Amino acids, proteins, enzymes, condensation polymerisation
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
give the name of the condensation polymer formed from amino acids
polypeptides
explain what happens when when 2 amino acids join to form a dipeptide
a hydrogen is removed from the N-H group and the O-H group is removed.
Forming a peptide link and releasing of molecule of water
give the name of the process when peptide links are made
condensation polymerisation
give the name of the process when peptide links are broken
hydrolysis
give the intermolecular forces and bonding peptides contain (4 marks)
van der waals forces
hydrogen bonding
peptide links
disulphide bridges (if the R group contains Sulfur)
explain how disulphide bridges may arise in peptides
disulphide bridges are formed if the R group in 2 amino acids contains Sulfur
Sulfur is electronegative, but not as electronegative as oxygen
so it forms disulphide bridges between the sulfur atoms
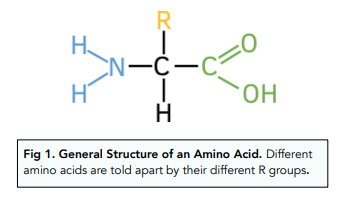
draw the general structure of an amino acid
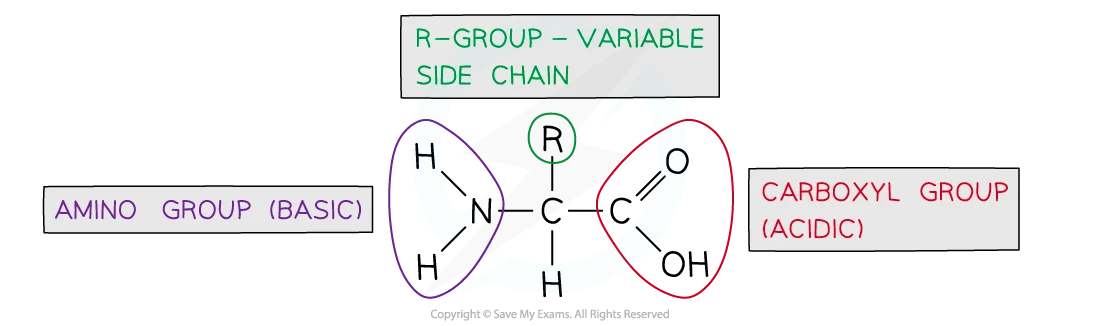
draw a circle and label which groups are acidic and basic in an amino acid
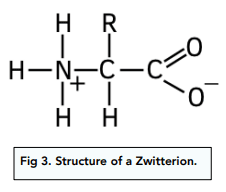
explain how zwitterions are formed from an amino acid
the H from the O-H group is attracted o the electronegative N in the amine group.
Forming a zwitterion with a positive N and negative O
There is no overall charge to the amino acid. This is called a zwitterion and is only formed at a specific pH for each amino acid. The pH at which a zwitterion is formed is called the isoelectric point.
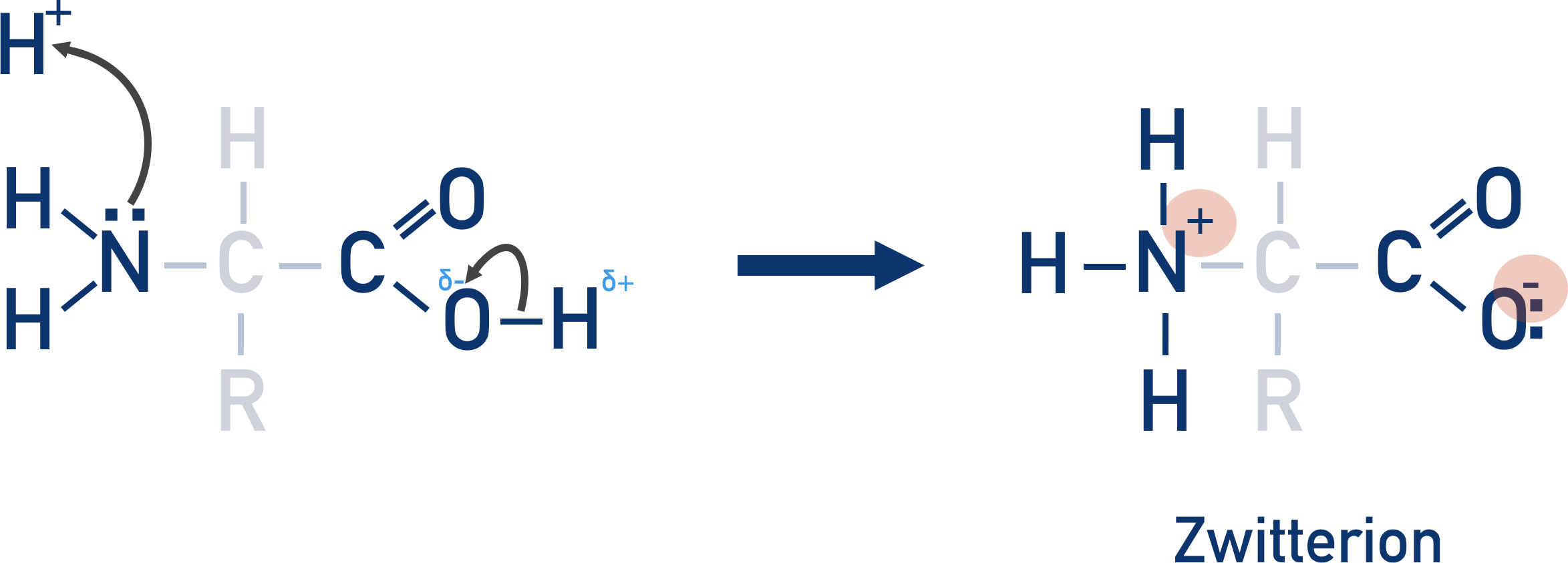
explain what happens to an an amino acid in acidic conditions
the amine group will accept a proton
the N in the amino acid will become positively charged.
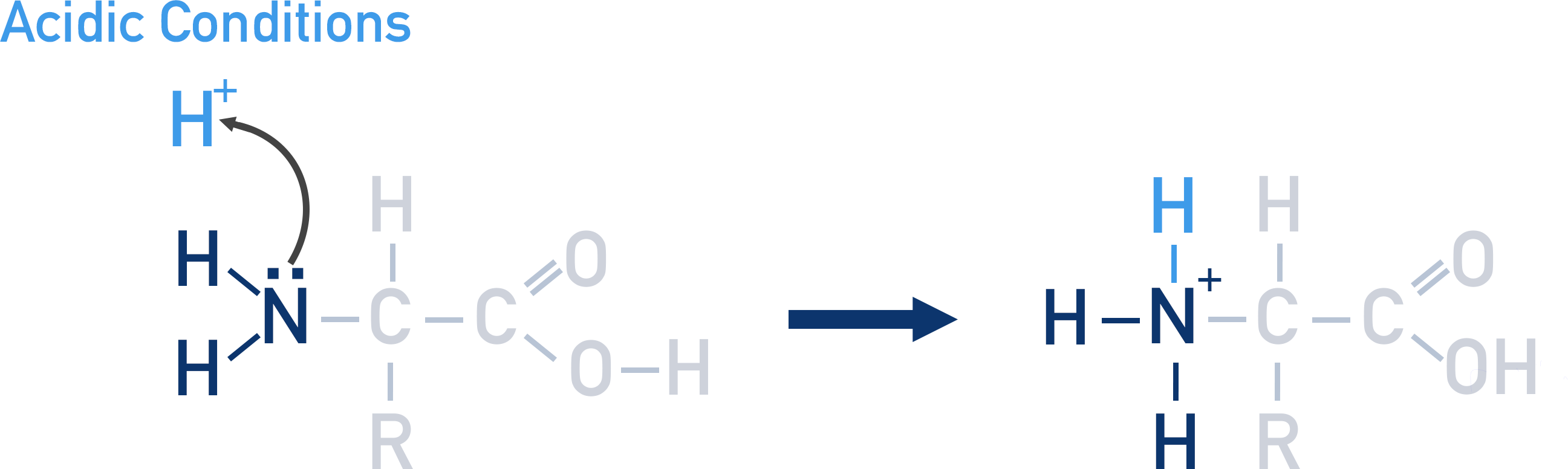
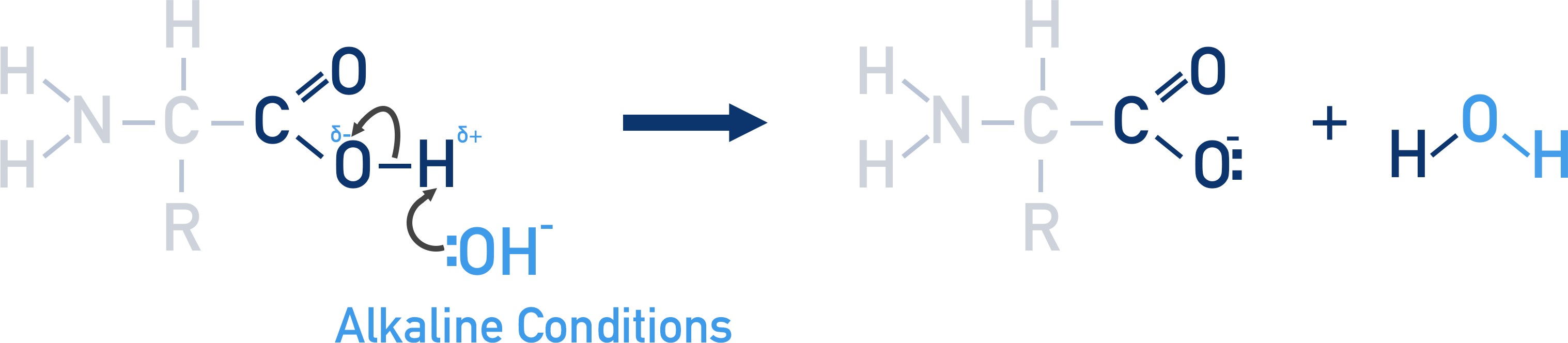
explain what happens to an amino acid in alkaline conditions
the carboxylic acid will lose a proton
the O in the amino acid will become negatively charged.
