Immuno lecture 18
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
what processes in the body protect our healthy tissues?
negative selection
anergy
suppression of T cell activaiton
Induction of inflammation
immunological inflammation
compliment regulation
negative selection
Deletion of self-reactive lymphocytes leads to Central Tolerance (no creation to self in central lymphoid organs) and prevents self binding tissues
describe negative selection in T cells
Tightly regulated
AIRE allows expression of many self-peptides in thymus
T cells that bind to self-peptides are deleted
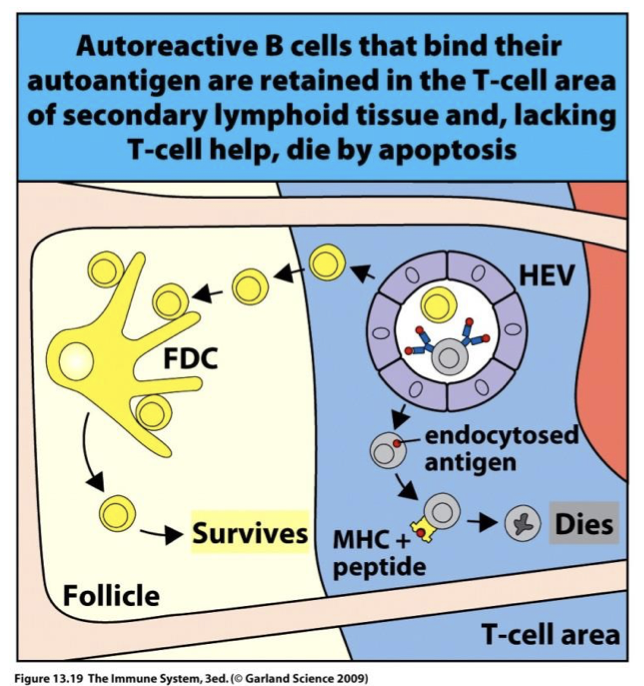
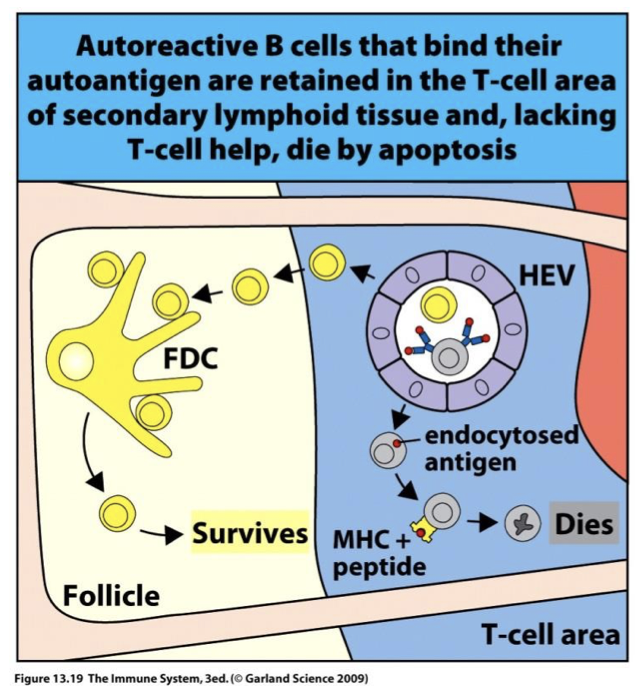
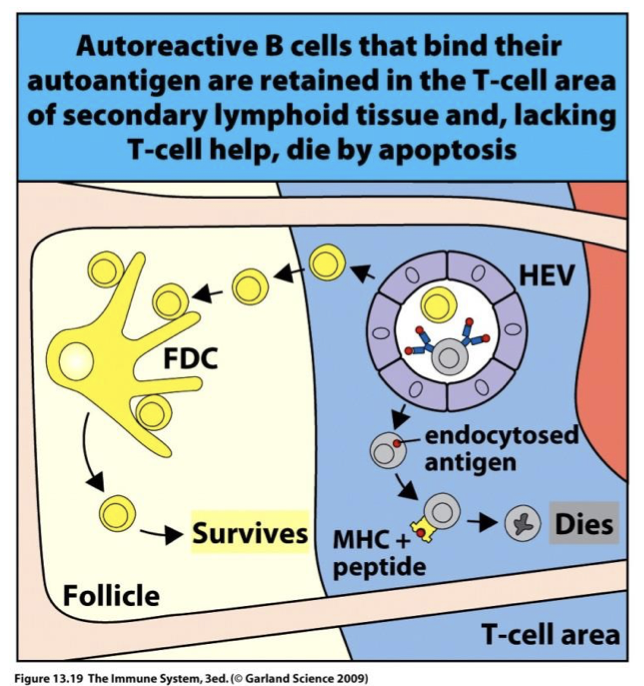
describe negative selection in B cells
less tightly regulated
B cells that bind multi-valent self-peptides are deleted
B cells that are self-reactive are likely to get stuck in T cell zone and die before they find a self-reactive T cell
how does anergy protect healthy tissues
ceases responses in cells
how does anergy occur in T cells? how does it occur in B cells?
T cells – TCR binds w/o co-stimulation
B cells – BCR binds monovalent Ag in BM
Suppression of T cell Activation is done by which two molecules? (how?)
CTLA-4 on T reg cells– competes with CD28 (off switch for costimulation) (used in healthy situations or to suppress cells)
TREG – suppress proliferation of TCR binding autoantigen (Peripheral Tolerance)
how does induction of inflammation protect healthy tissues?
only starts inflammation process during infection or injury
how does immunological privilege protect healthy tissues
sites in body w/o immune response (purposely) as tissue damage risk outweighs immune response benefits
-seen in brain, eyes, uterus
how does complement regulation protect healthy tissues (with what molecules?)
prevents complement on self cells
-uses DAF and MCP
loss of tolerance leads to?
