Ammonia Assimiliation and Toxicty
1/87
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
88 Terms
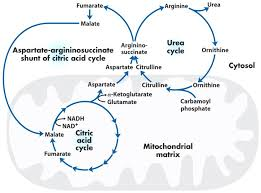
What does the aspartate-arginosuccinate shunt demonstrate
how the body avoids isolating biochemical processes and instead creates efficient systems where pathways communicate and share intermediates
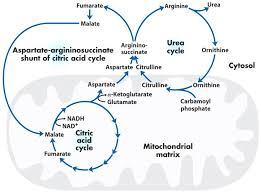
What is linked to the urea cycle in relation to the aspartate-argininosuccinate shunt
the urea cycle and the TCA cycle
What is the urea cycle responsible for
detoxifying ammonia by converting it into urea
what is the TCA cycle responsible for
energy production
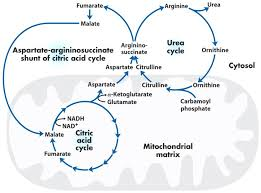
How does the aspartate-argininosuccinate shunt connect the TCA cycle and the urea cycle
it allows intermediates to move between them
What is a key TCA cycle intermediate that can be converted into aspartate via transamination
oxaloacetate
What happens to the aspartate that was converted from the oxaloacetate via transamination
it exits the mitochondria and enters the urea cycle
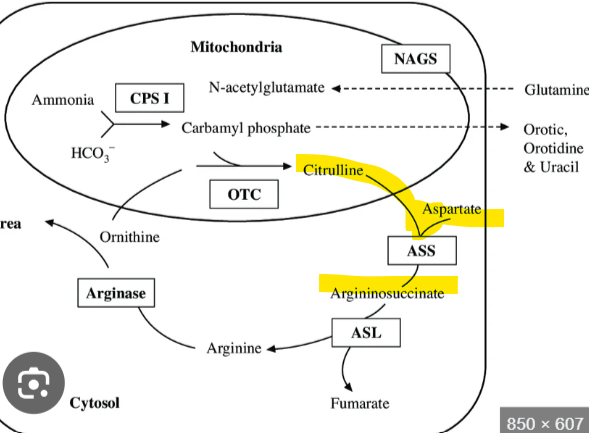
The oxaloacetate from the TCA cycle gets converted to asparate via transamination. It then exits the mitochondria and enters the urea cycle, what happens after this
it combines with citrulline to form argininosuccinate which is then later split into arginine to release fumarate as a byproduct
Specifically, oxaloacetate, a key TCA intermediate, can be converted into aspartate via transamination. This aspartate then exits the mitochondria and enters the urea cycle, where it combines with citrulline to form argininosuccinate. When argininosuccinate is later split into arginine, it releases fumarate as a byproduct.
What happens to the fumarate
it re enter the TCA cycle by being converted into malate and then back int oxaloacetate, completing the loop
Why is the Aspartate–Argininosuccinate Shunt system efficient
because it ensures thaty nitrogen disposal ( urea cycle) and energy metabolism ( TCA cycle) are synchronized and supports gluconeogensis when needed
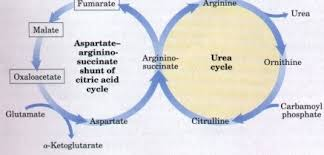
What does the term Krebs bicycle emphasize
that metabolism is not linear but interconnected
Why is ammonia toxic
becasue ammonia, although a normal byproduct of metabolism , is highly disruptive to cellular function, particularly in the brain
Under physiological conditions what does ammonia primarily exist as
ammonium (NH4+)

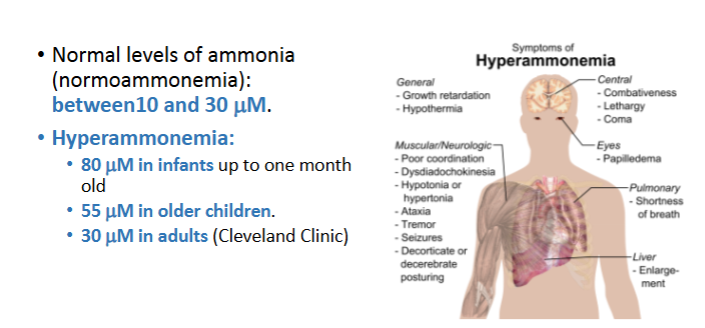
What is relevent about the concentration of ammonium in the blood
it is tightly regulated within a narrow range
Slight increases of ammonium above the range (approximately 10–30 micromolar) can lead to what condition
hyperammonia
why is hyperammonia so dangerous
ammonia can freely cross biological membranes, including the blood-brain barrier
Due to hyperammonia, ammonia can freely cross biological membranes, including the bl;ood brain barrier. What happens when ammonia gets inside cells
ammonia interferes with proton gradients across membranes - particularly in the mitochondria which are essential for ATP production
What does the disruption from ammonia interfering with intracellular membranes cause
it impairs oxidative phosphorylation, meaning cells cannot efficiently generate energy
Why is the brain partiocularly sensitive when hyperammonia happens
neurons have high energy demands and limited capacity to detoxify ammonia locally.
