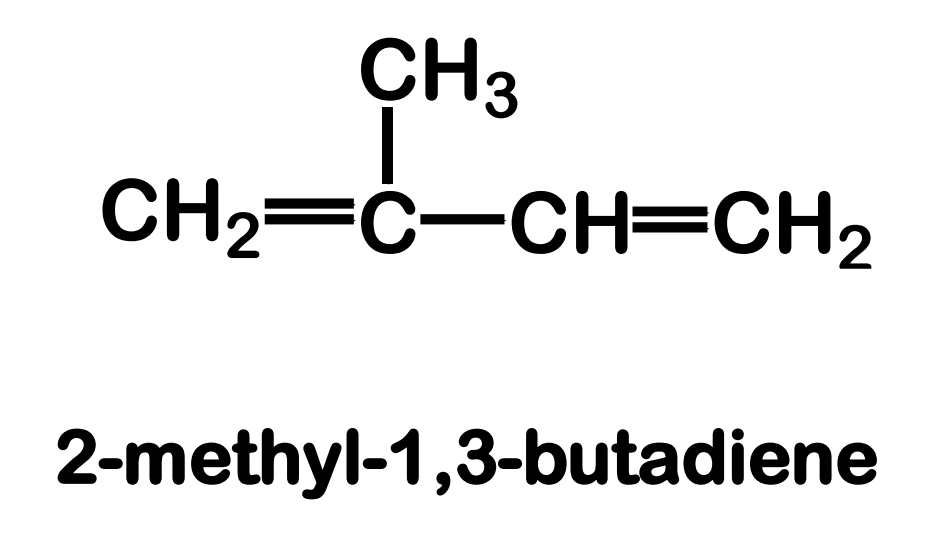
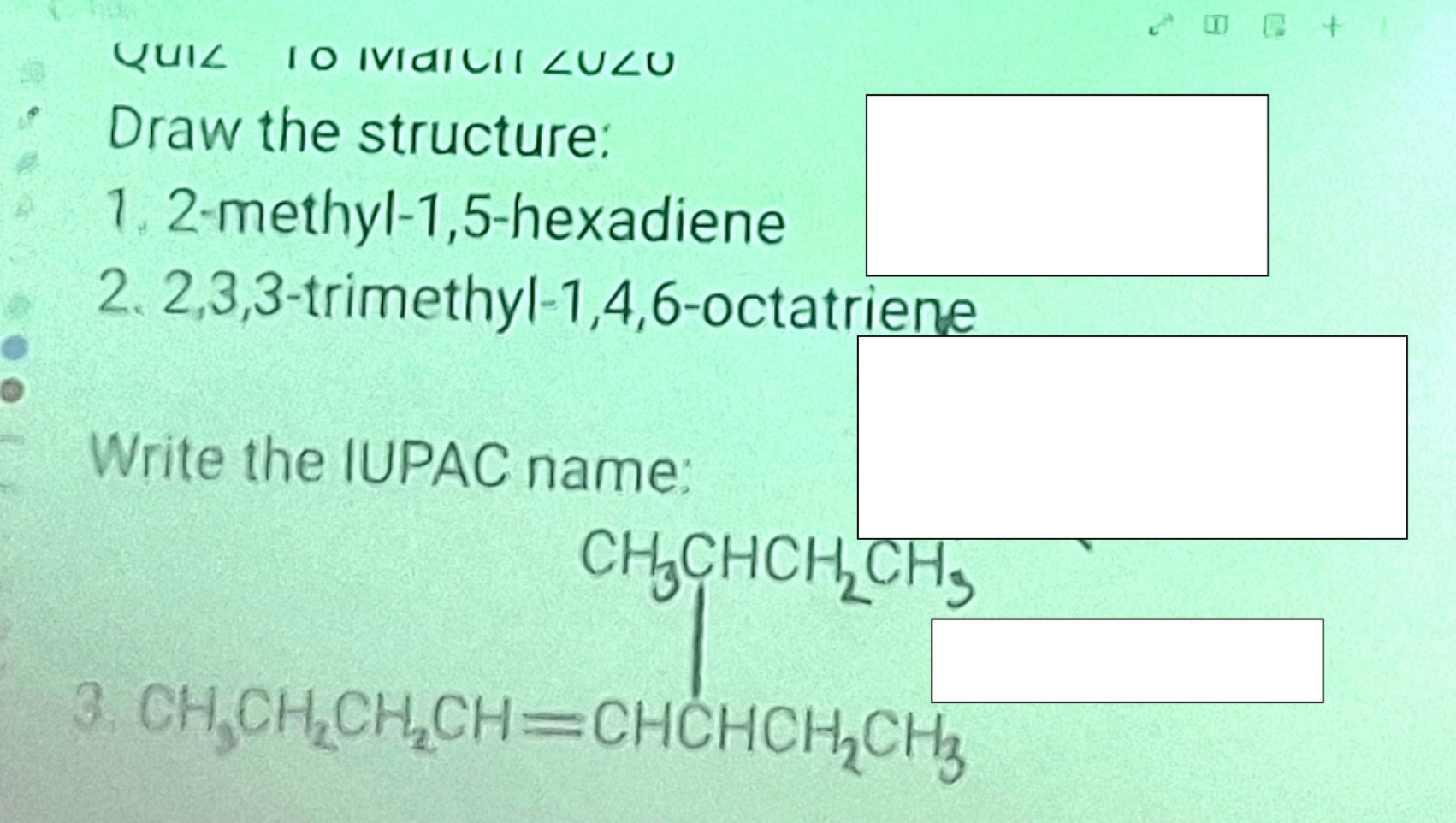
Systematic Names of Alkadienes
if two carbon to carbon double bonds are present, indicate the position of each and use the suffix -diene
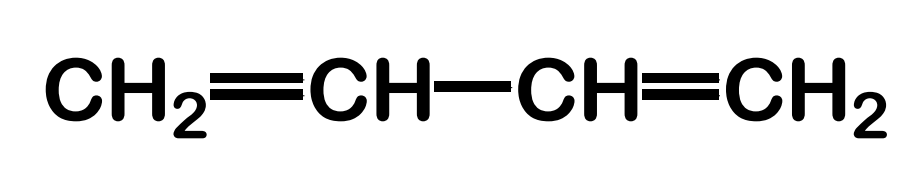
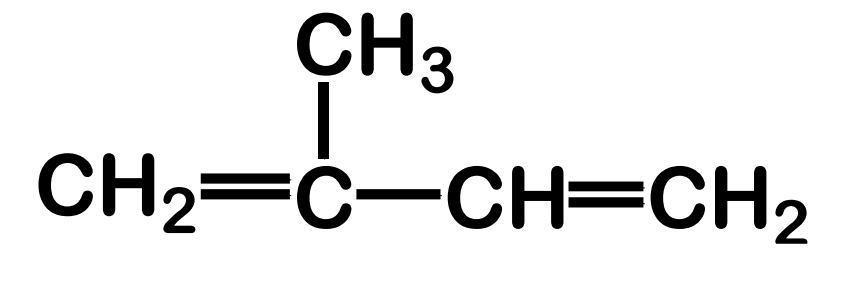
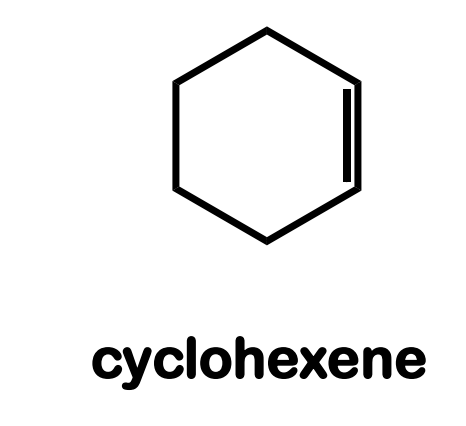
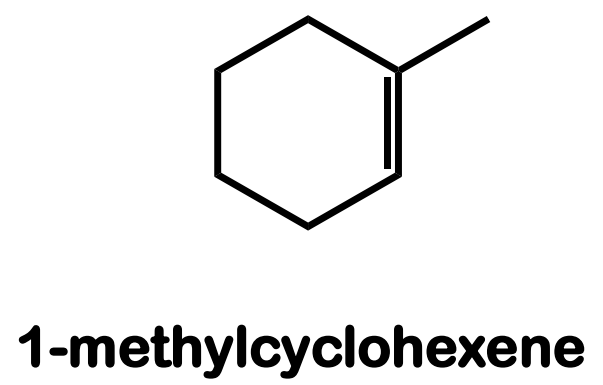
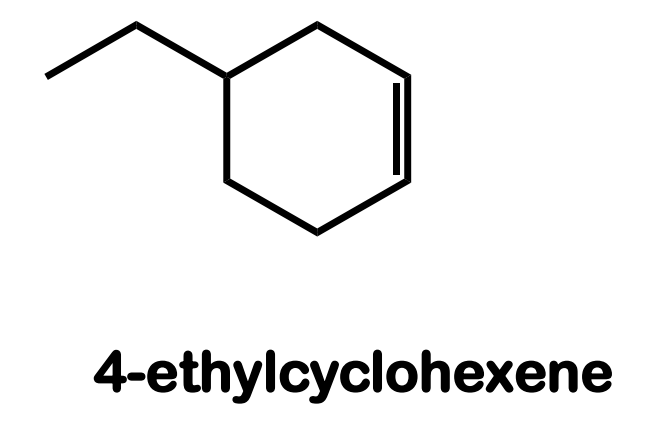
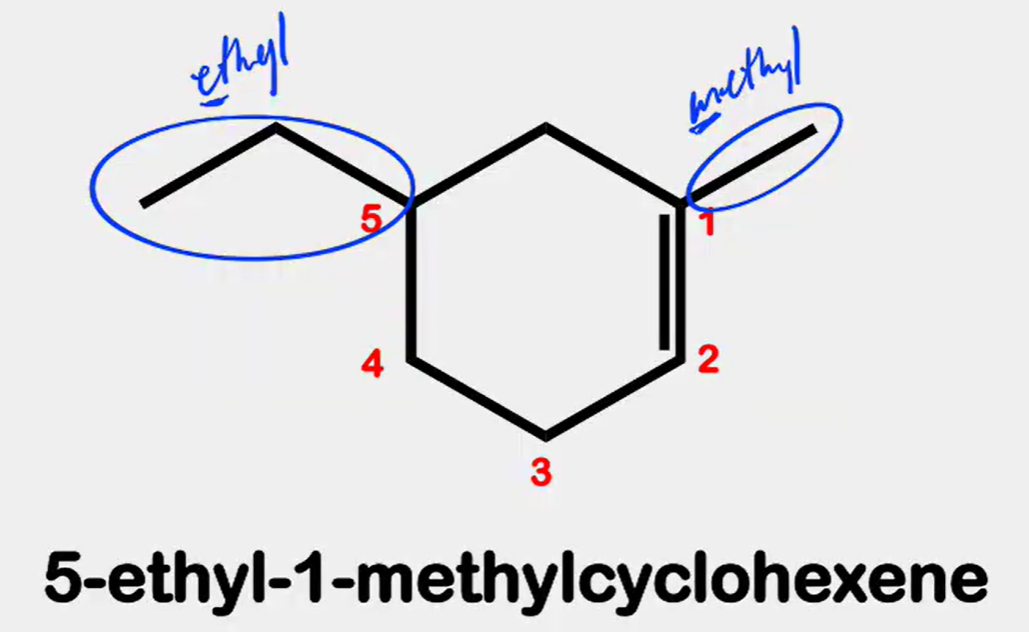
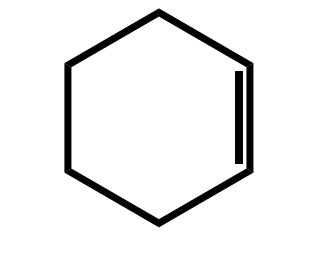
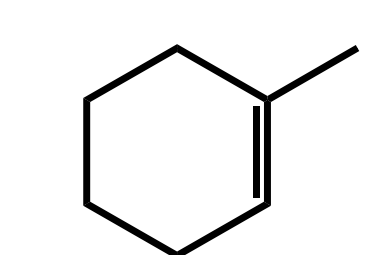
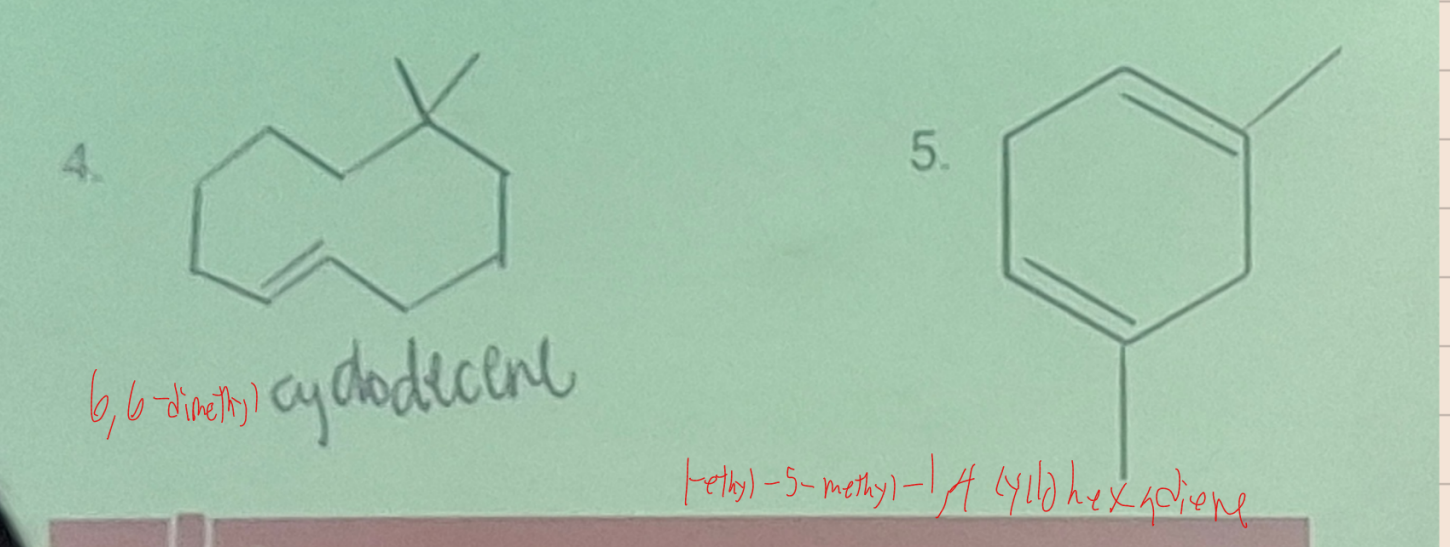
Systematic Names of Cycloalkenes
the double bond is located between C1 and C2
the ring is numbered clockwise or counterclockwise to give the first substituent the lower number
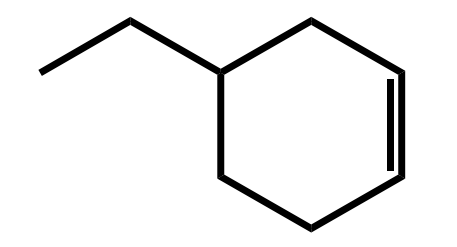
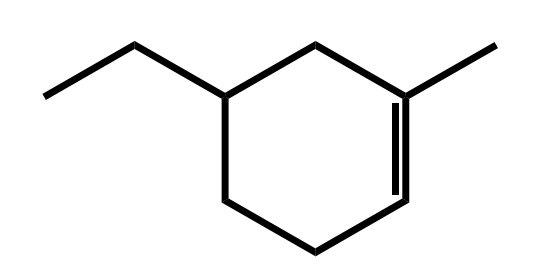
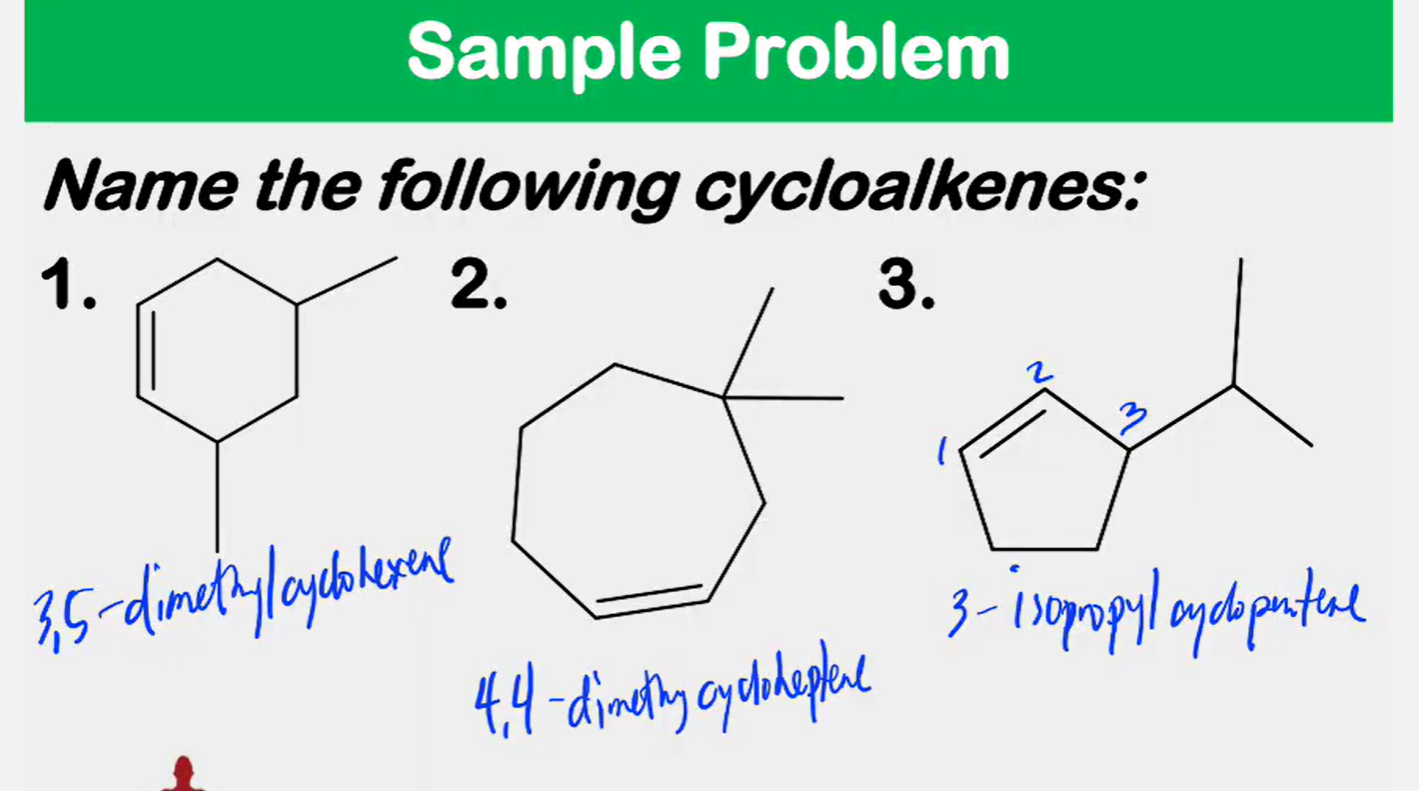
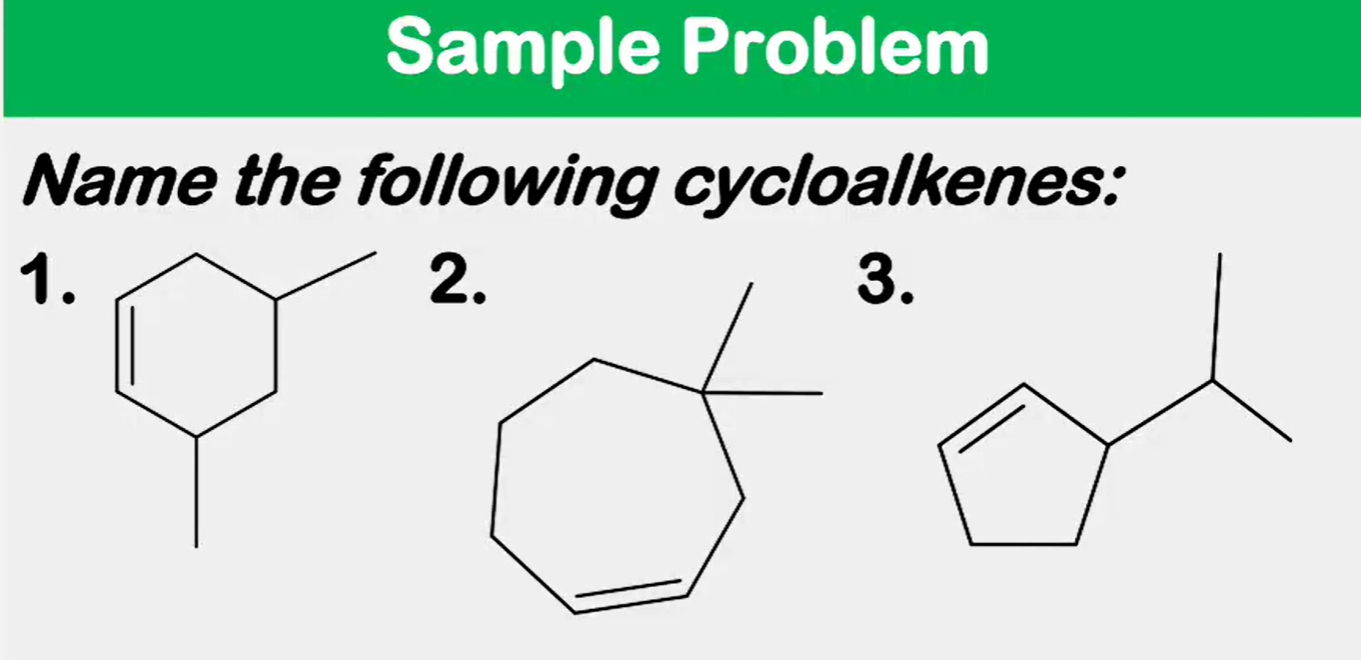
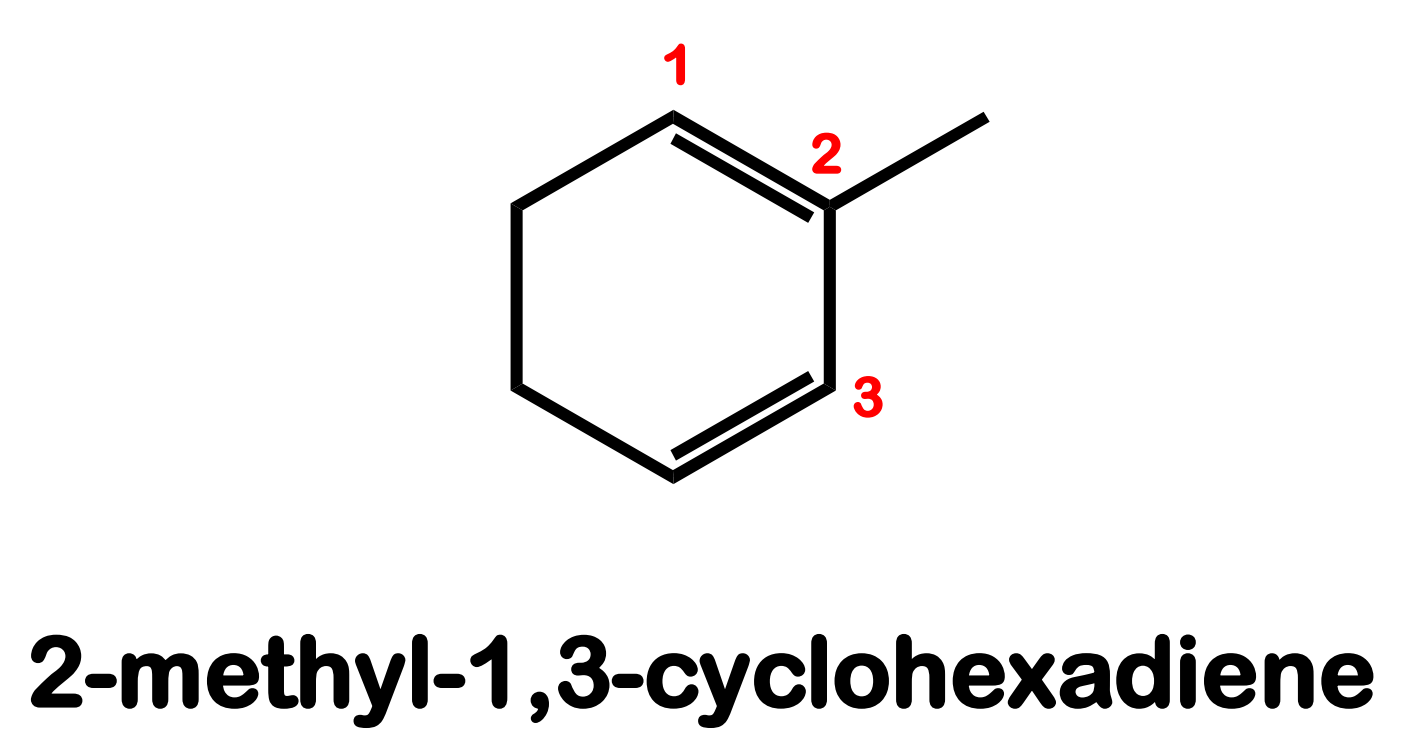
Systematic Names of Cycloalkadienes
the lowest possible set of locants are assigned to the two carbon to carbon double bonds, with one of the double bonds located between C1 and C2
trans - farther
cis - nearer
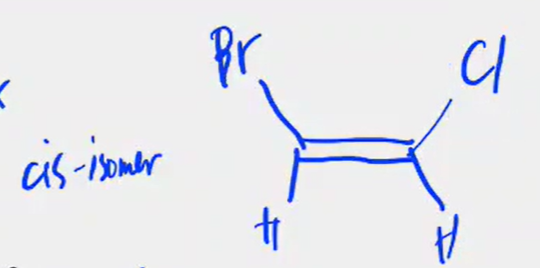
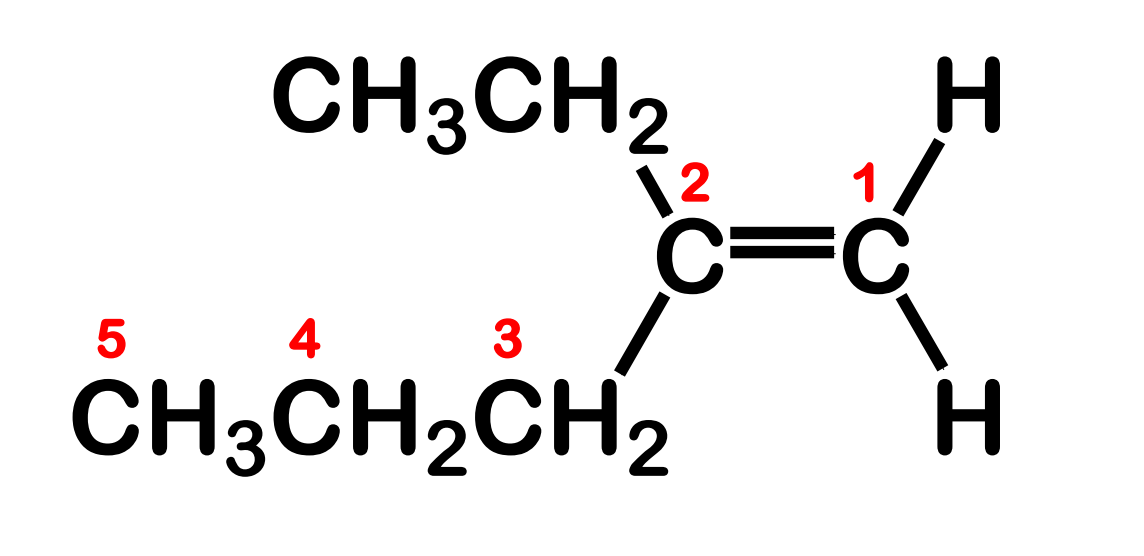
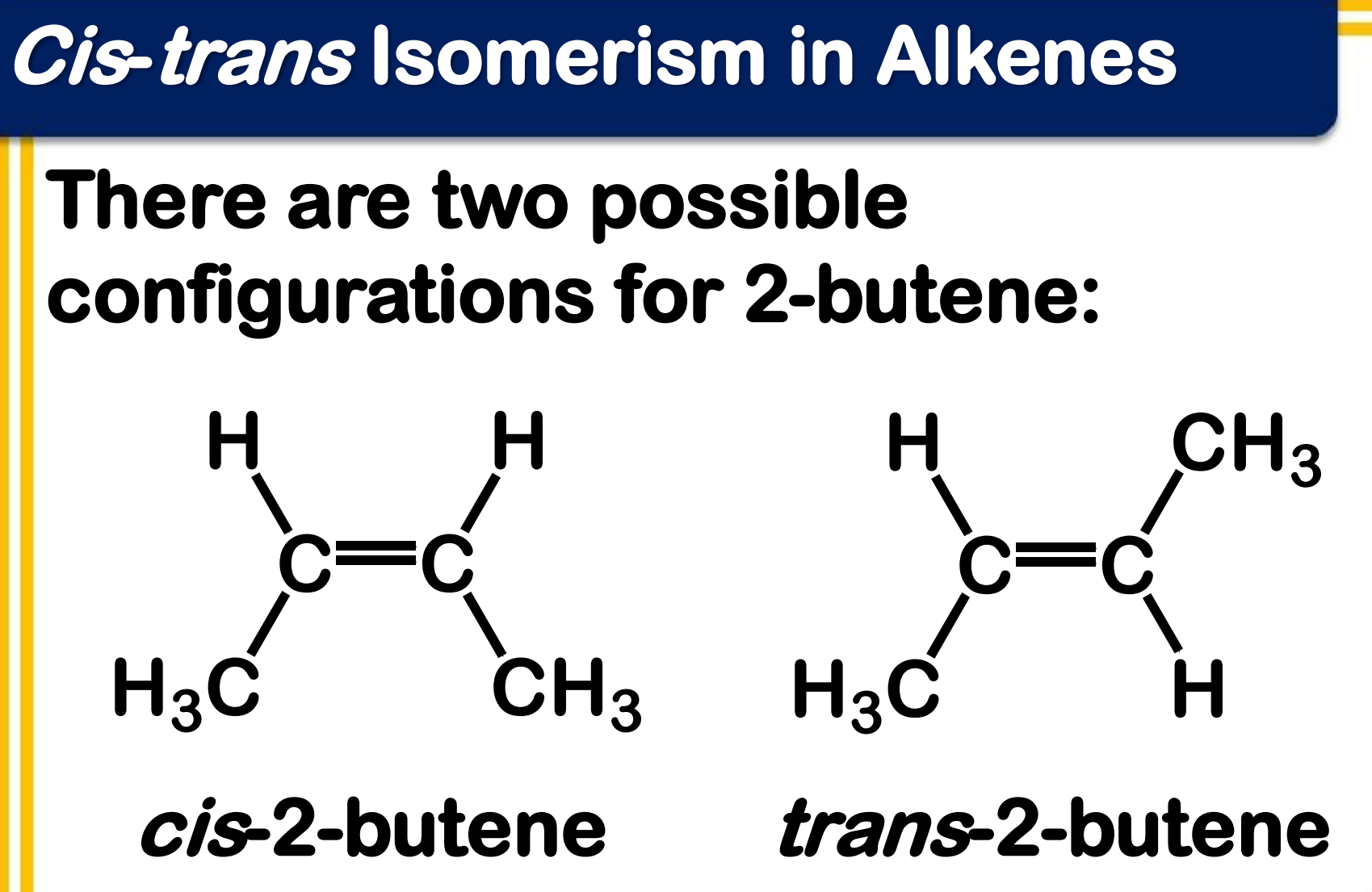
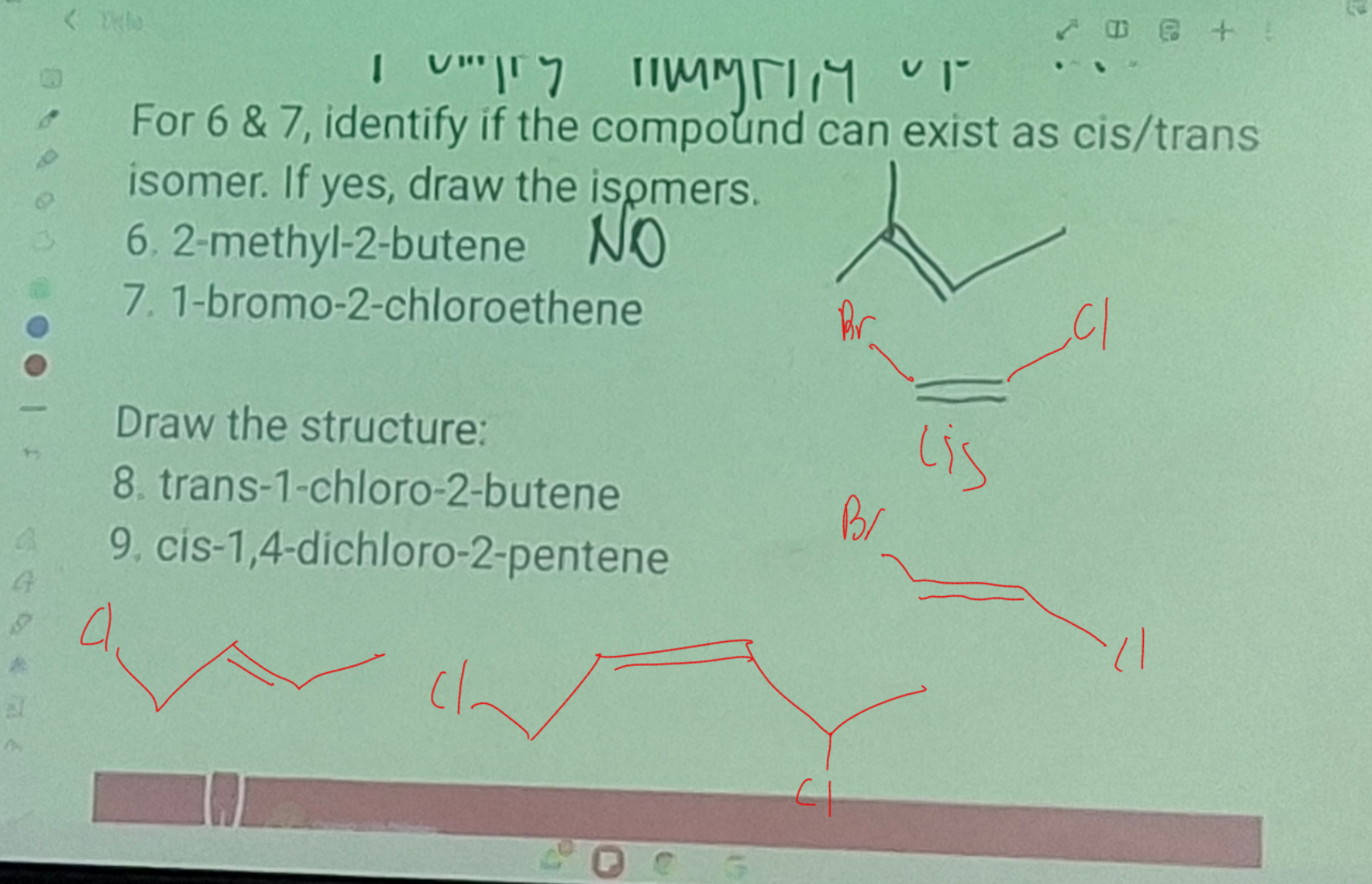
Cis and trans isomerism in Alkenes
the C-C double bond is not capable of free rotation
the energy required to break the π bond in ethene is approximately 268 kJ/mol
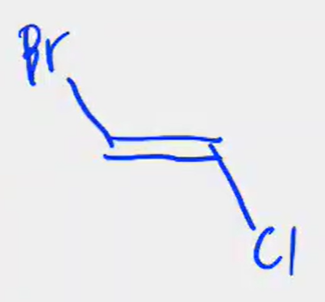
Between the two which C1 and C4 are farther from each other and which is nearer?
its as if you just flipped the entire molecule
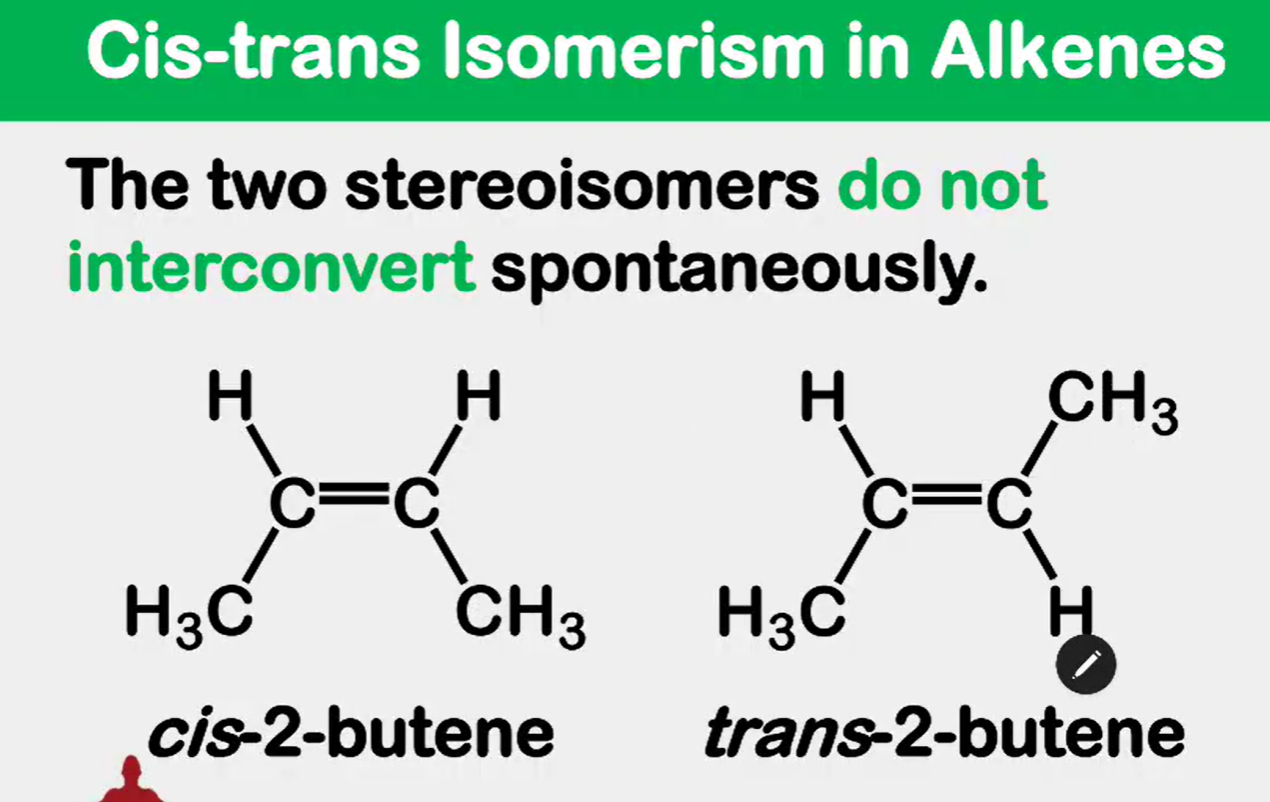
to rotate you’d have to break the double bond into a single bond, rotate, then reform the double bond.
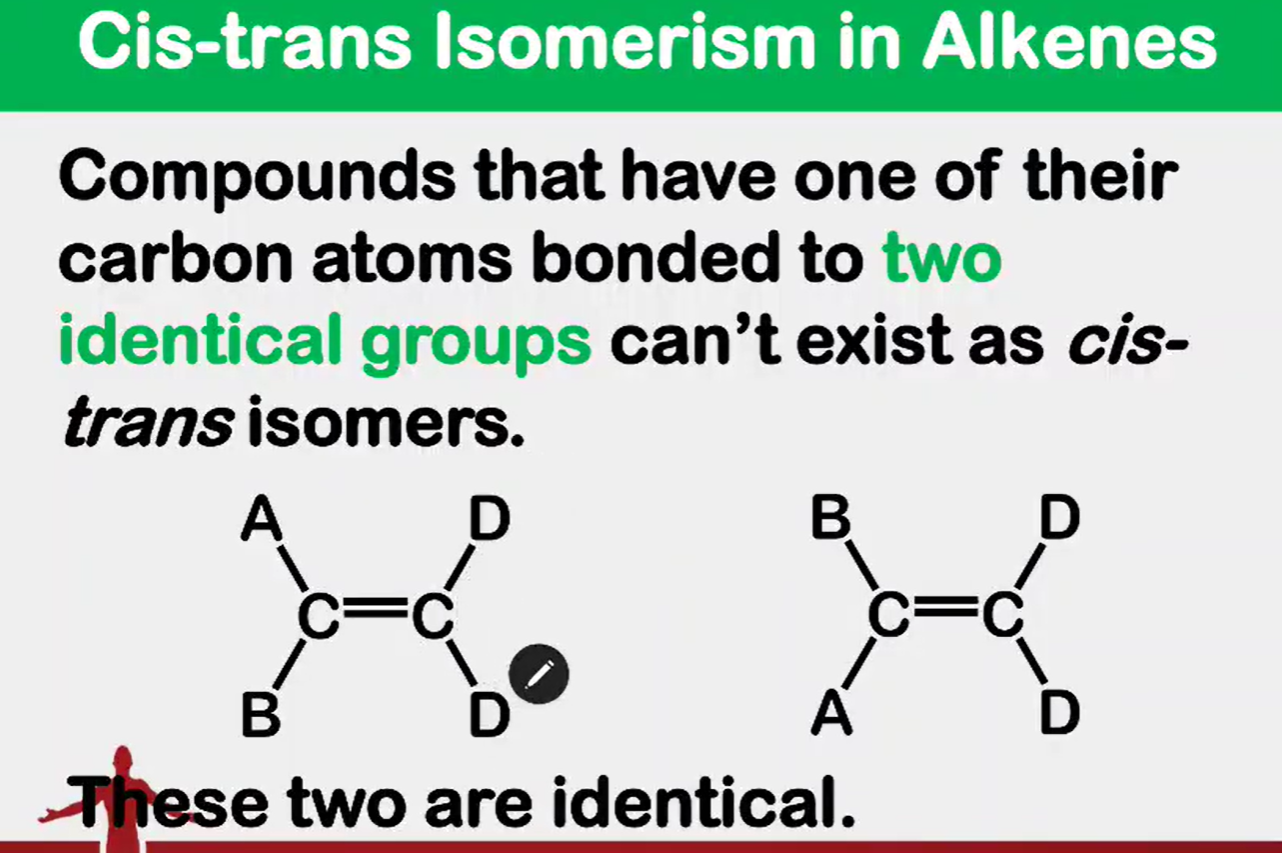
Why are they identical or can’t exist as cis-trans isomers?
cannot exist as cis-trans isomer