autoimmunity
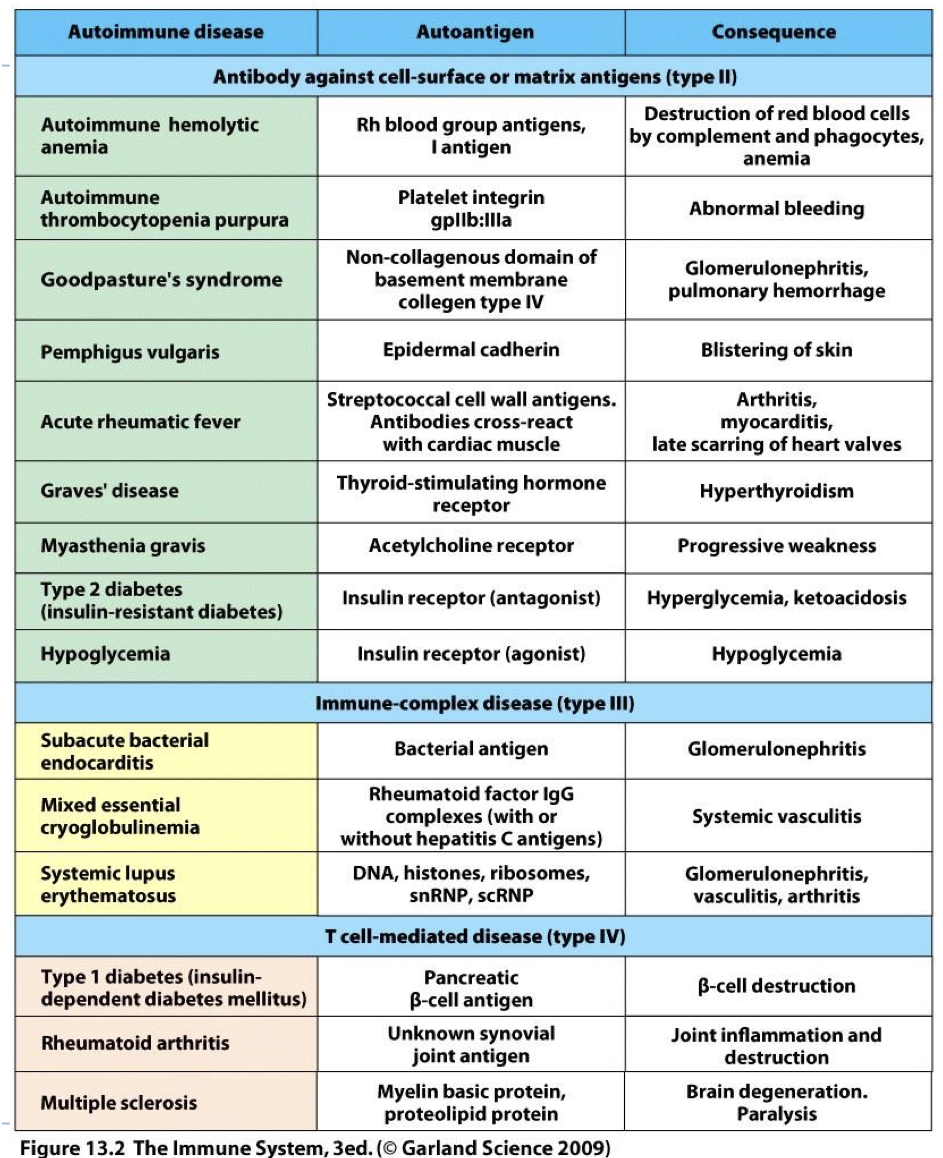
Autoimmune (AI) Disease
Disease in which pathology is caused by an immune response to normal components of healthy tissue.
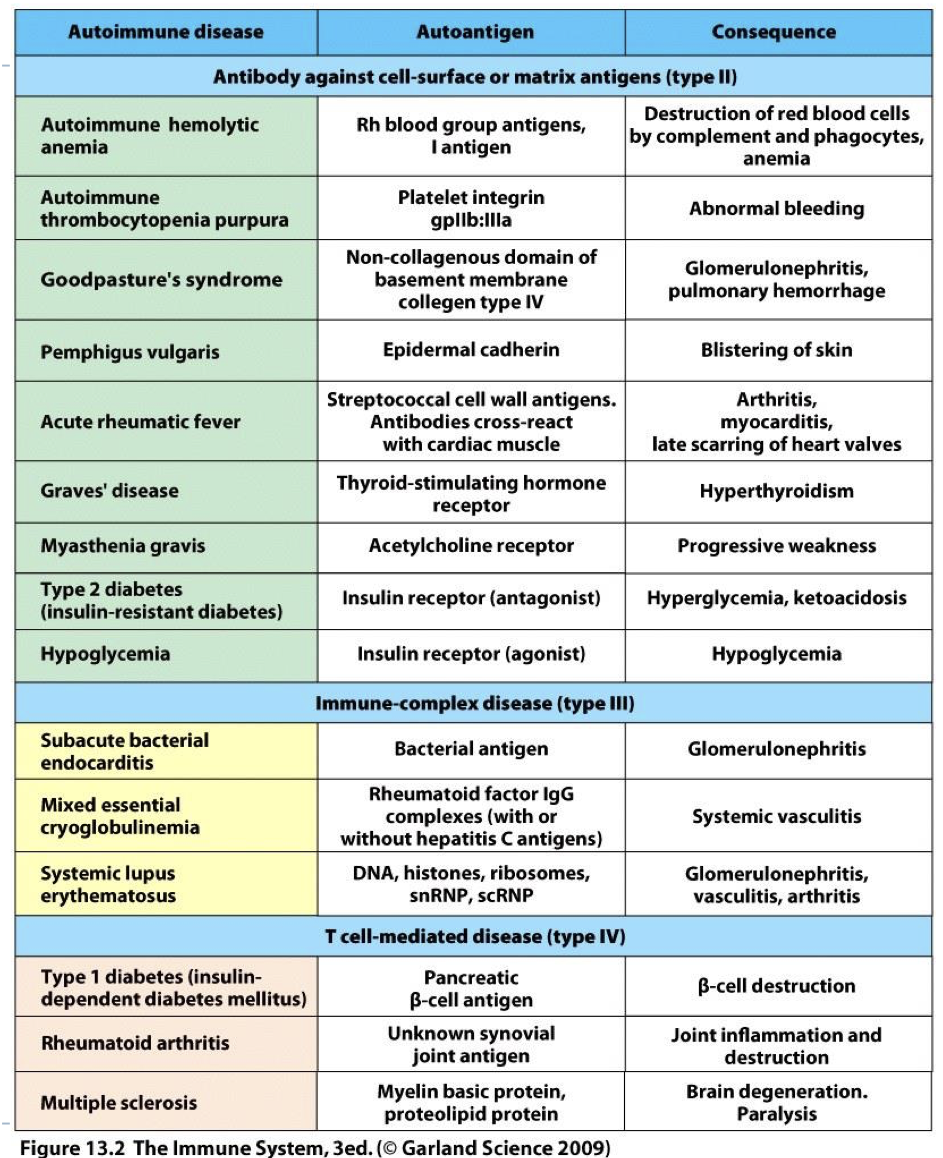
how do we classify autoimmune disease?
fall into 3 categories that parallel hypersensitivity classification by mechanism (II, III, IV).
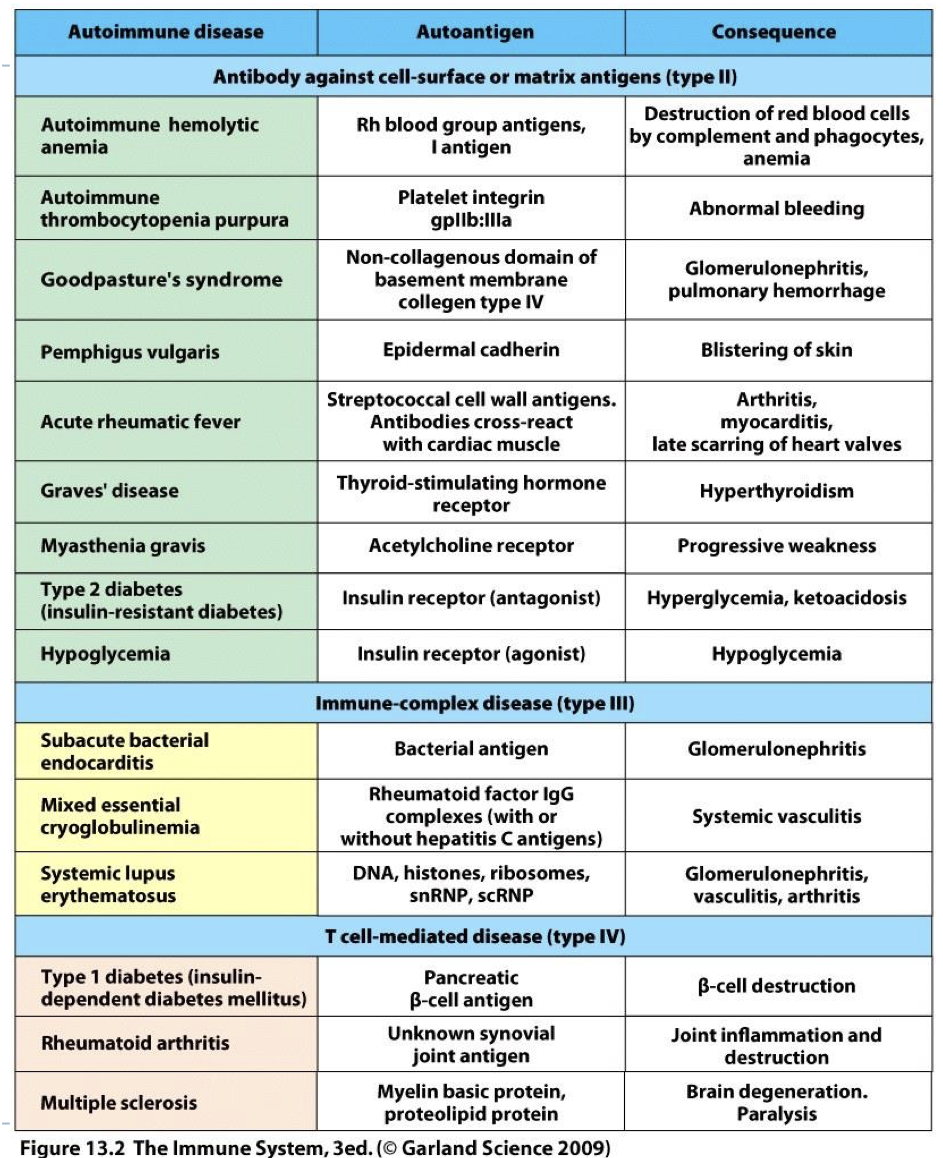
why are autoimmune diseases classified in a way that parallels HS classification? why isn’t there a type one AI class?