ammonia shifts neurotransmitter balance by affecting glutamate and glutamine levels, potentially leading to neuronal dysfunction, swelling (cerebral edema), and even coma
True or False:Clinically, hyperammonemia thresholds differ by age because enzyme systems involved in ammonia detoxification, such as those in the urea cycle, are not fully developed in newborns
true
Clinically, hyperammonemia thresholds differ by age because enzyme systems involved in ammonia detoxification, such as those in the urea cycle, are not fully developed in newborns
Therefore,
infants tolerate higher levels before diagnosis , but they are also more vulnerable to rapid neurological damge
Overall, why is ammonia is toxicity dangerous?
because it is constantly being produced and must be continuously managed to prevent systemic and neurological damage
Ammonia is continuously generated in the body as a natural consequence of nitrogen metabolism— what does this mean
its production is unavoidable and must be carefully controlled.
what is the primary source of ammonia
the breakdown of amino acids
when does the breakdown of amino acids take place
during normal protein turnover or increased protein metabolism ( the more you breakdown proteins the more you have to breakdown for amino acids) in which transamination and deamination reactions take place to free ammonia
In addition to amino acids, ammonia is also produced during the metabolism of nitrogen containing compounds such as: _________
purines and pyrimidines - components of DNA and RNA
Aside from the breakdown of amino acids and the metabolism of nitrogen-containing compounds what is another significant source of ammonia
the gastrointestinal tract
How is the gastrointestinal tract a source of ammonia
intestinal bacteria produce ammonia by breaking down urea through the enzyme urease.
What is significant about the bacterial contribution of ammonia
increased protein availability in the gut leads to increased bacterial ammonia production
True or false: ammonia production is not localized to a single organ—it occurs in virtually all tissues
True
Due to most tissues lackingthe capacity to safely dispose of ammonia what is needed
specialized transport and detoxification systems
What condition can result from a variety of physiological and pathological conditions, all of which ultimately either increase ammonia production or impair its removal.
Hyperammonemia
What major cause of hyperammonemia is being described:
GENETIC EFFECTS IN UREA CYCLE ENZYMES
In these conditions, the body cannot effectively convert ammonia into urea, leading to accumulation
The severity depends on which enzyme is affected and whether the deficiecy is partial or complete
Urea cycle disorders (UCD)
What major cause of hyperammonemia is being described:
This causes a heavy impact becasue this organ is the primary site of ammonia detopxification
Acute _____ failure
Cirrhosis
Drug induced ___ damage
These examples can impar this organs ability to process ammonia
liver dysfunction
How can drug induced liver damage and chemotherapy agents impair the liver’s ability to process ammonia
by destroying the hepatocytes responsible for the urea cycle , distrupting mitochondiral function
ex-acetaminophen overdose or chemotherapy agents like 5-fluorouracil
What major cause of hyperammonemia is being described:
excess protein in the gut is metabolized into ammonia
gastrointestinal bleeding
What major cause of hyperammonemia is being described:
amino acid breakdown is elevated
intense muscle exertion
What major cause of hyperammonemia is being described:
low potassium levels, can stimulate enzymes like glutaminase, increasing ammonia release from glutamine.
electrolyte imbalances
What major cause of hyperammonemia is being described:
elevate ammonia levels by increasing its production in the gut
infections with urease-producing bacteria
What major cause of hyperammonemia is being described:
impairing urea excretion, leading to altered nitrogen balance.
Kidney failure
What doe the diverse causes of hyperammoneia highlight
hyperammonemia is not a single disease but a metabolic consequence of many different disruptions, all converging on the imbalance between ammonia production and detoxification.
Because ammonia is toxic and produced throughout the body, there must be a safe way to transport it from peripheral tissues to the liver, where it can be detoxified.
