MedChem of Agents Acting on Cell Walls & Membranes
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
Lactam
cyclic amide
Penicillin-binding Protein (PBP)
- type of transpeptidase enzyme involved in crosslinking peptidoglycan chains in the bacterial cell wall
- drug target of beta-lactam antibiotics
Siderophore
iron-chelating compound, strategy used to enhance gram (-)
Current CWM agents
- agents interfering with the synthesis of cell wall precursors starting materials
- inhibits the formation/elongation of petidoglycan chains
- agents inhibiting the cross-linking of peptidoglycan chains (PBP/transpeptidase inhibitors)
- agents used in combo with beta-lactam antibiotics
- agents acting on other cell wall & membrane targets
Agents Targeting CWM Precursors: interfere with synthesis of cell wall precursors
- clycloserine
- fosfomycin
Fosfomycin CC
epoxide or phosphonic acid (phosphoenolpyruvate analog)
Cycloserine CC
cyclic amino acid (D-alanine analog)
MOA of cycloserine
inhibits alanine racemase & D-Ala-D-Ala ligase
MOA of fosfomycin
inhibits enolpyruvate transferase
Spectrum of cycloserine and fosfomycin
both agents have borad-spectrum effects on both Gram(+) and Gram(-) microorganisms
Cycloserine activity
- can be either bactericidal or bacteriostatic
- concentration-dependent
Fosfomycin activity
bactericidal when used at therapeutic doses