-tissue damaging mechanisms are similar to HS mechanisms (Autoimmunity II similar to HS II)
-no AI mechanism stimulates mast cells/granules release seen in autoimmunity (like HS I)
whats the antigen and response called In a HS reaction
antigen: allergen
Response: allergic reaction
whats the antigen and response called In a AI reaction
Antigen: autoantigen
Allergen: autoimmunity
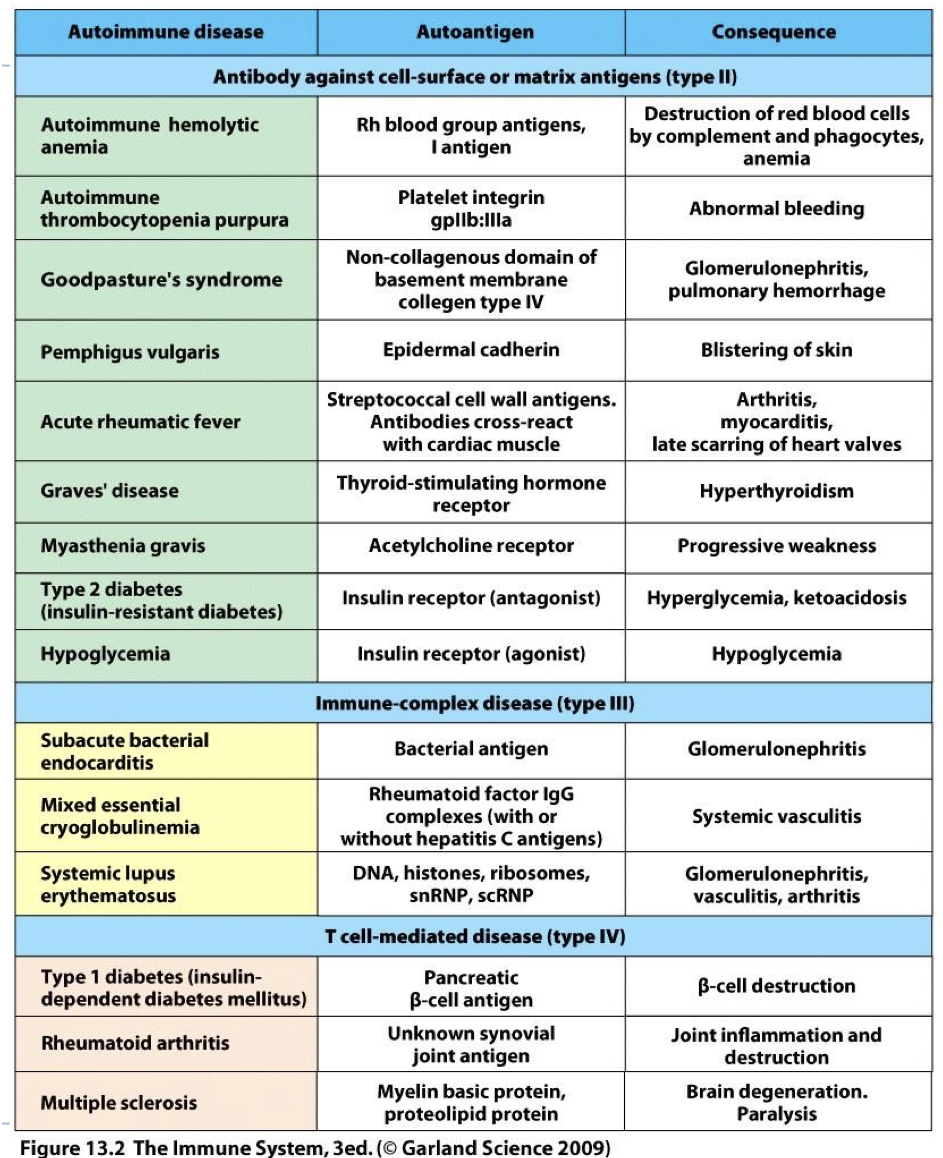
what is the auto antigen in graves disease? what is the consequence?
autoantigen: thyroid stimulating hormone receptor
consequence: hyperthyroidism
what is the auto antigen in type two diabetes (insulin resistant diabetes)? what is the consequence?
autoantigen: insluin receptor (antagonist)
consequence: hyperglycemia, ketoacidosis
what type AI respnose is graves disease?