and water is formed
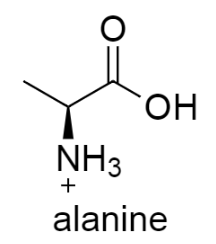
draw the structure of alanine in acidic conditions
amine group gains a proton. The amino group has a positive charge; the overall molecule thus has a net charge of +1.
draw the mechanims of alanine in alkaline conditions
Carbonyl group loses a hydroxyl group. oxygen becomes negative with 2 lone pairs
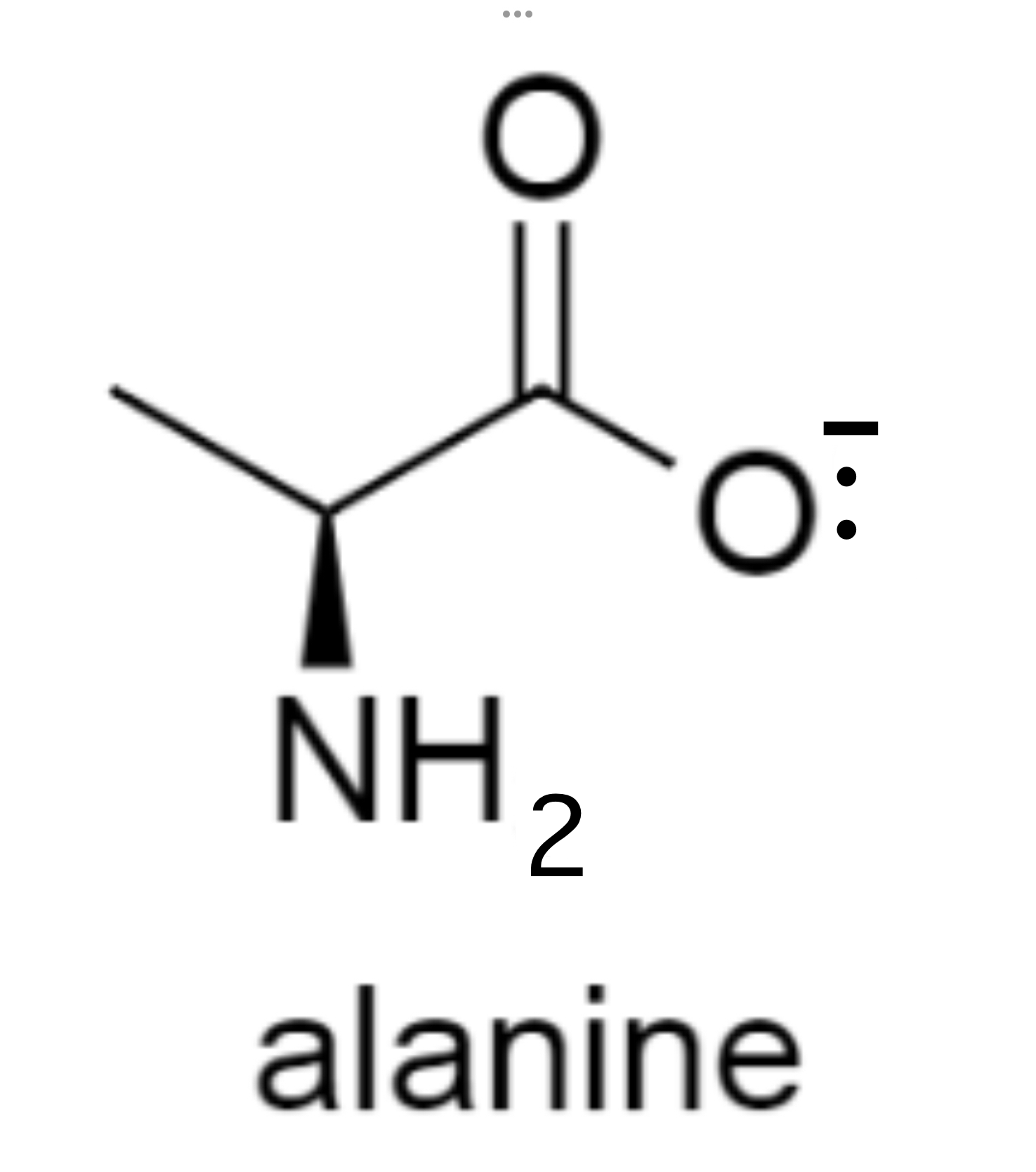
define isoelectric point
pH at which an amino acid exists as a zwitterion
define zwitterion (1 mark)
ion with a (permanent) negative and positive charge but overall the compound is neutral
state 2 physical properties of amino acids at room temperature (2 marks)
high melting point or solids
