Organic practicals 5
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Draw labelled distillation apparatus
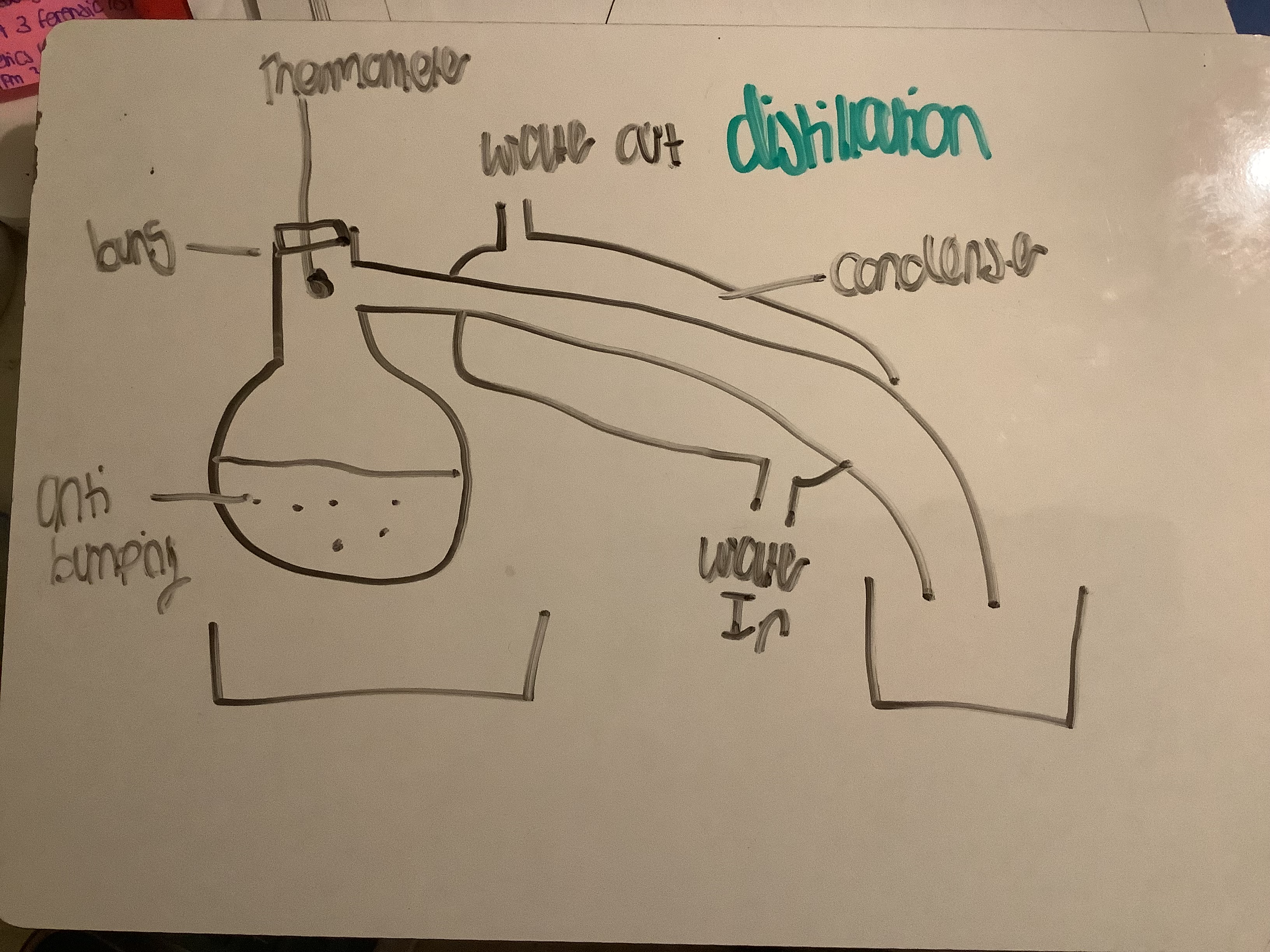
Explain why we use anti bumping granules
Create smaller bubbles
Explain why we use bung distillation
Closed system
Prevent gases of product escaping
Allows collection
Which direction should water be added
Water in at bottom
Out at top
Why do we add water at the bottom
So condenser is fully filled with water
So condenser fully cool
So product can condense
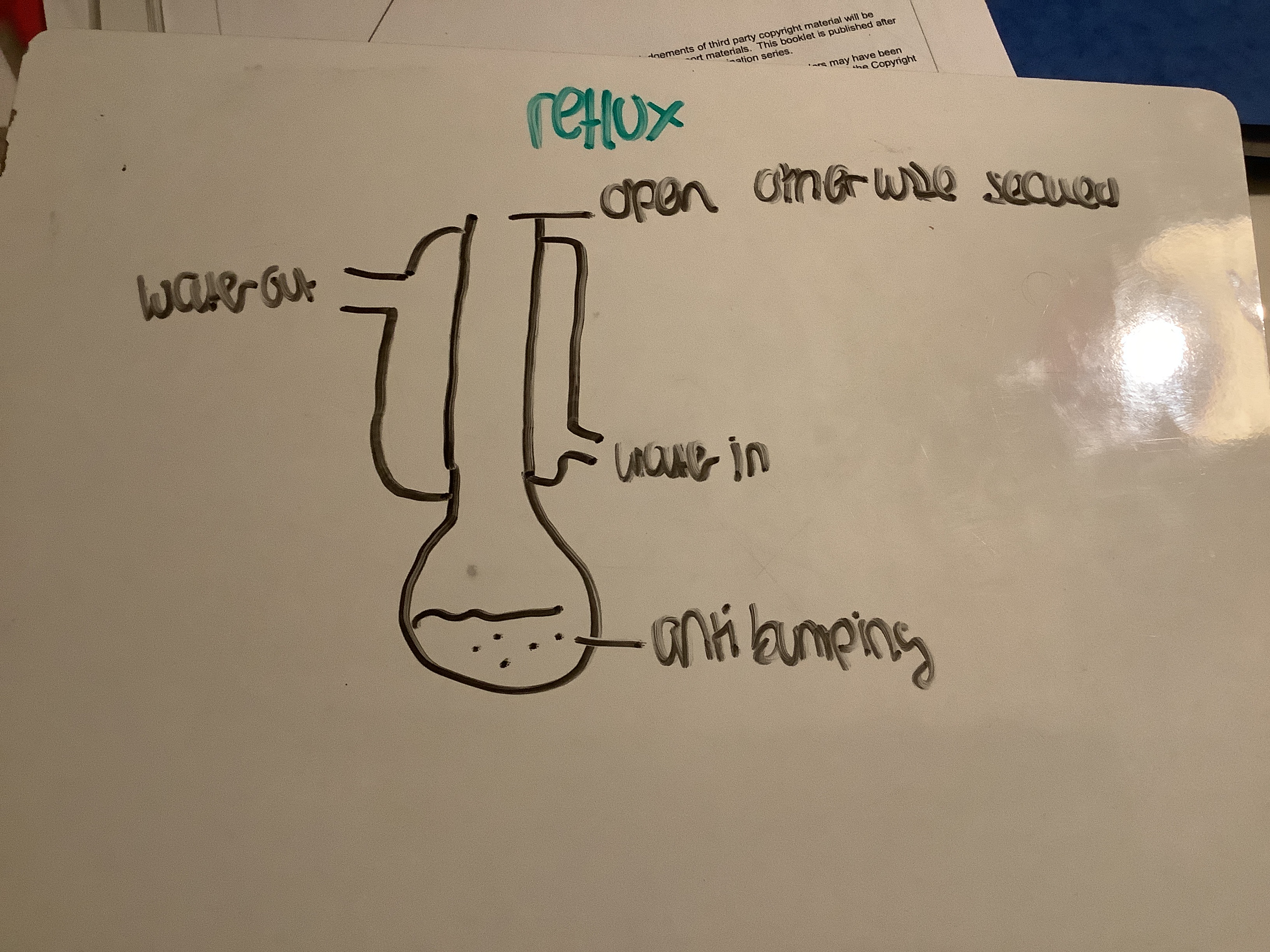
SAFTEY concern s reflux
Round bottom flask not clamped
Bung-build up of pressure
Suggest why a water bath is better than direct heating with a Bunsen burner
Alcohol/organic product
Flammable
Draw labelled reflux apparatus
What can acidified potassium dichromate be used to distinguish between
-primary alcohols
Secondary alcohols
Aldehydes
Expected observation when oxidising with acidified potassium dichromate
Orange to green
What can tollens reagent be used to distinguish and observations
Aldehyde
Silver mirror
How can you distinguish between two primary alcohols
comapre to known database
Have different fingerprint regions
Why might mass spectrometry using electrosoray ionisation not be a suitable method of distinguishing between isomers
They have the same precise mr
What can you use to test for a carboxylic acid
Sodium carbonate
Produce co2 effervescence
Outline method used for recrystallisation
Dissolve product in minimum volume of hot solvent -create saturated solution
Hot filtration to remove soluble impurities
Cool to recrystallise
Filter under reduced pressure
Wash with cold solvent and dry-between two pieces of filter paper
Explain why:
Flask cooled to room temp before crystals filtered off
Crystals were compressed in funnel
Cold water was poured through crystals
Yield lower if warm
Air passes through sample so better drying
Wash soluble impurities
Outline how you would show recrystallised product is pure
Measure melting point
Using mp apparatus
Match mp to data boom
Suggest two reasons why recrystallised product % yield is not 100%
Sample lost during recrystallisation
Sample still wet.