Type II AI
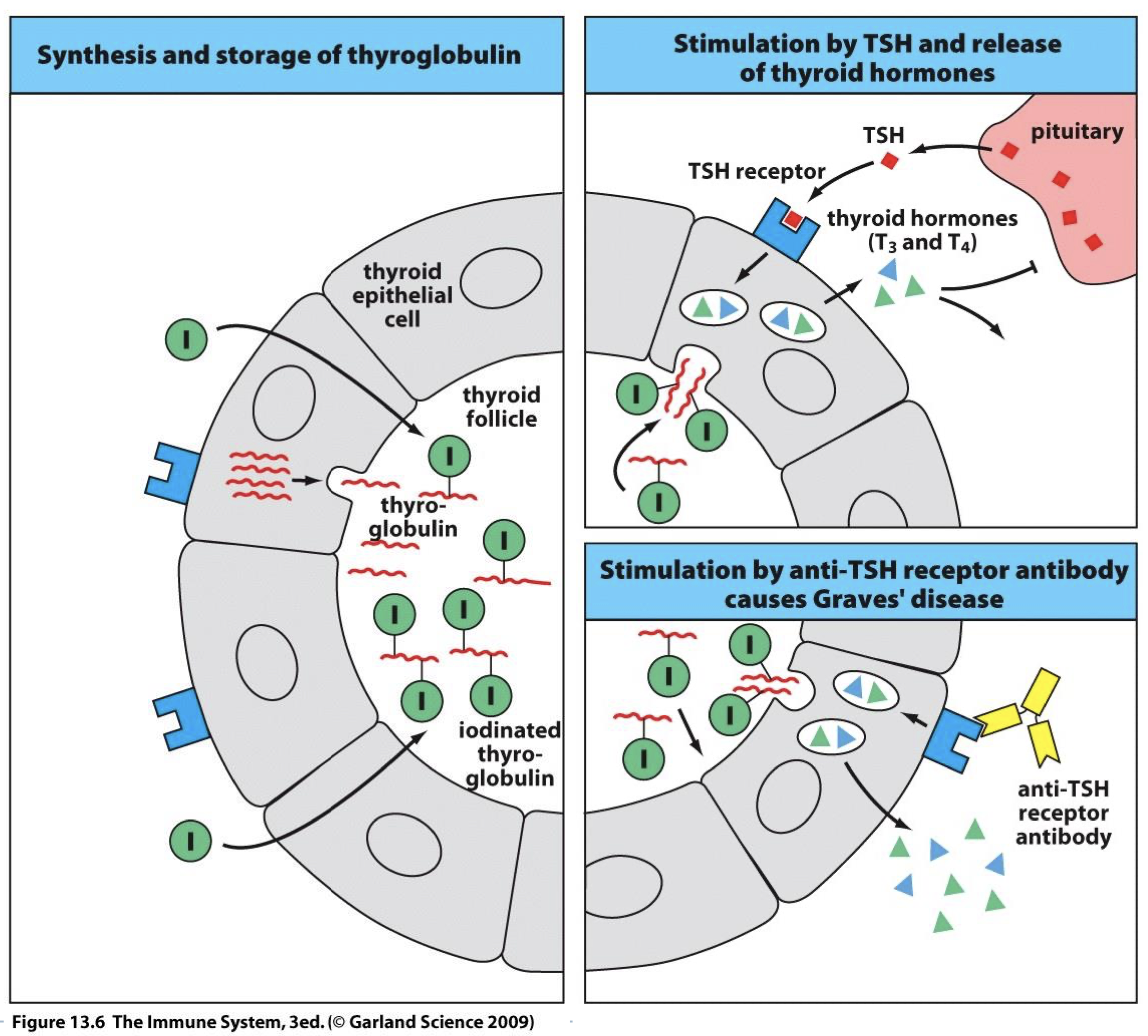
describe normal thyroid function
-epithelial cells of thyroid secrete thyroglobulin into the follicle
-reaction with iodine forms iodinated thyroglobulin
-when metabolism increases we stimulate T3/T4 which binds to TSH receptor on thyroid cells
-T3/T4 stimulates epithelial cells to endocytose iodinated thyroglobulin and allows cells to produce T3/T4
-sends feedback to turn off pituitary since product is made and we stop stimulating epithelial cells
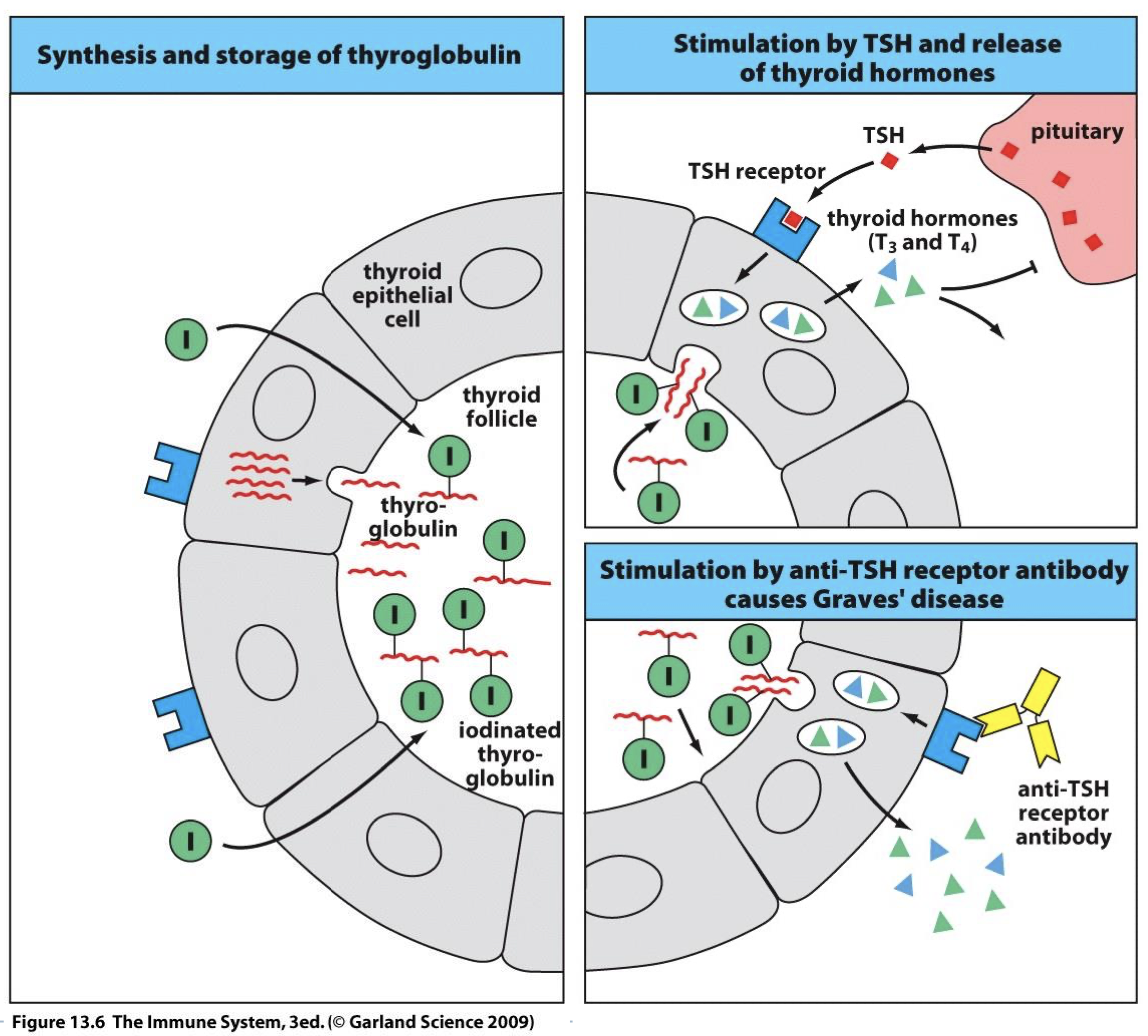
describe thyroid function with graves disease
auto antibody binds auto-Ag (TSH receptor) causing constant production of T3 & T4 that’s independent from pituitary signals leading to hyperthyroidism
hyperthyroidism symptoms
Heat intolerance, nervousness, irritability, warm moist skin, weight loss, enlarged thyroid, bulging eyes
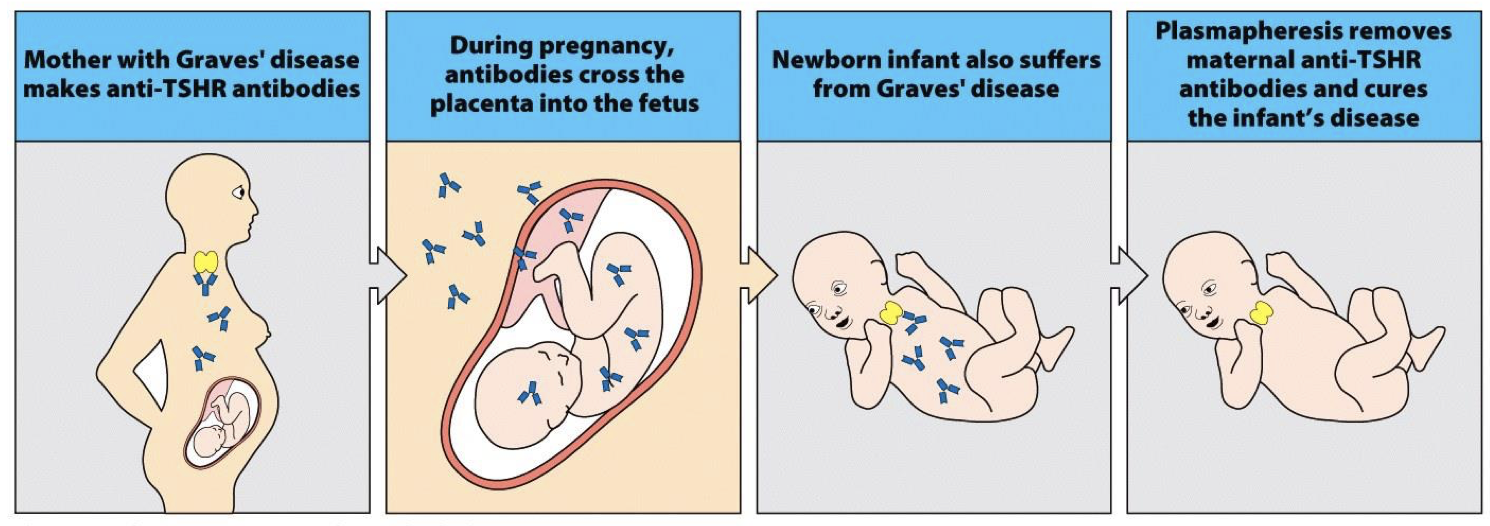
explain how pregnancy can determine the cause of a disease
those with AI diseases are healthy for a long time but once tissue destruction begins, its damage has become irreversible by the time of diagnosis and we become unsure if damage was from disease or cause of disease →
-but we learned from AI that pregnant women with AI pass auto-IgG and anti TSHR to baby → baby born with autoimmnune disease will exhibit symptoms till 6 months (when mothers Ab degrades and produces own Ab) and baby will be symptom free →lymphocytes can’t cross placenta, telling us this was an autoimmune disease
describe the diabetes epidemic in the US
29.1 million Americans have diabetes (as of 2012)
90-95% of the cases are Type II Diabetes
The number of new cases of diabetes has tripled from 1980 to 2011.