ADRs of cycloserine
- nasty side-effect profile (d/t partial agonism at CNS NMDA receptors)
- includes acute psychosis & convulsions
- not really used
ADRs of fosfomycin
minimal, probably d/t single-dose regimen
T/F: Cycloserine is commonly used to treat uncomplicated UTIs
FALSE its rarely used d/t side effect profile
Uses of cycloserine and fosfomycin
- cycloserine is typically limited to second-line therapy for resistant M. tuberculosis infections
- fosfomycin usually limited to second- or third-line therapy for uncomplicated UTIs
Agents inhibiting the formation/elongation of peptidoglycan chains: Chain inhibitors
- Cyclic peptides: bacitracin
- glycopeptides/lipoglycopeptides: vancomycin, dalbavancin, oritavancin, telavancin
Chain inhibitor agents
- bacitracin
- dalbavancin
- oritavancin
- telavancin
- vancomycin
SAR of bacitracin & vancomycin
- natural products
- newer glycopeptides have lipophilic additions to improve PK profiles
MOA/PC of bacitracin
prevents dephosphorylation of lipid carrier that transfers peptidoglycan subunits to the cell wall
MOA/PC of glycopeptides
bind to D-Ala-D-Ala subunits, preventing transglycosylation & transpeptidation
Spectrum of bacitracin
primarily gram (+)
Spectrum of glycopeptides
gram (+) incl. MRSA
Activity of bacitracin
bactericidal
Activity of Glycopeptides