cannot exist aas cis-trans isomers because the two CH3 attached ti the 2nd carbon are the same.

can exist, drawn below is the trans (top) and cis (bottom) isomers.

same reason as number 2.

can exist, cis and trans drawn below.






C is double bonded to O so imagine 2 O’s and the O is double bonded to the C so imagine 2 C’s.

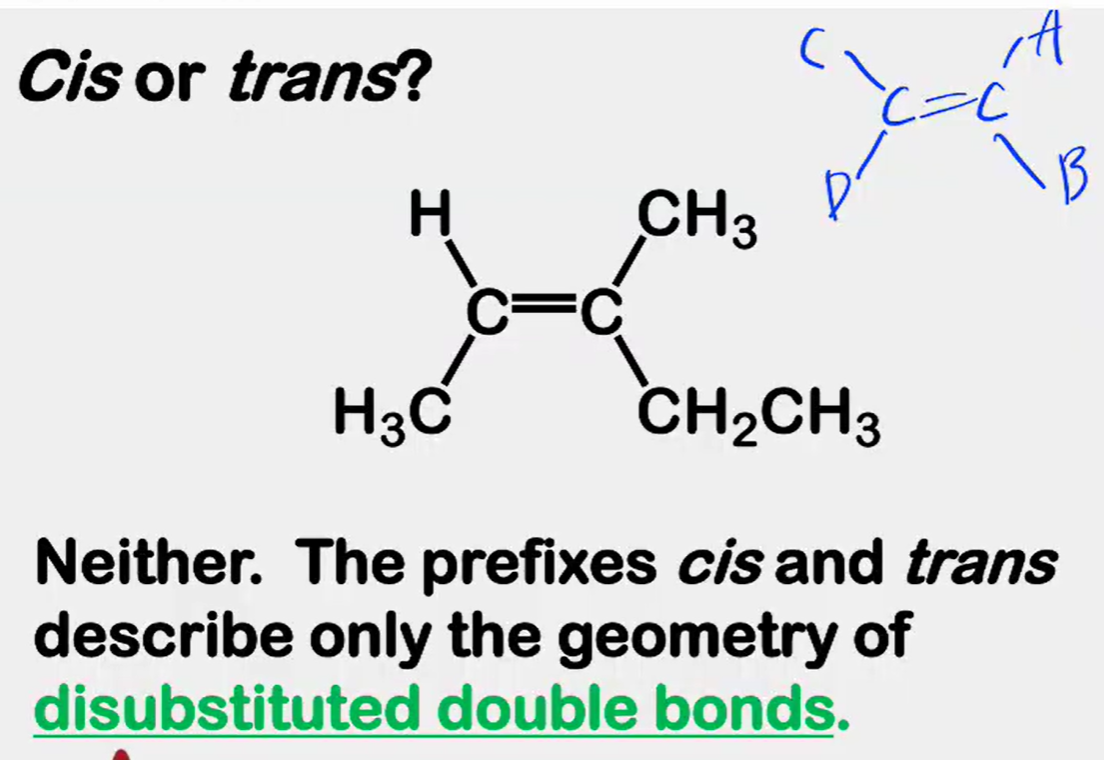
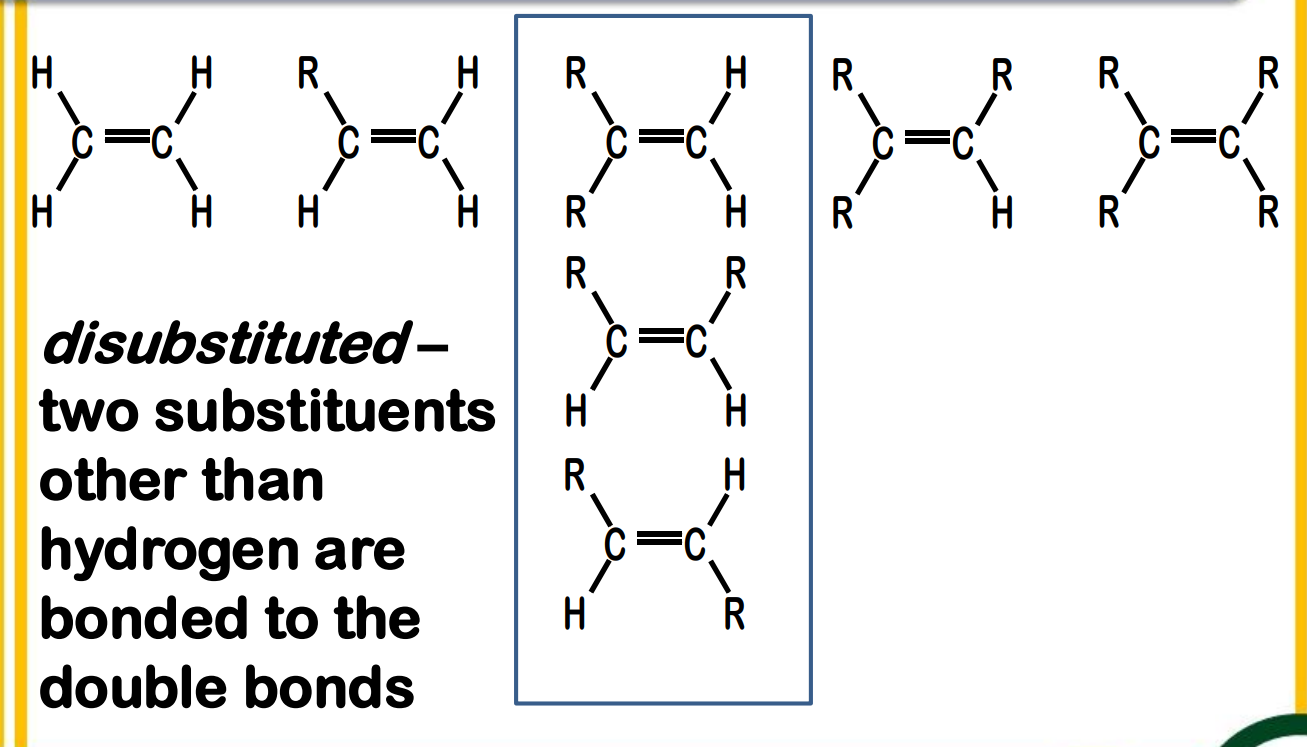