Diabetes is the 7th leading cause of death in the US in 2010 (70,000 people)
For more than 3x as many deaths diabetes was listed as a condition contributing to death (complications from diabetes kill)
what complications arise from diabetes
Hypoglycemia, hypertension, high LDL, cardiovascular disease, heart attacks, stroke, blindness, kidney disease, amputations
how does type two diabetes work?
Type II diabetes is a disease that affects the way the body metabolizes glucose. The body resists the effects of insulin, or doesn’t produce enough insulin.
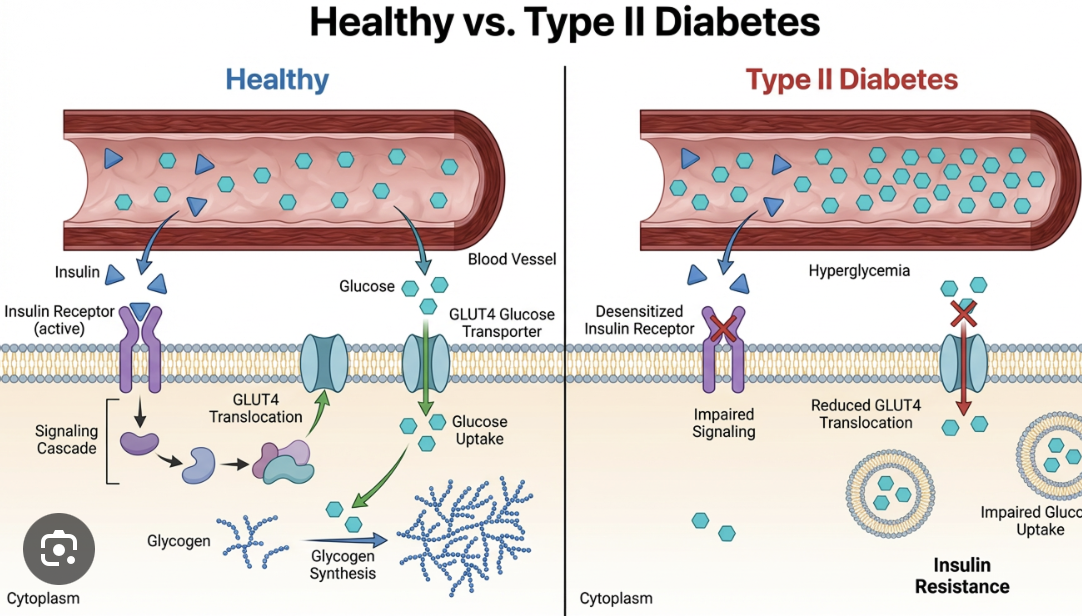
what is diabetes II being classified as now? why?
Previously this was thought to be a metabolic disorder associated with obesity.
Now there is evidence that it is an autoimmune disease.
Mice with Type II diabetes normalized blood sugar levels through treatment with anti-CD20 (depleting B cells) (Winer DA., Nature Medicine 2011) (CD20=CD19 in Mice B cells (lineage)→ able to normalize blood sugar w/o B cells (Big news!))
B cells produce IgG auto-Ab that bind to targets linked to insulinresistance. (Winer DA., Cell Mol Life Sci 2014)
what type AI is diabetes II
its a type II AI
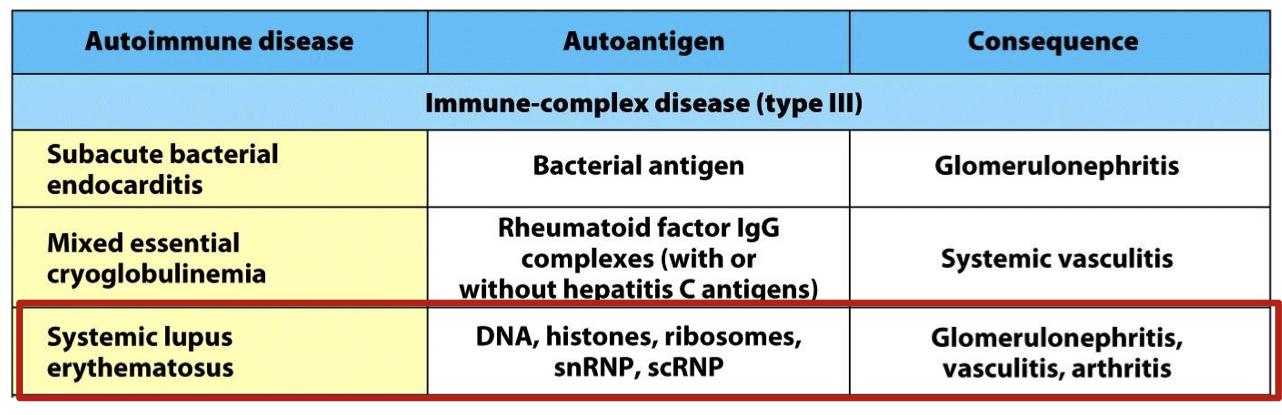
what type AI is systemic lupus erythematous
AI type III
what is the auto antigen and consequence of systemic lupus erythematous
autoantigen: DNA, Histones, ribosomes, snRNP, scRNP
Consequence: Glomerulonephritis, vasculitis, arthritis
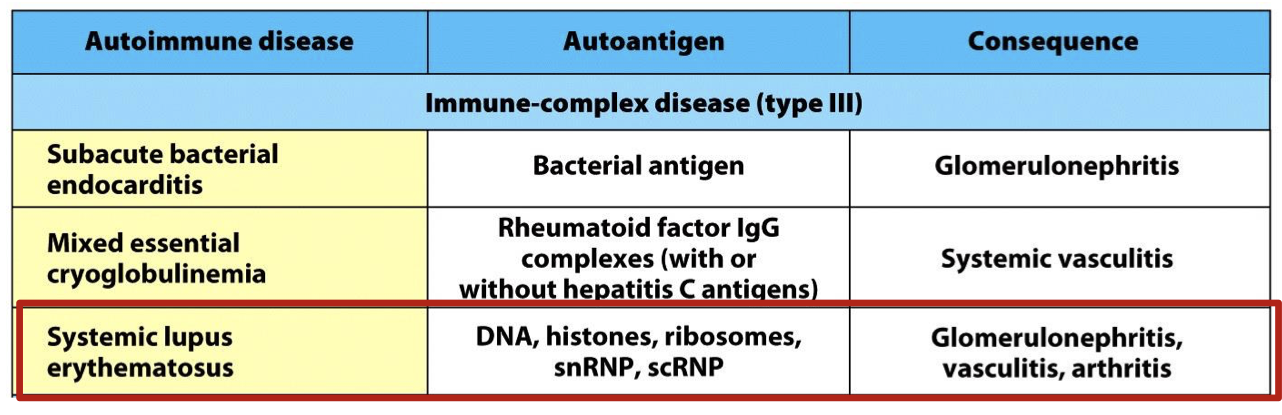
how does systemic lupus erythematous arise?