usually bactericidal against susceptible microorganisms ("last resort" agents)
Why does bacitracin have minimal toxicity?
d/t topical usage
Glycopeptides ROA
PO (vancomycin) and IV (vanco, dalba, ortia, tela)
When administered orally, the BA of vancomycin is less than ____________, making it ineffective for treating _______________
- less than 10%
- ineffective for treating systemic infections
ADRs of vancomycin
- anaphylaxis
- red-man syndrome
- nephrotoxicity
- ototoxicity (can be permanent)
Telavancin BBW
increased mortality in HABP/VABP pts w/ pre-existing moderate-severe renal impairment, nephrotoxicity, and embrofetal toxicity
Typical usage of bacitracin
topical wound care
What is PO vancomycin used for? What is IV vancomycin used for?
- PO for treatment of C. diff diarrhea & enterocolitis d/t S. aureus
- IV for treatment of various serious infections incl. septicemia, endocarditis, SSTIs, bone infections, LRTIs
What are dalbavancin, ortavancin, and telavancin used for?
treating ABSSSIs caused by suscpetible gram (+) organisms in adult & pediatric pts
Bacitracin Characteristics
- PC/MOA: binds to undecaprenyl pyrophosphate
- CC: (mixture of) cyclic peptides
Glycopeptides characteristics
- PC/MOA: binds to D-Ala-D-Ala subunits
- CC: glycopeptide (natural product)
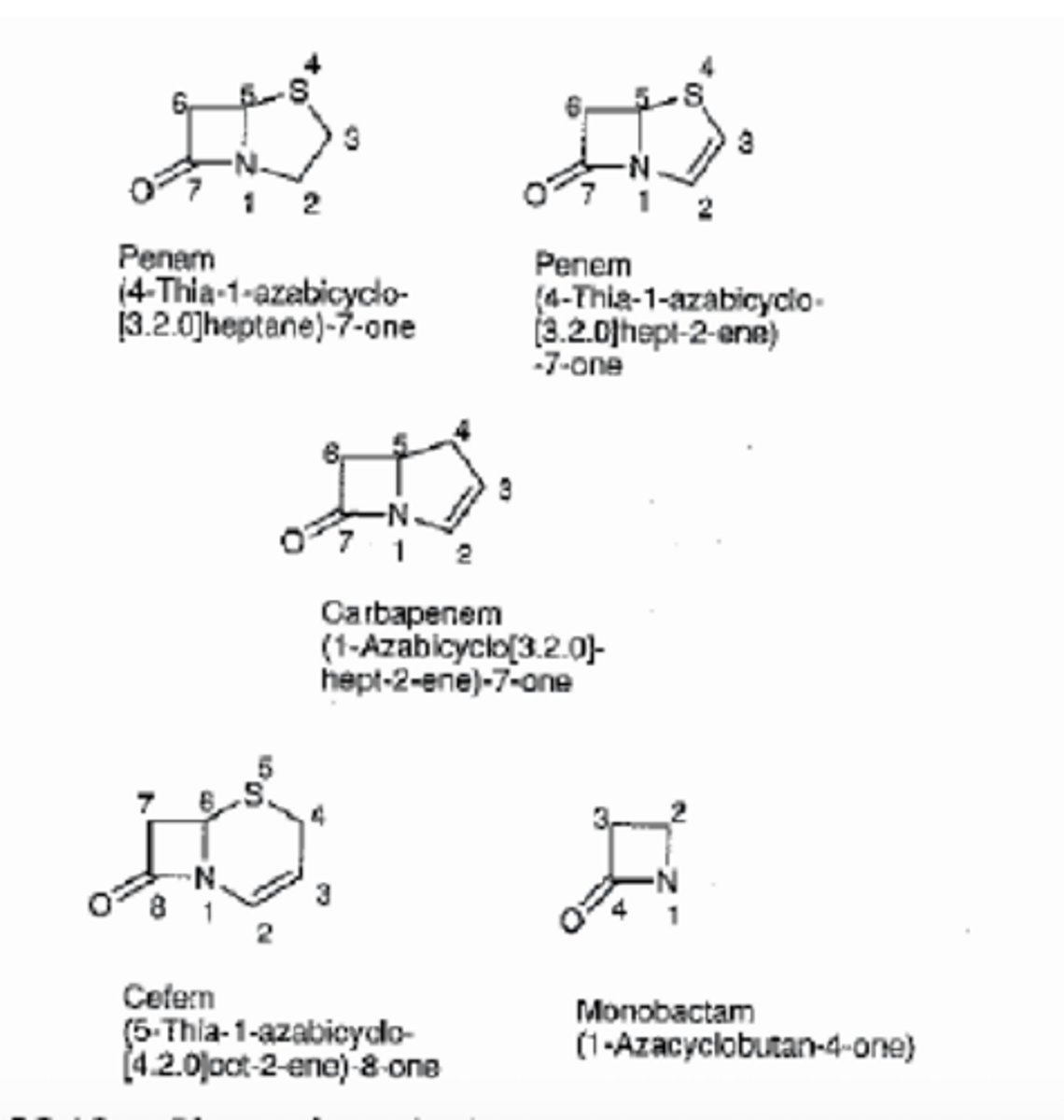
Cross-Linking Inhibitors: Inhibit cross-linking of peptidoglycan chains