How is this accomplished ?
primarily through the formation of glutamine
Because ammonia is toxic and produced throughout the body, there must be a safe way to transport it from peripheral tissues to the liver, where it can be detoxified.
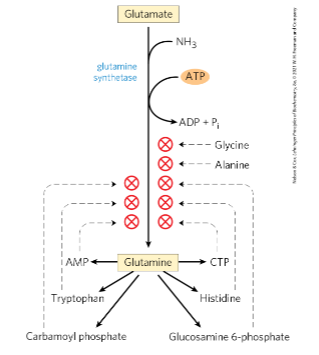
This is accomplished primarily through the formation of glutamine. In peripheral tissues such as muscle and brain
What enzyme catalyzes the incorporatuon of ammonia into glutamate to form glutamine
glutamine synthetase
In the formation of glutamine why is it critical
because it converts free ammonia, which is toxic, into a non-toxic, transportable form
Glutamine acts as a “nitrogen carrier,” safely transporting ammonia through the bloodstream to the liver.
What happens in th eliver
glutaminase removes the ammonia from glutamine, releasing it for entry into the urea cycle
Why is the glutamine system especially important to the brain
the brain lacks a urea cyce and therefore cannot directly detoxify ammonia
By converting ammonia into glutamine, the brain protects itself from toxicity, why must this system be tightlt regulated
because excessive glutamine accumulation can lead to osmotic imbalances and contribute to brain swelling
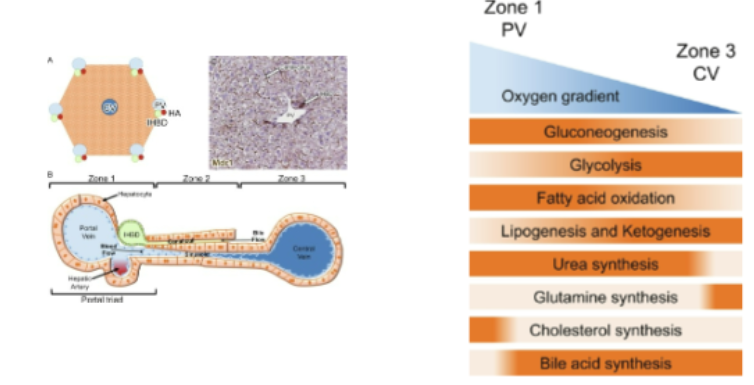
The liver is uniquely structured to maximize its ability to detoxify ammonia through what concept
zonation
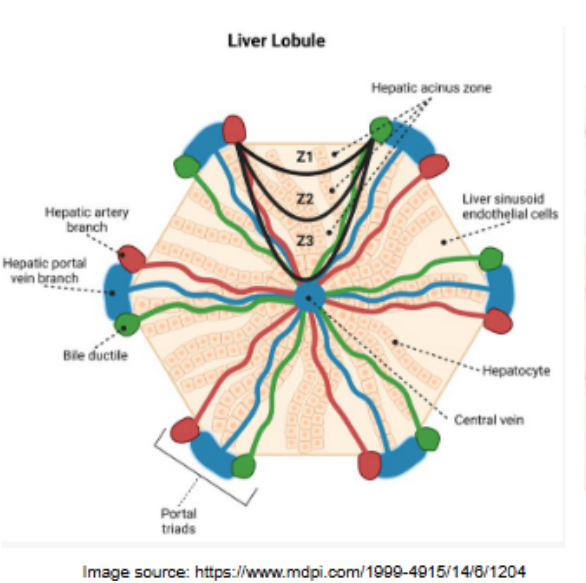
where does zonation take place
the liver acinus
what is the liver acinus
the functional unit of the liver=
it is going to be oriented around this, this vascular system of where the blood enters, and then the blood leaves the liver to then go to the kidneys for filtration, and then come back in.
What is the pathway of the blood through the liver
it enters through the portal vein to the central vein
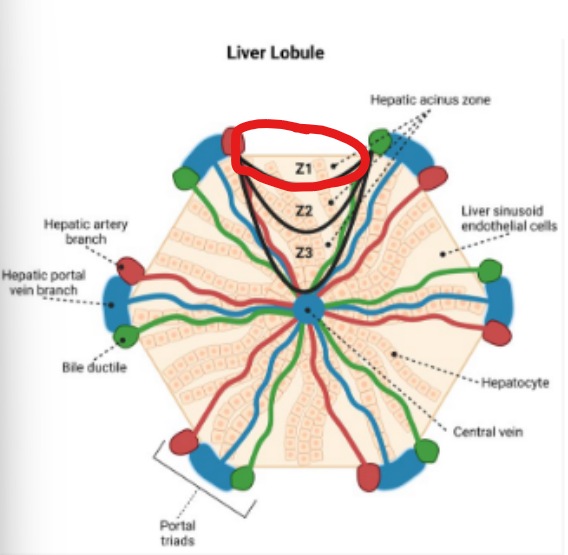
Blood enters the liver through the portal vein, carrying nutrients and toxins, including ammonia, and flows toward the central vein.