Auto-Ab react with Auto-Ag throughout the body, destroying self tissues and releasing soluble Ag.
Soluble Ag and Auto-Ab = Immune Complex deposited & inciting inflammation
what does SLE lead to (couse of disease)
Variable disease course, leads to death due to failure of vital organs (depending on where it occurs)
what are examples of the consequences of Type III AI: Systemic Lupus Erythematosus (SLE) what causes them?
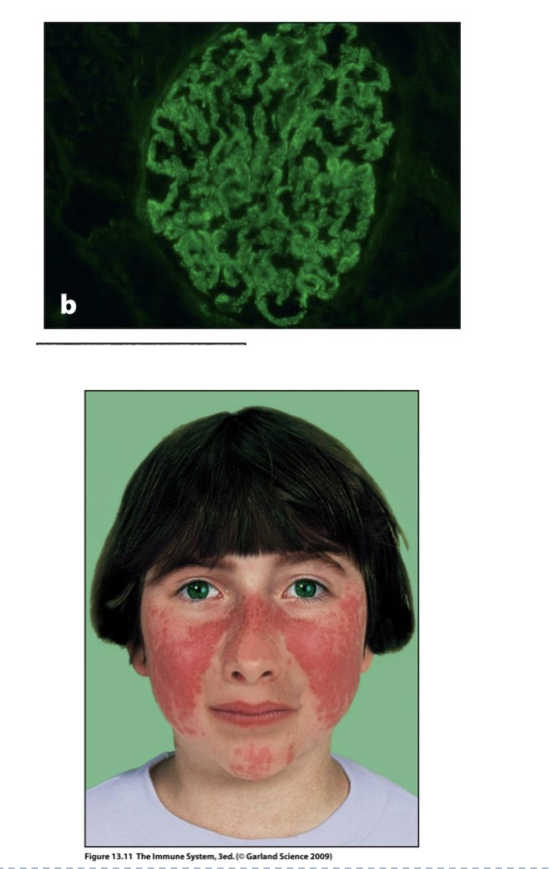
Green = anti-Ig Ab shows Auto-antibodies in green, deposited on basement membrane of kidney section.
Butterfly shaped skin rash (erythema) can appear on face due to immune complexes in the skin
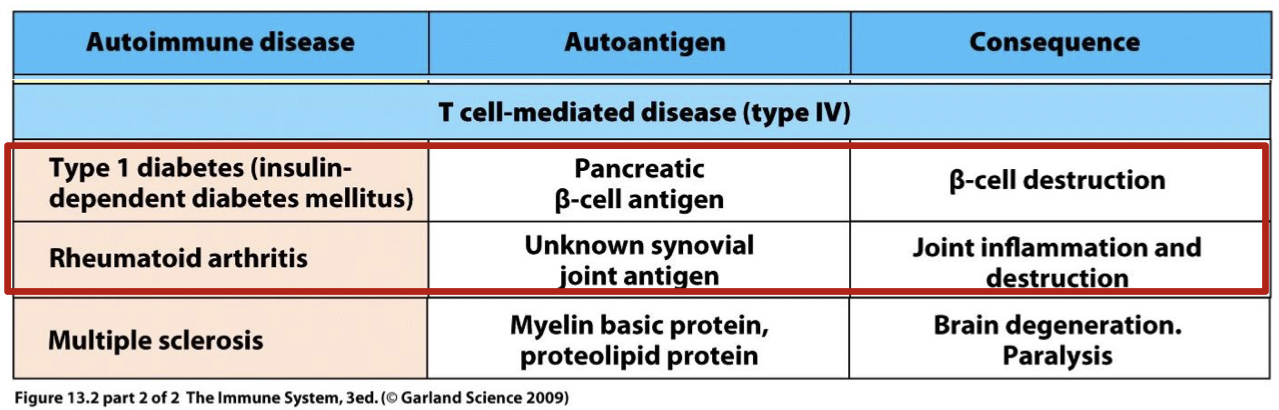
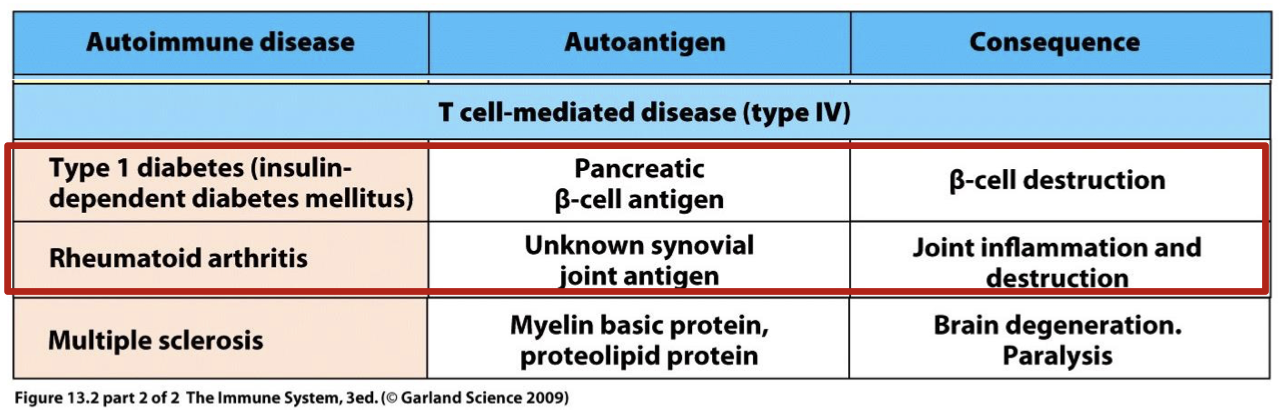
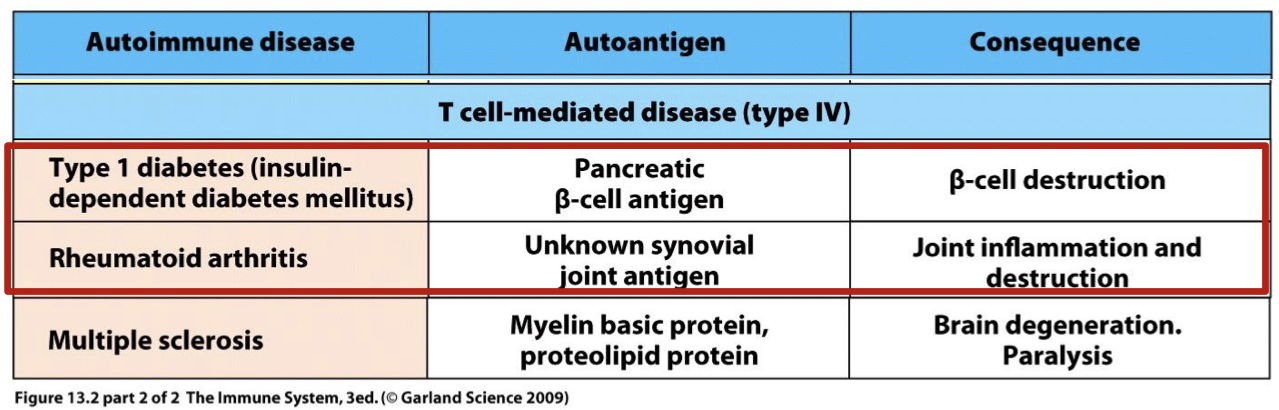
unlike types II, and III type IV AI is? example diseases are?