- Beta-lactams, including:
- penicillins
- cephalosporins
- carbapenems & thiopenems
- monobactams
Penicillin CC
beta-lactams
SAR of penicillin: Fundamental requirements for beta-lactam antibiotic activity
- highly-constrained, fused B-lactam* structure
- carboxylic acid (or similar acidic) group at C3
- substituted amide side chain --> g-APA inactive*
Groups of penicillins
- natural (or fermentation-derived) penicillins
- anti-staphylococcal (penicillinase-resistant) penicillins
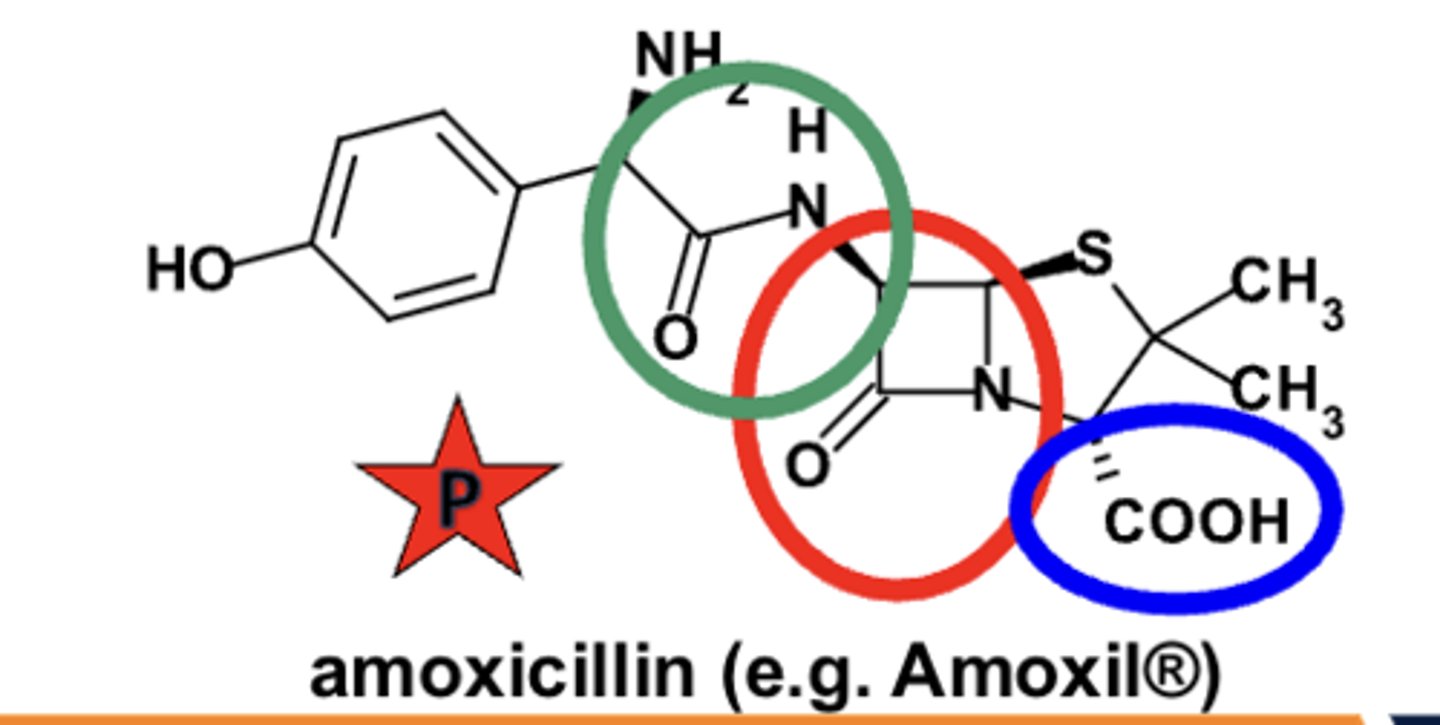
- extended-spectrum penicillins (amoxicillin)
- combinations w/beta-lactamase inhibitors
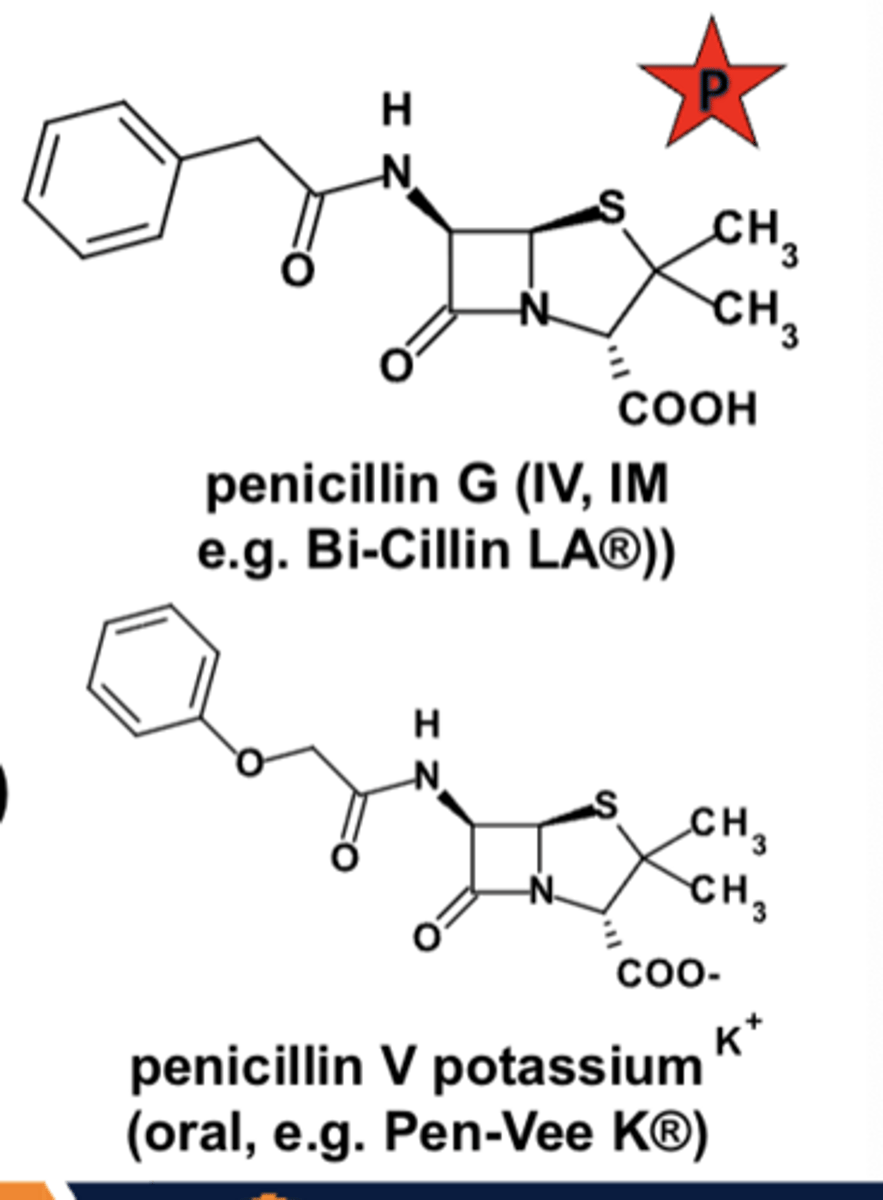
Natural (or fermentation-derived) penicillins
- PO, IV, IM (depot preps)
- penicillin G, penicillin V
- bactericidal
- spectrum: primarily non-lactamase-producing Gram(+) cocci, still used as first-line therapy
- sensitive to beta-lactamases
- ADRs primarily limited to hypersensitivity and injection-site reactions
Extended-spectrum penicillins
- PO & IV
- ureido-(piperacillin)
- amino-(ampicillin, amoxicillin)
- sensitive to beta-lactamases
- ADRs primarily limited to hypersensitivity, injection-site reactions, and GI upset
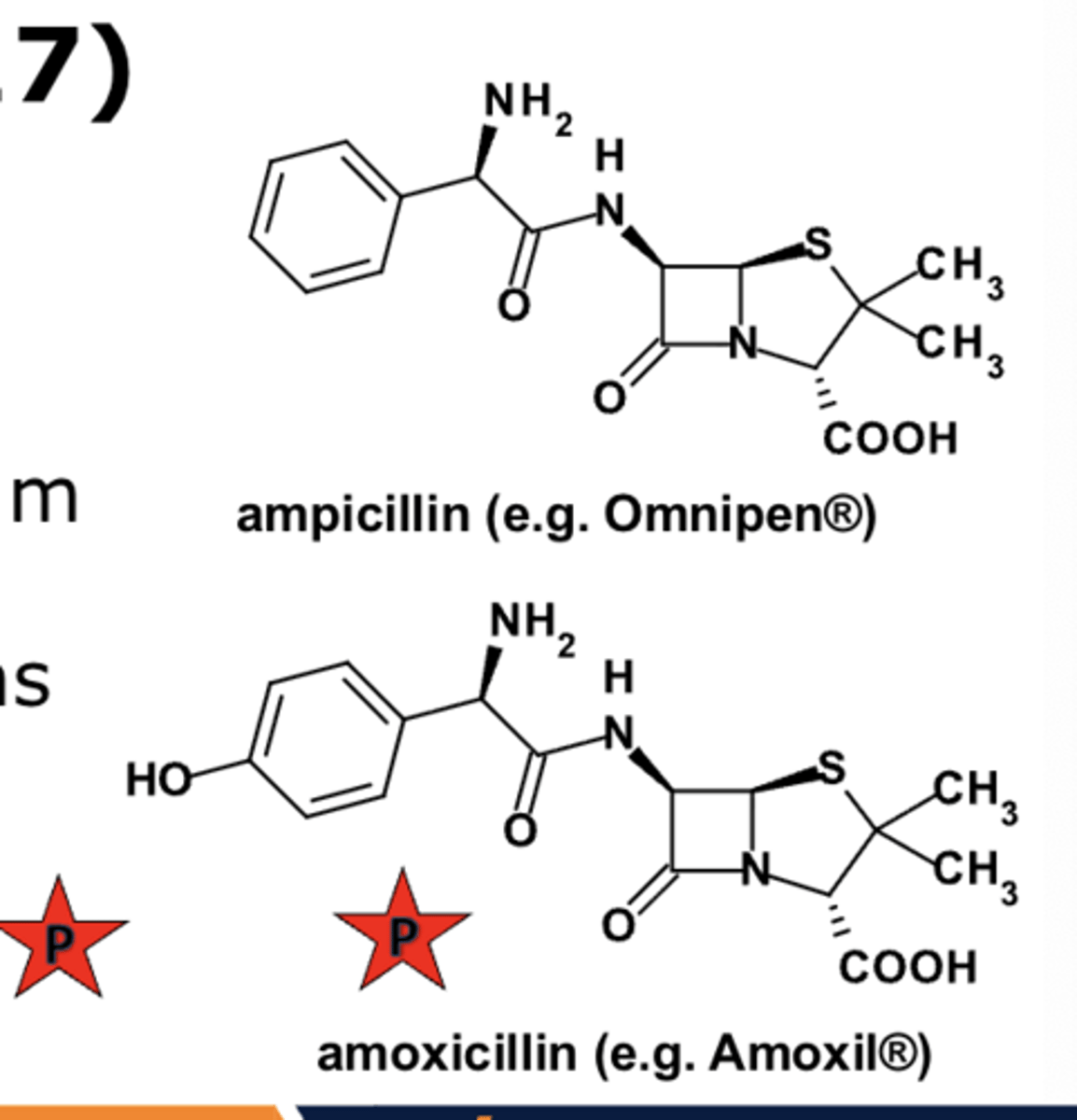
Extended-spectrum penicillins Usage: amoxicillins