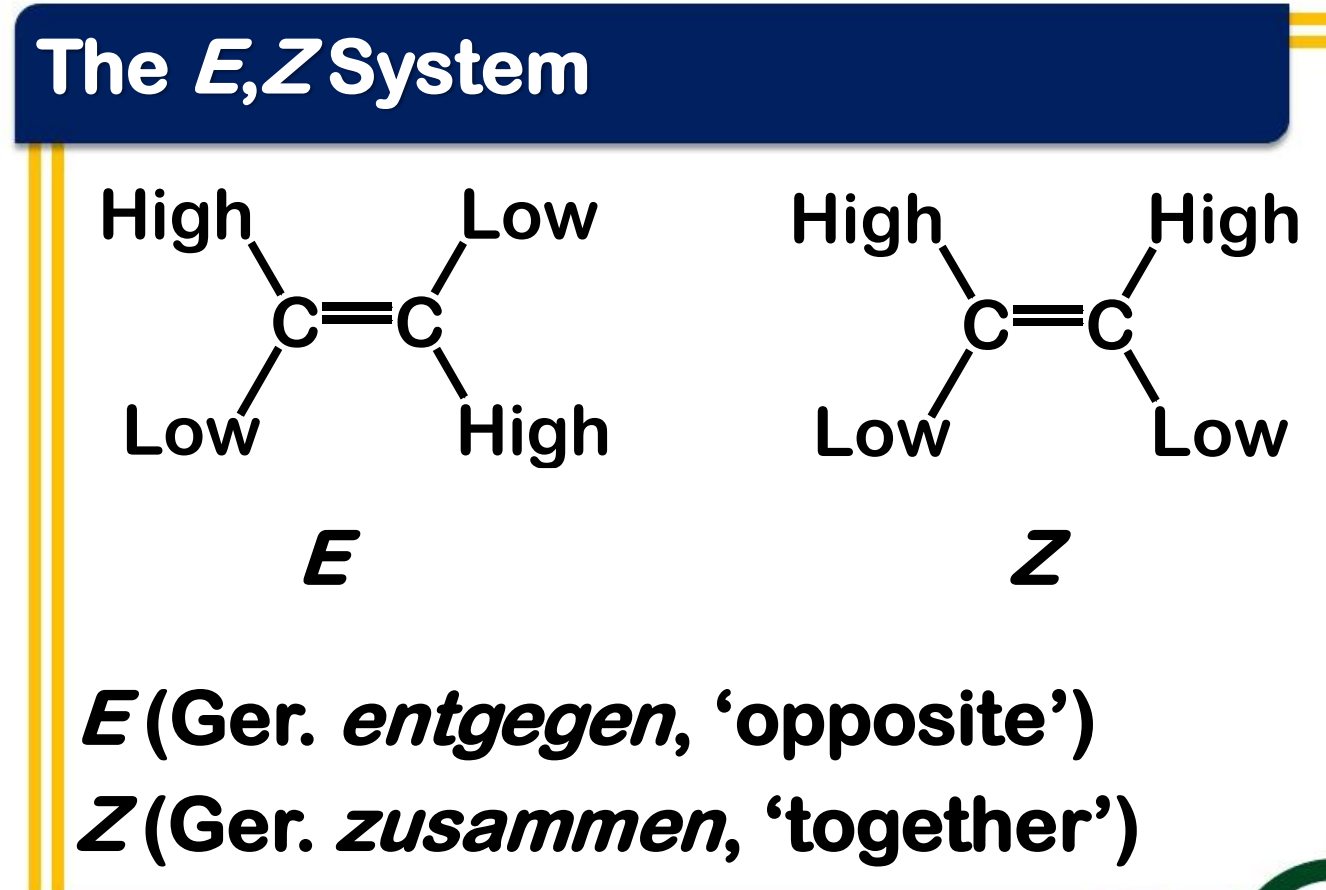
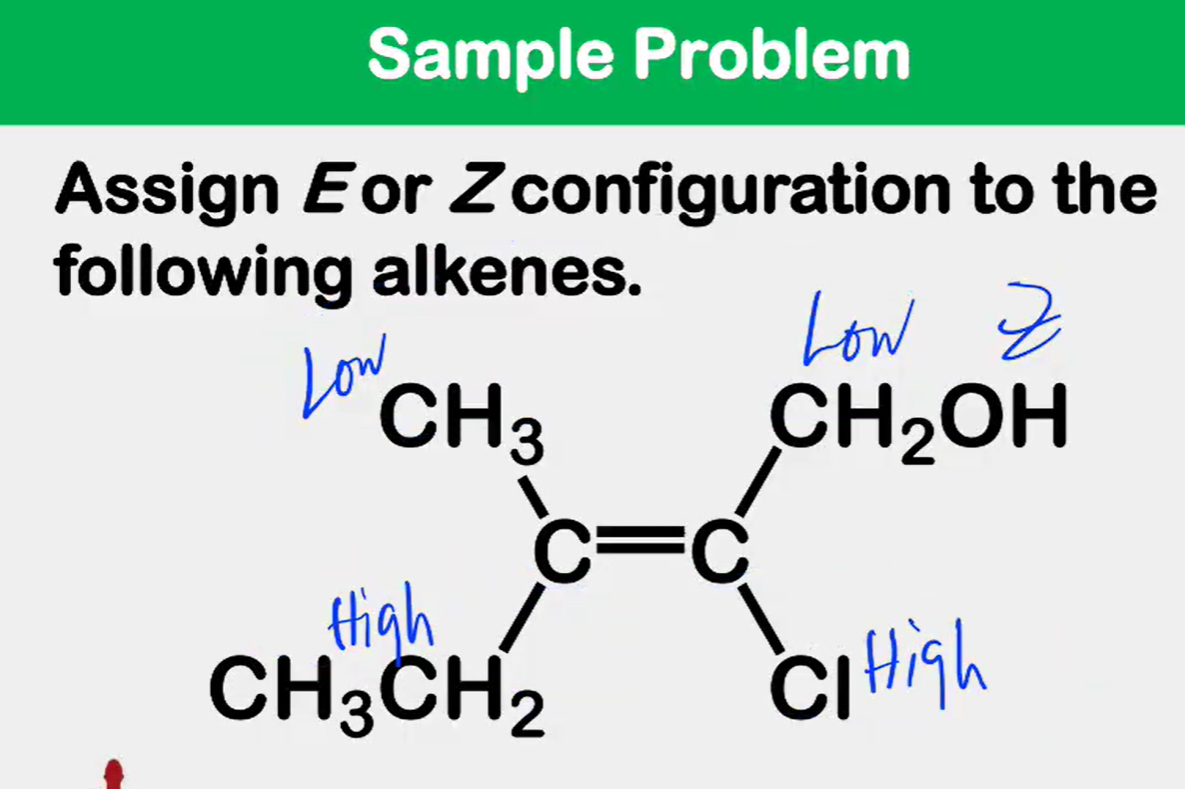
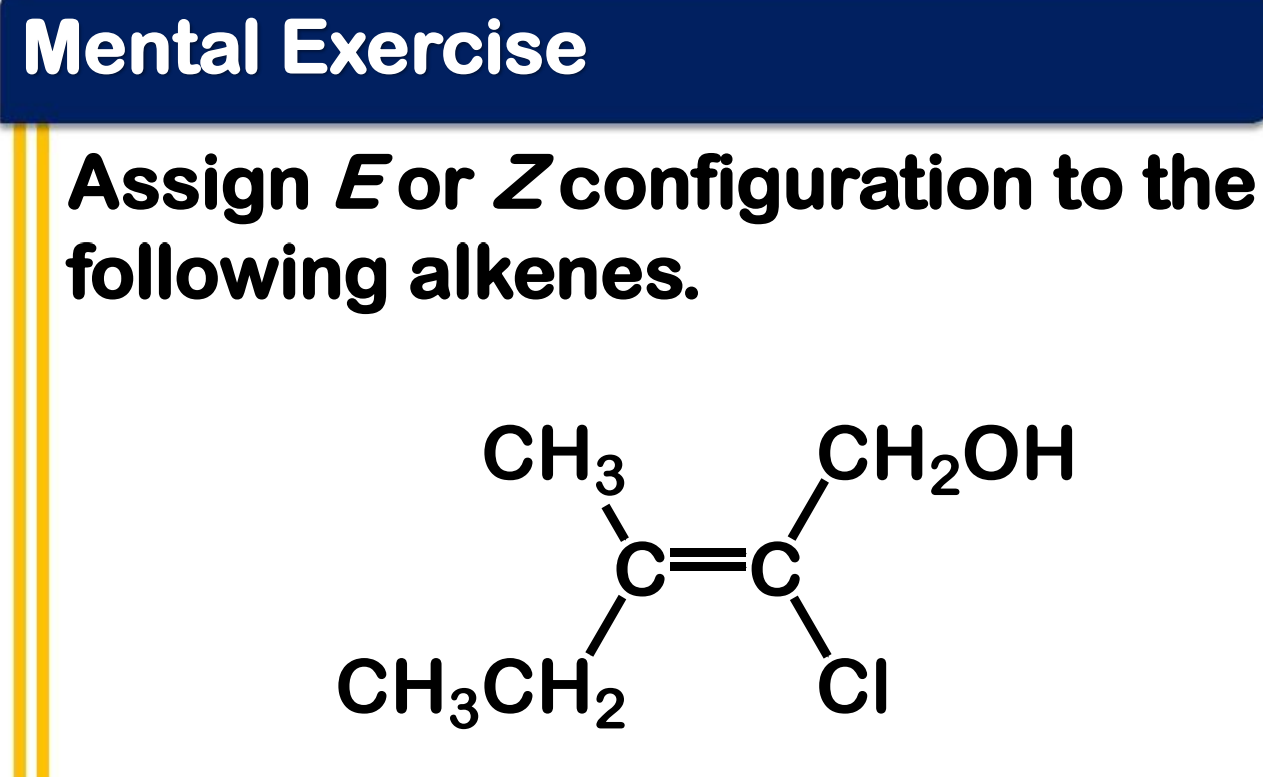
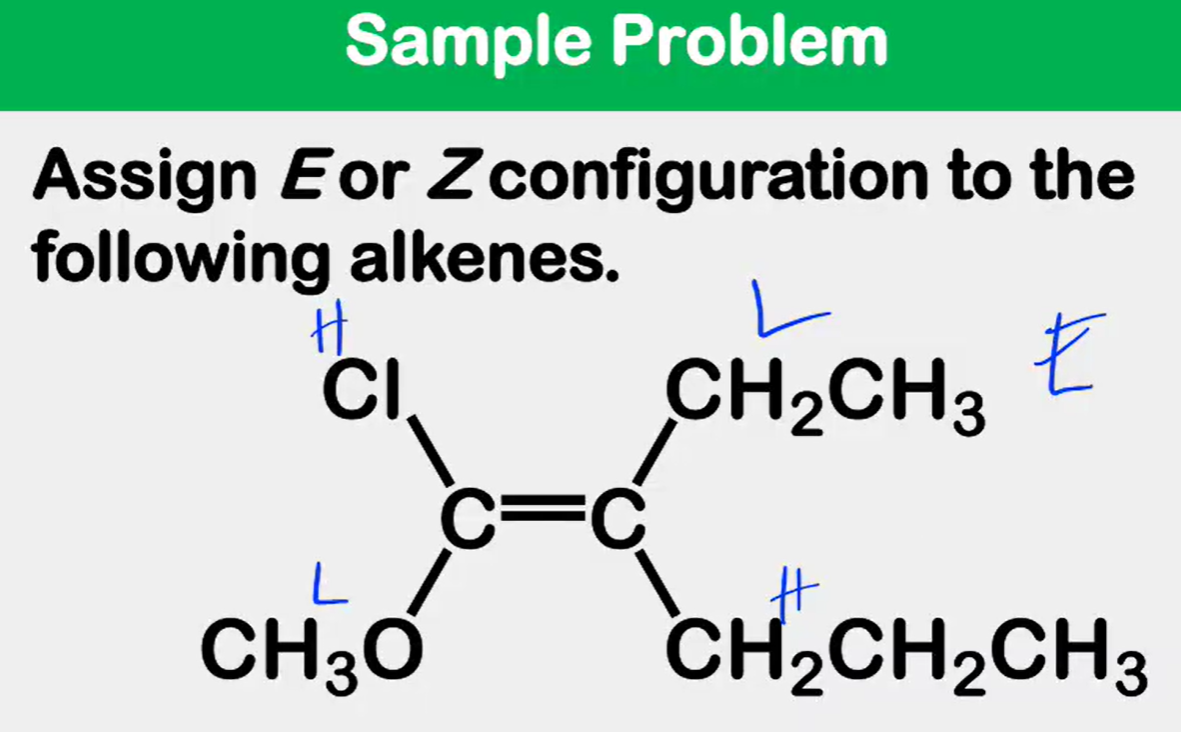
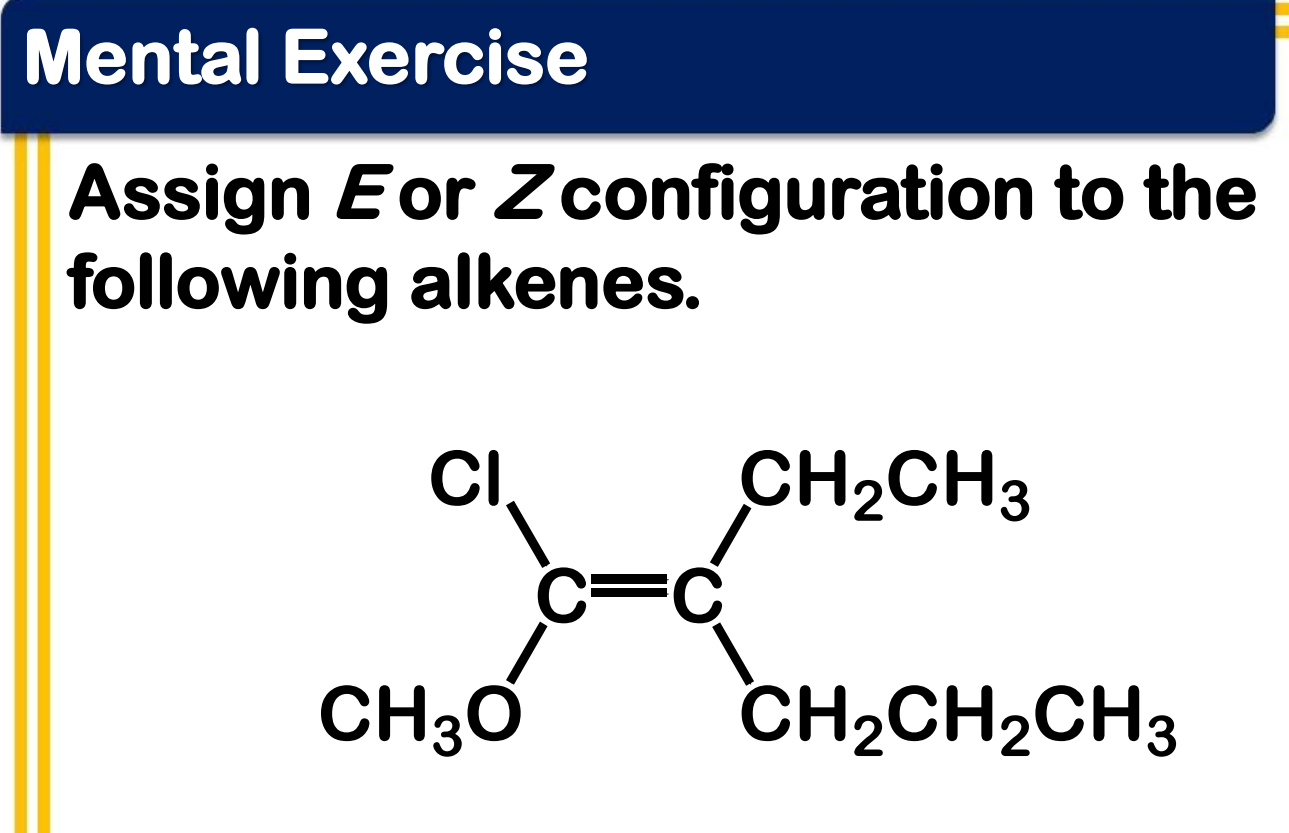
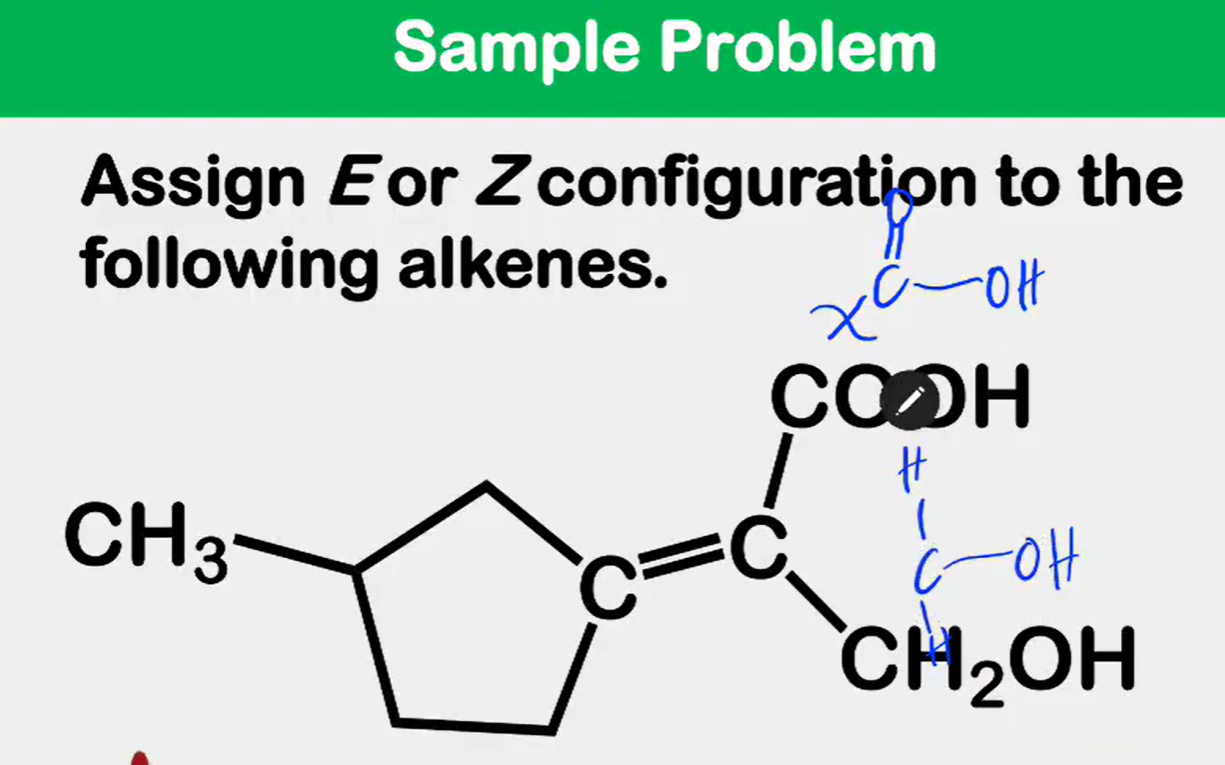
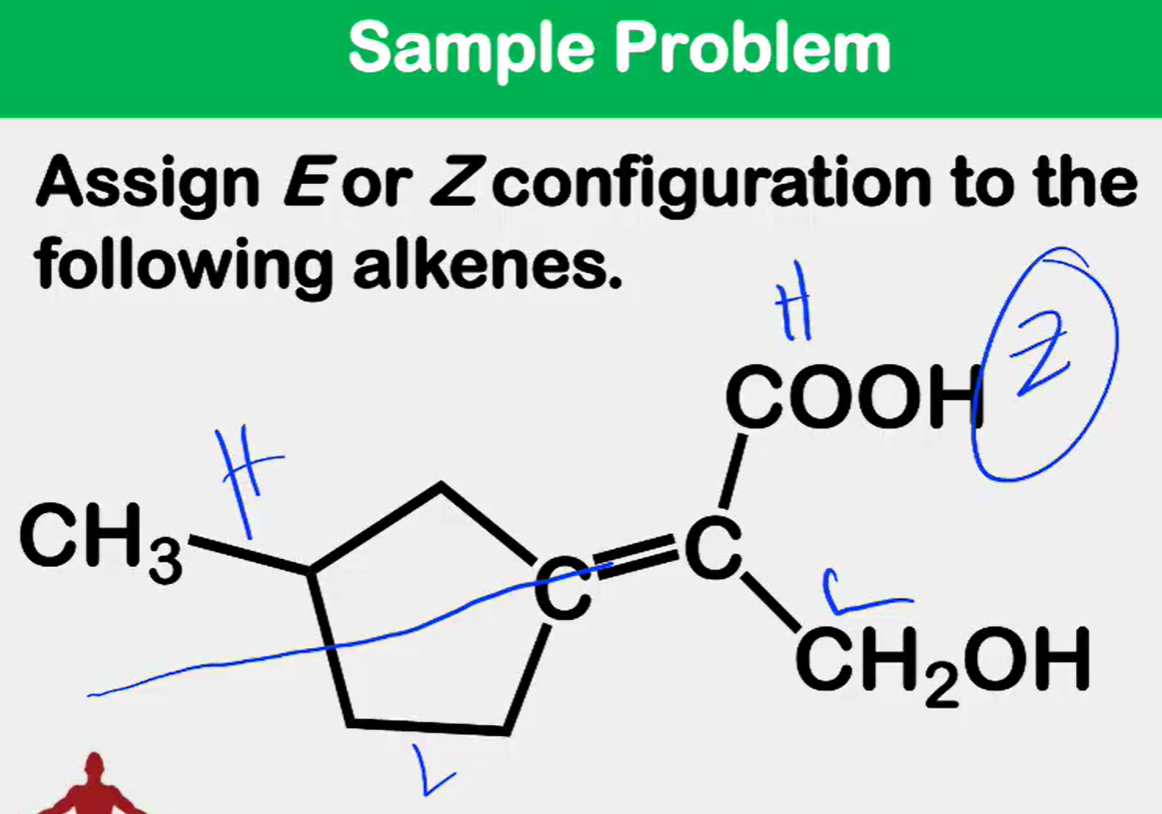
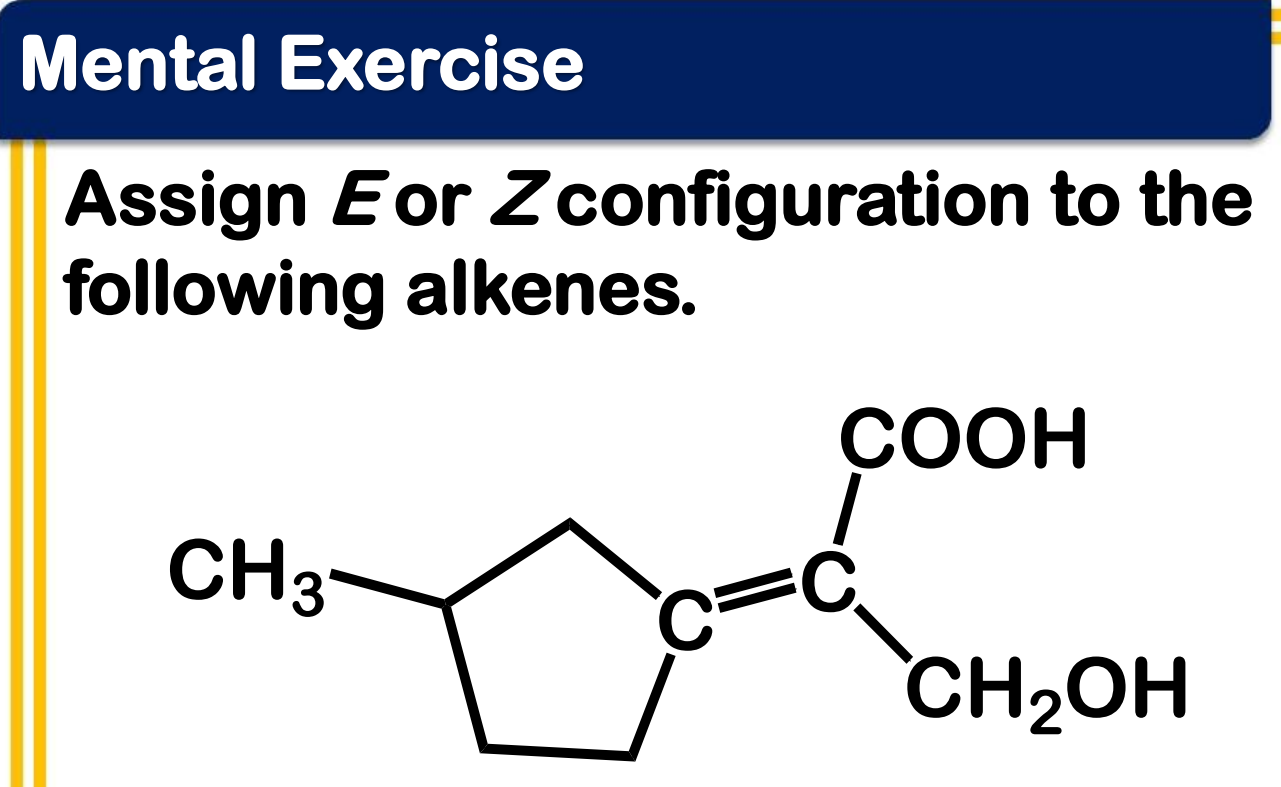
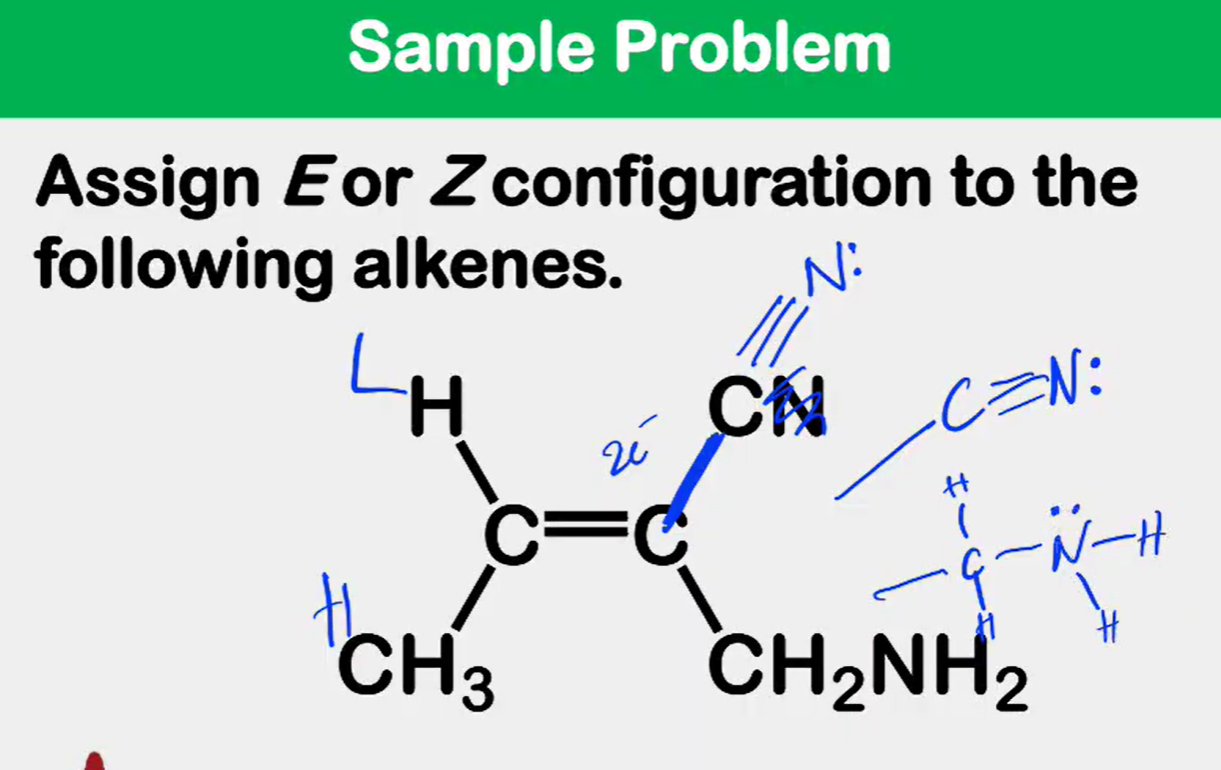
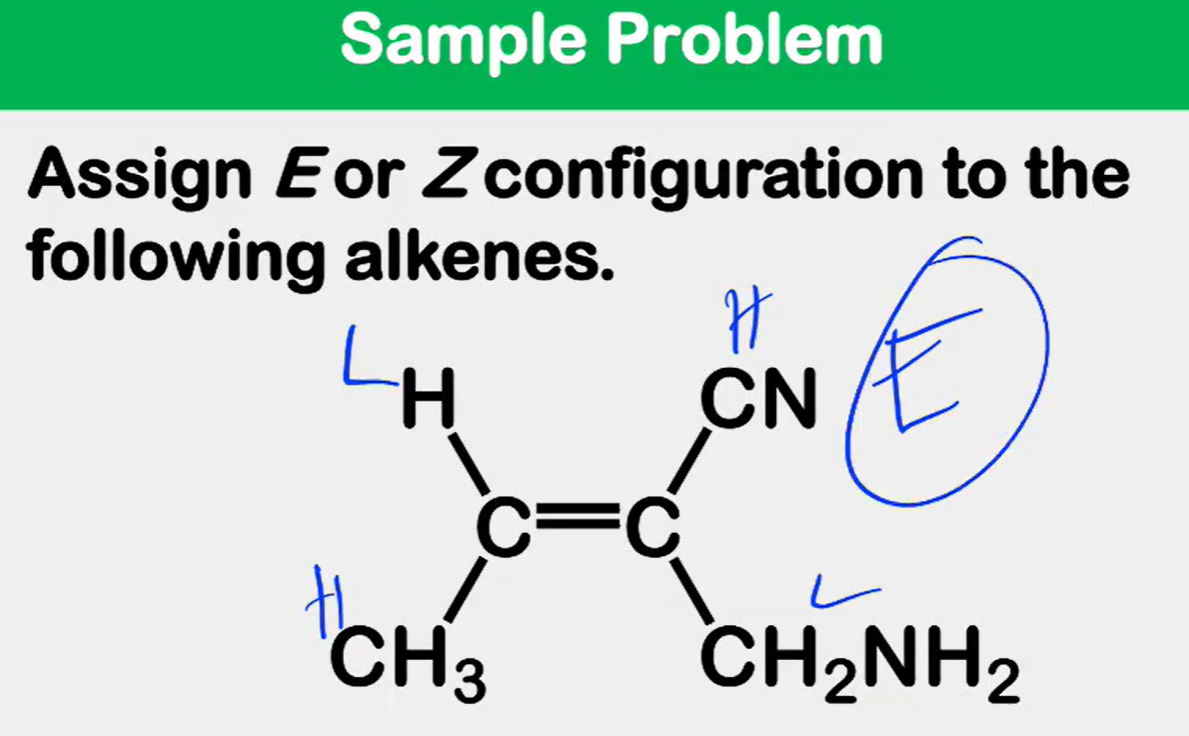
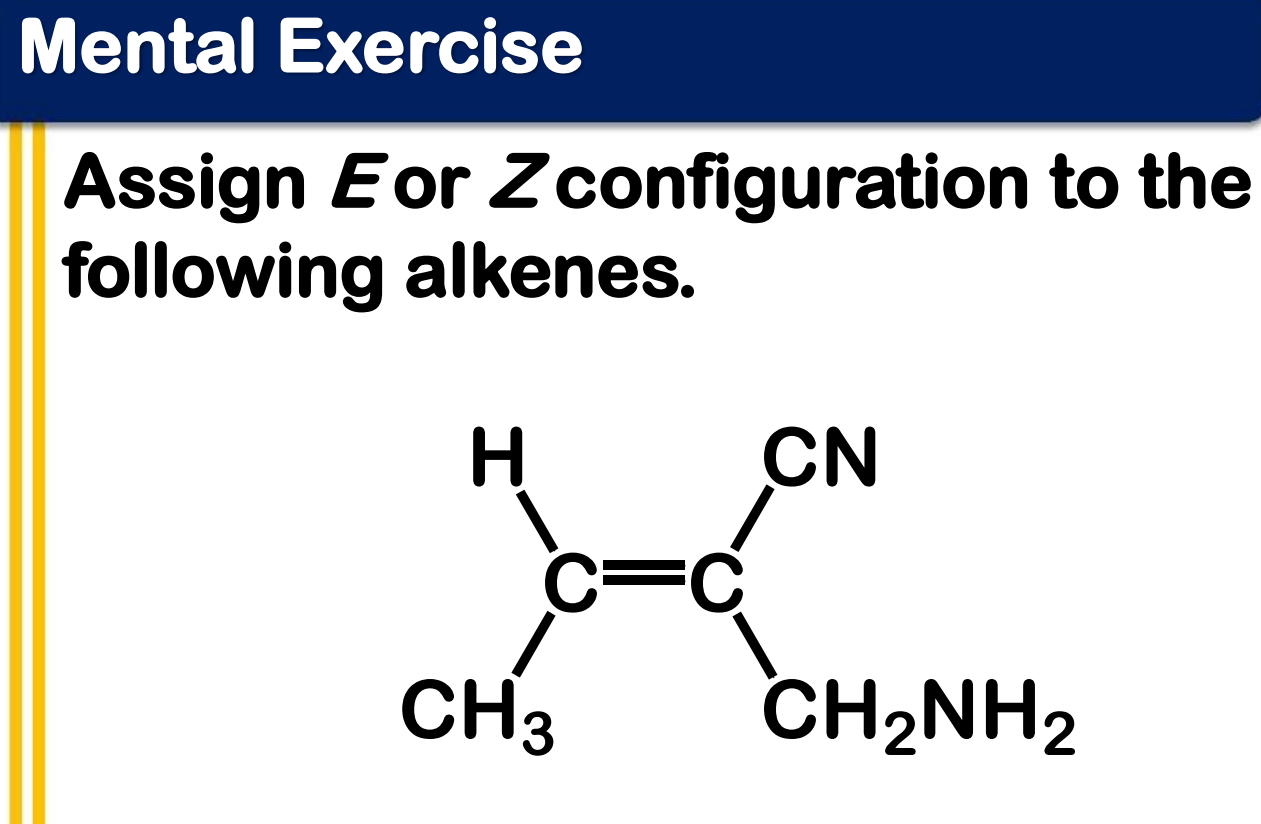

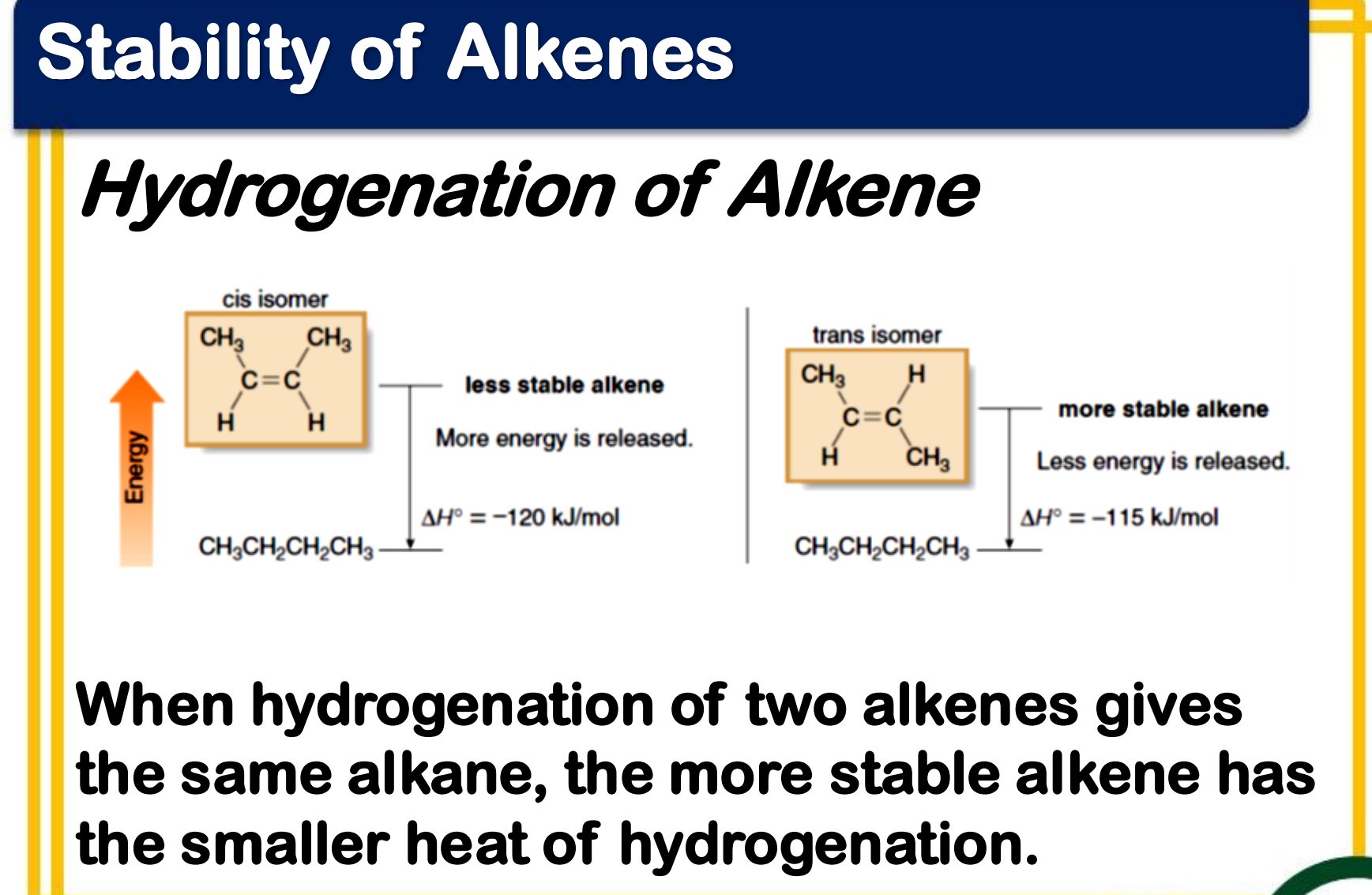
Alkene Stability
Stereochemistry - trans alkenes are generally more stable than cis alkenes because the groups bonded to the double bond carbons are farther apart, reducing steric interactions
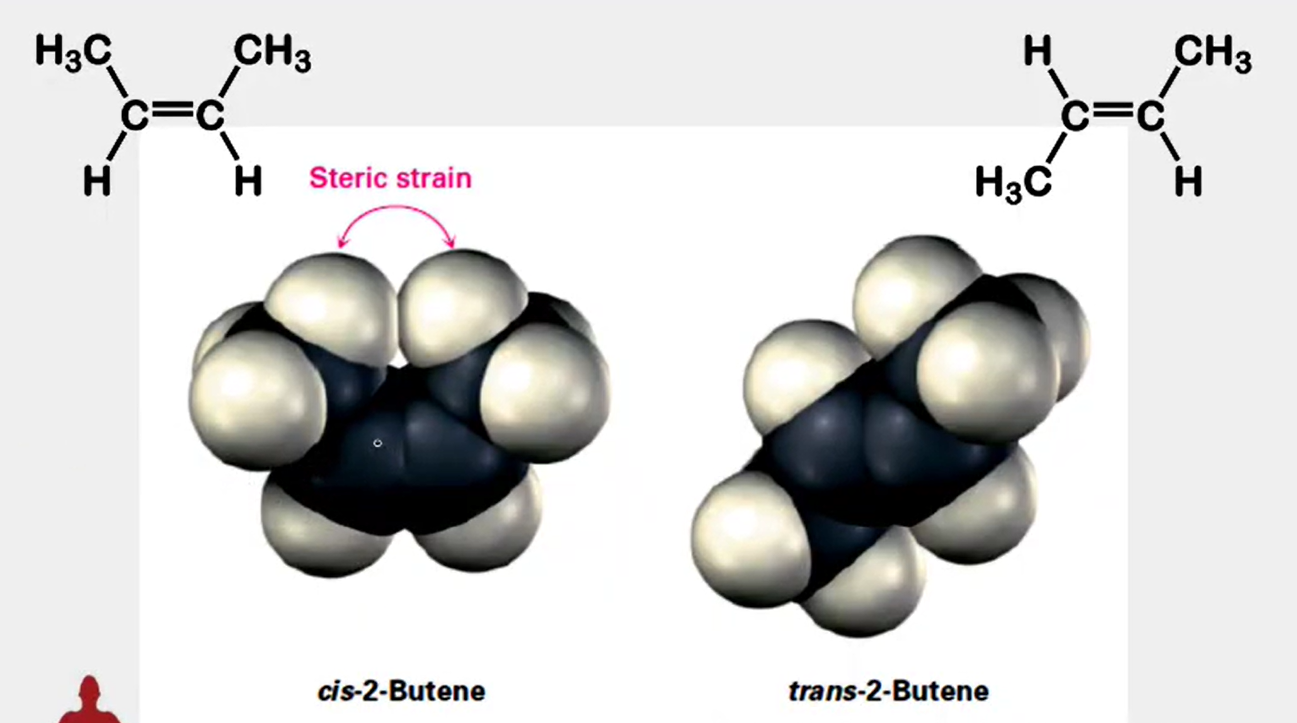
which is more stable trans alkenes or cis alkenes? Why?
Number of substituents across the double bond
alkene stability increases as the number of alkyl groups bonded to the double bond carbons increases
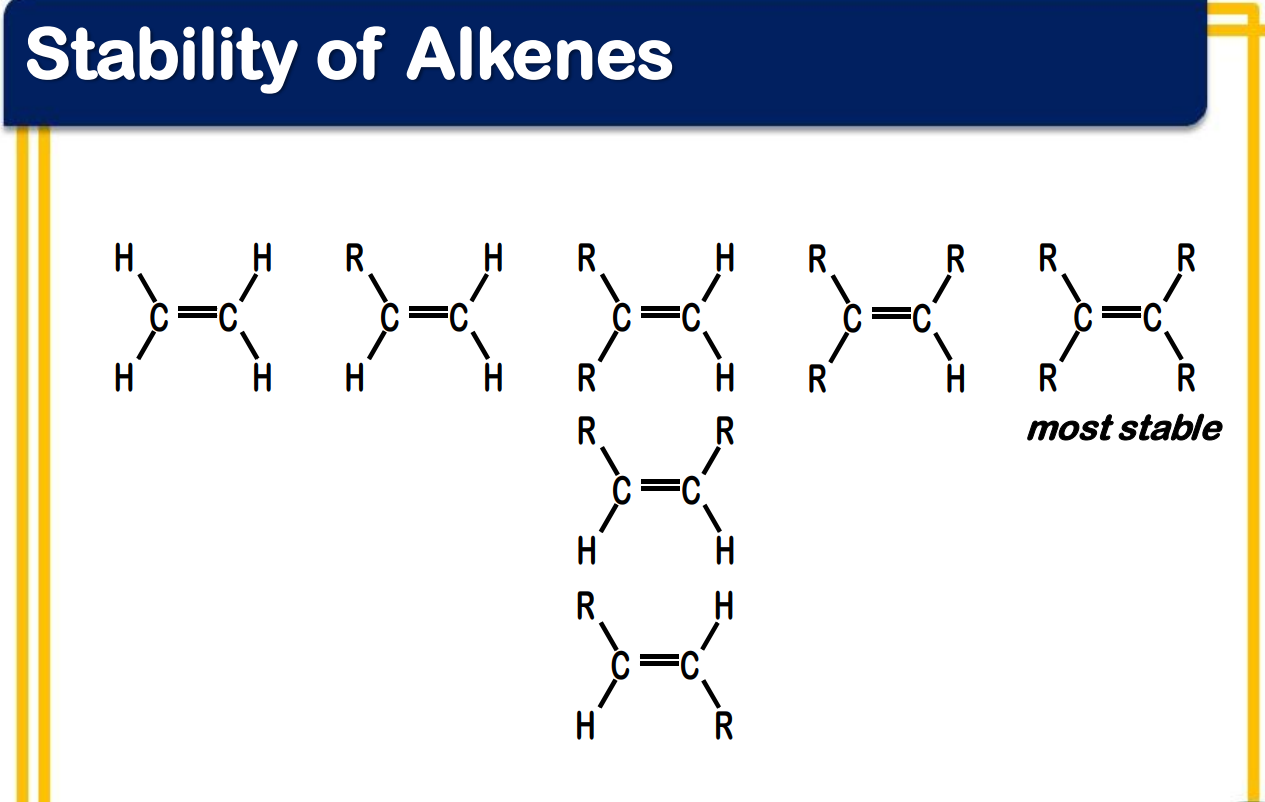