Along this pathway hepatocytes are organized into zones with different metabolic rates
What accurately describes Zone 1
closest to the incoming blood supply
Here, enzymes like carbamoyl phosphate synthetase I (CPS1) and glutaminase are highly active
These enzymes have low affinity but high capacity- meaning they are designed to handle large amounts of ammonia quickly without becoming saturated
This is known as the first line of defense
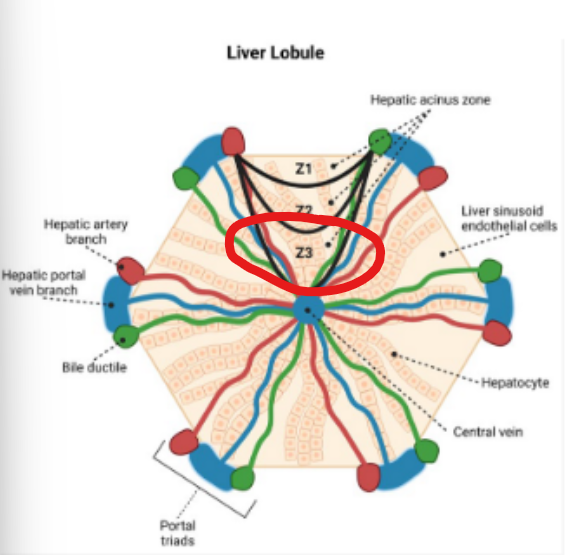
Blood enters the liver through the portal vein, carrying nutrients and toxins, including ammonia, and flows toward the central vein.
Along this pathway hepatocytes are organized into zones with different metabolic rates
What accurately describes Zone 3
ammonia levels are much lower
glutamie synthetase us highly active
enzyme has high affinity but low capacity - meaning it is extreemly effective at capturing even small amounts of residual ammonia
creates a second filtering systen that ensures almost no ammonia escapes the systemic circulation
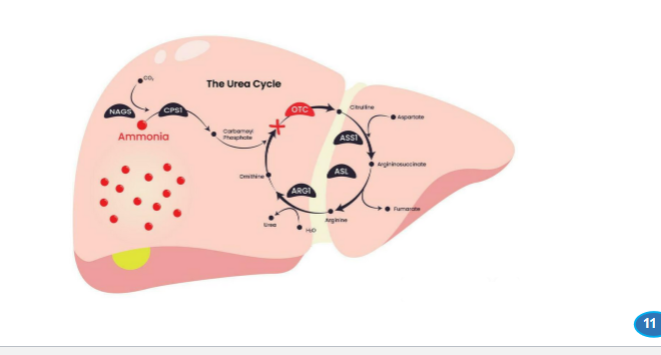
Urea cycle disorders (UCD)are inherited conditions that impair the body’s ability to detoxify ammonia, leading to its accumulation in the blood
What is the most important example?
ornithine transcarbamylase (OTC) deficiency.
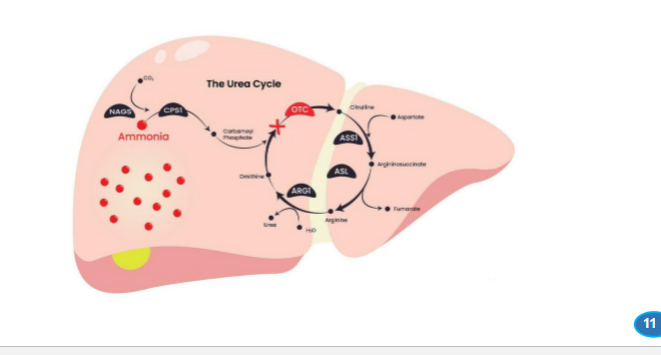
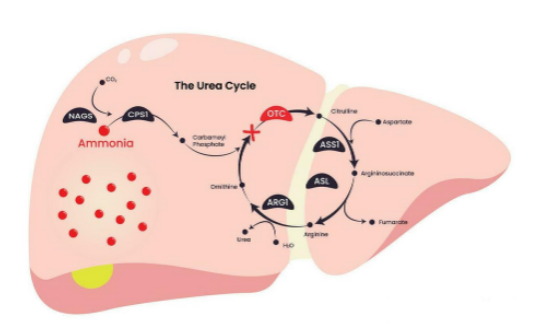
What describes ornithine transcarbamylase (OTC) deficiency?