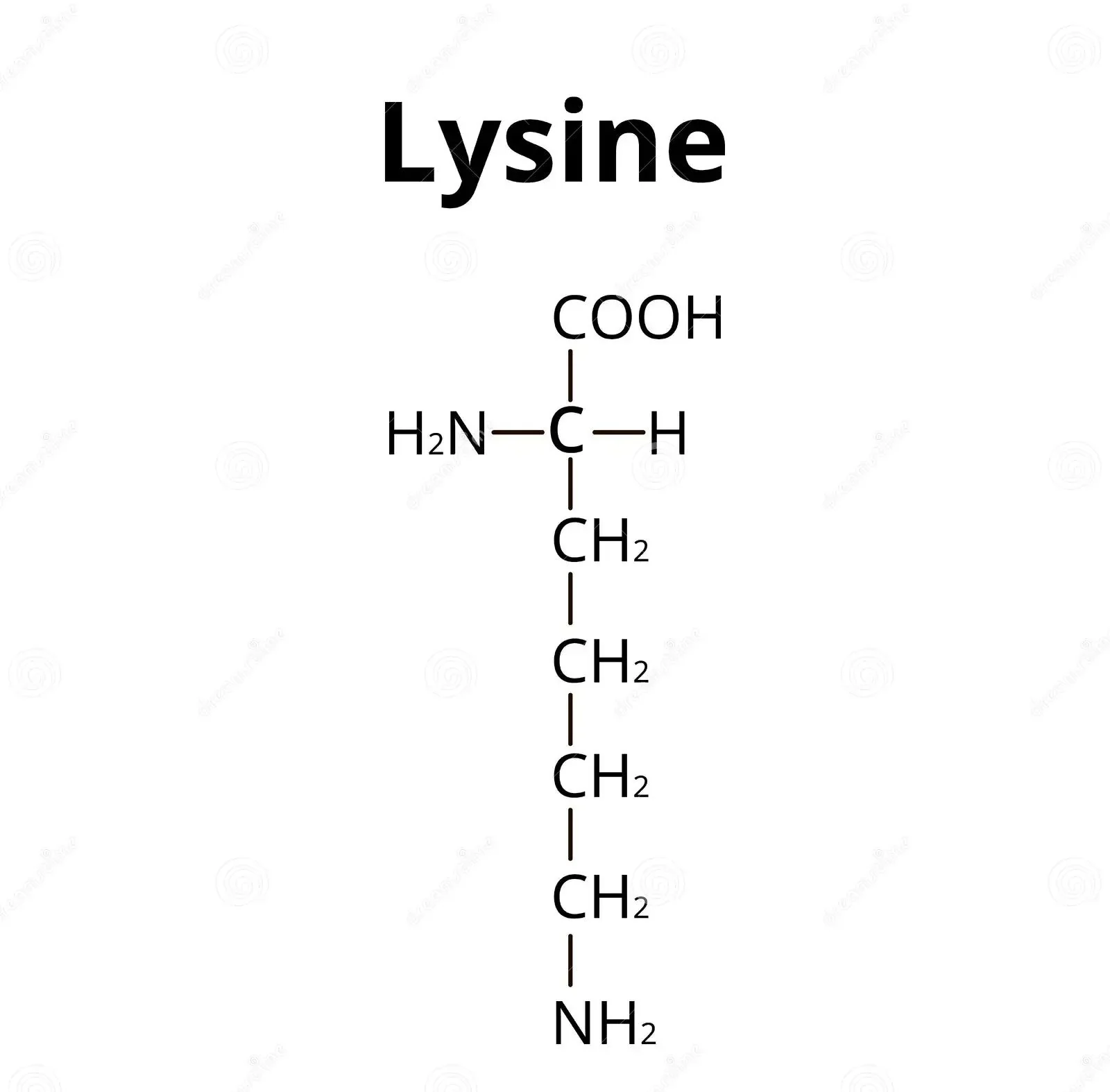
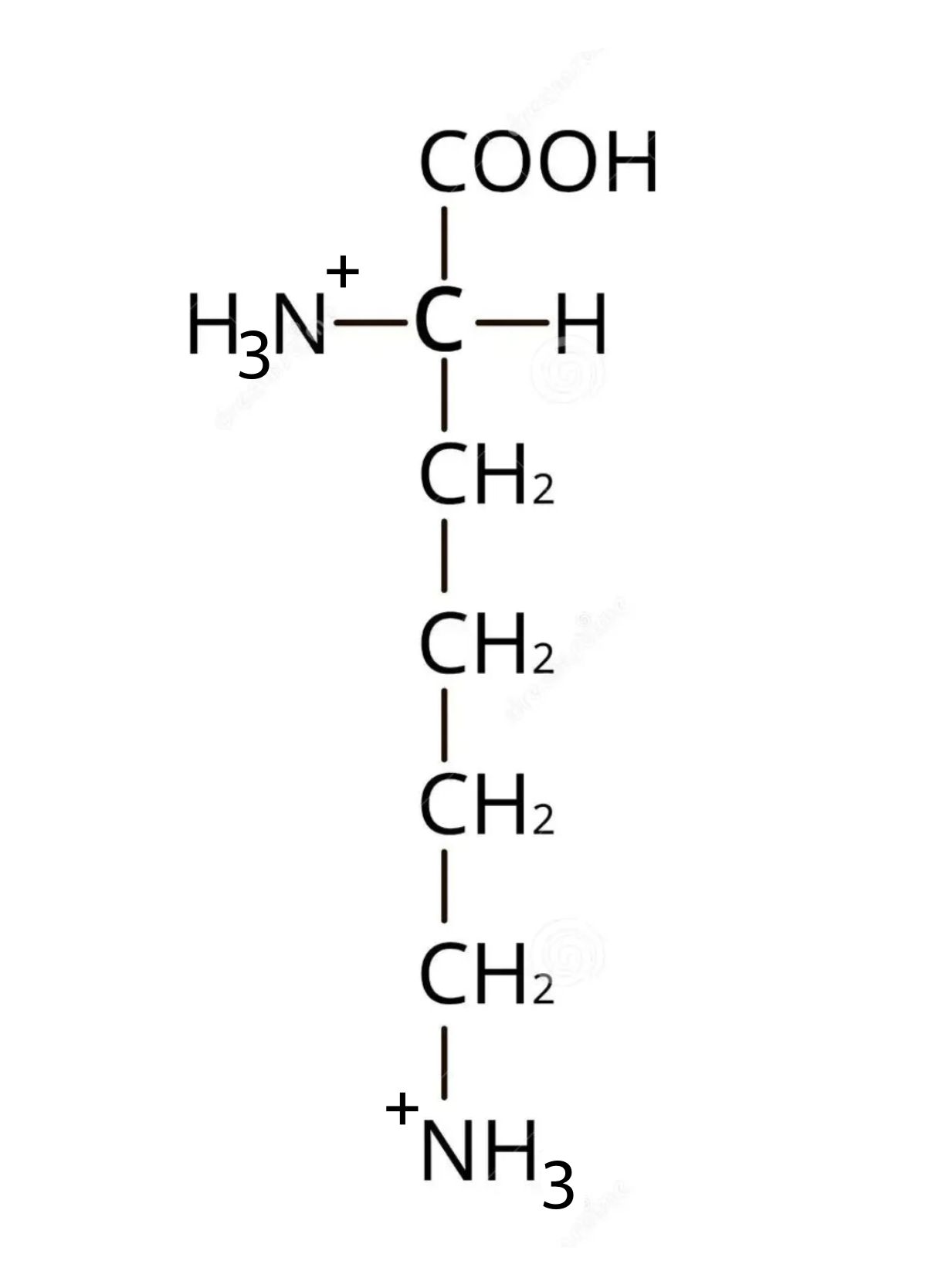
Draw the structure of lysine in a solution of pH=12
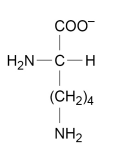
![<p>Give the structures of the two products formed after boiling this dipeptide in an excess of hydrochloric acid. [2 marks]</p>](https://assets.knowt.com/user-attachments/c96aa647-b8fd-44be-b43a-c7fa8d2d5584.png)