T cell mediated
type one diabetes and rheumatoid arthritis
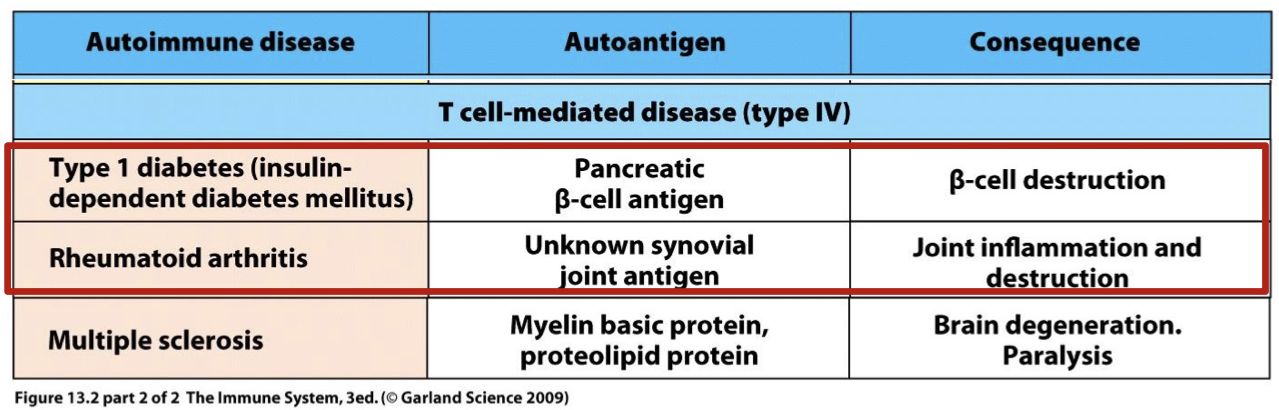
what is the auto antigen and consequence in type one diabetes (insulin dependent diabetes mellitus)
autoantigen: Pancreatic (beta) cell antigen
consequence: Beta cell destruction
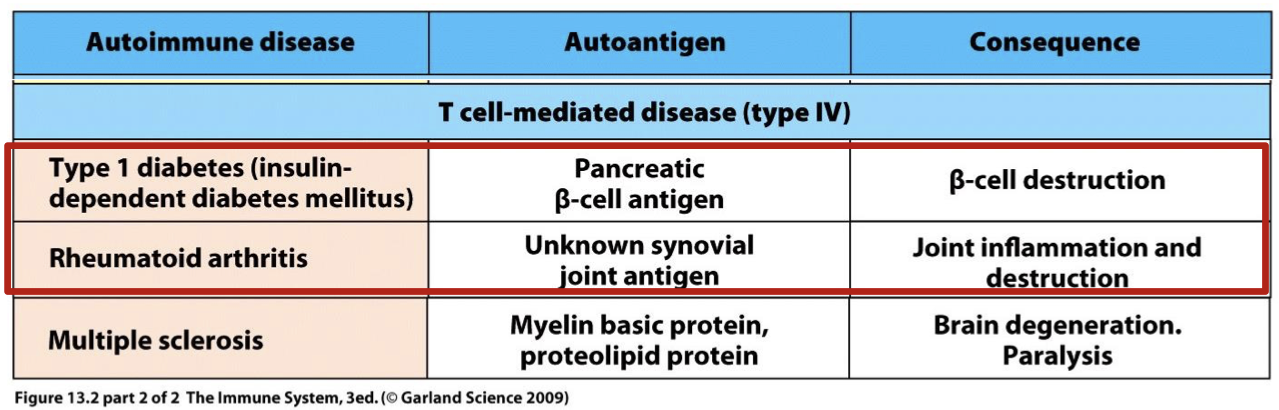
what is the auto antigen and consequence in rheumatoid arthritis
autoantigen: unknown synovial joint antigen
consequence: joint inflammation and destruction
what causes type one diabetes?
Caused by T cell responses that destroy insulin producing pancreatic cells (NO INSULIN PRODUCED AT ALL → unlike type 2)
what causes Rheumatoid Arthritis (RA)
Caused by T & B cell responses in joints that cause debilitating inflammation in joints & surrounding tissues
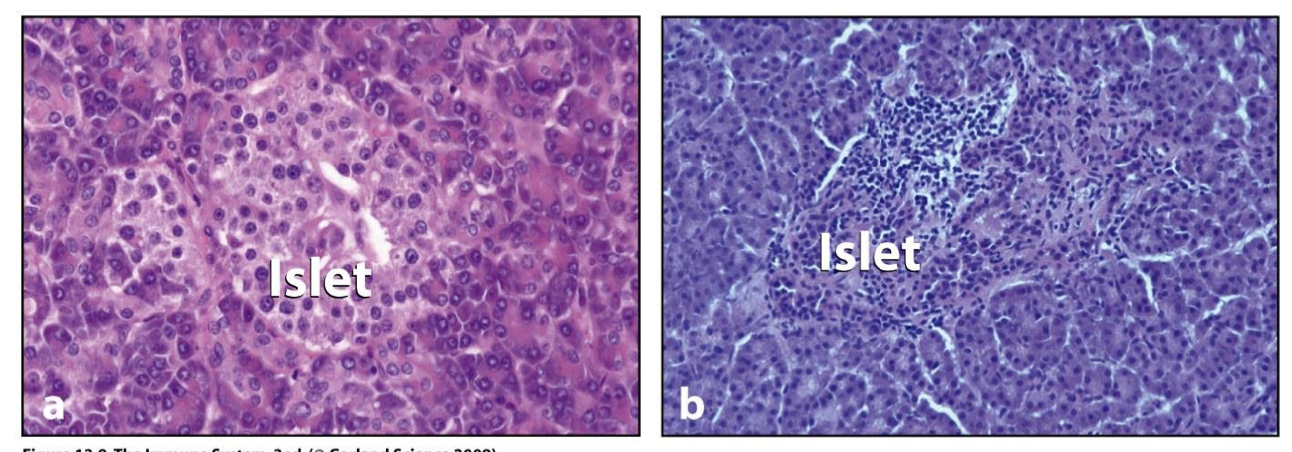
describe the process that causes type one diabetes
Islets in pancreas make hormones such as alpha cells – glucagon, beta cells – insulin, delta- somatostatin