the enzyme responsible for combining carbamoyl phosphate with ornithine to form citrulline is defective
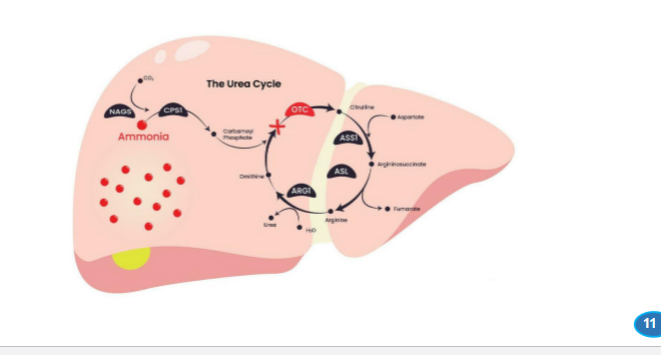
What is the result of ornithine transcarbamylase (OTC) deficiency in which the enzyme responsible for combining carbamoyl phosphate with ornithine to form citrulline is defective
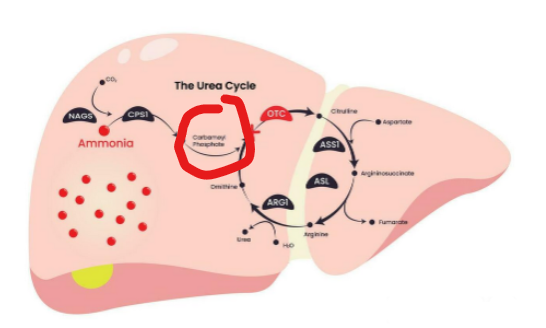
carbamoyl phosphate accumulates in the mitochondria.
One of the most important examples is ornithine transcarbamylase (OTC) deficiency. In this condition, the enzyme responsible for combining carbamoyl phosphate with ornithine to form citrulline is defective. As a result, carbamoyl phosphate accumulates in the mitochondria.
What are the 2 major consequences of this
ammonia cannot be efficiently incorporated into the urea cycle, leading to hyperammonemia
excess carbamoyl phosphate leaks into the cytosol, where it is diverted into pyrimidine synthesis pathways, leading to the production of orotic acid.
this can lead to oroticx acid uria
When excess carbamoyl phosphate leaks into the cytosol, diverting into pyrimidine synthesis pathways, leading to the production of orotic acid, what does this result in
orotic aciduria
What is a key diagnostic distinction of orotic aciduria caused by OTC deficiency compared to orotic aciduria caused by defects in pyrimidine synthesis that is clinically important for diagnosis ?
orotic aciduria caused by OTC deficiency is accompanied by hyperammonia
Where does OTC deficiency often present
in newbvorns with severe symptoms within the first few days of life:
vommitting
neurological impairment
lethargy
What does the severity of OTC deficiency depend on
the degree of enzyme deficiency
In terms of the the severity of OTC deficiency depending on enzyme deficiency , what does it mean when there is complete deficiencies
it becomes life threatening
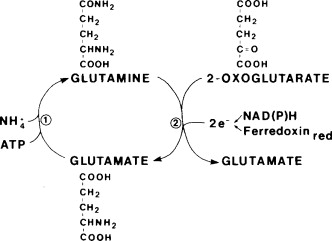
What is ammonia assimilation
to the incorporation of free ammonia into organic molecules to reduce its toxicity
What is one key enzyme involved in the process od ammonia assimilation
glutamate dehydrogenase (GDH)—which catalyzes a reversible reaction.
How does GDH do a reversible reaction
In the reverse direction, it incorporates ammonia into alpha-ketoglutarate to form glutamate (reductive amination), using NADPH.