primary drug of choice for the PO treatment of non-lactamase-producing infections by susceptible strains
Anti-staphylococcal penicillins
- PO (diclo) & IV (naf, oxa)
- bactericidal
- primarily used for susceptible Staph. infections
- nafcillin & the isoxazolyl penicillins (oxacillin, dicloxacillin)
- resistant to some beta-lactamases
- ADRs primarily limited to hypersensitivity and injection-site reactions
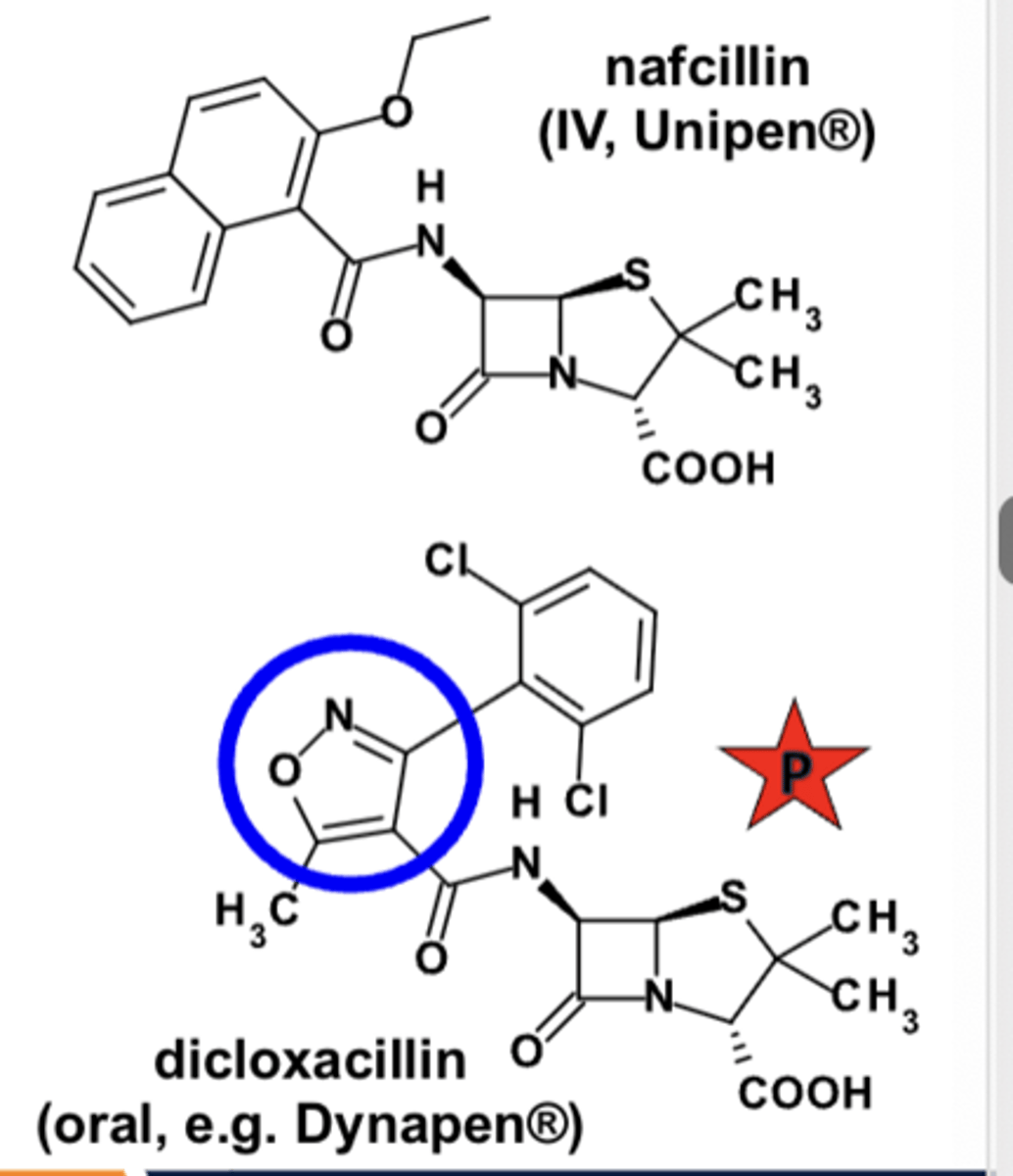
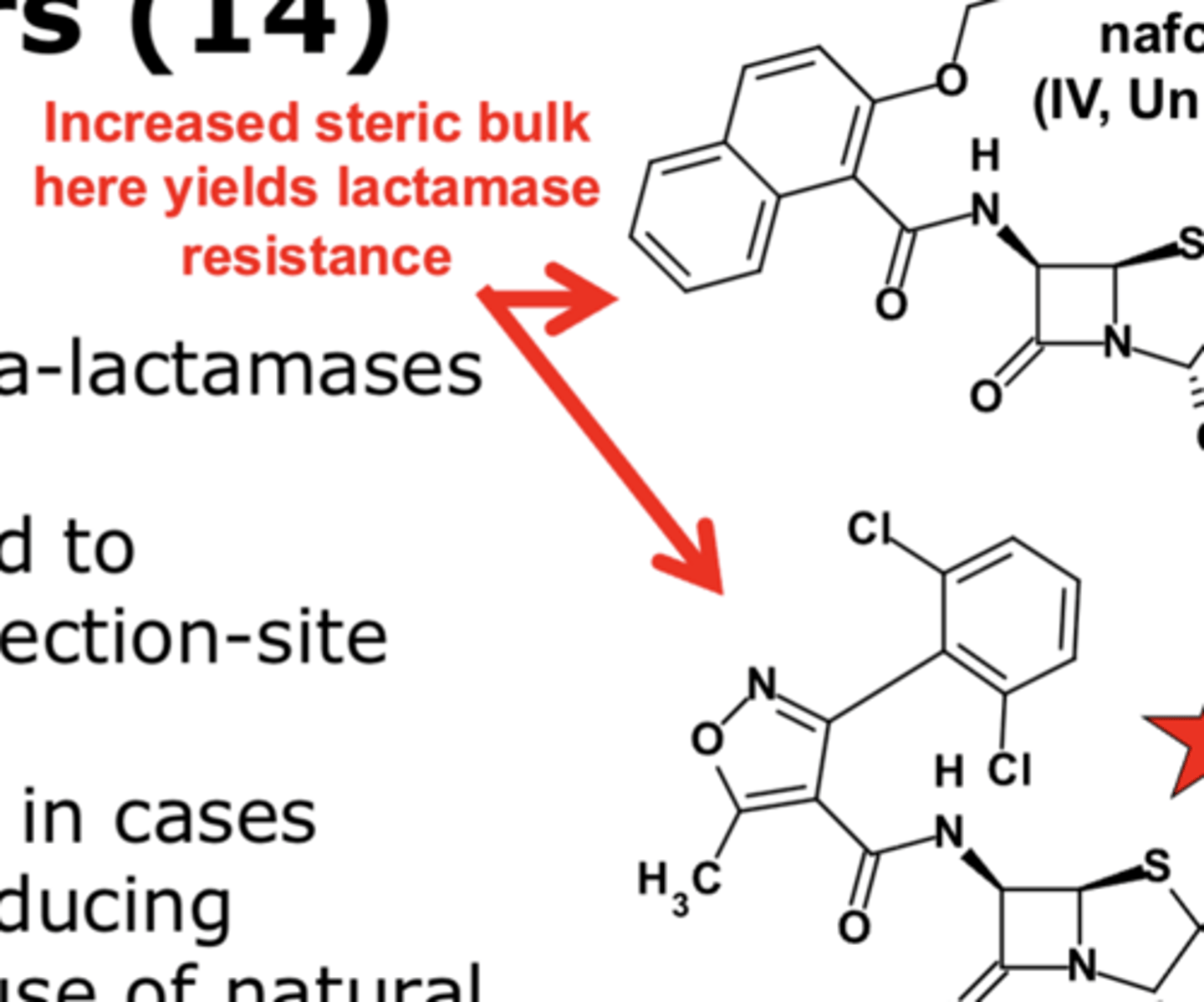
Anti-staphylococcal penicillins: What structure change yields lactamase resistance?
increased steric bulk
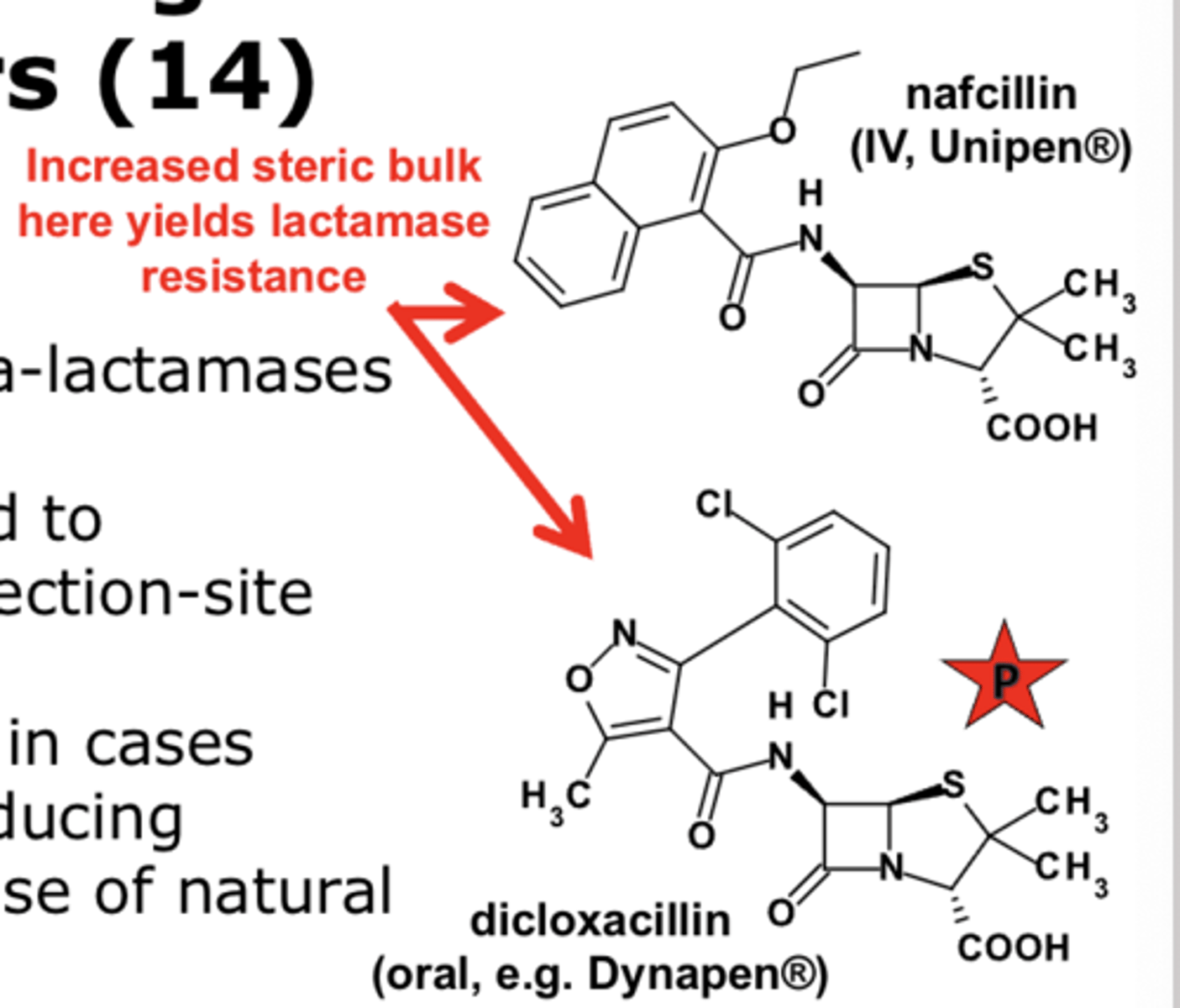
Usage of Anti-staphylococcal Penicillins
primarily used in cases where penicillinase-producing infections prevent the use of natural PCNs
T/F: Newer generation cephalosporins typically have better resistance to beta-lactamases and better Gram(-) coverage
TRUE
1st generation cephalosporins
- PO, IV, IM
- bactericidal
- primarily non-lactamase-producing Gram(+)
- sensitive to beta-lactamases
- used as second- or third-line therapy for susceptible infections
- ADRs primarily limited to hypersensitivity & injection-site reactions
- cephalexin, cefadroxil, cefazolin
1st generation cephalosporin spectrum
primarily non-lactamase-producing Gram(+)
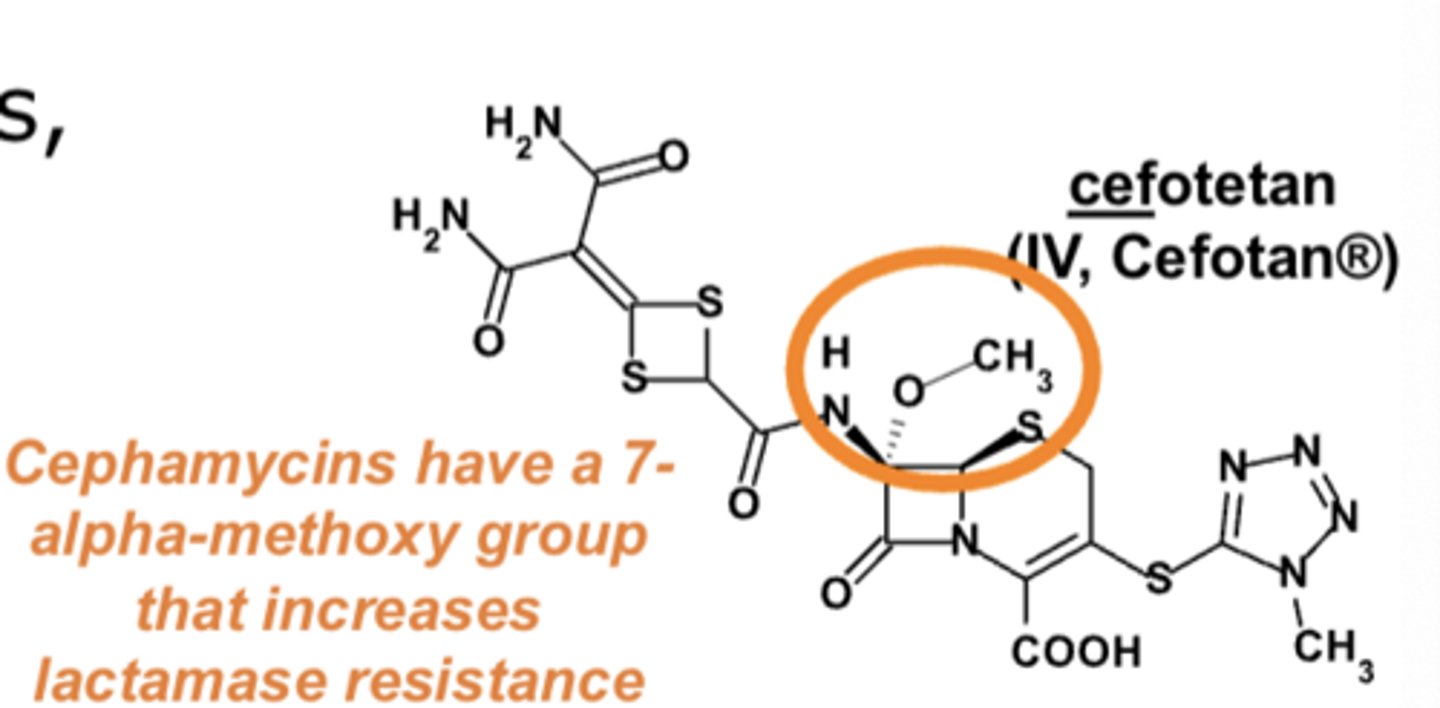
2nd generation cephalosporins
- PO, IV, IM
- bactericidal