Give the structures of the two products formed after boiling this dipeptide in an excess of hydrochloric acid. [2 marks]
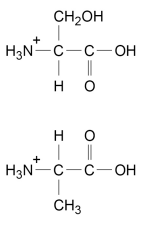
![<p>Draw the skeletal formula of the organic product formed when aspartic acid reacts with an excess of methanol and a little concentrated sulfuric acid. [1 mark]</p>](https://assets.knowt.com/user-attachments/9edce208-09db-4b9e-9cc2-b5db45098d0a.png)
Draw the skeletal formula of the organic product formed when aspartic acid reacts with an excess of methanol and a little concentrated sulfuric acid. [1 mark]
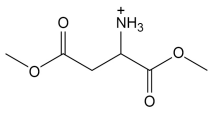
draw the structure of alanine when it is placed in a solution that is at the pH of it’s isoelectric point and state the product formed
a zwitterion
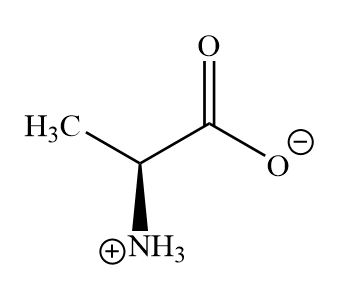
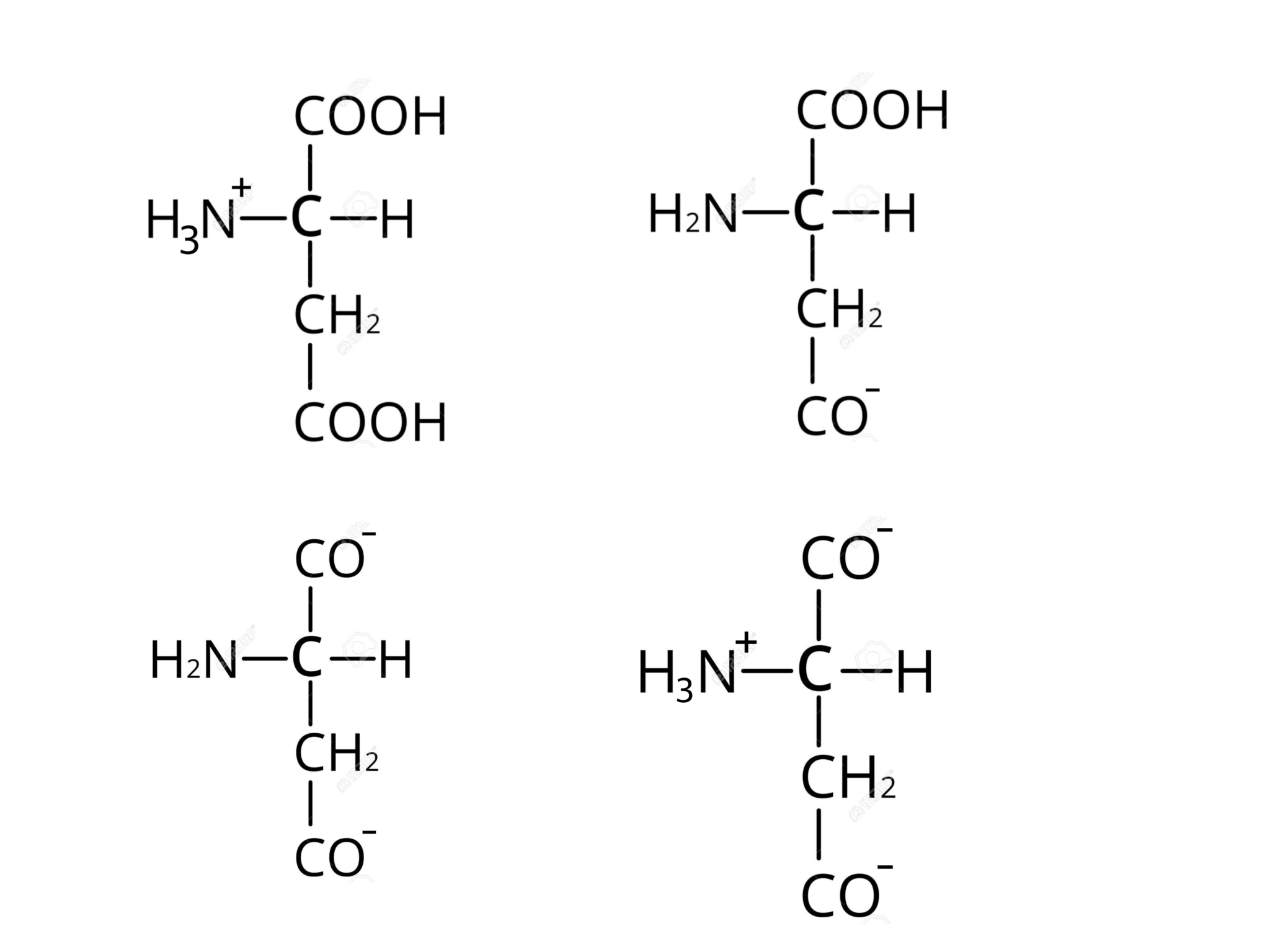
Which is the correct form of aspartic acid in alkaline conditions
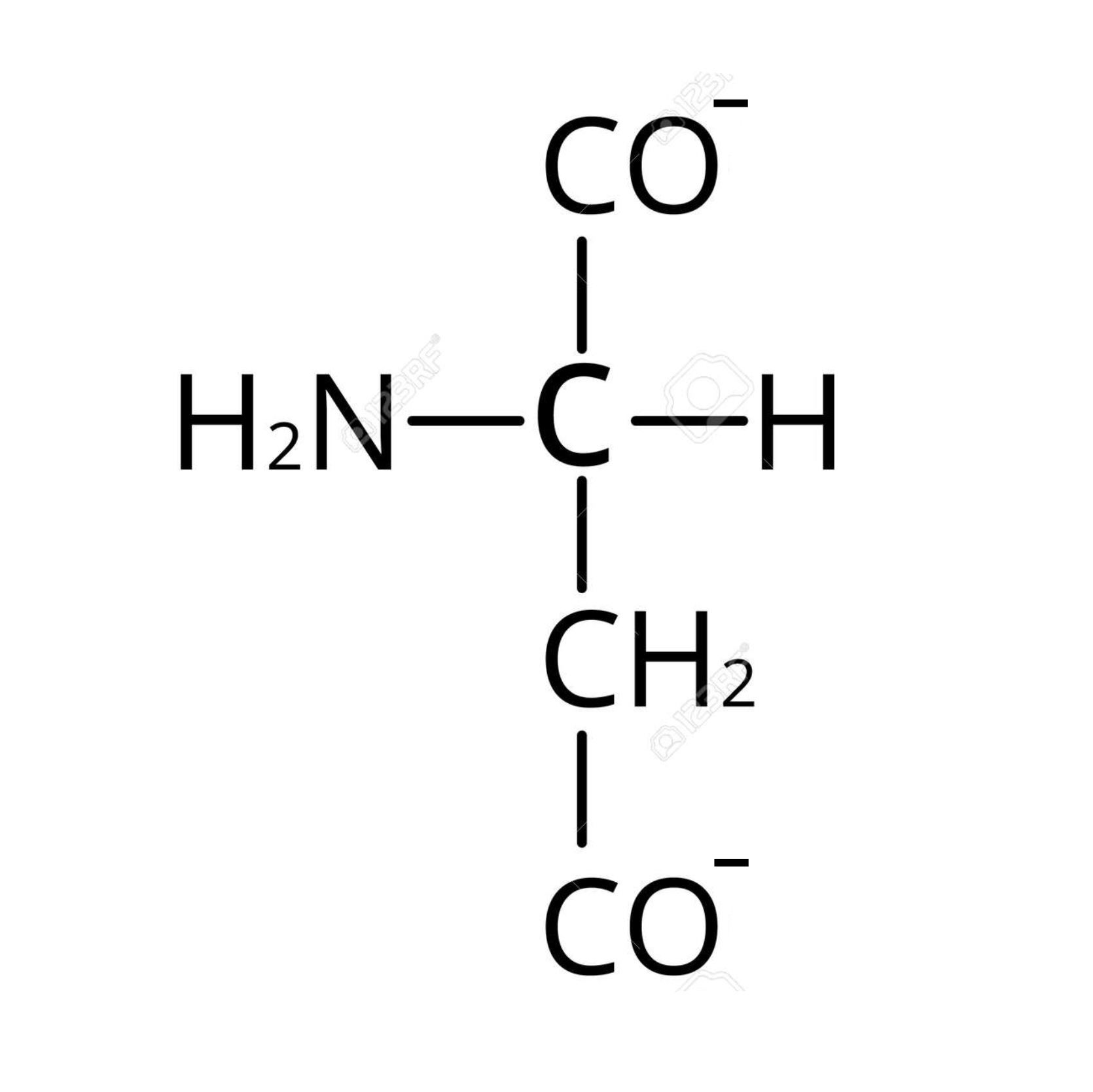
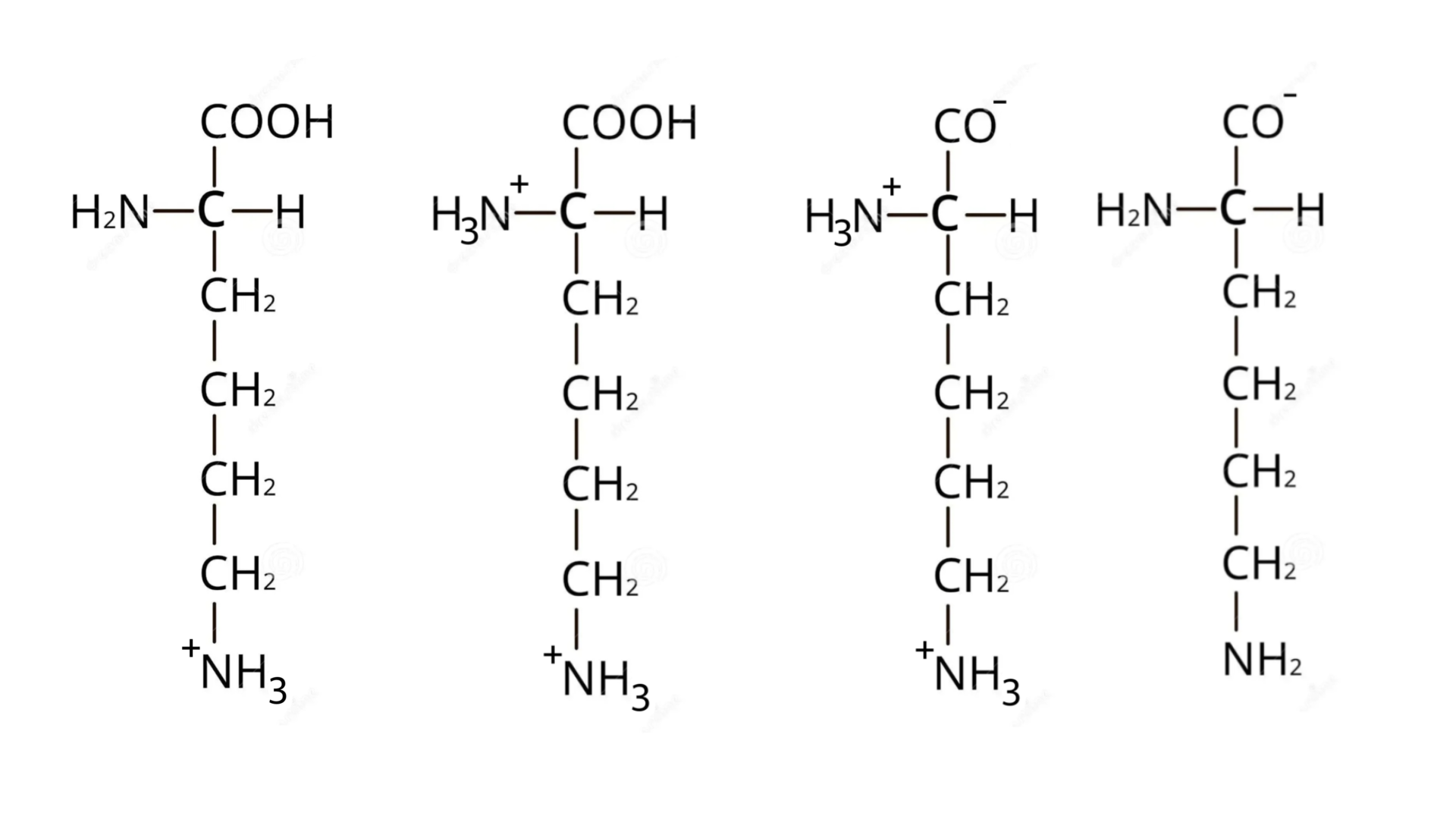
draw the structure of lysine in acidic conditions