Number of substituents across the double bond
alkene stability _____________ as the number of alkyl groups bonded to the double bond carbons increases
Hyperconjugation
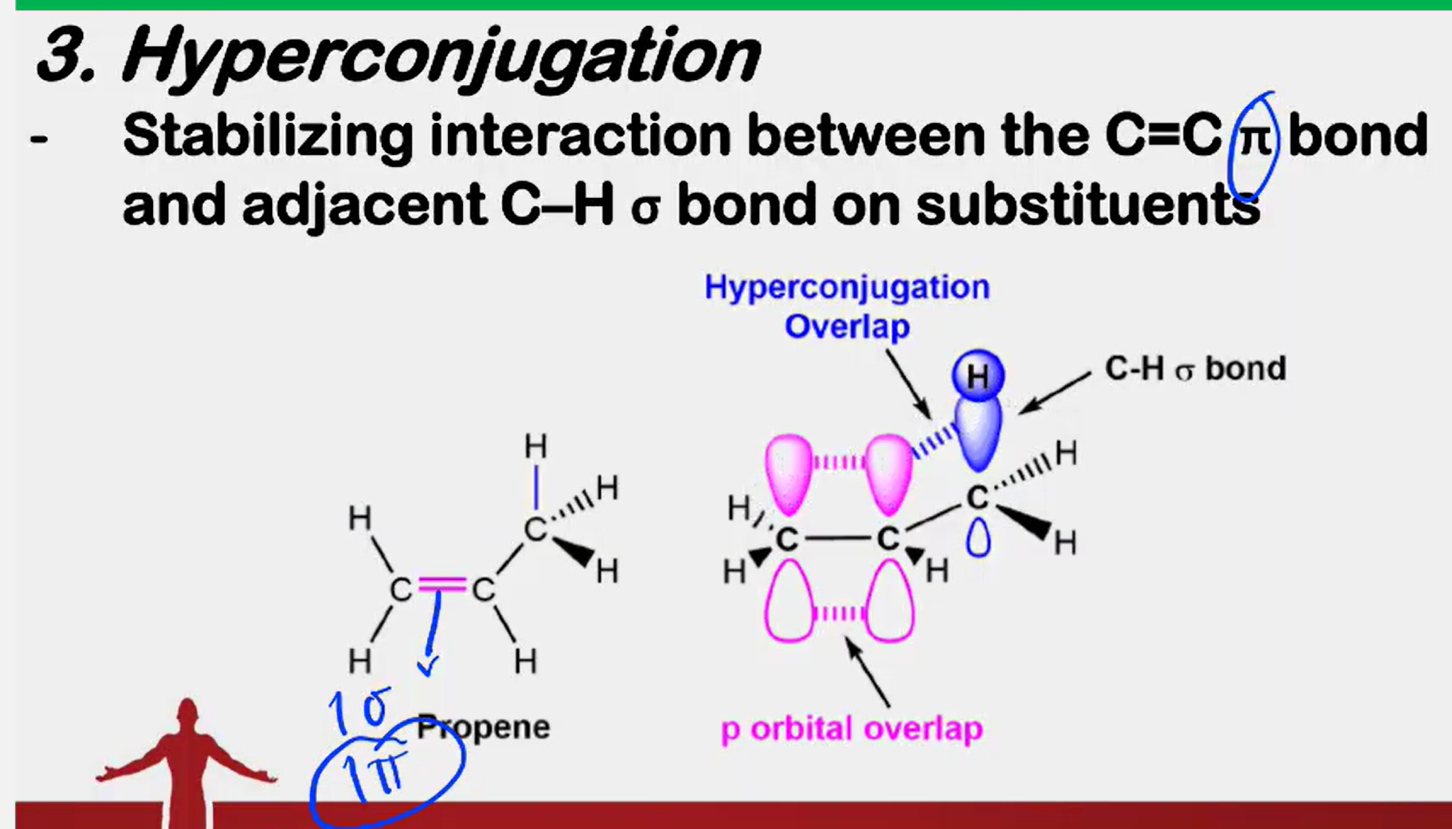
___________________ - Stabilizing interaction between the C=C π bond and adjacent C-H o bond on substituents
heat of hydrogenation
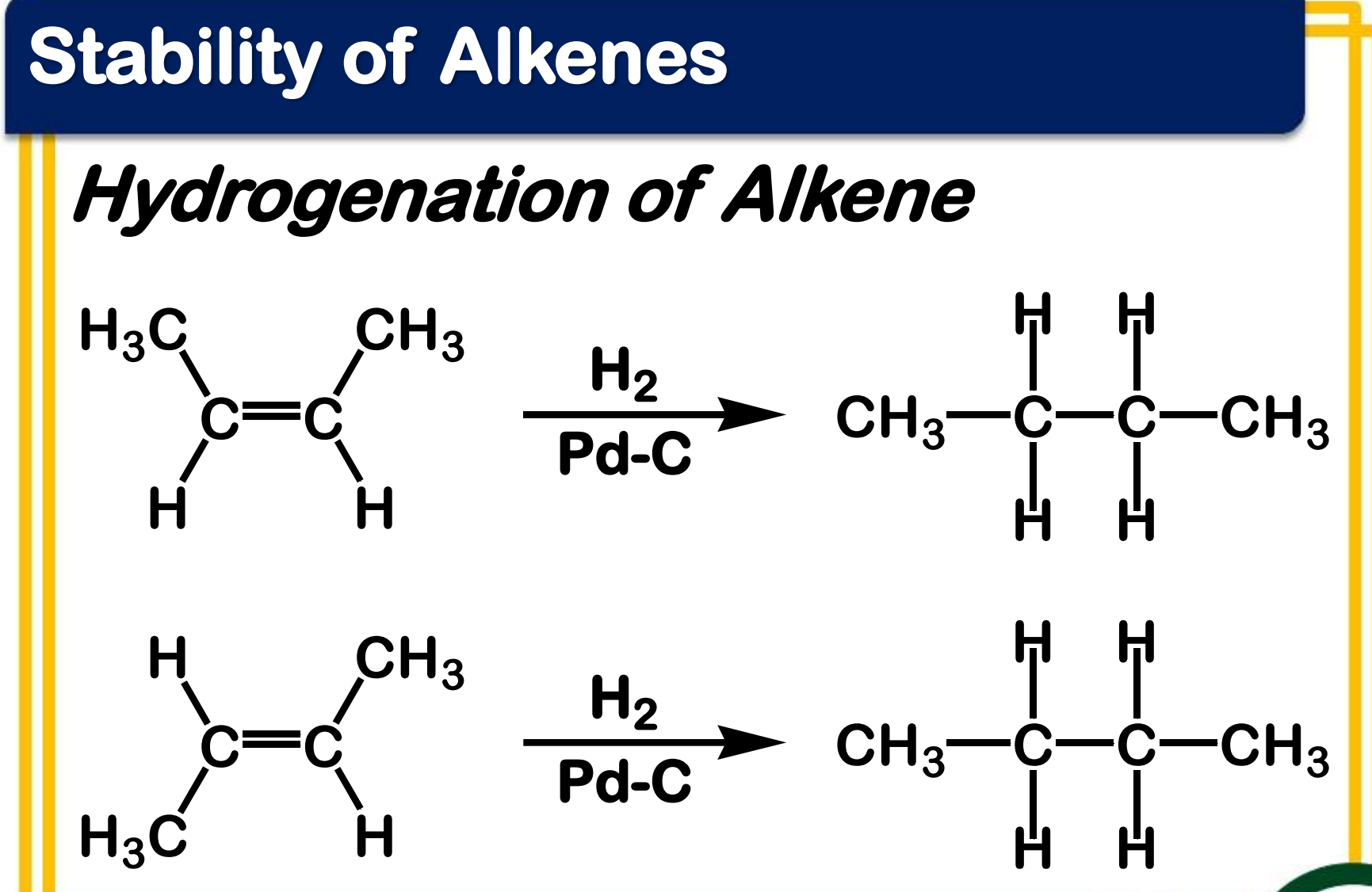
Alkene Stability - _____________ can be used to measure the relative stability of two different alkenes that are hydrogenated to the same alkane
smaller
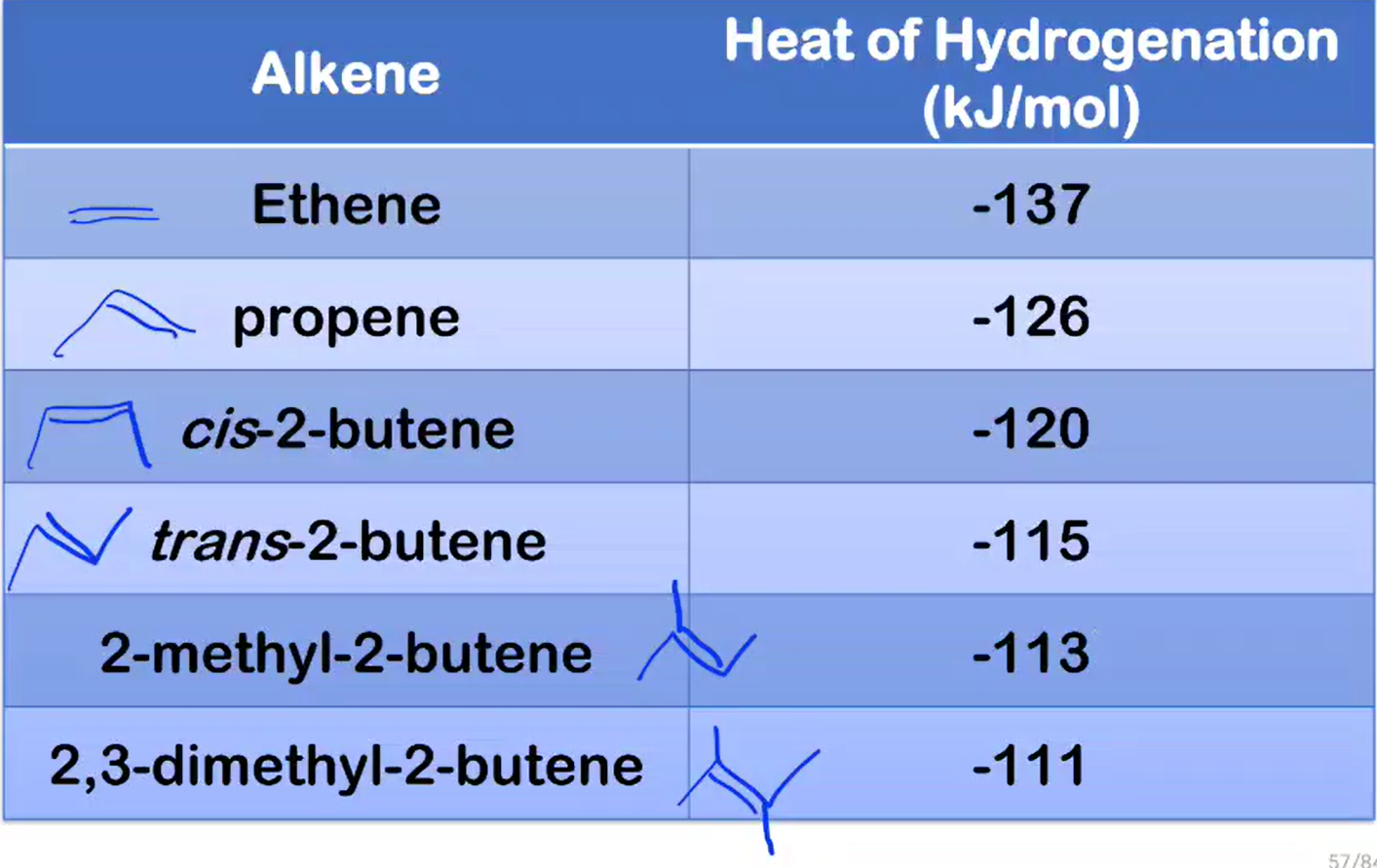
More substituted, more stable
When hydrogenation of two alkenes gives the same alkane, the more stable alkene has the __________ heat of hydrogenation.
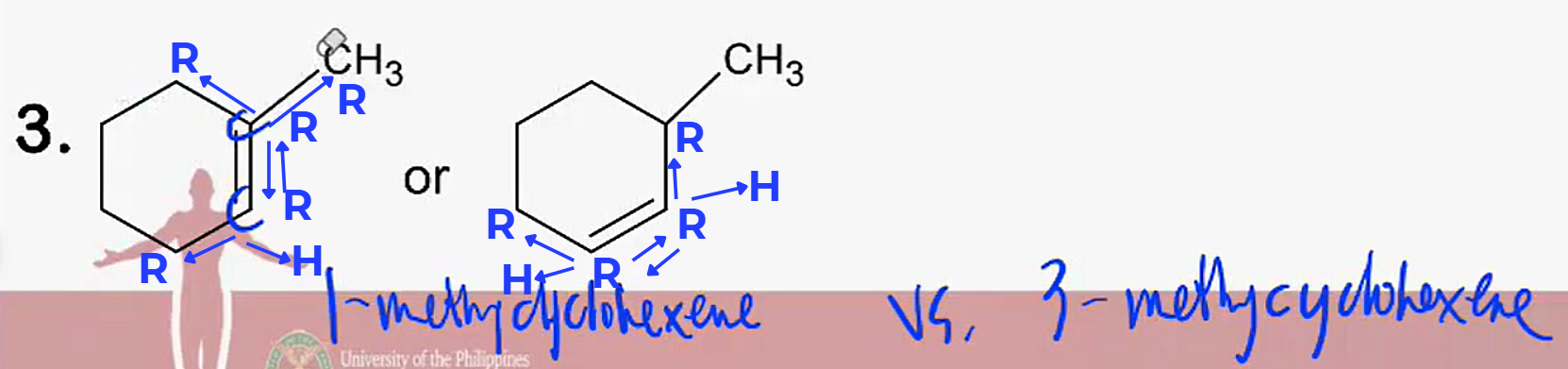
Dehydrohalogenation
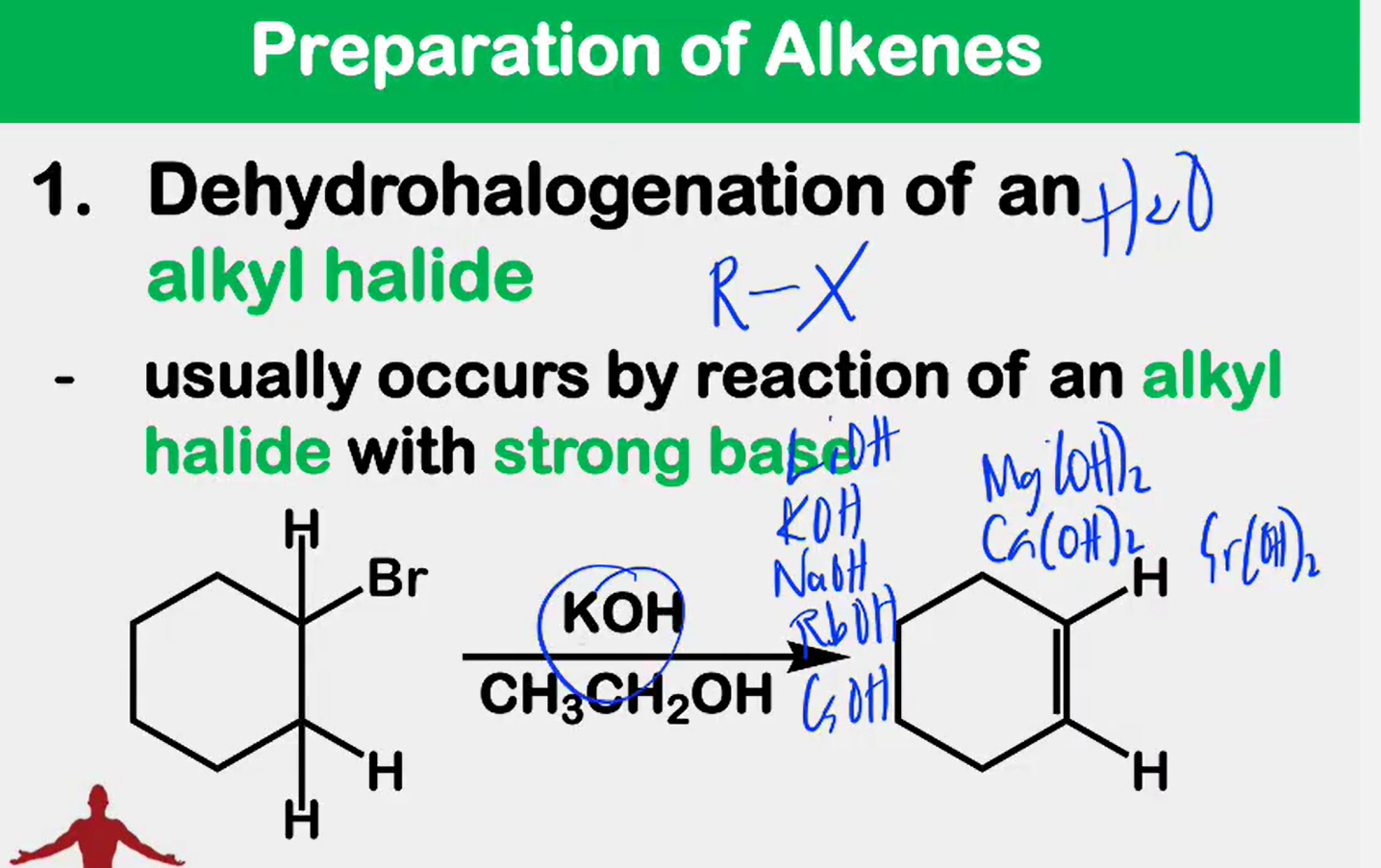
_________________ of an alkyl halide - usually occurs by reaction of an alkyl halide with strong base
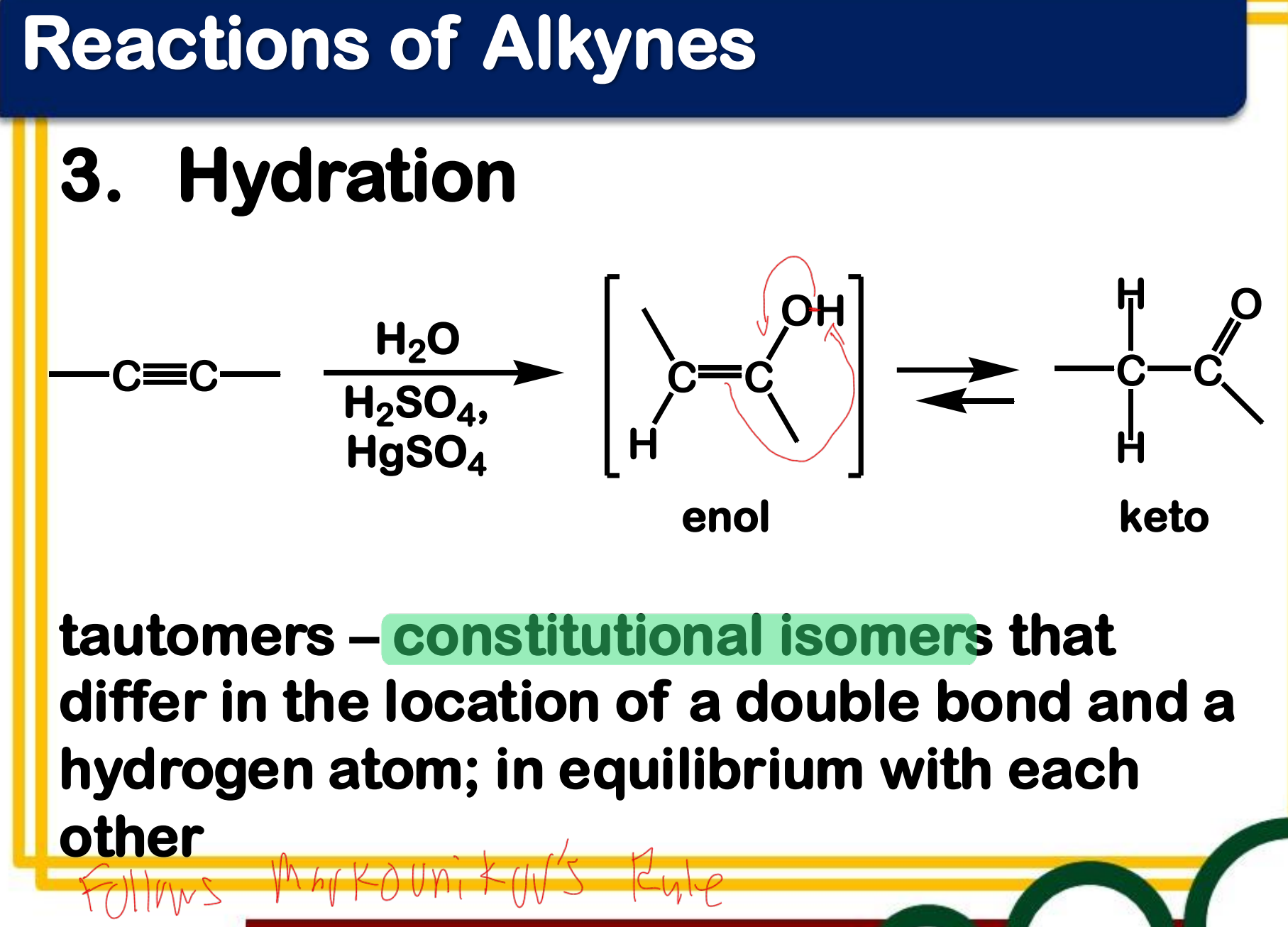
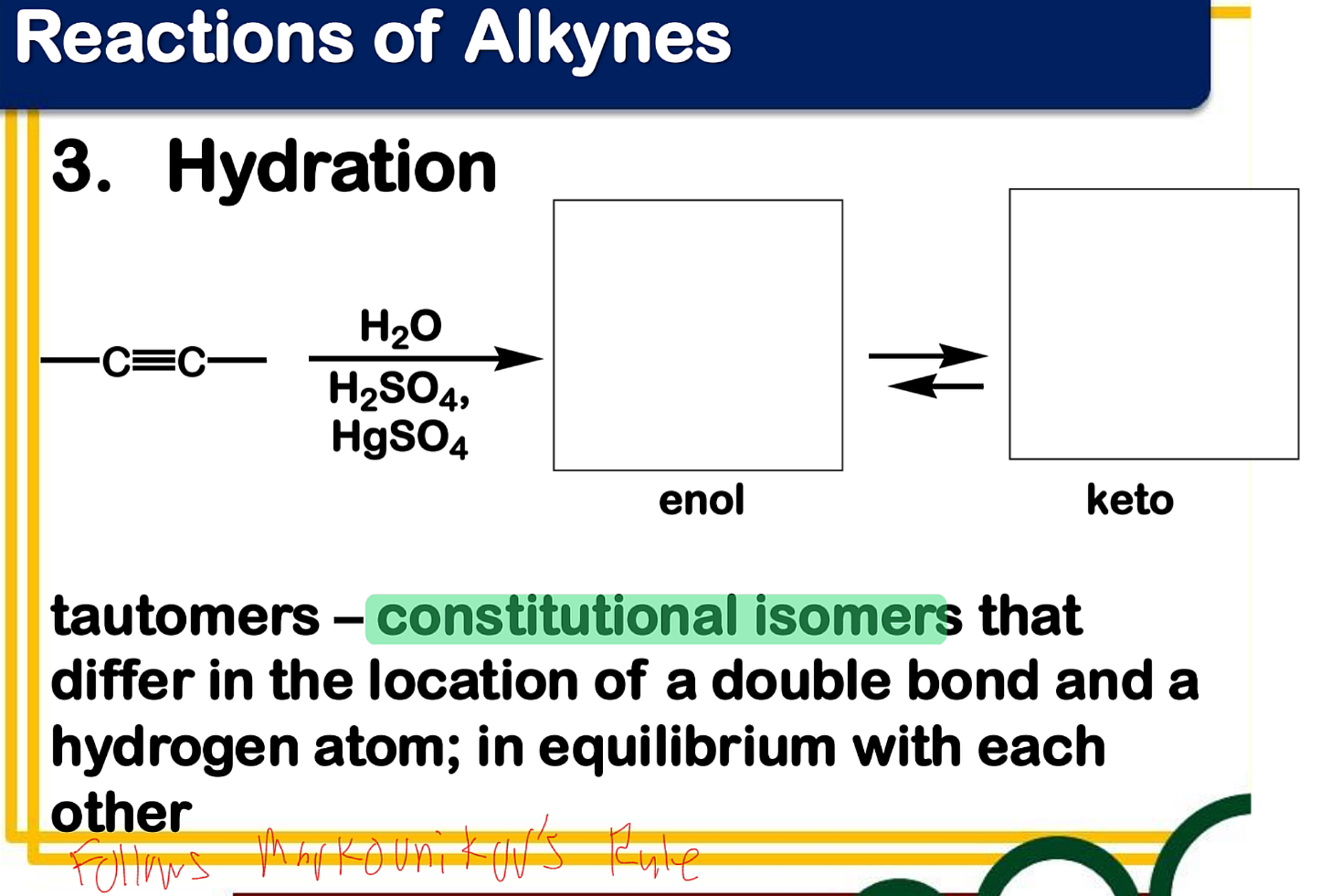
Dehydration
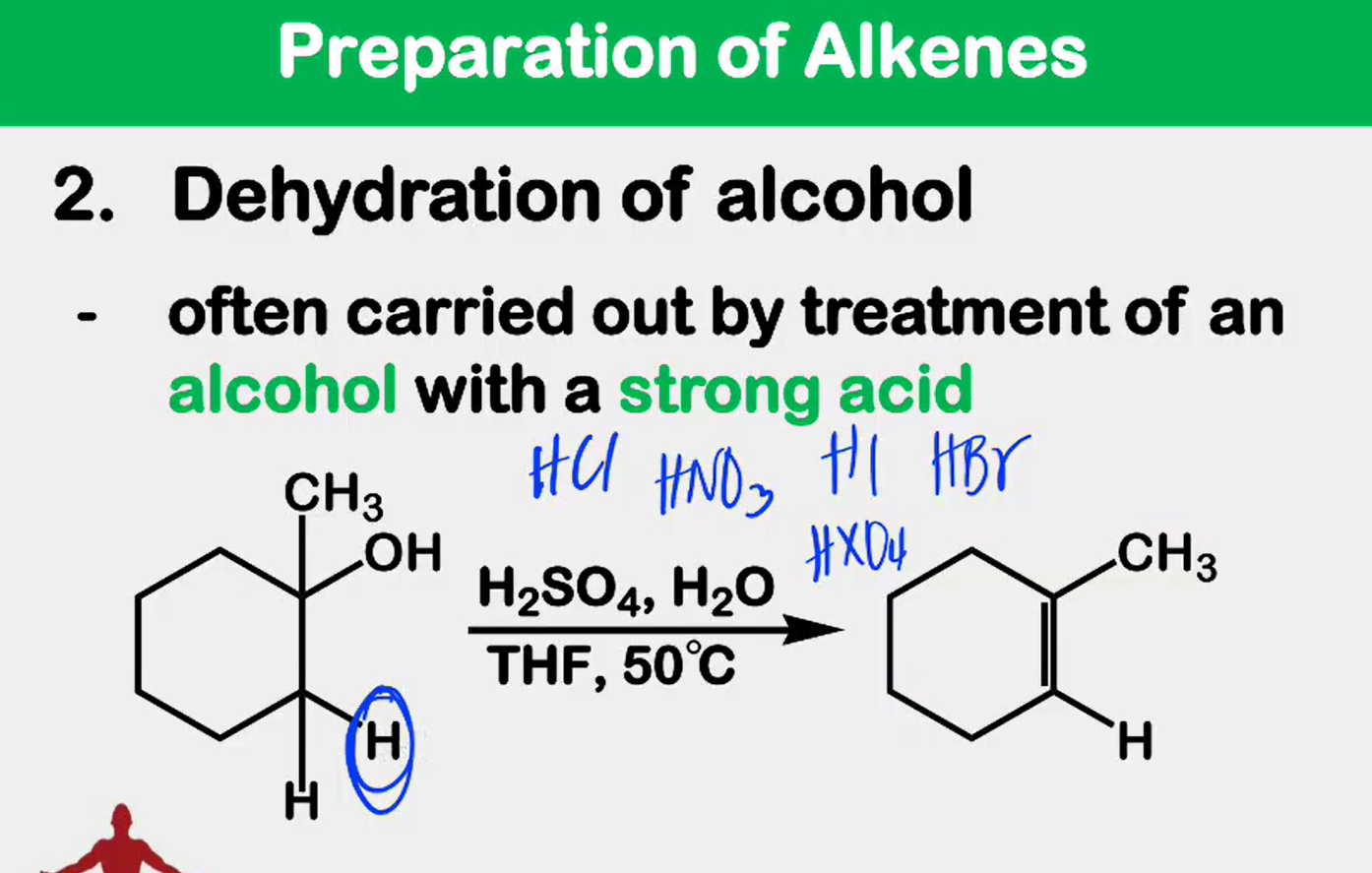
_______________ of alcohol - often carried out by treatment of an alcohol with a strong acid