- used as second- or third-line therapy for susceptible infections
- sensitive to some beta-lactamases
- ADRs primarily limited to hypersensitivity & injection-site reactions
- cefaclor, cefotetan, cefuroxime, cefoxitin, cefproxil
Cephamycins have a __________________ group that increases resistance
7-alpha-methoxy group
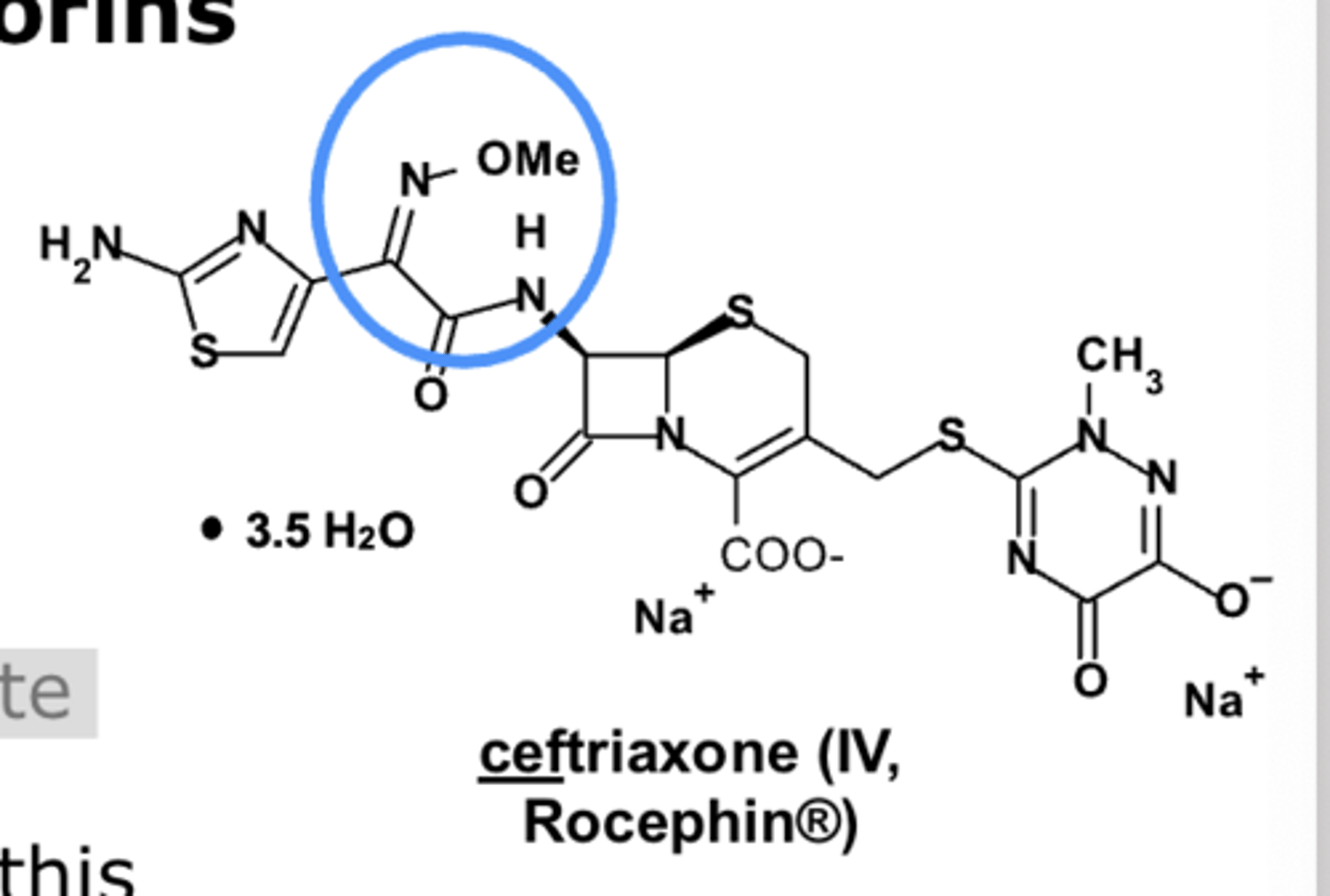
3rd-generation cephalosporins
- PO, IV, IM
- bactericidal
- sensitive to some beta-lactamases (oximes increase resistance)
- several agents are first-line therapy for different types of infections
- ADRs primarily limited to hypersensitivity & injection-site reactions
- cefpodoxime, cefixime, cefdinir, ceftriaxone, cefotaxime, ceftazidime, cefditoren
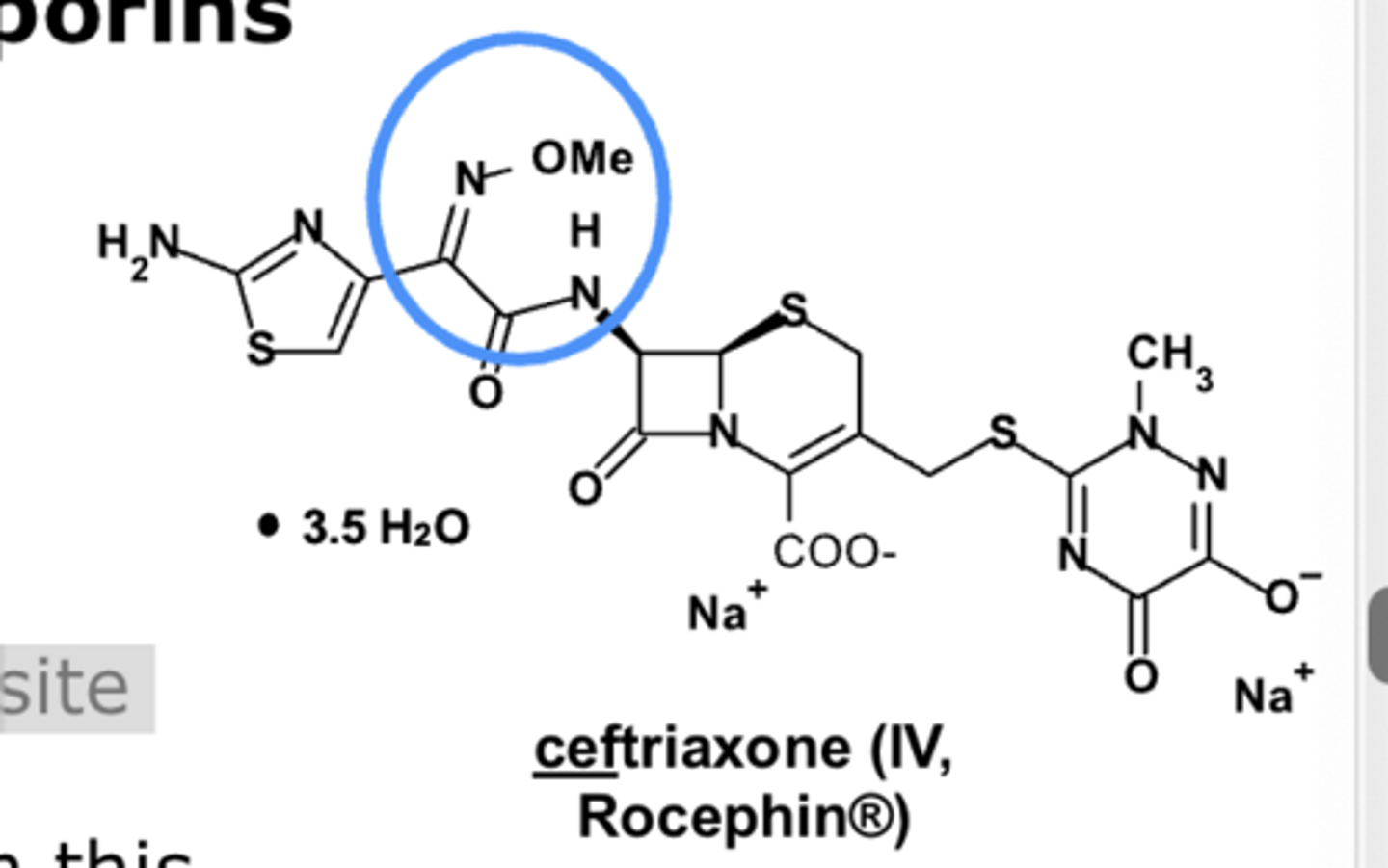
3rd-generation cephalosporins: What increases resistance to some beta-lactamases?
oximes
4th generation cephalosporins
- IV, IM
- oximes incr. resistance to beta-lactamases
- bactericidal
- potential second- or third-line therapy for infections d/t susceptible microorganisms
- ADRs primarily limited to hypersensitivity & infection-site reactions
- cefepime
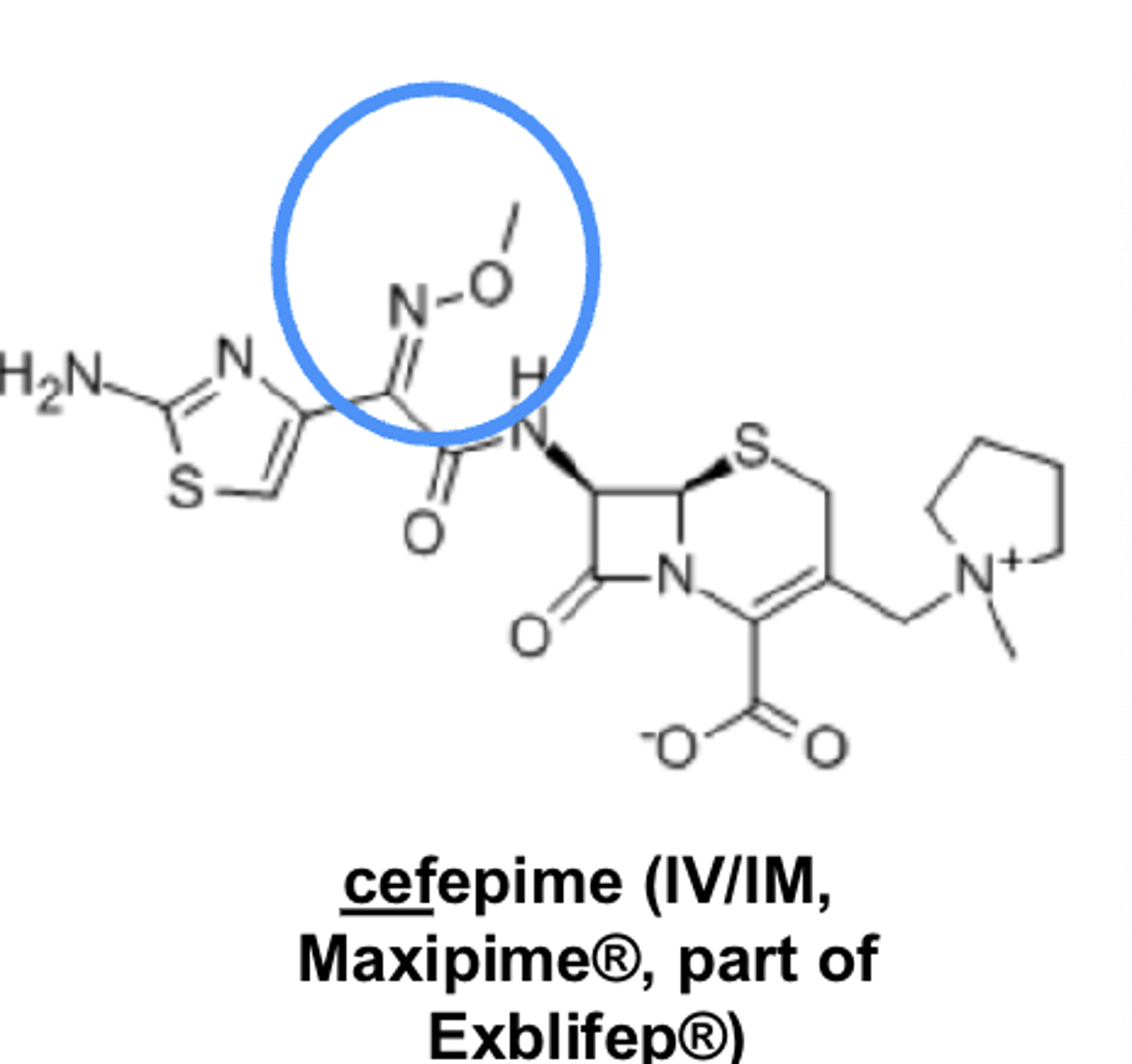
4th generation cephalosporins spectrum
similar to 3rd gen, but with better Gram(-) coverage & lactamase resistance
5th generation cephalosporins
- IV
- oximes incr. resistance to beta-lactamases
- bactericidal
