Organic Chemistry
1/74
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
What is a hydrocarbon?
compound containing only hydrogen and carbon atoms
What is a general formula?
chemical formula for series of compounds that differ from each other by constant unit, CnH2n
What does the structural formulae show?
shows atoms joined to each carbon atom, CH3CH2CH3 (propane)
What is the empirical formulae?
simplest ratio for atoms of different elements in compound
What is the molecular formulae?
actual number of atoms of different elements in one molecule of compound
What is a homologous series?
family of compounds with similar chemical properties, formulae differs by CH2, alkanes
What is a functional group?
determines properties and reactions of molecule, C=C (alkenes)
What is an isomer?
compounds with same molecular formulae but different structural formulae
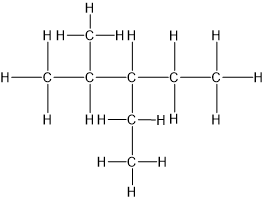
3-ethyl-2-methylpentane
What can you find out using the molecular formula?
if hydrocarbon, alkane, alkene or alcohol
What can you not find out using the molecular formula?
atom arrangement (branches, length or number), structural and displayed formula
What can you find out using the structural and displayed formula?
how atoms are arranged in molecule
What are the different classifications of reactions of organic compounds?
addition, substitution, combustion
What is crude oil?
mixture of hydrocarbons
Describe how crude oil is industrially processed by fractional distillation
fractional distillation, heated, vapour enters lower part of column, temperature gradient up column, vapour rises up column until it condenses at height where T lower than b.p., each fraction removed and collected at different heights
What are each of the fractions from top to bottom?
refinery gases, gasoline, kerosene, diesel, fuel oil, bitumen
Uses of Refinery Gases
fuel for heating or cooking
Uses of Gasoline
fuel for cars or vehicles, petrol
Uses of Kerosene (paraffin)
aeroplane fuel, fuel for heating
Uses of Diesel
fuel for vehicles (diesel engines)
Uses of Fuel oil
fuel for ships
Uses of Bitumen
road surfacing
How do the properties of the fractions change as the boiling range gets higher?
darker, more viscous, less flammable
What is fuel?
substance that releases energy when burned
Complete Combustion of Hydrocarbons
hydrocarbon + oxygen -> water vapour + carbon dioxide
Incomplete Combustion of Hydrocarbons
hydrocarbon + oxygen → water vapour + carbon monoxide
Why is carbon monoxide bad for humans?
toxic, poisonous, reduces capacity of blood to carry oxygen
Nitrogen and Oxygen in Car Engines
nitrogen and oxygen in air react at high temperatures in car engine, form oxides of nitrogen, dissolves in water, forms nitric acid
Combustion and Formation of Sulfur Dioxide
sulfur in fuel reacts with oxygen, forms sulfur dioxide, dissolves in water, forms sulfuric acid
Why is acid rain dangerous?
kills fish, damages plants, corrodes limestone or marble buildings or statues
What is catalytic cracking?
thermal decomposition of long-chain hydrocarbons into short-chain hydrocarbons using silica as catalyst at 600-700°C
Why is cracking necessary?
high demand for short-chain alkanes because more flammable, used for fuel, produces alkenes to make polymers because unsaturated, high supply of long-chain hydrocarbons
What is the general formulae for alkanes?
CnH2n+2
What are alkanes?
saturated hydrocarbons, only contain carbon-carbon single bonds
Structural and Molecular Formulae for Alkanes (Methane and Ethane)
CH4, CH4, C2H6, CH3CH3
Reactions of Alkanes and Halogens (methane and bromide)
substitution reaction, UV light, produces bromomethane and hydrogen bromide
What are alkenes?
unsaturated hydrocarbons, contain carbon-carbon double bond
What is the functional group, structural and general formulae of alkenes (ethene)?
C=C, CH2=CH2, CnH2n
Molecular and Structural of Alkenes (ethene)
C2H4, CH2=CH2
Reactions of Alkenes with Bromine (ethene)
addition reaction, produces colourless dibromoethane
How can you distinguish between alkenes and alkanes?
add bromine water, with alkanes bromine water stays yellow, with alkenes bromine water turns from yellow to colourless
Functional Group of Alcohols
-OH
Names, Molecular and Structural Formulae of Alcohols (meth)
methanol, CH4O, CH3OH
Complete Combustion of Ethanol
ethanol + oxygen -> carbon dioxide + water vapour
Microbial Oxidation of Ethanol
oxygen in air reacts with ethanol, forms ethanoic acid (using microorganisms, bacteria or yeast)
Heating with Potassium Dichromate (VI) in Dilute Sulfuric Acid
add oxidising agent [O] of potassium dichromate (VI) (K2Cr2O7) and dilute sulfuric acid, heat under reflux (boiling point) in water bath, ethanol present if solution turns from orange to green
Displayed Formulae of Heating with Potassium Dichromate (VI) in Dilute Sulfuric Acid

Fermentation to Produce Ethanol
fermentation of glucose, absense of air, 30°C, zymase (enzyme in yeast)
Hydration of Ethene
- Ethene from crude oil reacts with steam by hydration to form ethanol.