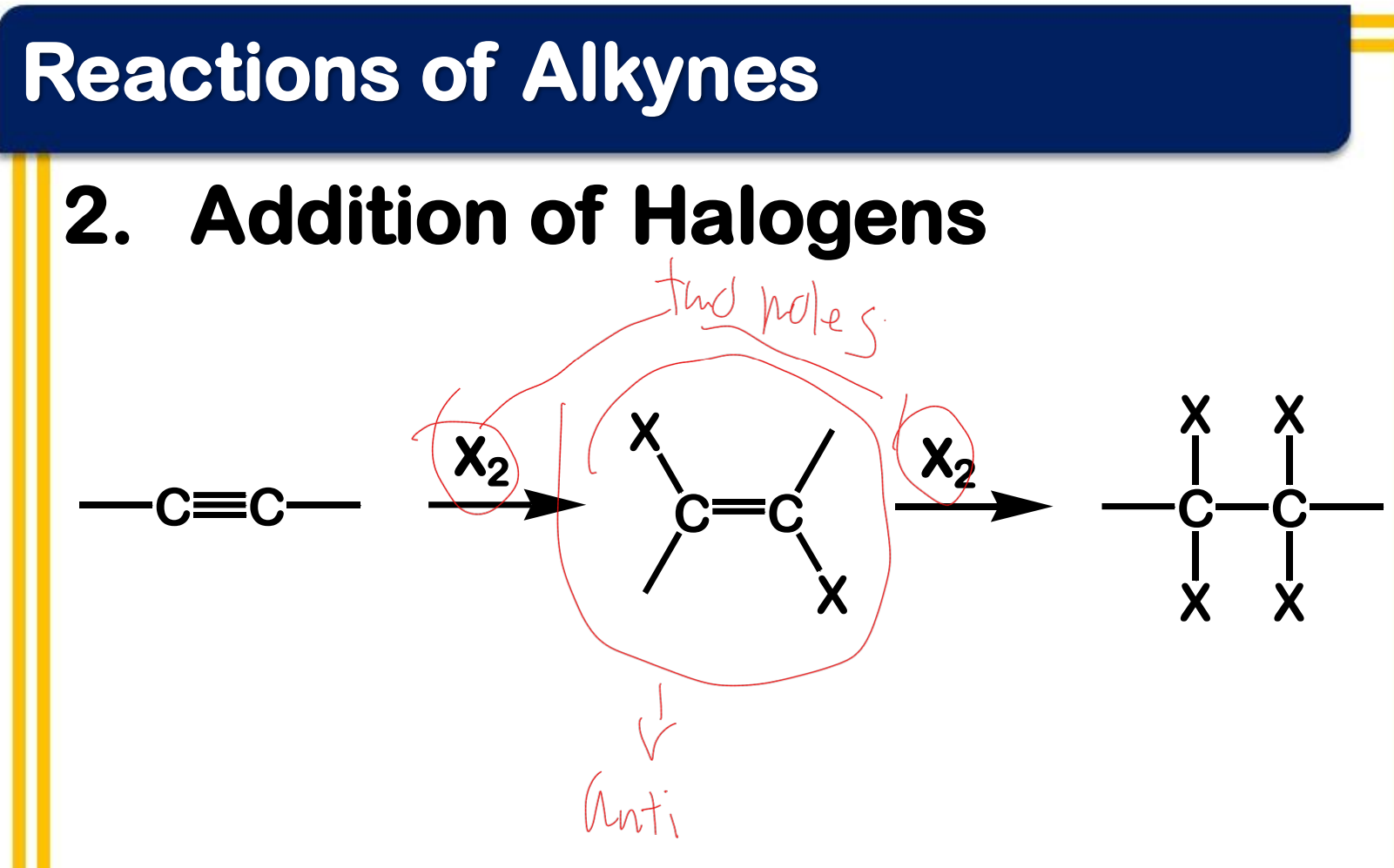
Electrophilic addition; Addition of halogens
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: Br2 or Cl2 in organic solvent, CCl4 or CH2Cl2
Electrophilic addition; Addition of halogens
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
positive visual result: disappearance of redorange bromine color

Electrophilic addition; Addition of halogens
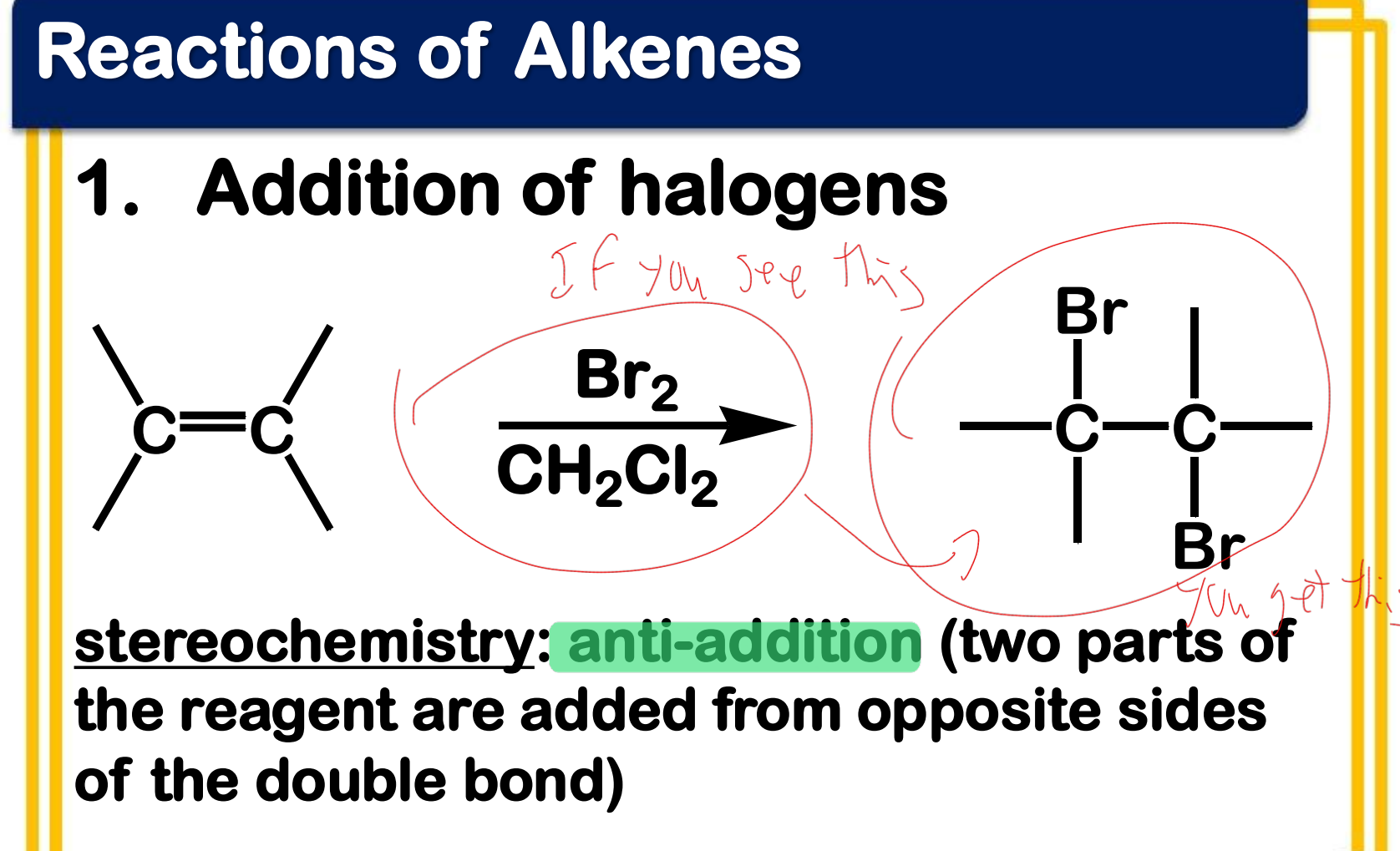
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
stereochemistry: anti-addition (two parts of the reagent are added from opposite sides of the double bond)
Electrophilic addition; Addition of HX
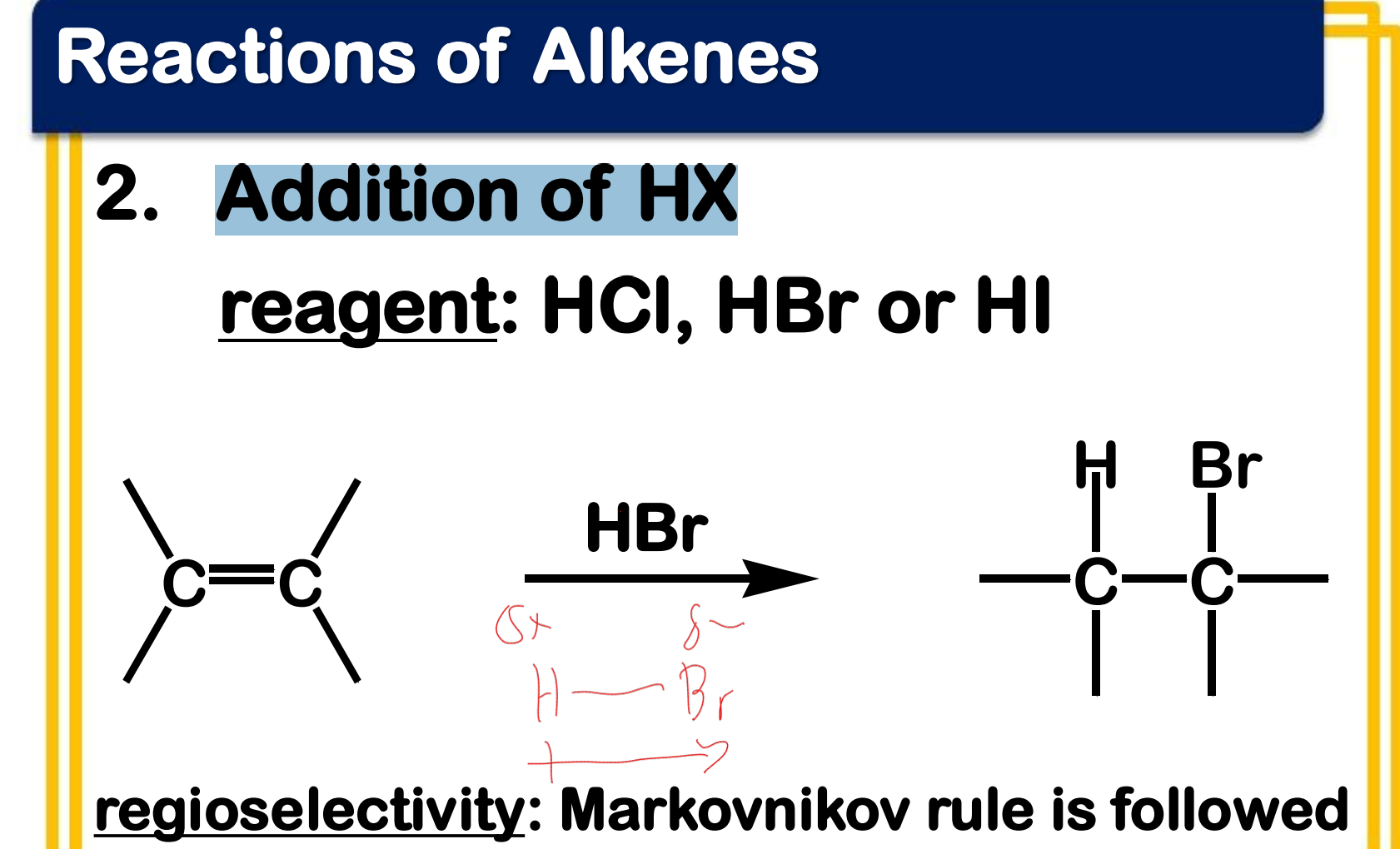
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: HCl, HBr or HI
Markovnikov Rule

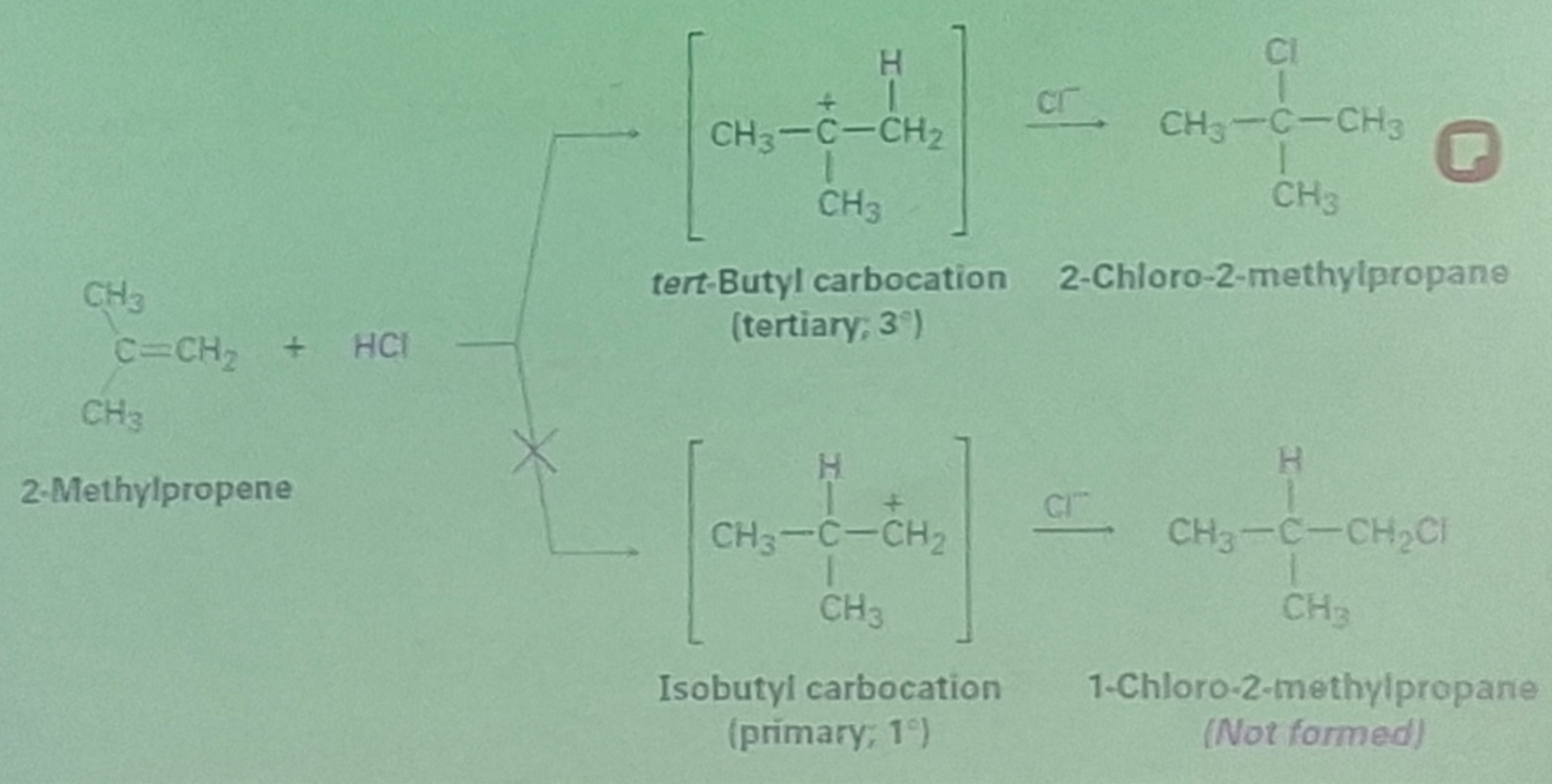
the rule that states in the addition of HX to an unsymmetrical alkene, the H atom bonds to the less substituted carbon atom
In an addition of HX to an alkene, the more highly substituted carbocation is formed as the intermediate rather than the less substituted one.
Inductive Effect
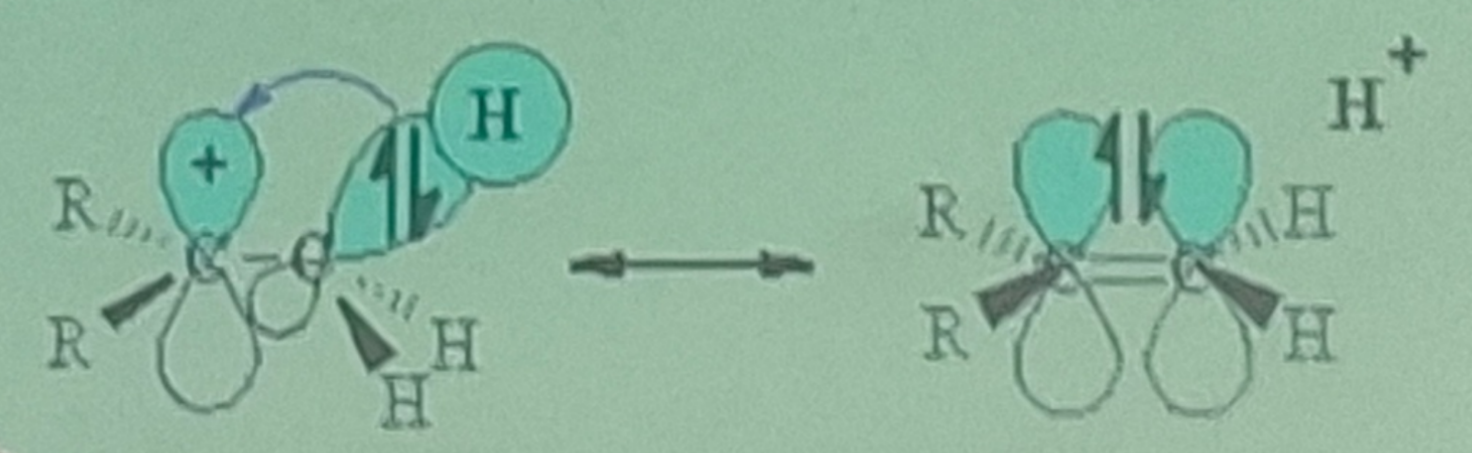
Two concepts to explain carbocation stability:
________________
shifting of electrons in a sigma bond in response to the electronegativity of nearby atoms
Hyperconjugation
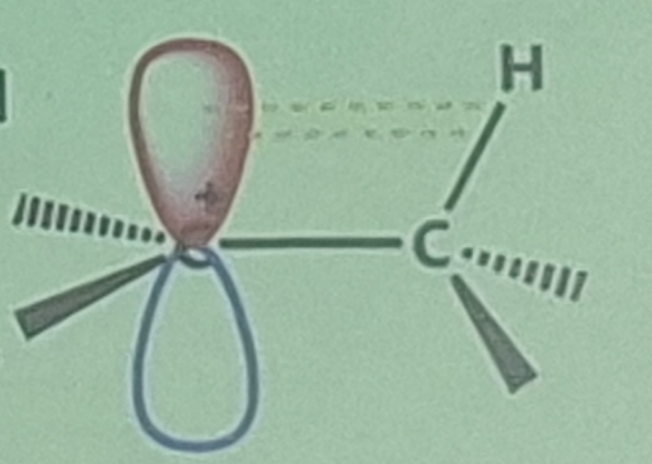
Two concepts to explain carbocation stability:
__________________
stabilizing interaction between a vacant p orbital and neighboring sigma bond electrons
Positive charge is spread over a greater volume of space
Electrophilic addition; Halohydrin formation
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: Br2 in H2O
Electrophilic addition; Halohydrin formation
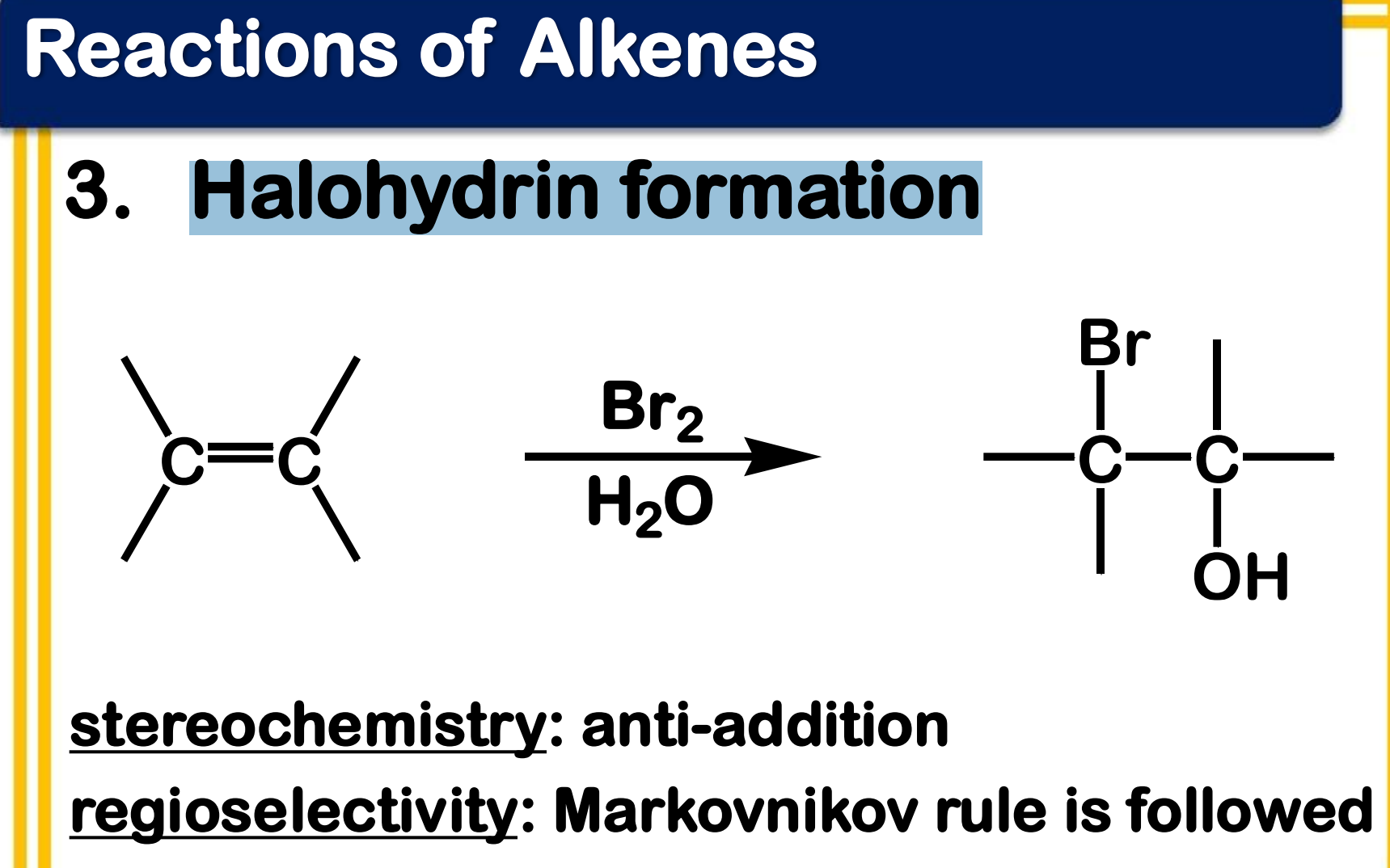
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
the solvent, water, will compete with bromide as nucleophile
stereochemistry: anti-addition
regioselectivity: Markovnikov rule is followed
Electrophilic addition; Reaction with cold, concentrated H2SO4

State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: cold, conc. H2SO4
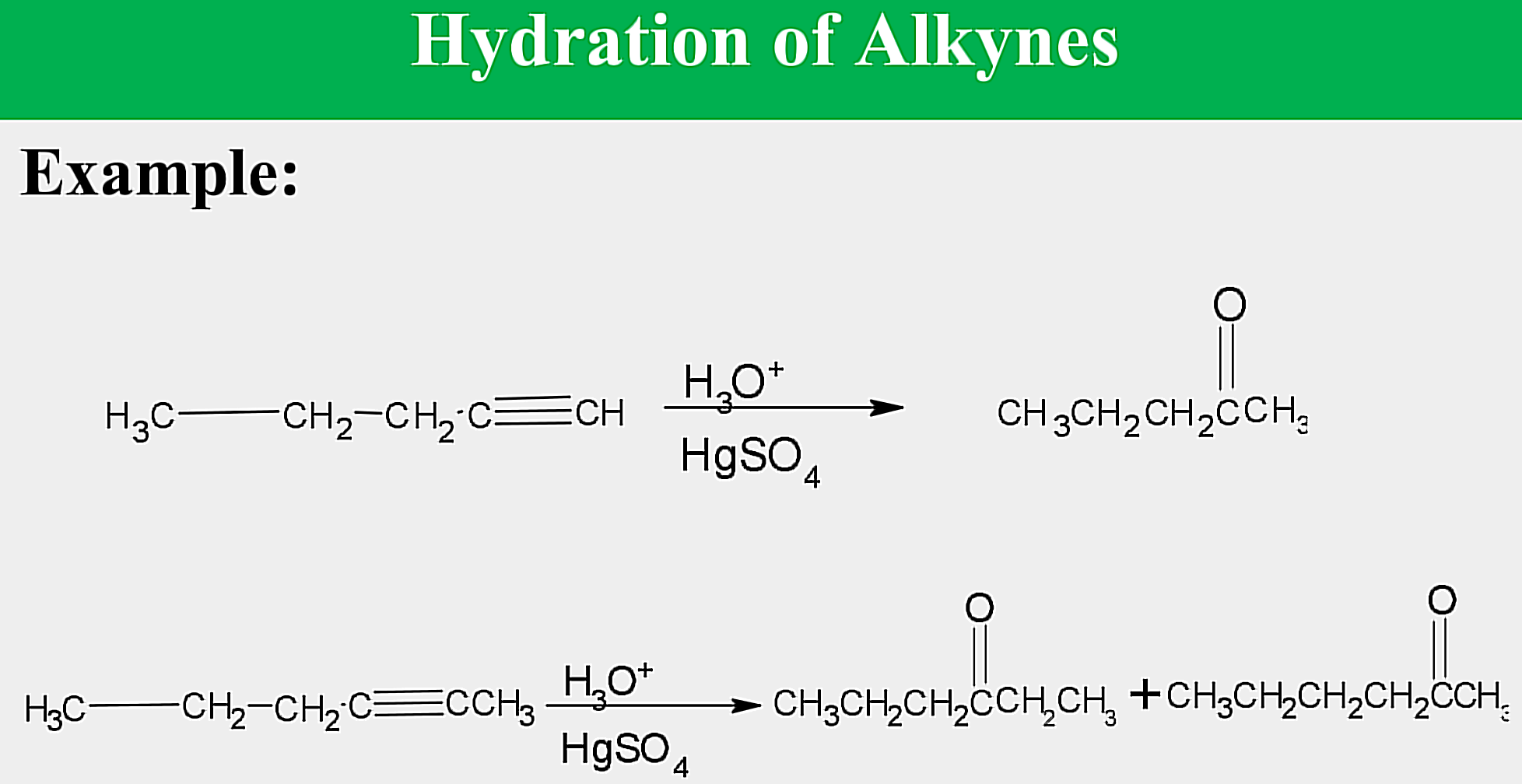
Electrophilic addition; Hydration
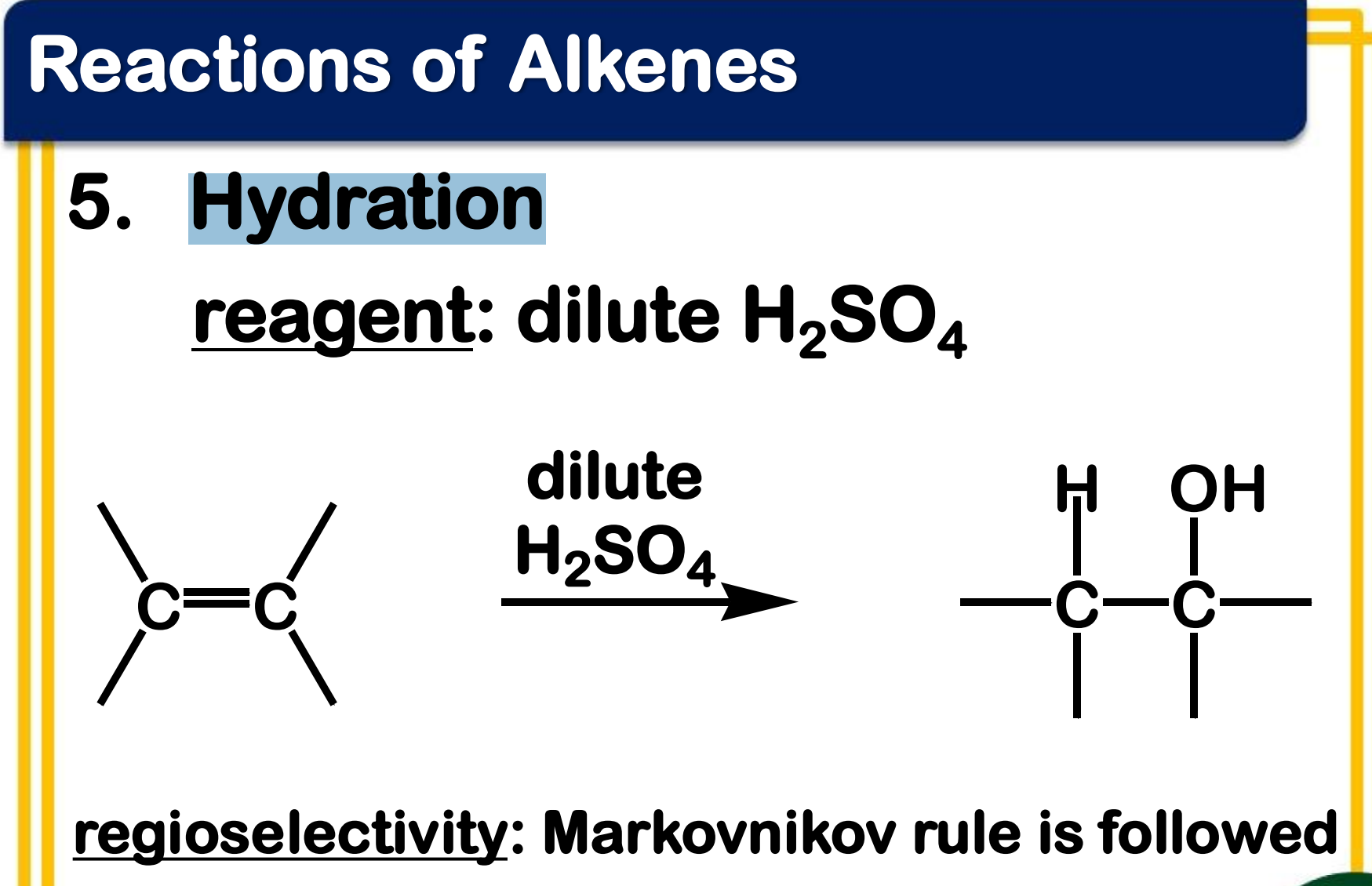
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: dilute H2SO4