- used as potential second- or third-line therapy for infections d/t susceptibel microorganisms
- ADRs primarily limited to hypersensitivity & injection-site reactions
- ceftaroline, ceftobiprole, ceftolozane
5th-generation cephalosporin spectrum
similar to 3rd & 4th gen agents, but with better Gram(-) coverage & lactamase resistance
Cefiderocol
- IV
- uses a siderophore as a "Trojan horse" to enhance penetratoin into Gram(-) bacteria
- bactericidal
- ADRs primarily infusion-site reactions, GI distress
- used for the treatment of susceptible Gram(-) infections (HABP/VABP, UTIs) in adult pts
Cefiderocol spectrum
Gram(-)
Carbapenems
- IV, NOT orally active
- BROAD
- inhibits some beta-lactamases
- bactericidal
- ADRs infusion-site reactions most common
- used for second- or third-line therapy for severe infections by susceptible organisms
Carbapenems spectrum
- among the widest spectrum of activity
- ULTRA-BROAD
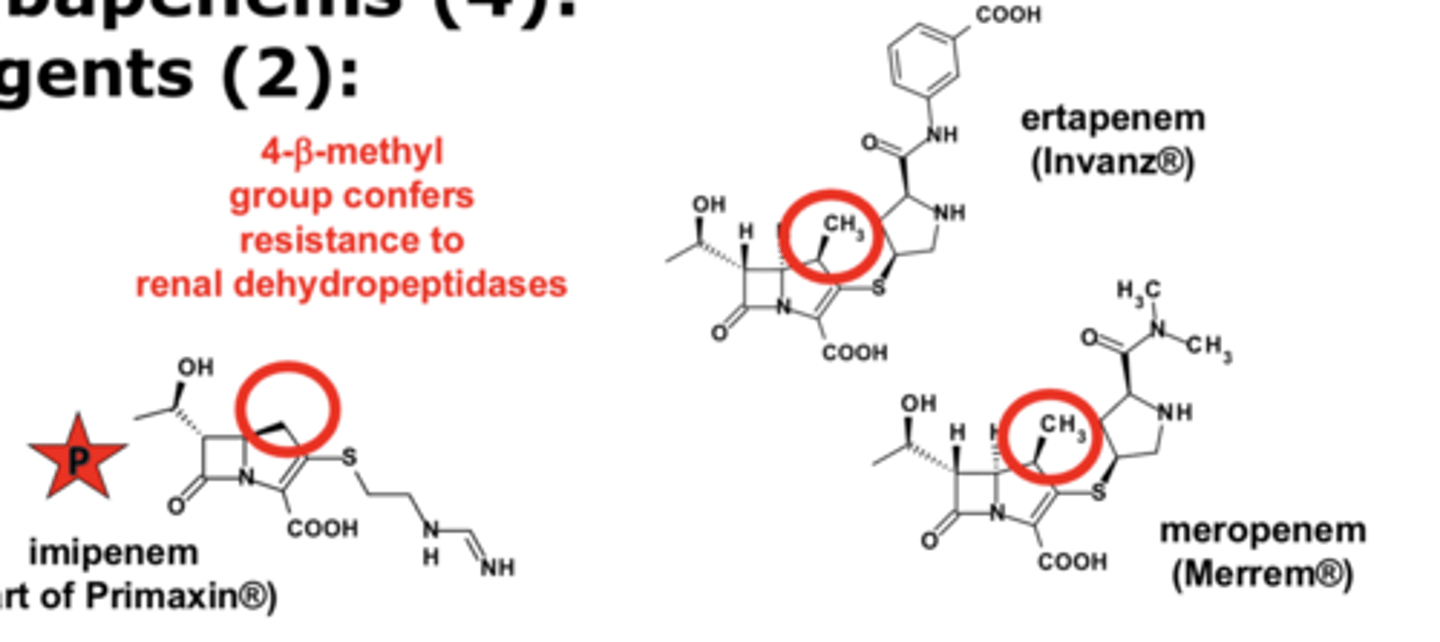
Imipenem is deactivated by renal dehydropeptides, so it is used in combination with....
cilastatin
1st generation carbapenem
imipenem
2nd generation carbapenem
- meropenem
- ertapenem
SAR of carbapenems: What conders resistance to renal dehydropeptidases?
4-B-methyl group confers resistance to renal dehydropeptidases
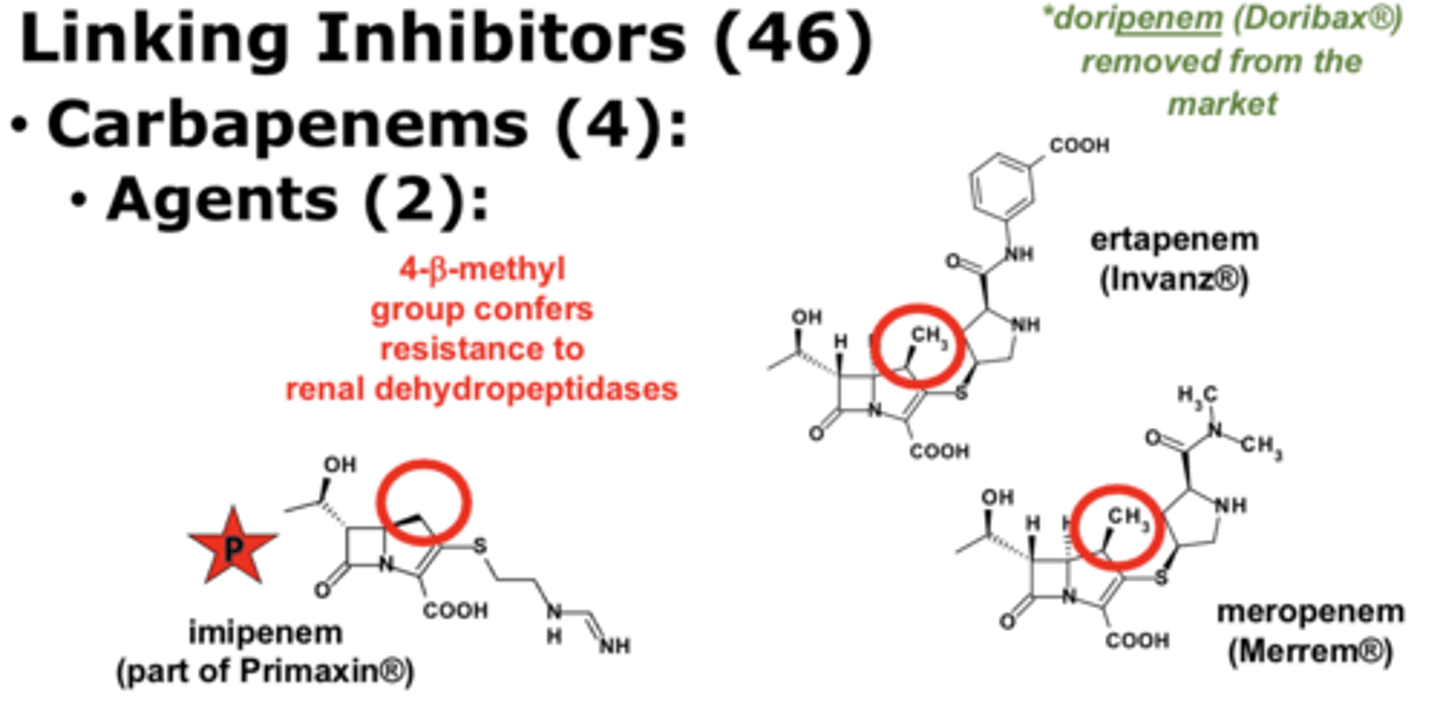
Monobactams
- IV, IM, or Inh.