What does the reversibility of GDH allow
allows GDH to function in both ammonia production and detoxification, depending on cellular needs
Aside from GDH what is another critical enzyme for ammonia assimilation
glutamine synthetase,
what does glutamine synthetase do
converts glutamate and ammonia into glutamine using ATP
Where and why is the reaction of glutamine synthetase converting glutamate and ammonia into glutamine using ATP important
peripheral tissues and the brain becasue it is the primary mechanism for ammonia detoxification
what is located in the mitochondria of liver cells and catalyzes the first committed step of the urea cycle to incorporate ammonia in to carbamoyl phosphate
CPS1
How does GDH, glutamine synthetase, and CPS1 form a coordinated system
GDH and glutamine synthetase manage ammonia at the cellular level, while CPS1 initiates its ultimate disposal in the liver.
What is:
one of the most important amino acids in the body, serving as both a nitrogen carrier and a metabolic hub.
the most abundant amino acid in the bloodstream and plays a critical role in transporting ammonia safely between tissue
Glutamine
Beyond its role in detoxification, glutamine is involved in numerous biosynthetic pathways, including the synthesis of _____ and _______
nucleotides and other amino acids
Glutamine is one of the most important amino acids in the body, serving as both a nitrogen carrier and a metabolic hub.
It is the most abundant amino acid in the bloodstream and plays a critical role in transporting ammonia safely between tissues.
Beyond its role in detoxification, glutamine is involved in numerous biosynthetic pathways, including the synthesis of nucleotides and other amino acids.
However, its central role also creates potential problems.
What are some examples of this?
In conditions of hyperammonemia, excessive glutamine production can occur, particularly in the brain. This can lead to osmotic imbalances, causing cells (especially astrocytes) to swell, contributing to cerebral edema
Glutamine can be converted back into glutamate in neurons, potentially leading to excitotoxicity due to excessive neurotransmitter activity.
What is the paradox of glutamine
while glutamine formation protects against ammonia toxicity, its overproduction can contribute to neurological damage
How can the body carefully regulate glutamine levels
through feedback inhibition mechanisms, where end products of glutamine metabolism inhibit glutamine synthetase
When amino acids are broken down, their nitrogen is removed and processed through the urea cycle, but their carbon skeletons remain and must be utilized efficiently.
What happens to those amino acids
These carbon skeletons can enter central metabolic pathways and are classified based on their metabolic fate
What are glucogenic amino acids
those whose carbon skeletons can be converted into intermediates of the TCA cycle or gluconeogenesis, allowing them to contribute to glucose productio
What are ketogenic amino acids
they ( their carbon skeletons) converted into acetyl-CoA or acetoacetate, which can be used for the production of ketone bodies but cannot be converted into glucose.
Some amino acids are both glucogenic and ketogenic—— what does this mean?
they ( their carbon skeletons) can contribute to both pathways depending on the body’s metabolic state
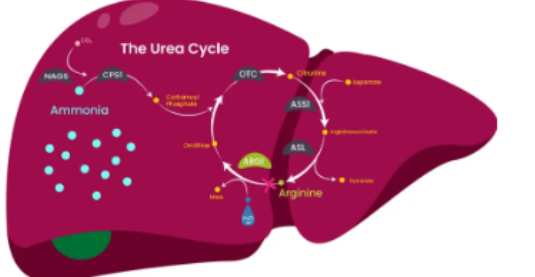
What UCD is when there is not enough arginine to progress within the urea cycle
Argininemia (ARG1 Deficiency)
What are symptoms that higlight arginase deficieny
feeding problems , vommitting, poor growth, seizures, anmd spasticity
may have developmental delay and intellectural disabilit\y
what ammonia acids are ketogenic
leucine and lysine
what amino acids are ketogenic AND glycogenic
Isoleucine
Phenylalanine
Tyrosine
Tryptophan
Threonine
What amino acids are glycogenic
alanine (pyruvate)
aspartate (oxaloacetate)
asparagine (oxaloacetate)
glutamate ( a-ketoglutarate)
glutamine (a-ketoglutarate)
What is the significance of ammonia scavenging and how does the liver acinus trap ammonai
The liver acinus traps ammonia through a spatially organized, two-stage system: high-capacity urea synthesis in the periportal zone (Zone 1) handles the bulk of ammonia, while a high-affinity glutamine synthetase system in the perivenous zone (Zone 3) scavenges any remaining ammonia
How does the liver acinus act as a double filter system
The liver acinus acts as a "double filter system" by utilizing a one-way portal-to-central vascular flow that creates a strict, cascading gradient of oxygen and nutrients. This structure drives metabolic zonation, whereby hepatocytes are specialized to handle different tasks based on their location along this gradient, effectively filtering blood twice: first for oxidative, high-energy nutrients (Zone 1) and subsequently for detoxification and low-oxygen processing (Zone 3).