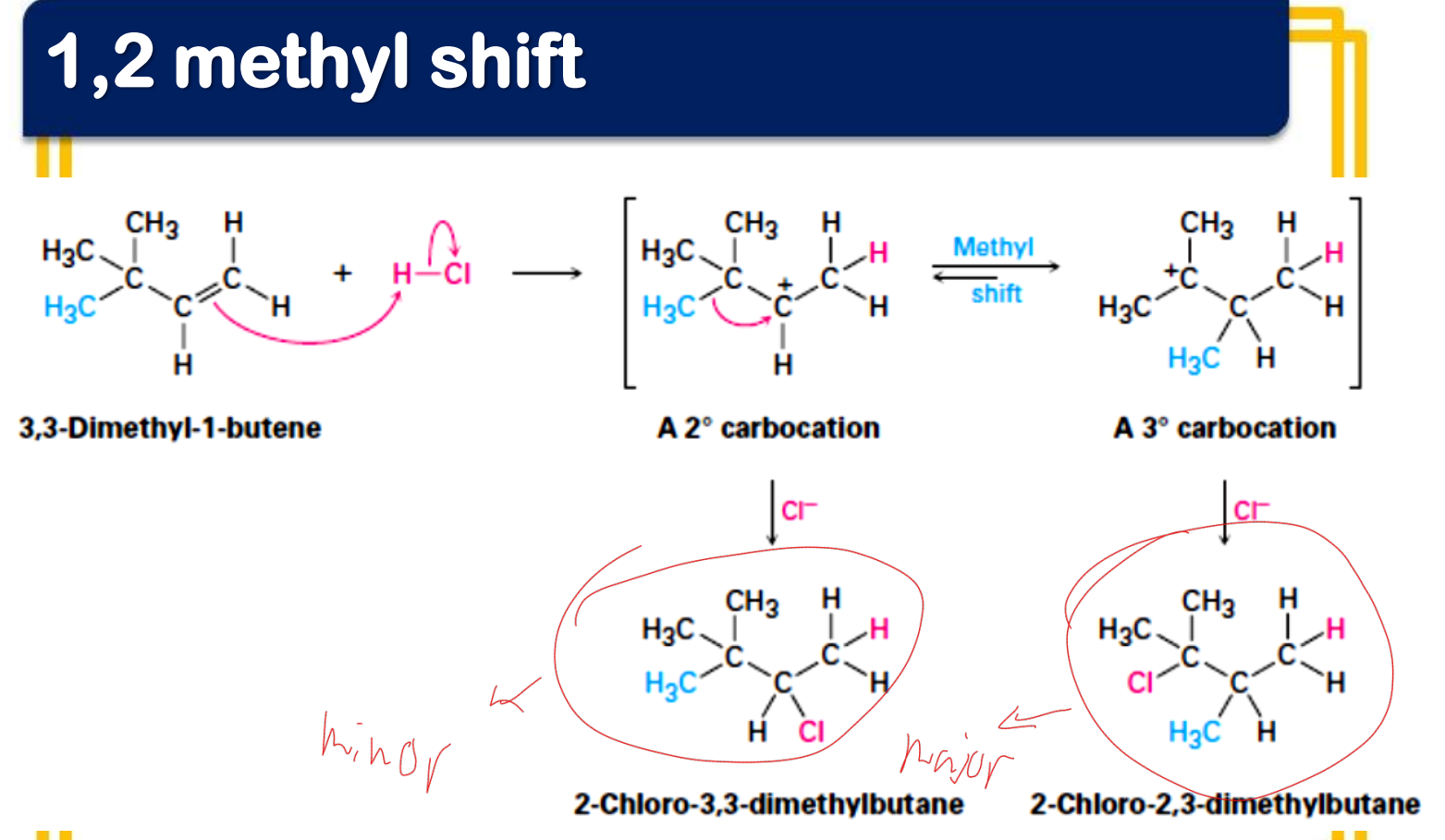
In electrophilic addition reactions, carbocations may rearrange forming ‘unexpected’ products. What are these changes called?
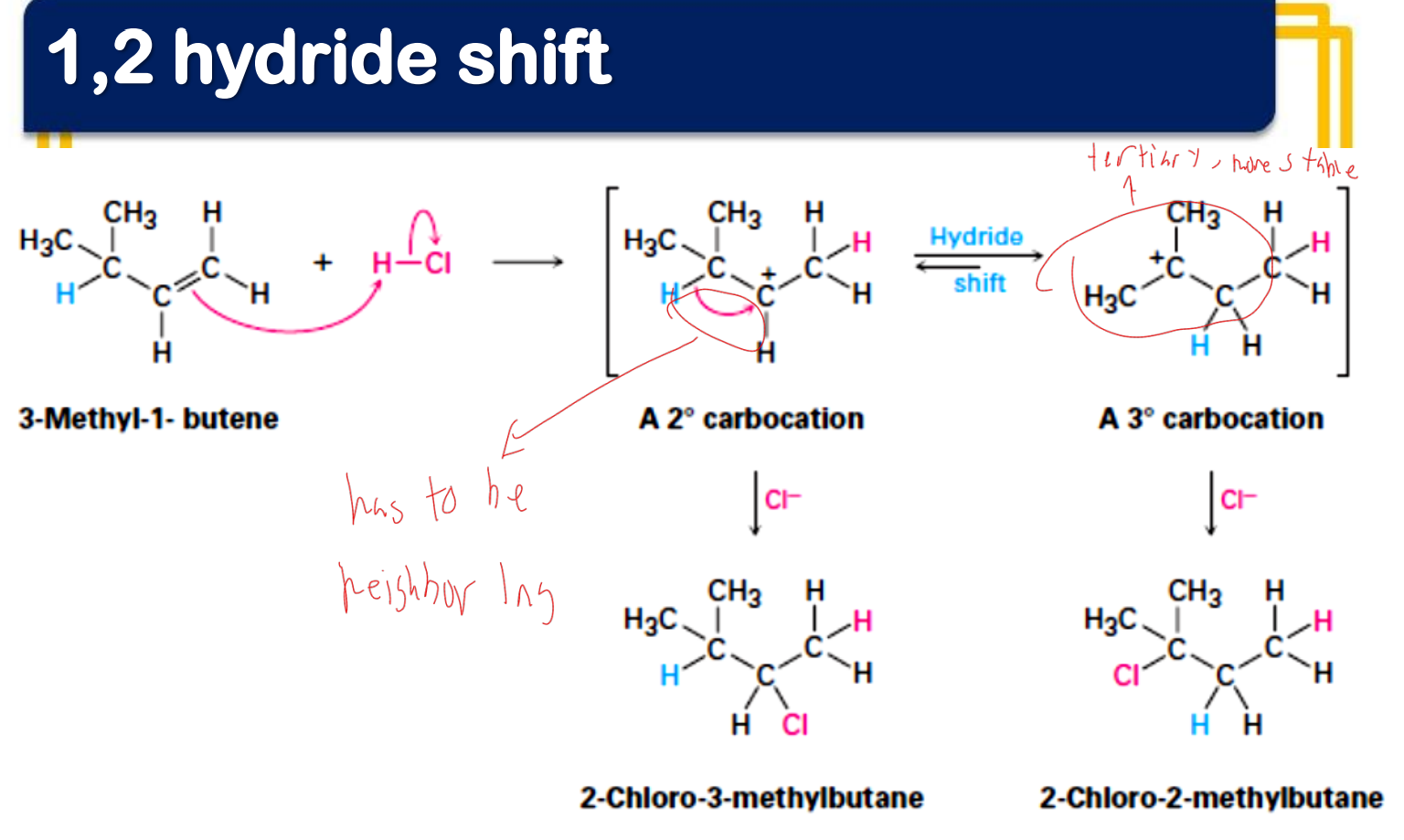
Given what are the minor and major products?
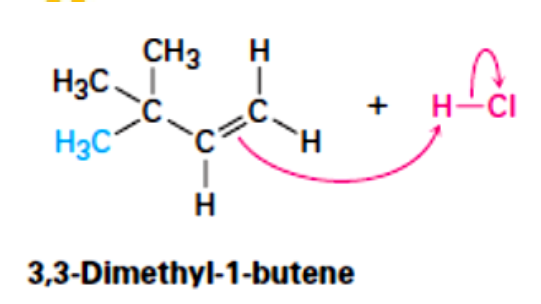
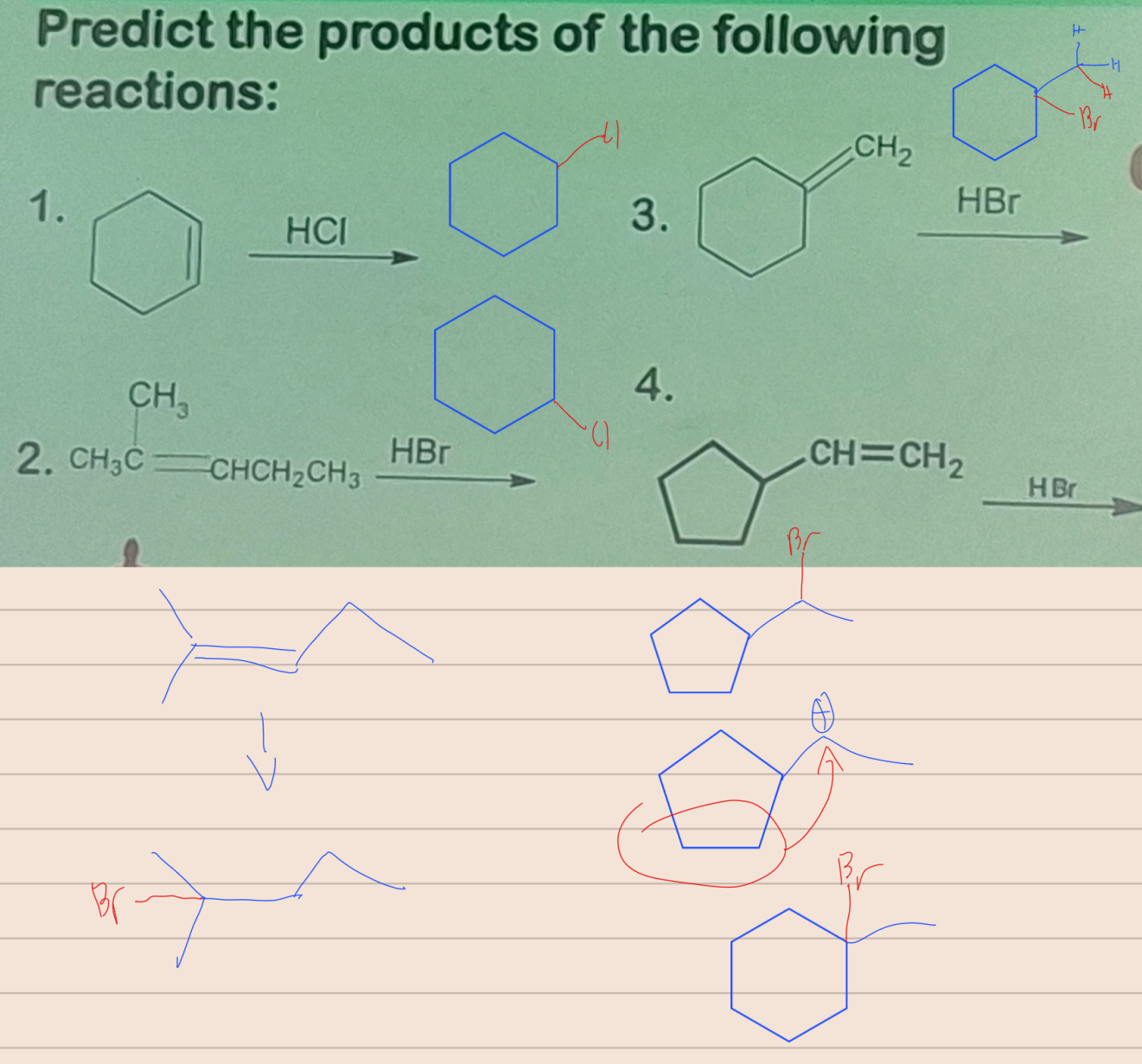
Free Radical Addition; Addition of HBr with peroxide
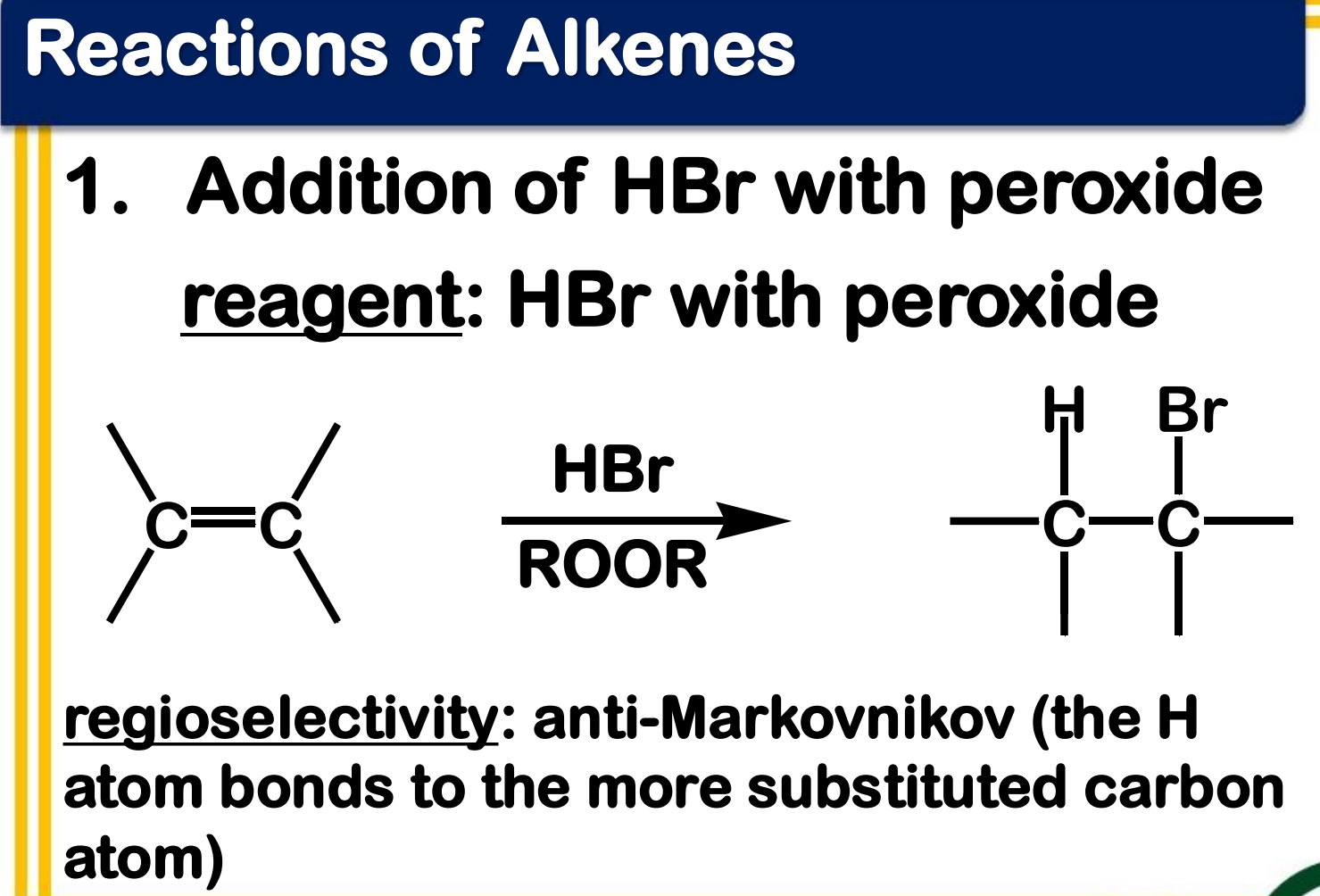
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: HBr with peroxide
regioselectivity: anti-Markovnikov (the H atom bonds to the more substituted carbon atom)
Free Radical Addition; Polymerization of alkenes
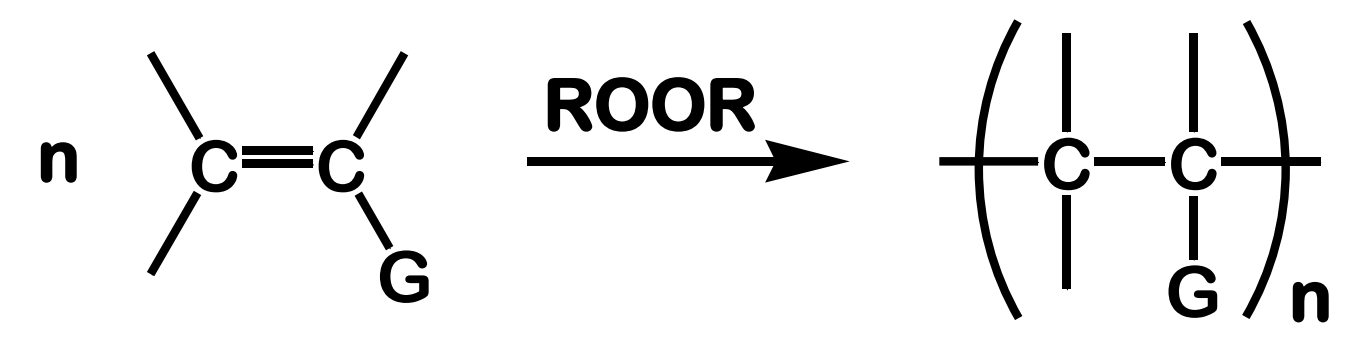
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: peroxide as initiator
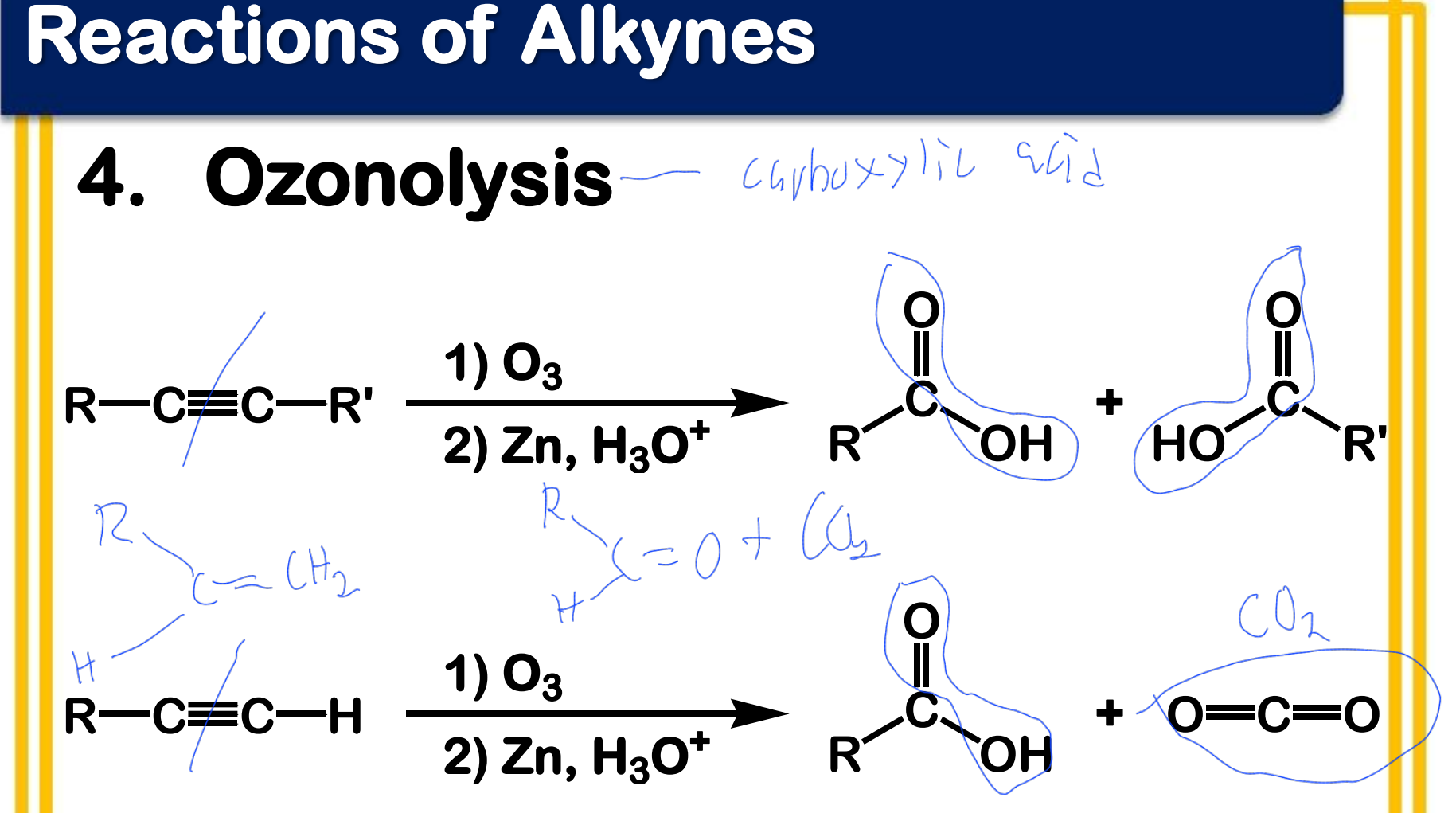
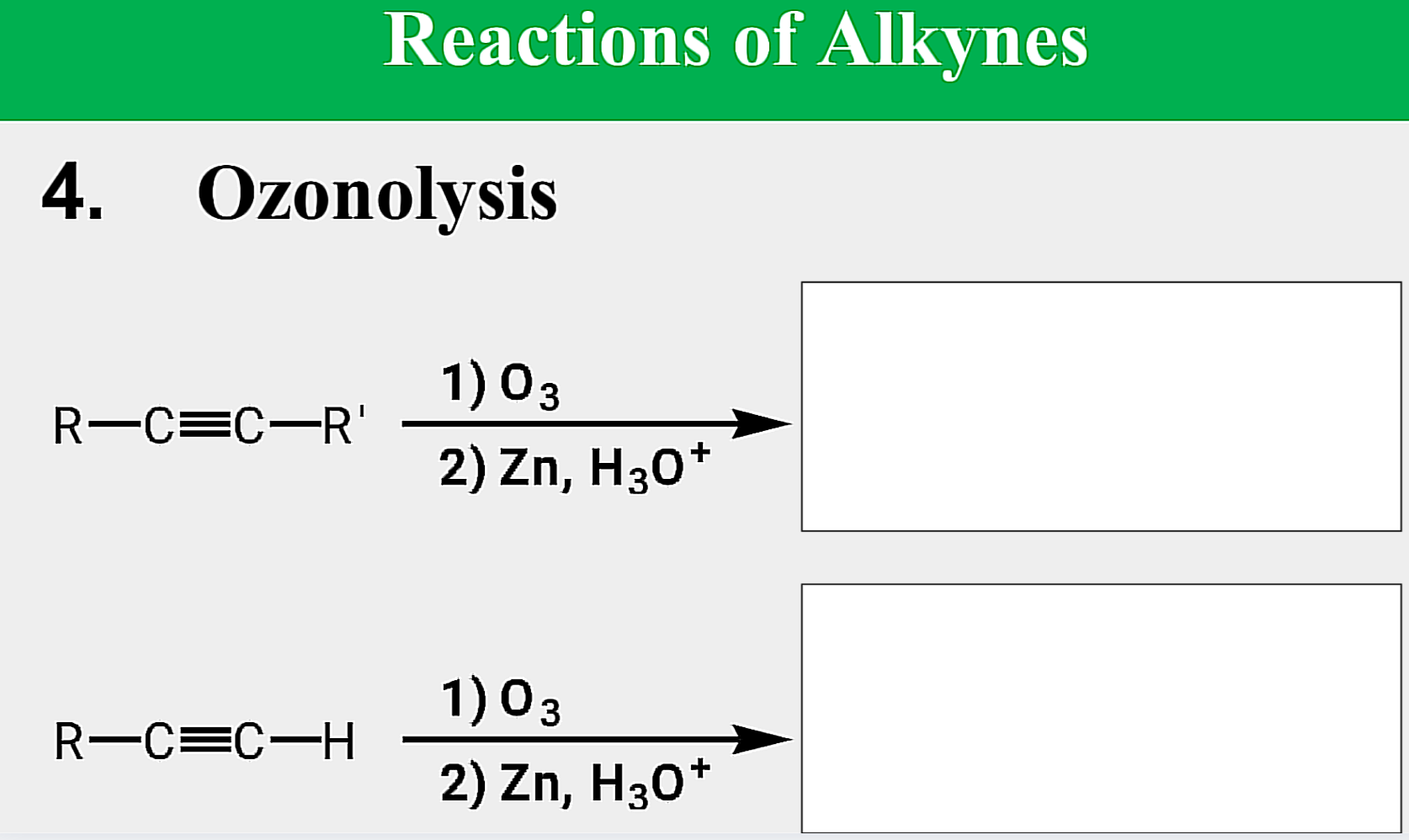
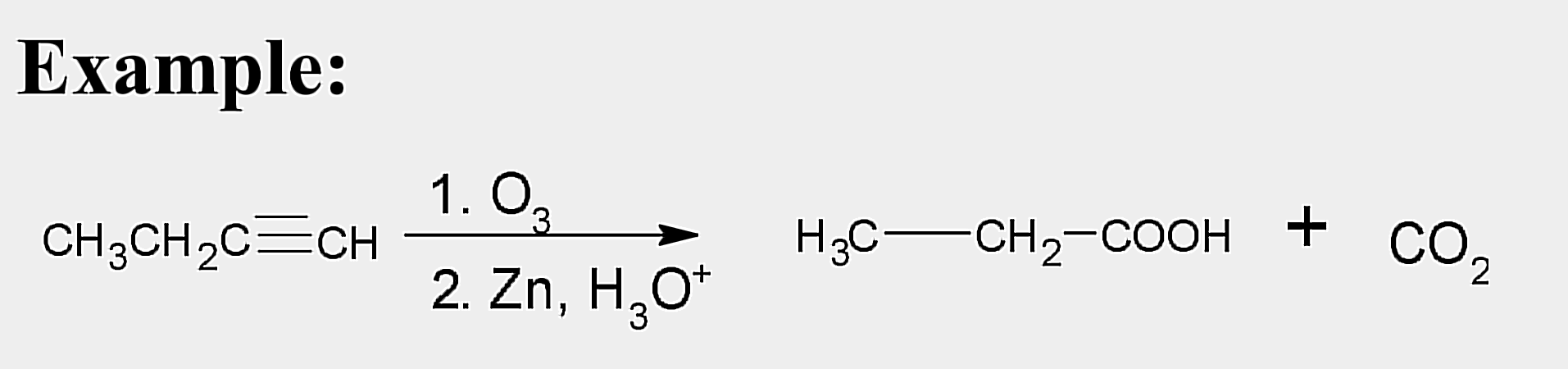
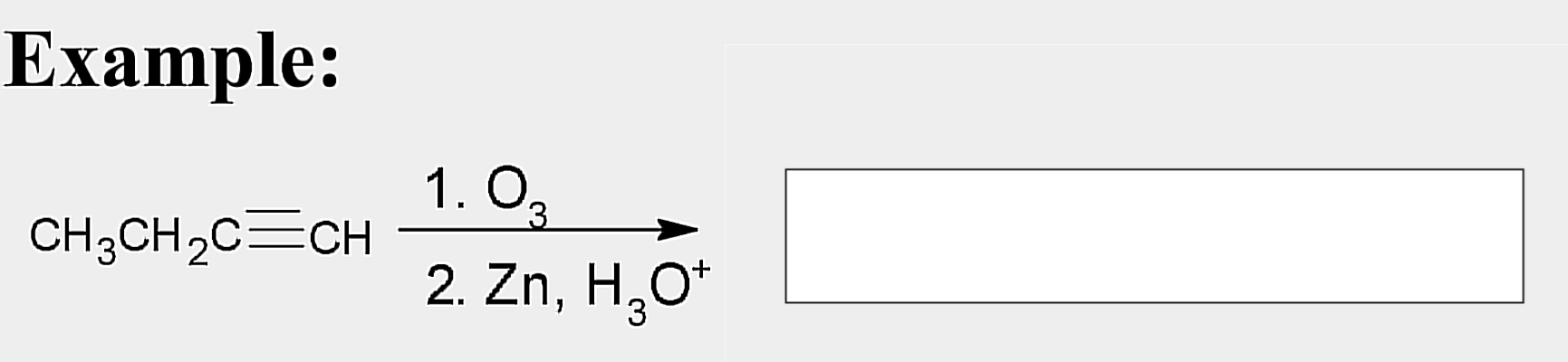
Oxidation; Ozonolysis

State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: (1) O3 ; (2) Zn, H3O+
aldehydes; ketones