- A temperature of 300°C, 60-70 atmospheres of pressure and the catalyst of phosphoric acid (H₃PO₄) is needed.
- CH₂=CH₂(g) + H₂O (g) → CH₃CH₂OH (g)
Advantages of Fermentation
- It is a batch process, which uses lower pressure and temperature so less energy is needed.
- The glucose from sugar cane is a natural resource, and therefore renewable.
- Yeast is a natural resource, wheras phosphoric acid (in hydration of ethene) is a manufactured catalyst.
Disadvantages of Fermentation
- Batch processes are slower and less efficient.
- Continuous process (in hydration of ethene) produce pure ethanol that does not need to be purified.
- Growing sugar cane takes up land that can be used to grow food crops.
Why does fermentation need to be anaerobic?
- It needs to anaerboic because in aerobic conditions, ethanol is not produced, only carbon dioxide and water will form.
- The ethanol produced will be oxidised, forming ethanoic acid.
Why does the temperature need to be between 25 and 40°C for fermentation?
- If the temperature goes above 40°C, enzymes become denatured, causing fermentation to slow down or stop.
- At a lower temperature, the reaction is too slow.
Functional Group of Carboxylic Acids
- Carboxylic acids contain the functional group -COOH.
- (Notion)
Structural, Displayed Formulae and Names for Carboxylic Acids
(Notion)
Reaction of Carboxylic Acid with Magesium
Dilute ethanoic acid reacts with magnesium with a lot of fizzing, producing a magnesium ethanoate salt which is soluble in water and therefore colourless.
Mg (s) + 2CH₃COOH (aq) → (CH₃COO)₂Mg (aq) + H₂ (g)
Reaction of Carboxylic Acids with Other Metals
- Sodium is dangerously reactive, zinc produced bubbles of H₂ more slowly than it does with HCl.
- Metals below hydrogen in reactivity series won’t react.
Reactions of Carboxylic Acids with Metal Carbonates
- Metal carbonates react with carboxylic acids (with a lot of fizzing) to produce a colourless solution of metal ethanoate, carbon dioxide and water.
- Na₂CO3 (s) + 2CH₃COOH (aq) → 2CH₃COONa (aq) + CO₂ (g)+ H₂O (l)
What is vinegar?
Vinegar is an aqueous solution containing ethanoic acid.
Function Group of Esters
- Esters have the functional group of -COO-.
- (Notion)
Production of Ethyl Ethanoate
- Ethyl ethanoate is produced by esterification, where ethanoic acid, ethanl and a few drops of concentrated sulfuric acid (as the catalyst) are heated.
- CH₃COOH (l) + CH₃CH₂OH (l) ⇌ CH₃COOCH₂CH₃ (l) + H₂O (l)
Structural and Displayed Formulae of Ethyl Ethanoate
Structural Formulae: CH₃COOCH₂CH₃
Displayed Formulae: (Notion)
Structural, Displayed Formulae and Names of Esters
(Notion)
Uses of Esters
Esters are used in food flavourings and perfumes because they are volatile liquids (turn to vapour easily) with distinctive smells.
How to prepare an ester?
1. Put 1cm³ of ethanoic acid and 1cm³ of ethanol into a boiling tube.
2. Mix and add a few drops of concentrated sulfuric acid.
3. Place the boiling tube in a beaker of hot water at about 80°C for 5 minutes.
4. Allow the contents of the tube to cool.
5. When cool, pour the mixture into a beaker half-full of 0.5 mol/dm³ sodium carbonate solution.
6. Smell the product by gently wafting the odour towards you nose with your hand.
What is addition polymerisation?
Addition polymerisation is where lots of monomers join up to make a polymer.
Repeat Units for Addition Polymers
(Notion)
How to deduce monomer from repeat unit?
1. Find the repeat unit by taking any two adjacent carbon atoms in the main polymer chain.
2. Put a double bond between the two middle carbon atoms of the repeat unit, and get rid of the continuation bonds.
Advantages and Disdvantages of Addition Polymers
- Addition polymers are cheaper and stronger than polymers from corn starch.
- It frees up land to grow crops.
- Disposal of addition polymers is a problem as it is non-renewable, inert and non-biodegradable.
- It causes problems with litter and the burning of it creates greehouse gases.
What is condensation polymerisation?
- Condensation polymerisation is where a dicarboxylic acid and a diol reac to produce a polyester and water.
Reaction of Ethanedioic Acid and Ethanediol
(Notion)
What are biopolyesters
Biopolyesters are polyesters that are biodegradable.