- aztreonam
- bactericidal
- used as possible second- or third-line option for severe infections by susceptible strains
- ADRs minimal, but primarily limited to ROA issues
- have good CNS penetration
Monobactams are resistnat to __________________, but susceptibel to _________________________
- beta-lactamases
- hydrolysis by BSLs and ESBLs
Beta-Lactamase characteristics
- PC/MOA: PBP inhibitor
- CC: beta-lactam with either penam core, cefem core, carbapenem core, monobactam core
Beta-lactamase Inhibitors
- avibactam
- vlavulanic acid
- durlobactam
- enmetazobactam
- relebactam
- sulbactam
- tazobactam
- vaborbactam
Beta-lactam-based Beta-lactamase Inhibitors
- clavulanic acid
- enmetazobactam
- sulbactam
- tazobactam
Diazabicyclooctane-based beta-lactamase Inhibitors
- avibactam
- durlobactam
- relebactam
Boronic-acid-based beta-lactamase Inhibitors
vaborbactam
MOA of beta-lactamase inhibitors
- beta-lactams- covalent irreversible inhibition
- boronic acids, diazabicyclooctanes- covalent reversible inhibitions
Beta-lactamase Inhibitors activity
- designed to slow metabolism of beta-lactam-based antibiotics
- may have some antibiotic (PBP-inhibiting) effects on their own (sulbactam)
ADRs of beta-lactamase inhibitors
GI upset (N/D) common
Beta-lactamase inhibitor characteristics
- PC/MOA: beta-lactamase inhibitor
- CC: beta-lactam
Agents that act on the cell membrane
- lipopeptides: daptomycin
- polymyxins: polymyxin B, colistimethate
- both are bactericidal
MOA of daptomycin
causes membrane depolarization & blocks cell wall synthesis
MOA of polymyxins
causes membrane disruption
Spectrum of daptomycin
primarily on Gram(+)
Spectrum of polymyxins
primarily on Gram(-)
Daptomycin
- IV
- ADRs incl. possible myopathy
- used as possible second-line therapy for various infections, especially in cases of vancomycin resistance
Polymyxins
- ophthalmic, otic, topical
- ADRs minimal d/t ROA
- used topically (in combo) for wound care; via opthalmic & otic routes for eye & ear infections
Identify the major limitation associated with the systemic use of many antibiotics based on cyclic peptides
- systemic toxicity
- limited BA (PO vanco BA <10%)
Identify the chemical features of beta-lactam antibiotics that resemble the D-Ala-D-Ala dipeptide
- beta-lactam ring
- carboxylic acid group
- spatial arrangement
- substituted side chain
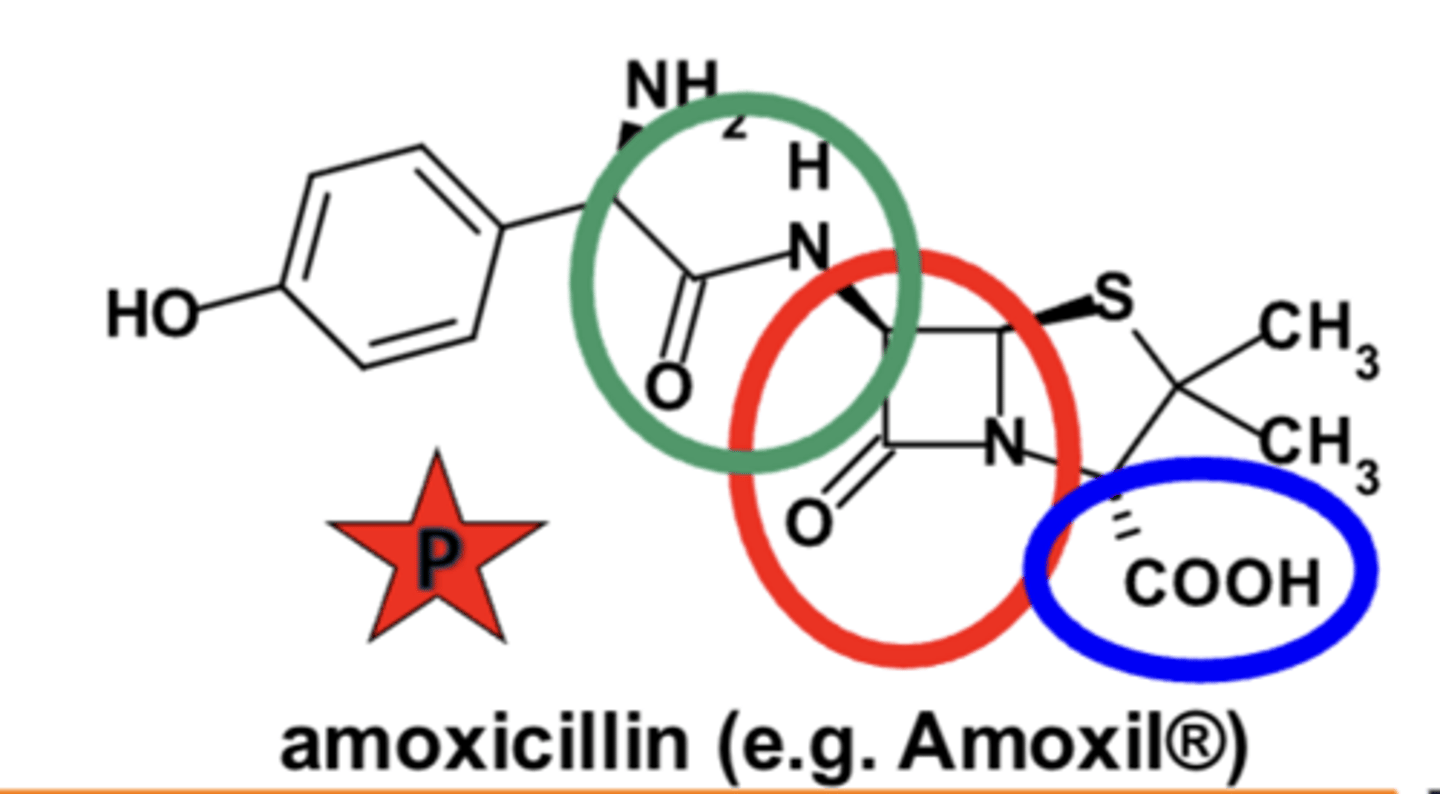
Briefly explain the major SAR points of the beta-lactam antibiotics
• a highly-constrained, fused β-lactam* structure
• carboxylic acid (or similar acidic) group at C3
• substituted amide side chain --> 6-APA inactive*Briefly
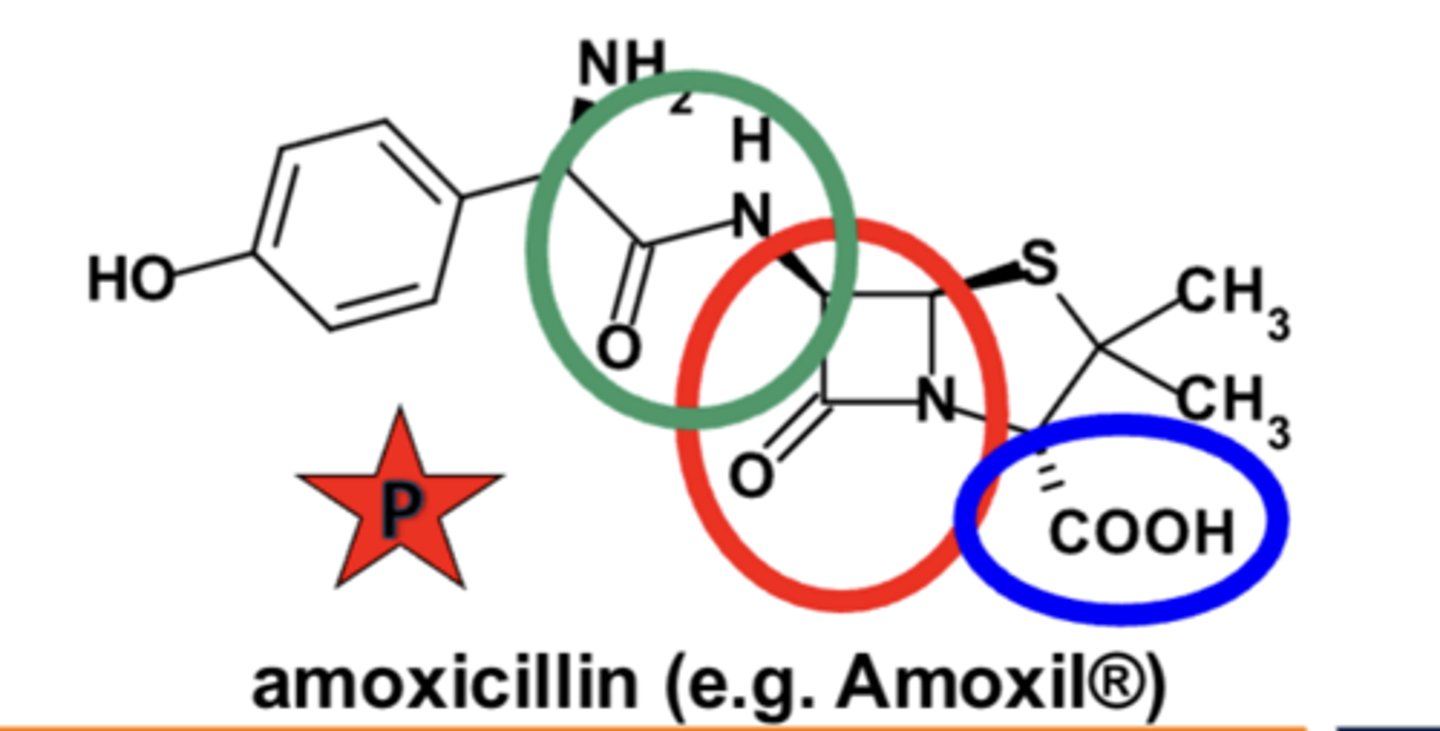
Why are beta-lactam antibiotics not typically stored or shipped as aqueous solutions?
- beta-lactam hydrolysis
- water reacts w/ beta-lactam and opens ring. once water is added, breakdown starts
What is the structural feature in anti-staphylococcal penicillins that confers resistance to some beta-lactamases?
increased steric bulk in the amide side chain
What is the structural feature in 2nd- generation CARBAPENEMS that confers resistance to renal dehydropeptidases?
4-β-methyl group confers resistance to renal dehydropeptidases
The different β-lactam cores