Products of ozonolysis of alkenes are __________ and/or ___________ depending on the structure.
Oxidation: Reaction with acidic KMnO4
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: KMnO4 , H3O+
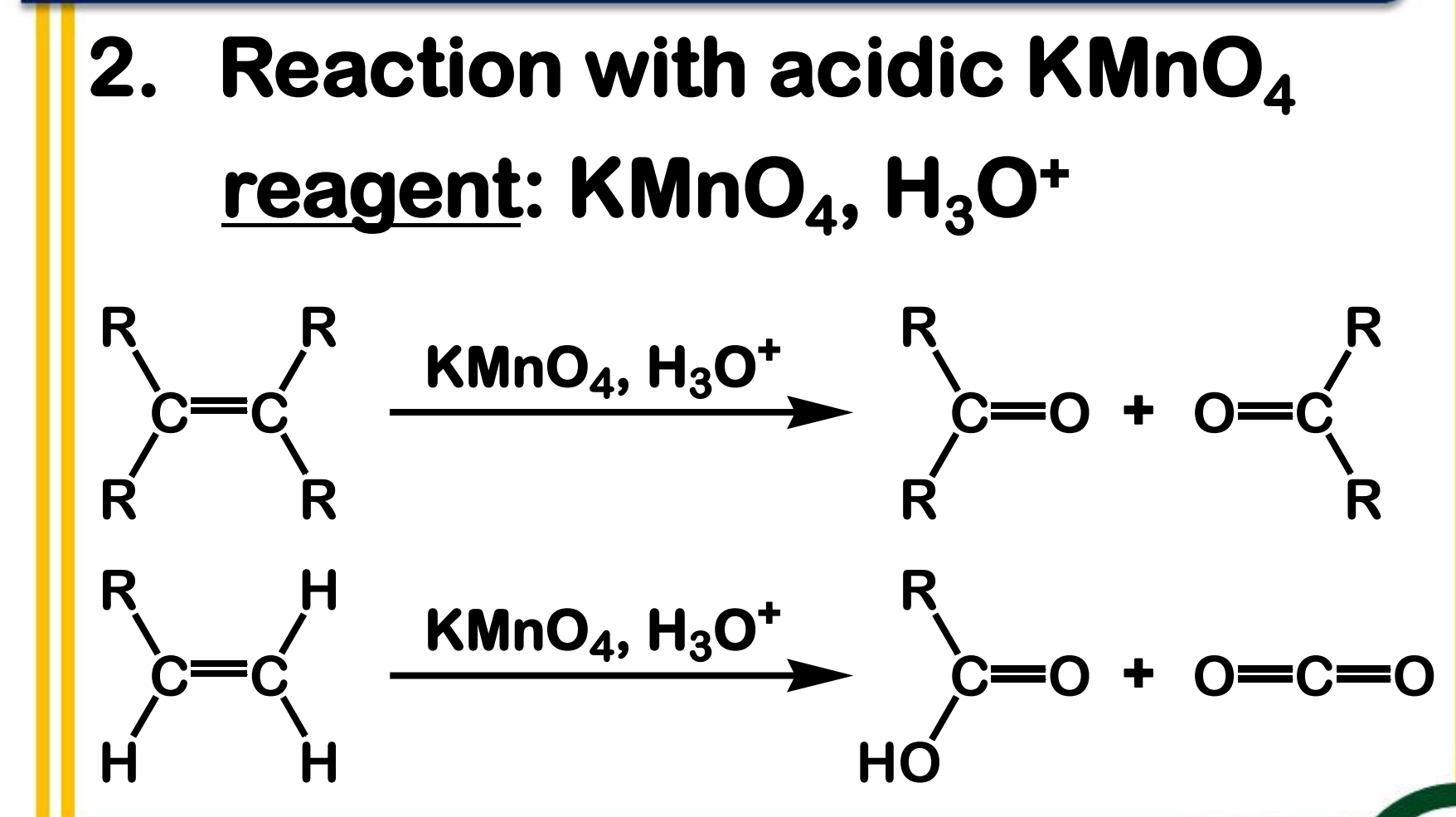
What are the respective products of each reactant in Oxidation: Reaction with acidic KMnO4?
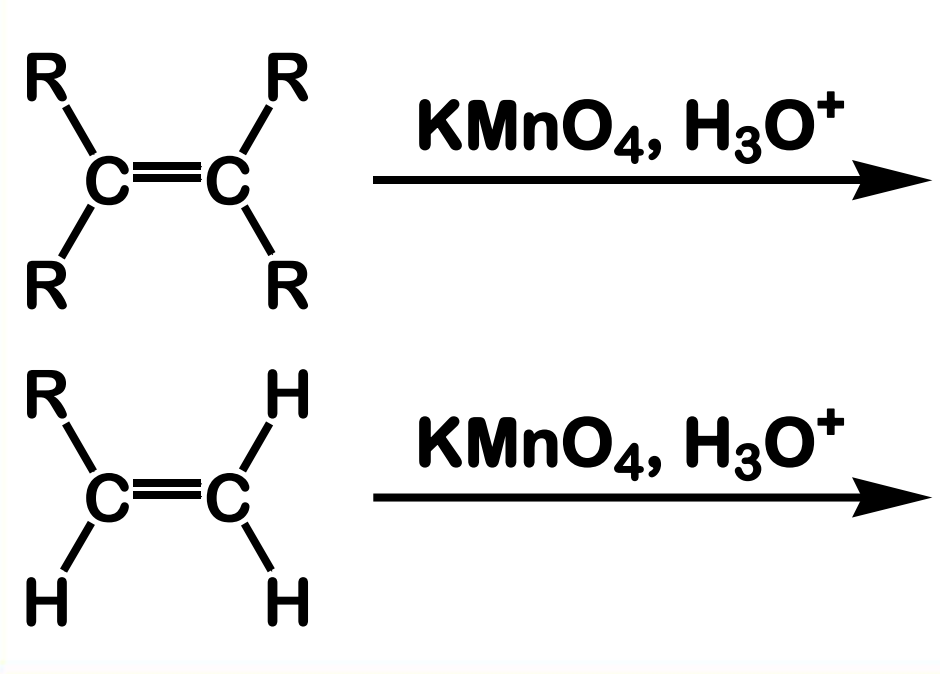

Oxidation with KMnO4
reagent: KMnO4
What is the positive visual result of this process.
Oxidation; Hydroxylation
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: cold, dilute, neutral KMnO4
stereochemistry: syn addition
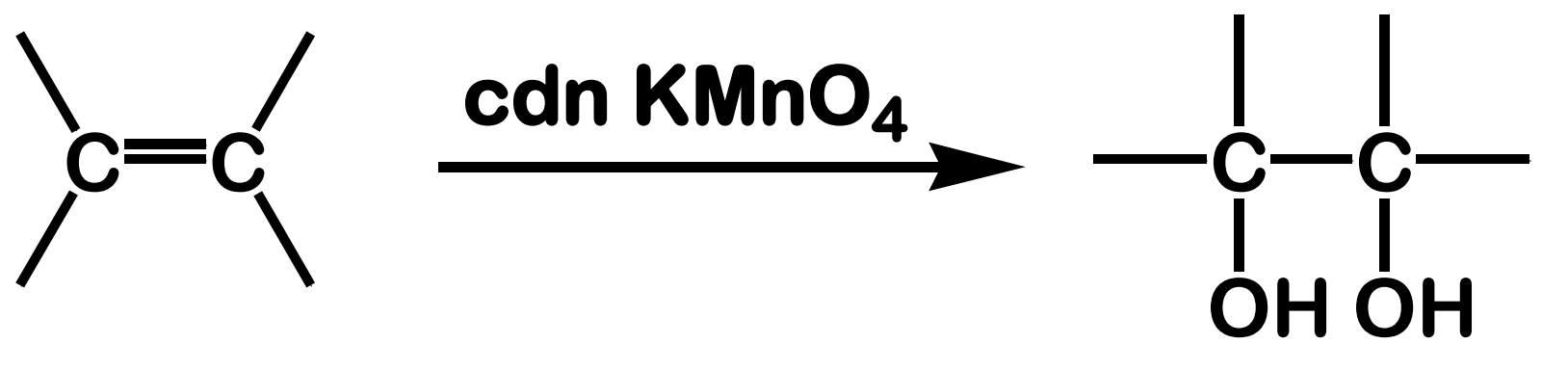
What is the product of this Oxidation; Hydroxylation reaction?
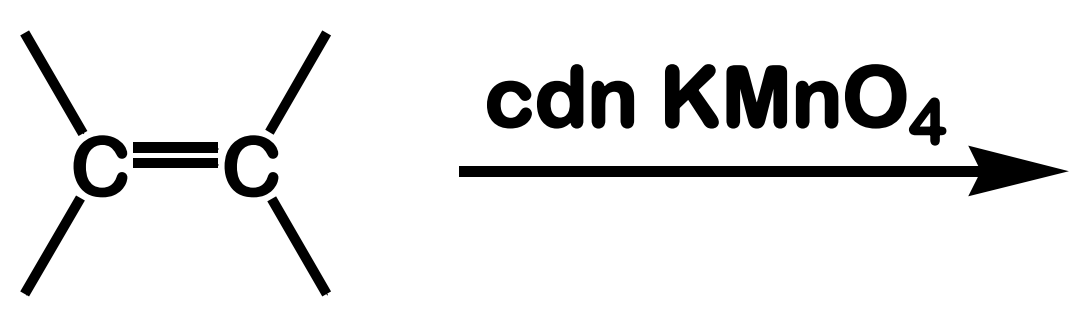
Oxidation; Hydroxylation
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
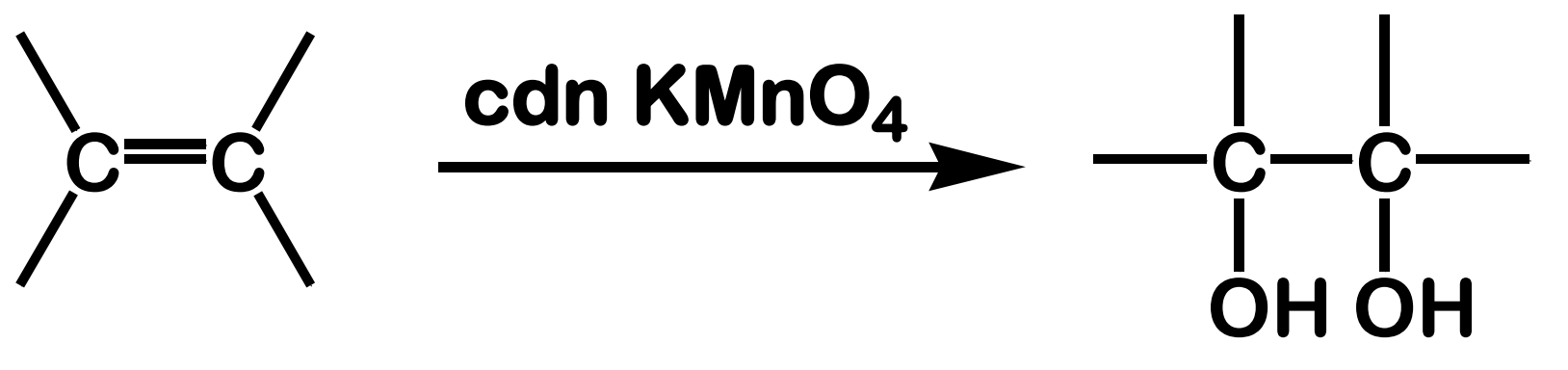
Oxidation; Epoxidation
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.
reagent: m-chloroperoxybenzoic acid
Oxidation; Epoxidation
State which type of Reactions of Alkenes (Electrophilic addition, Free Radical Addition, Oxidation) and which specific reaction under each category.


What is the product of this Oxidation; Epoxidation reaction?
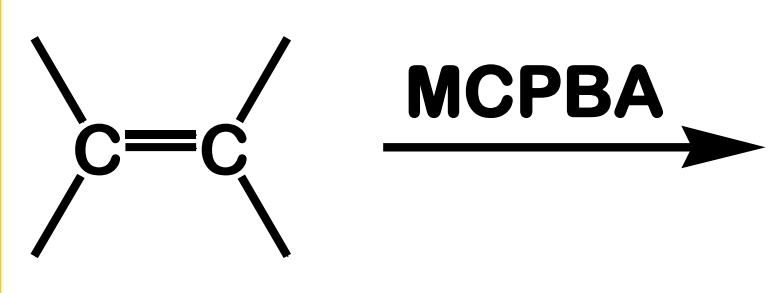
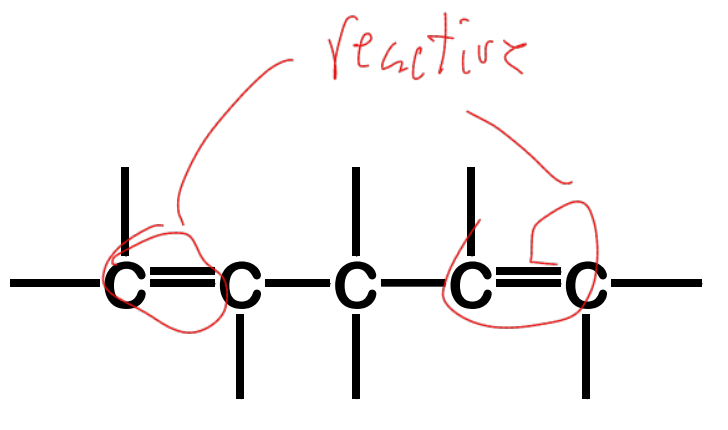
conjugated
Alkadienes - have two sets of C-C double bonds
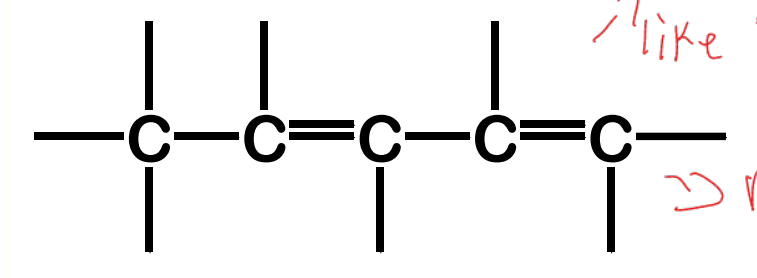
cumulated
Alkadienes - have two sets of C-C double bonds
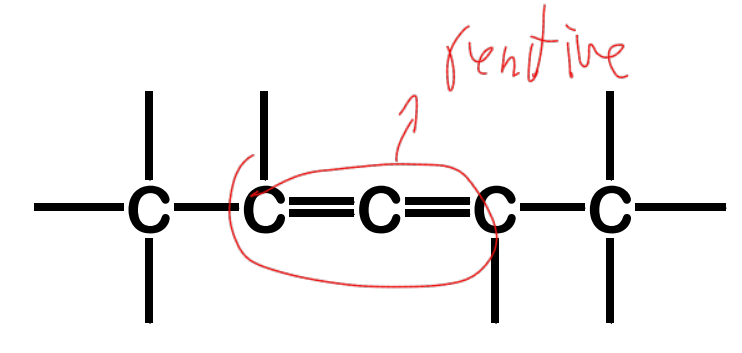
isolated
Alkadienes - have two sets of C-C double bonds
Conjugation
1,3-diene
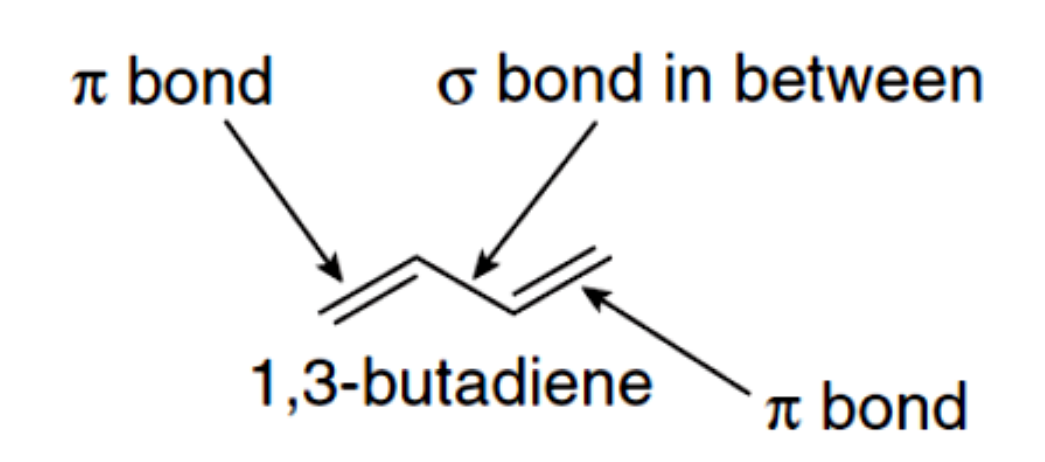
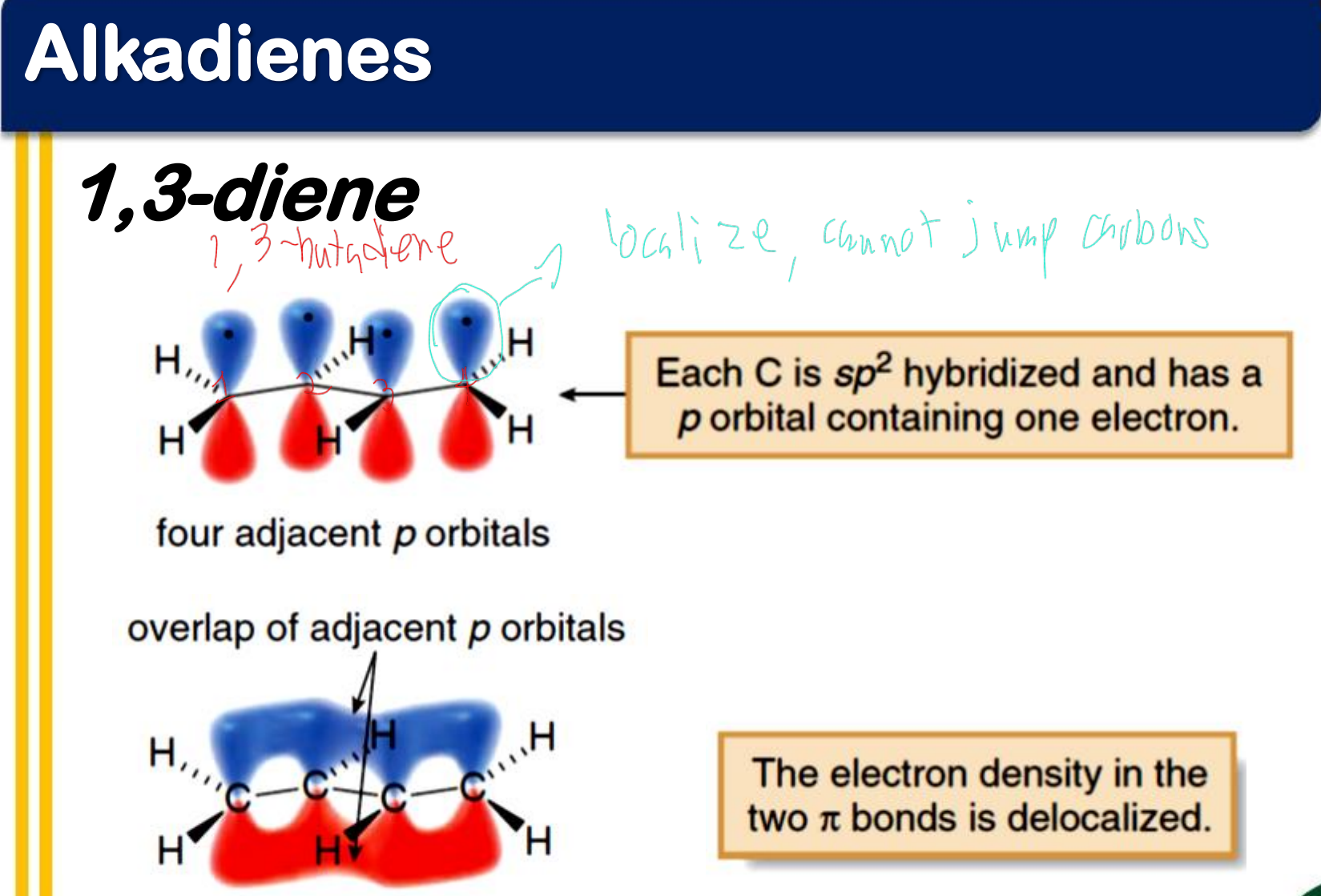
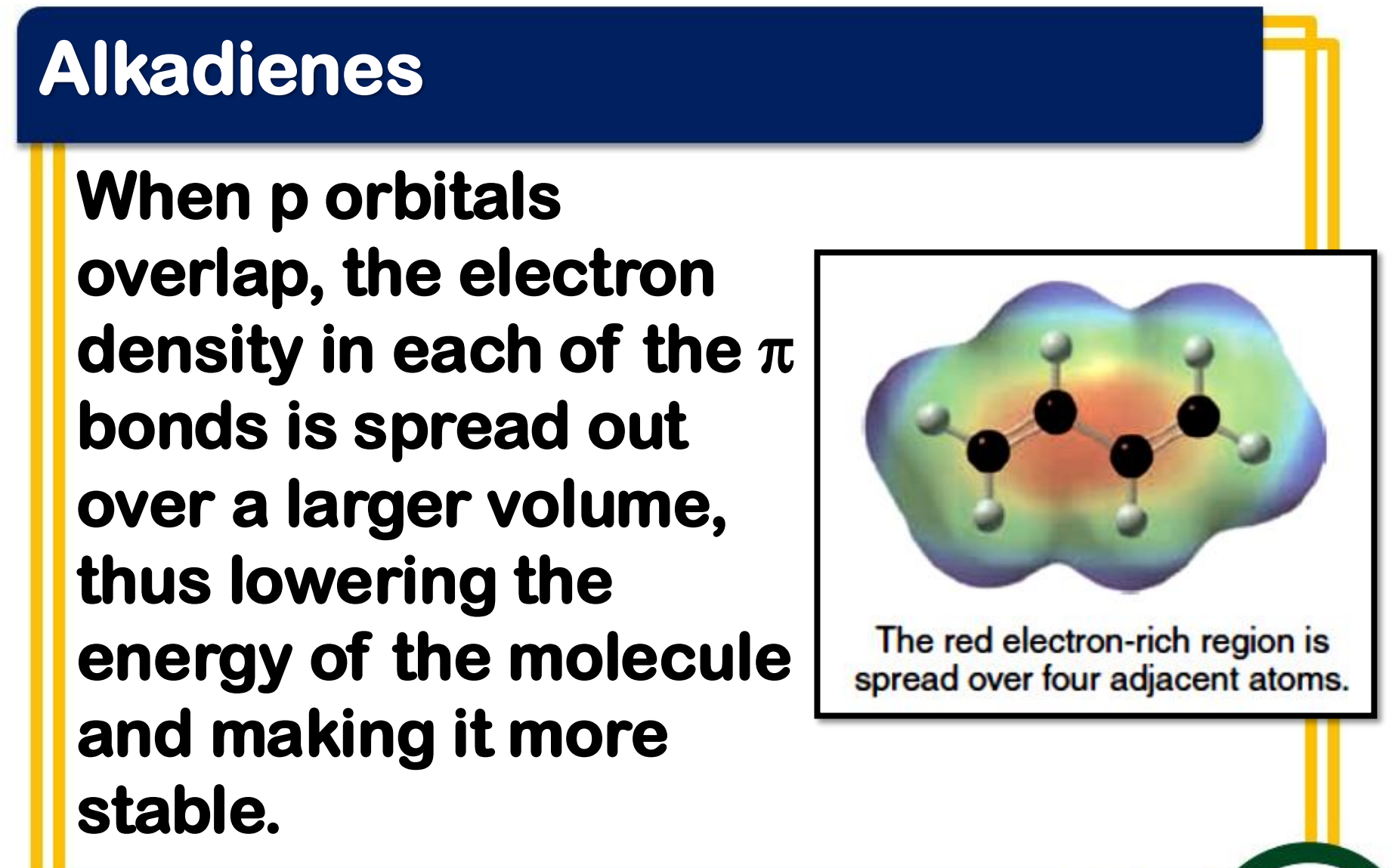
_______________ - occurs when p orbitals can overlap on three or more adjacent atoms
_____________ - contains two carbon-carbon double bonds joined by a single σ bond
Allylic carbocation
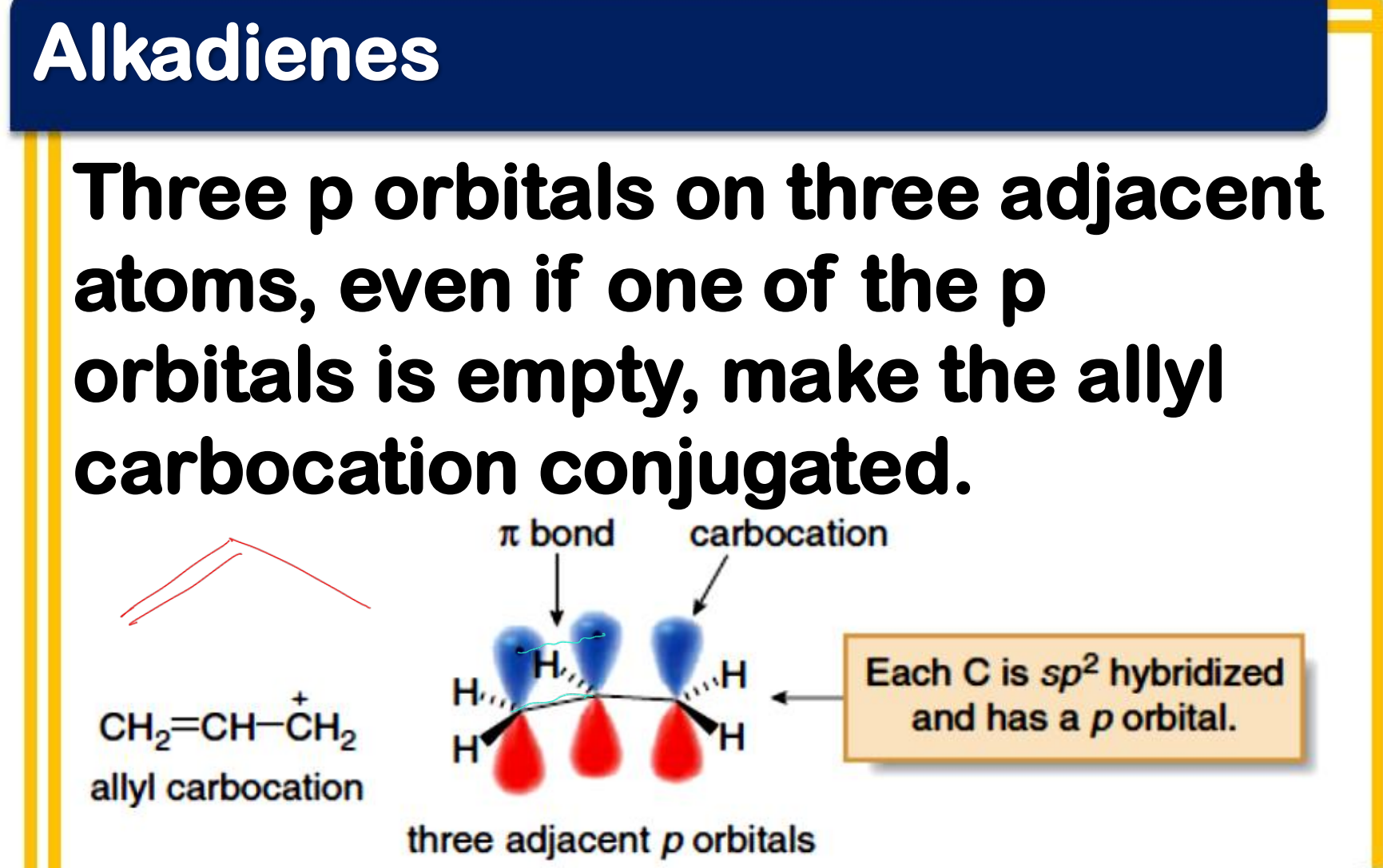
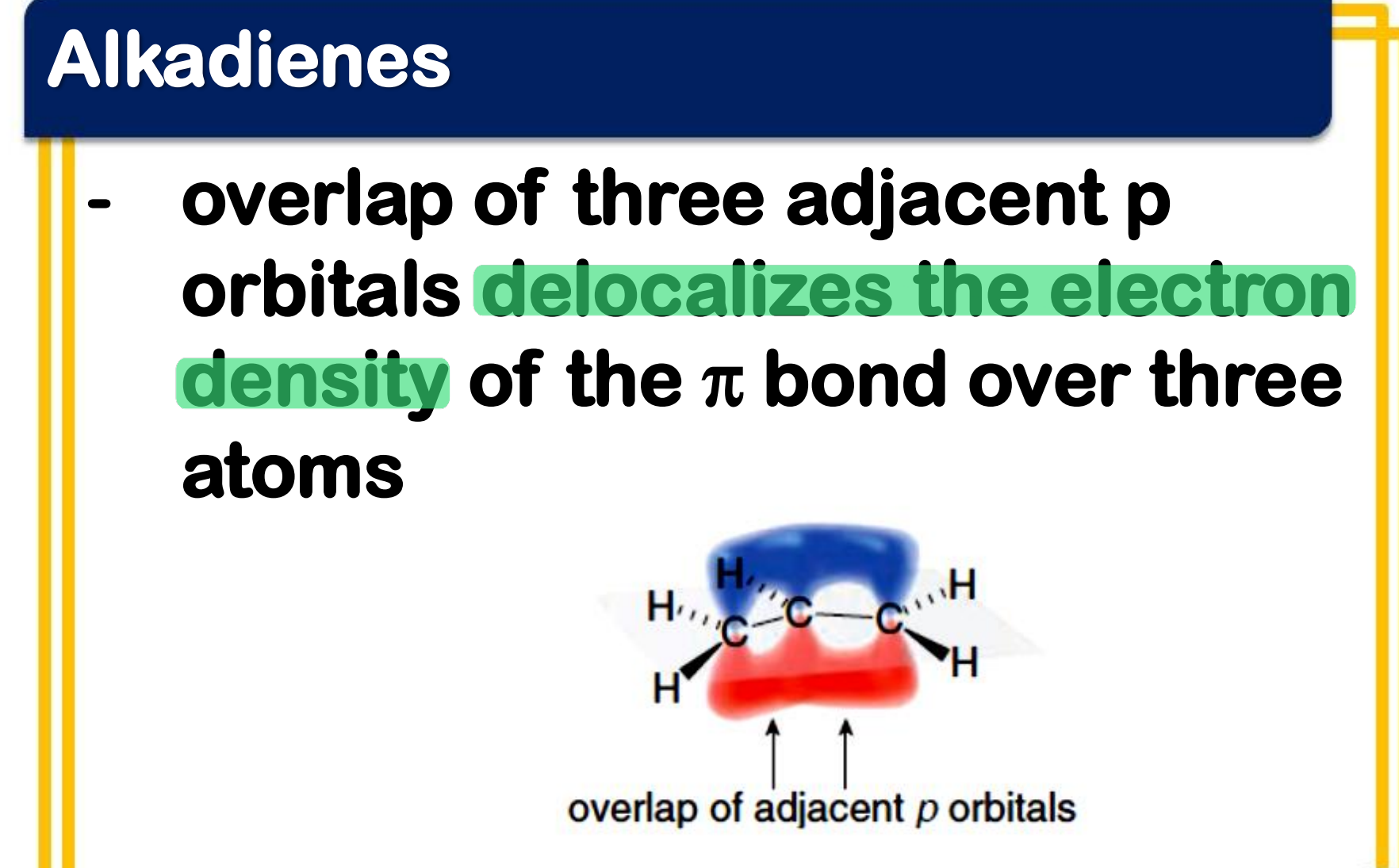
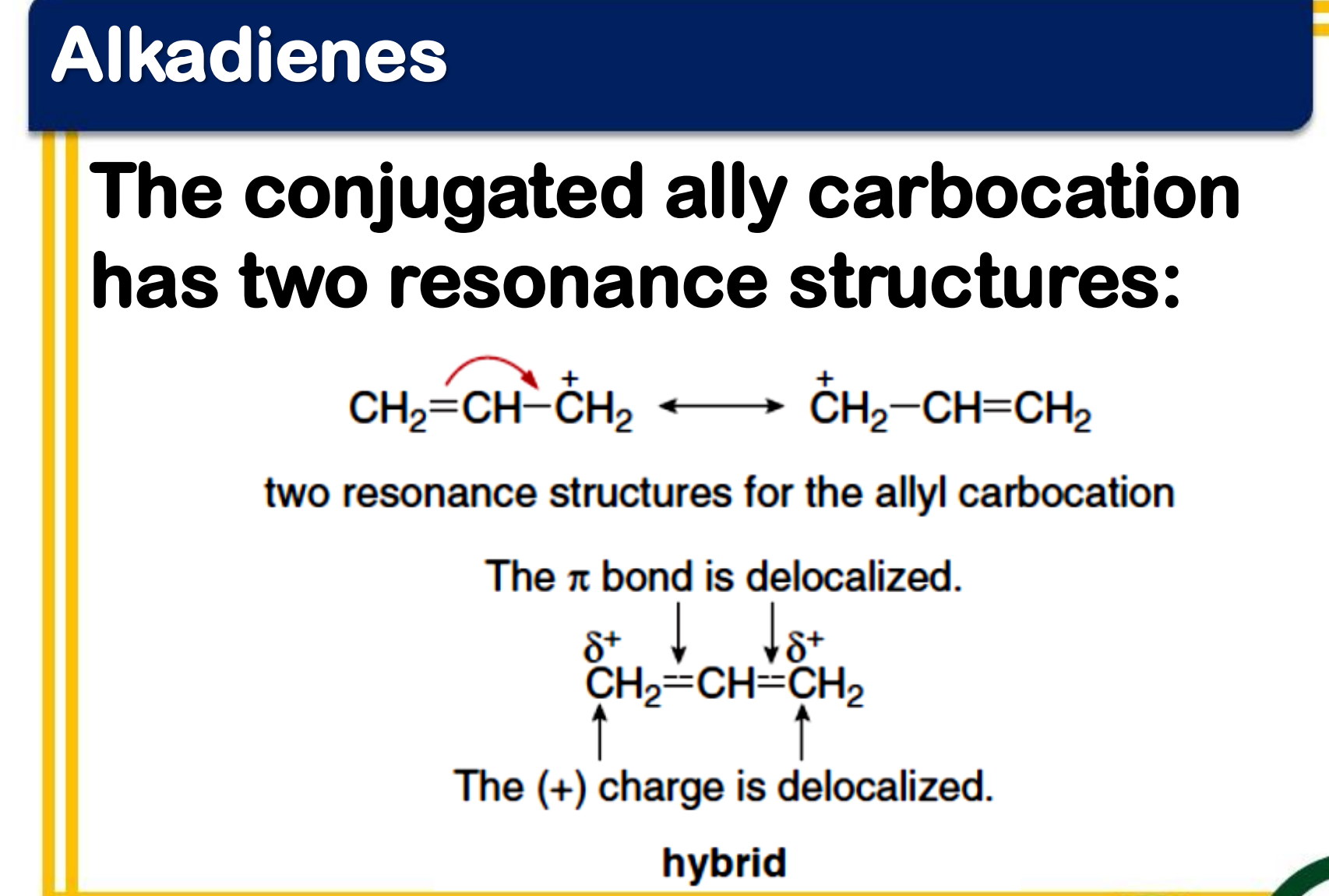
___________________
- a conjugated system
- the p orbitals of the double bond carbons each contain an electron, while the p orbital of the carbocation is empty
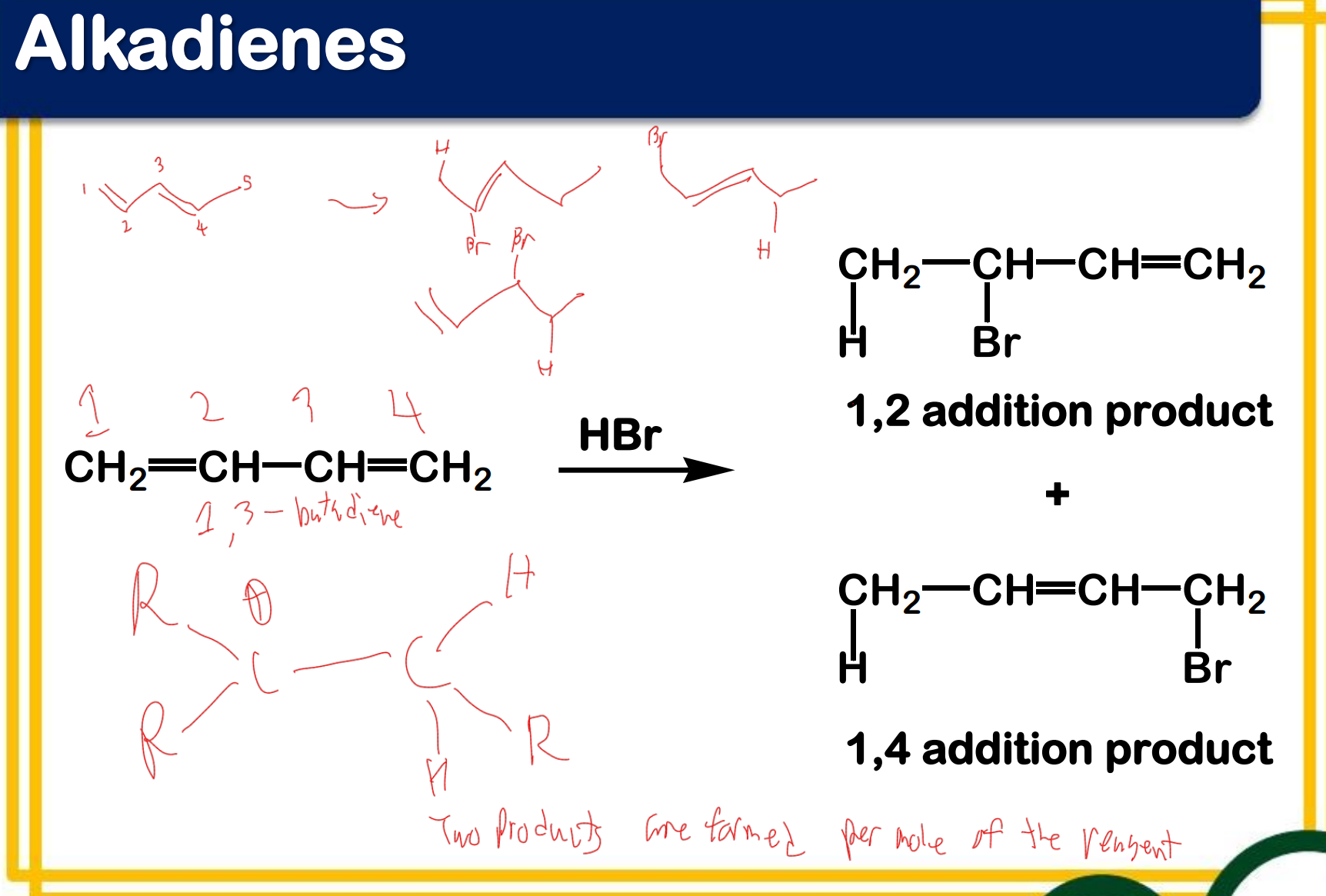
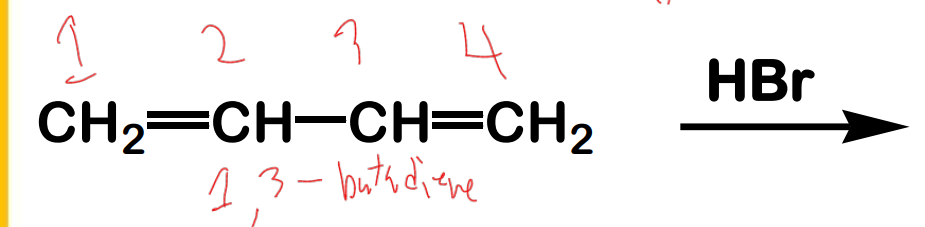
What are the products?
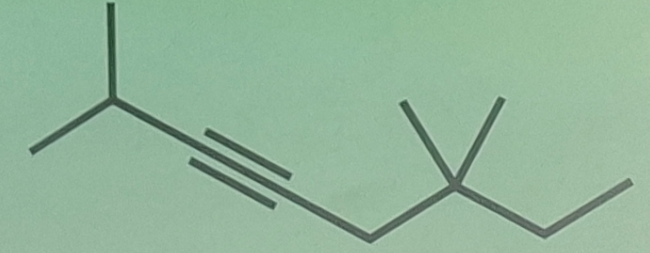
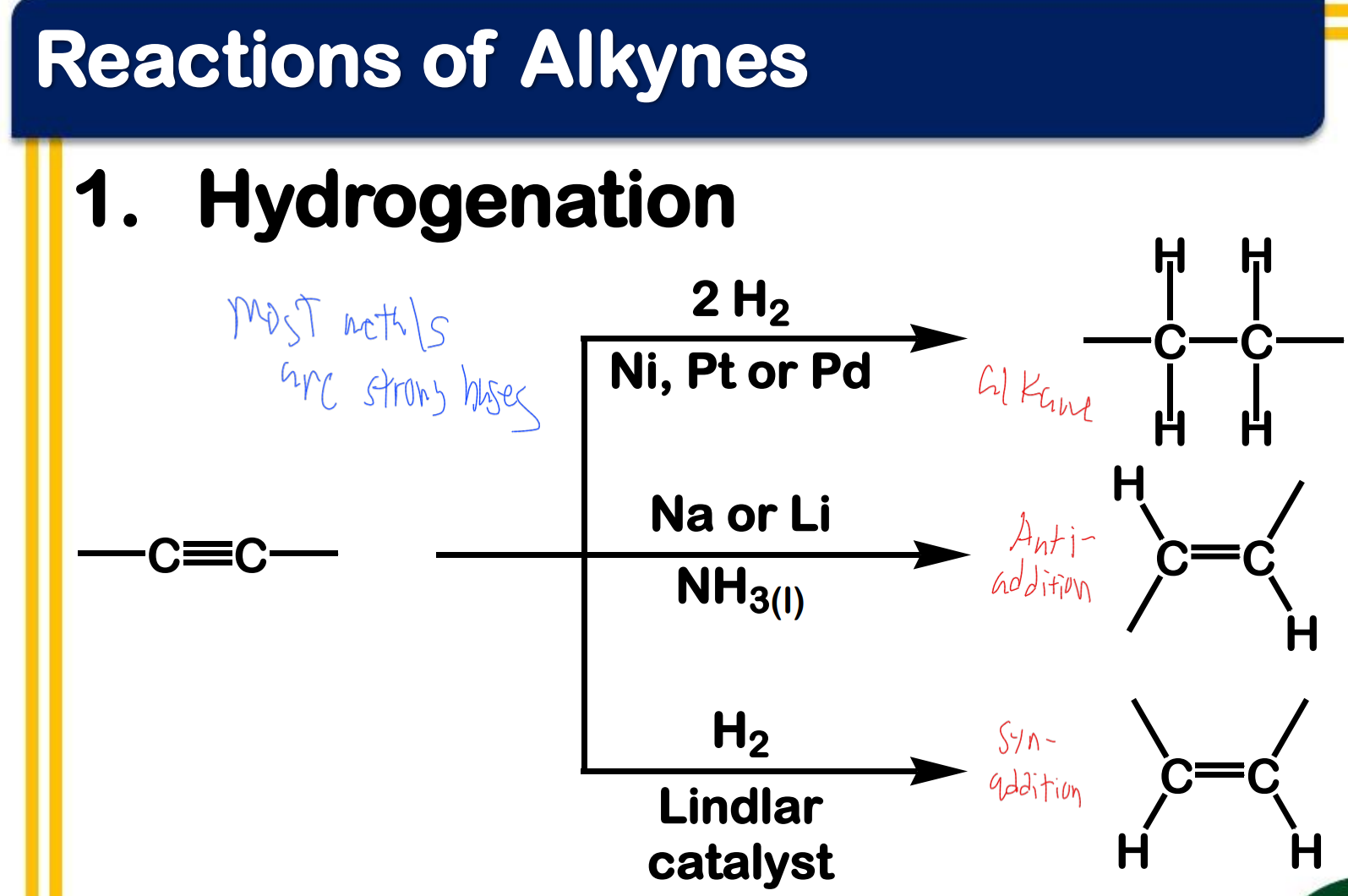
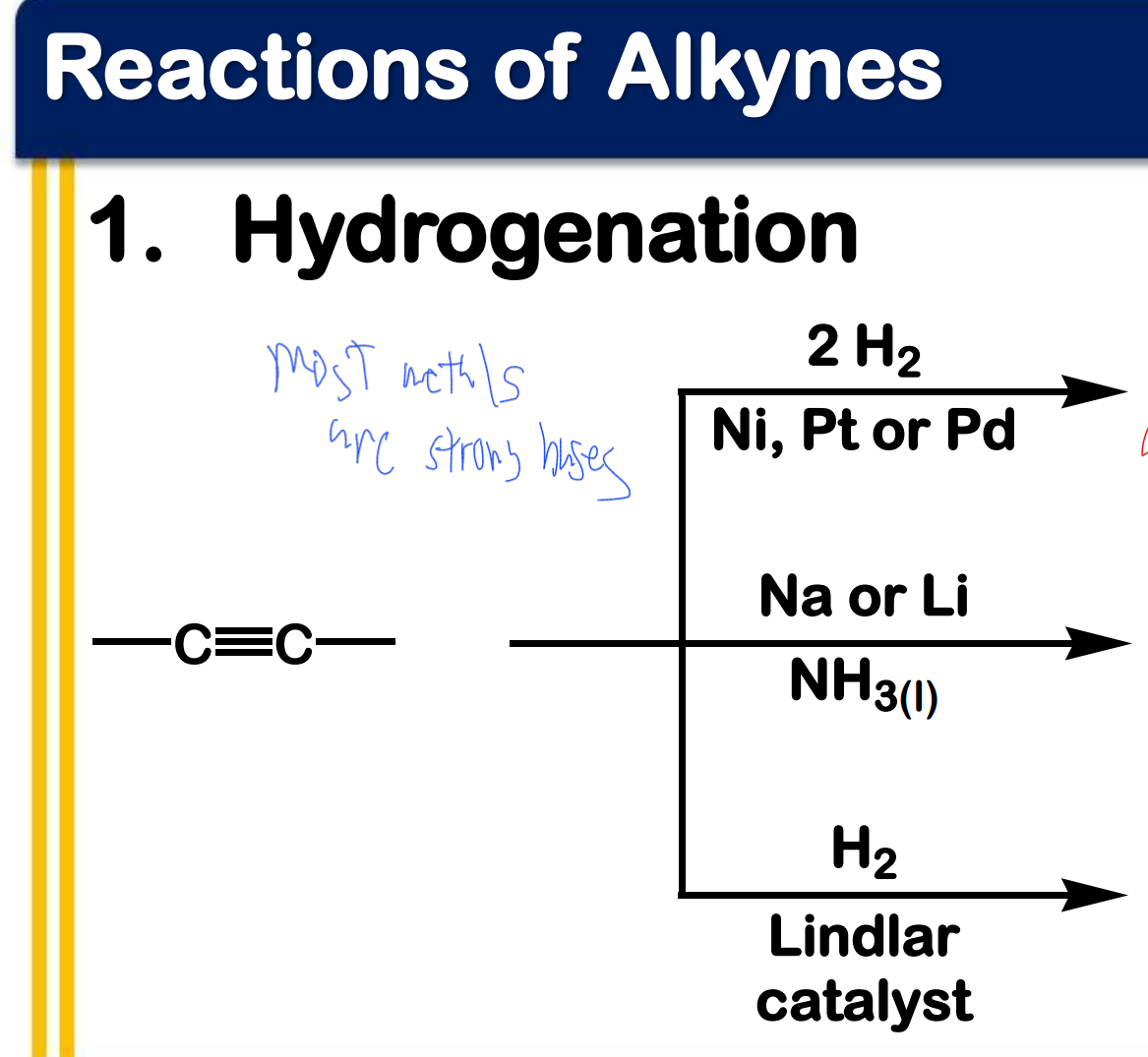
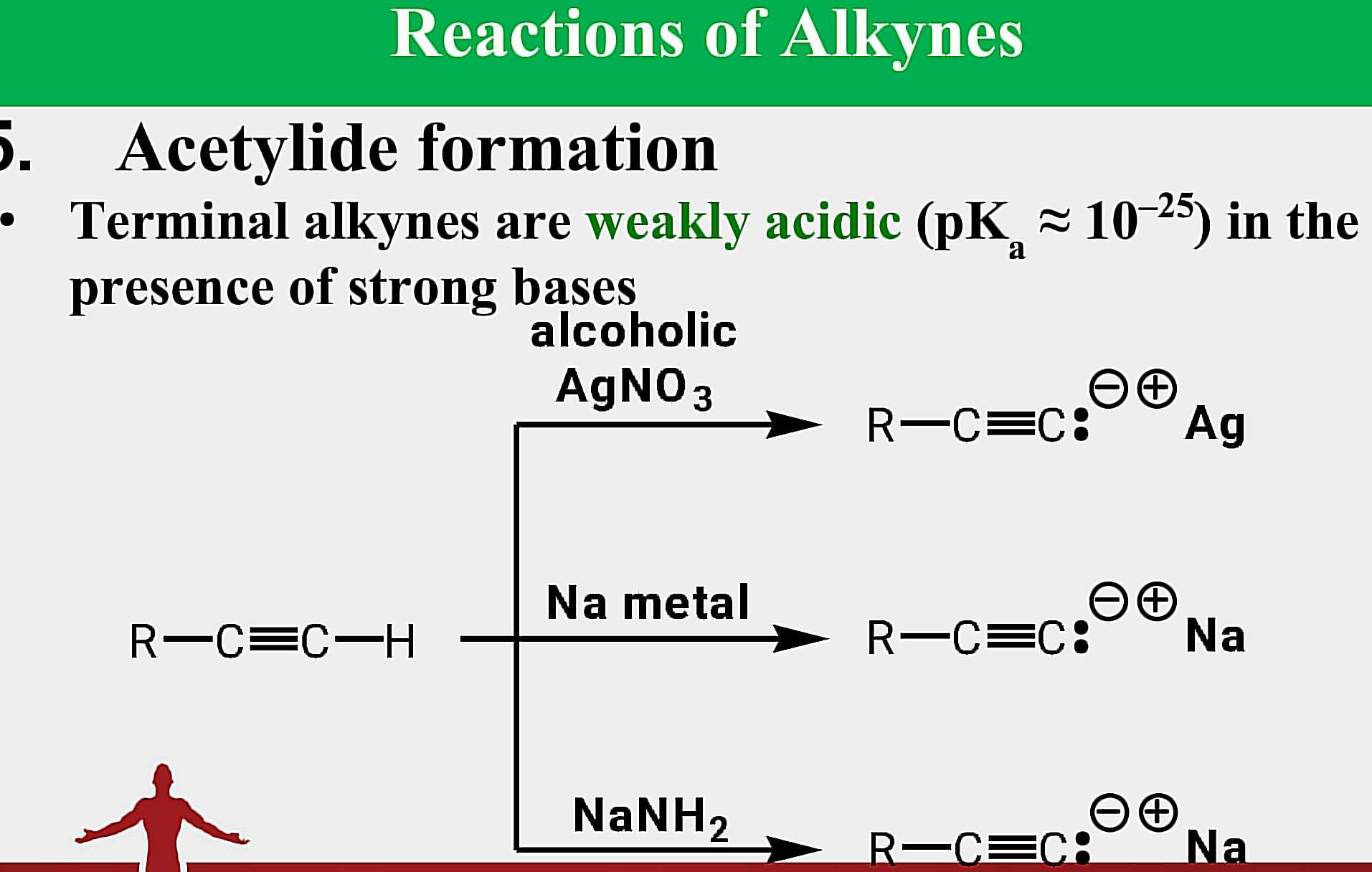
Alkynes
____________ - have C-C triple bonds as functional group
ethyne; propyne
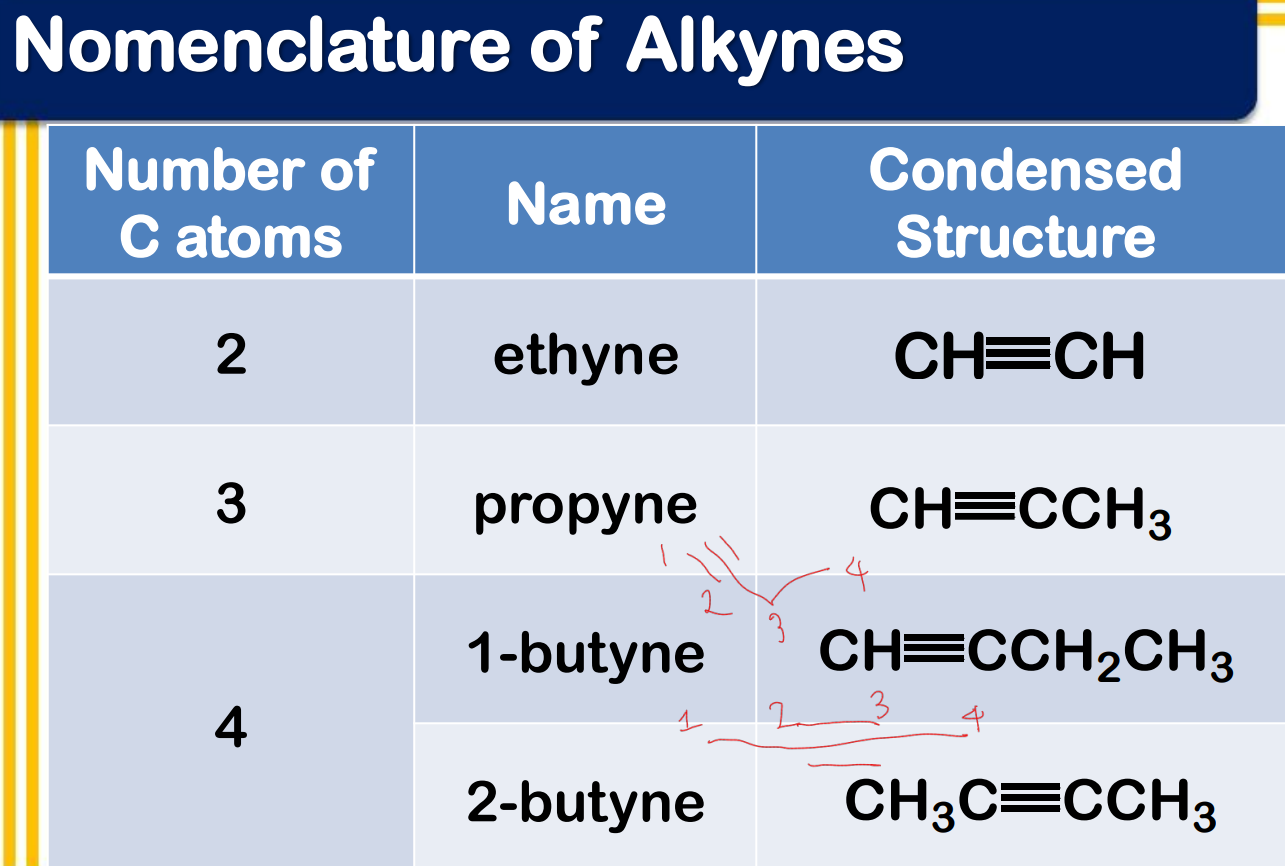

Systematic Names of Linear Alkynes
names end in –yne
except for _________ and ___________, locants are used to indicate the position of the carbon to carbon triple bond
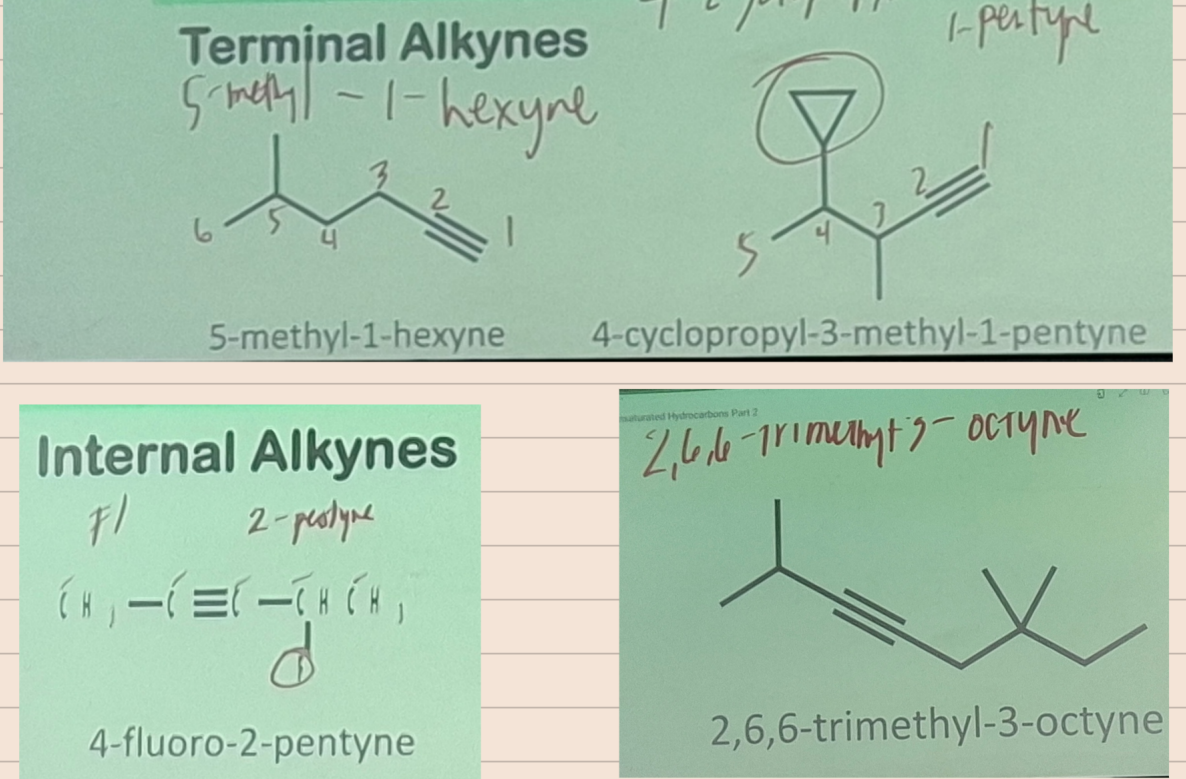

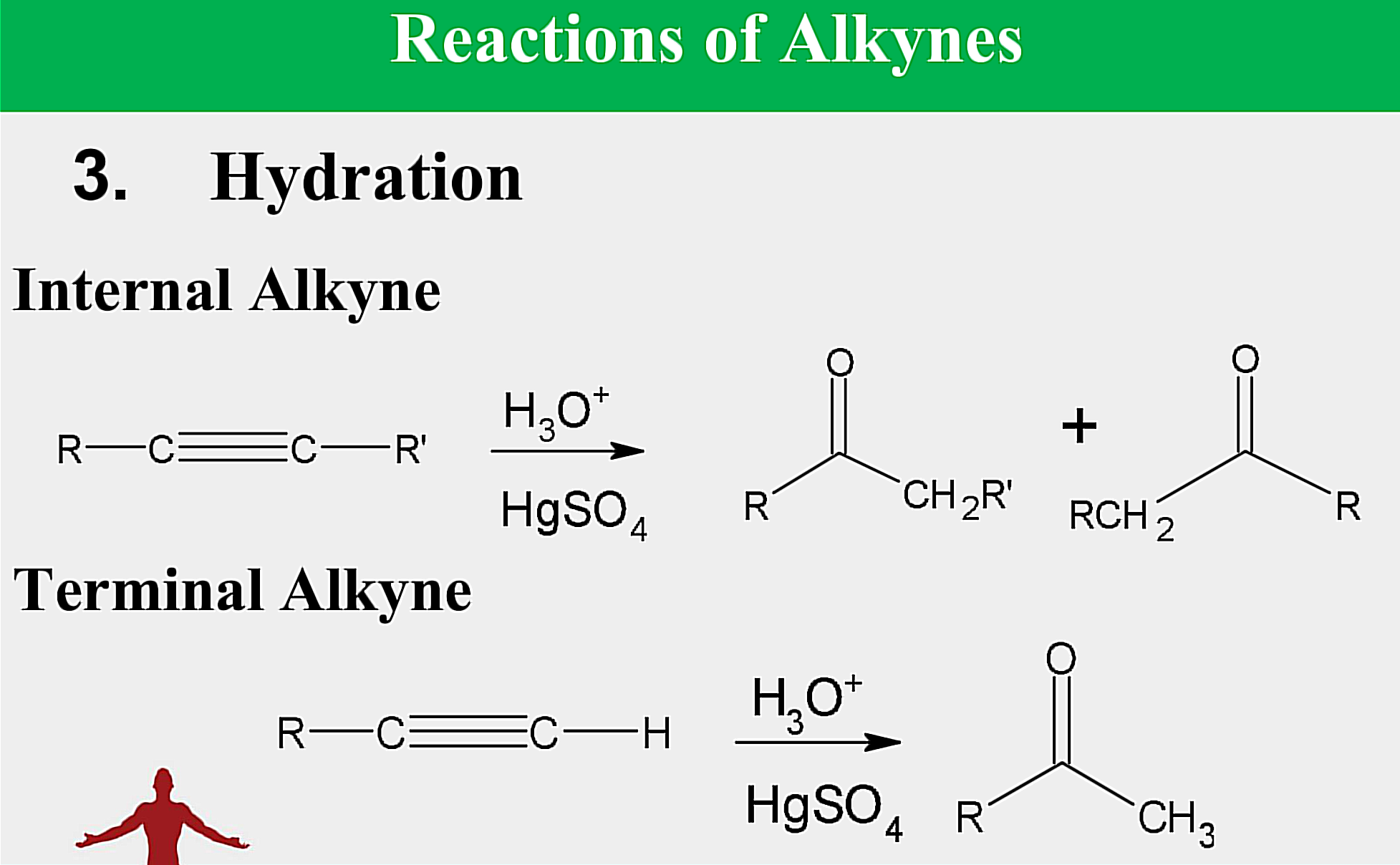
Internal Alkynes