CD8T targets B cells causing Insulitis
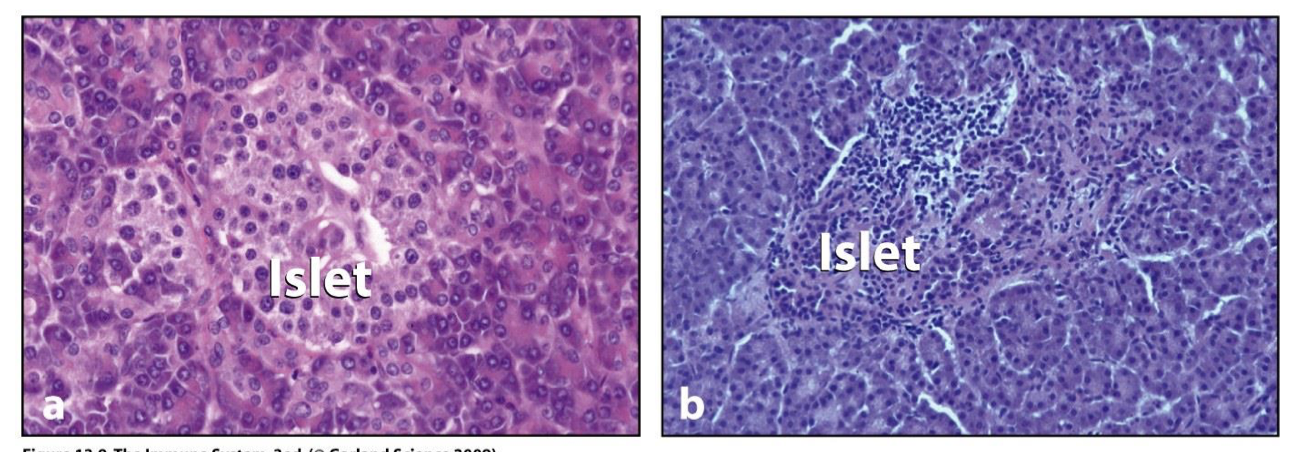
islets
Pancreas composed of small clumps of endocrine cells (islets) that make hormones
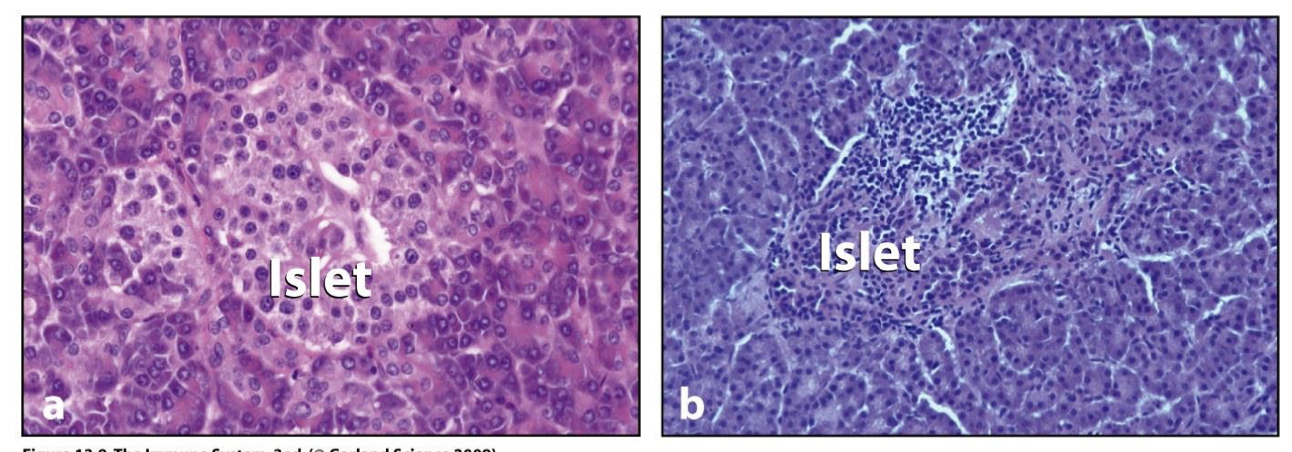
Insulitis
islets infiltrated with lymphocytes
alpha cells/Glucagon function
control glucose levels and prevents them from getting low (stimulates glycogen conversion to glucose)
beta cells/ insulin function
lowers glucose levels (10^8 B cells per islet and produce a LOT of insulin in healthy islets)
delta cells/somatostatin function
helps regulate endocrine function, inhibits insulin, growth hormones and gastrin
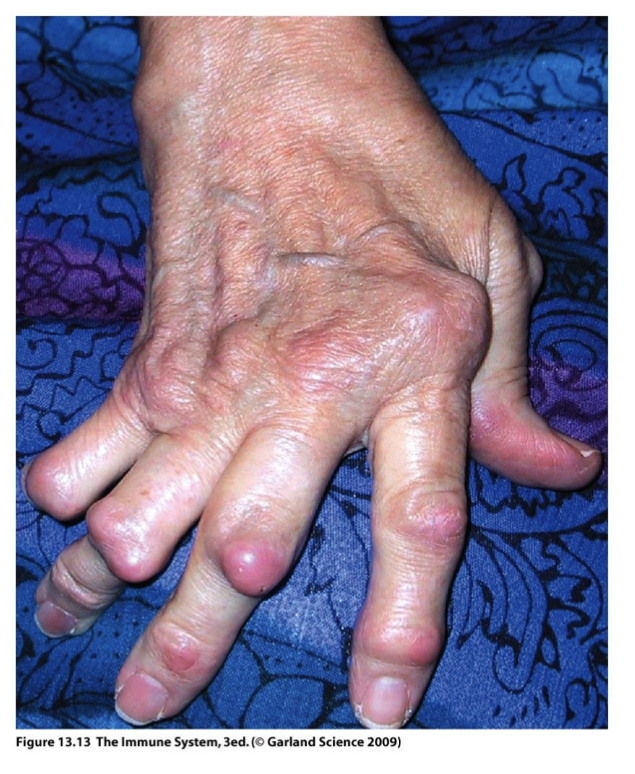
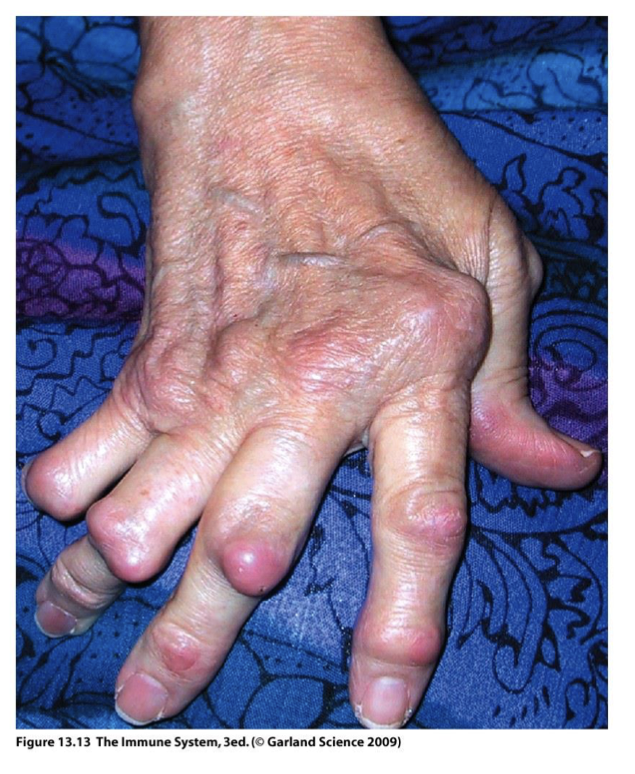
Type IV AI: Rheumatoid Arthritis (RA)
chronic inflammation and flares caused by accumulation of immune complexes in joints
Inflammation damages joint, cartilage, ligaments, tendons, & bones.
in joints affecting 1-3% of population (3:1 women to men) in ages 20-40 years old
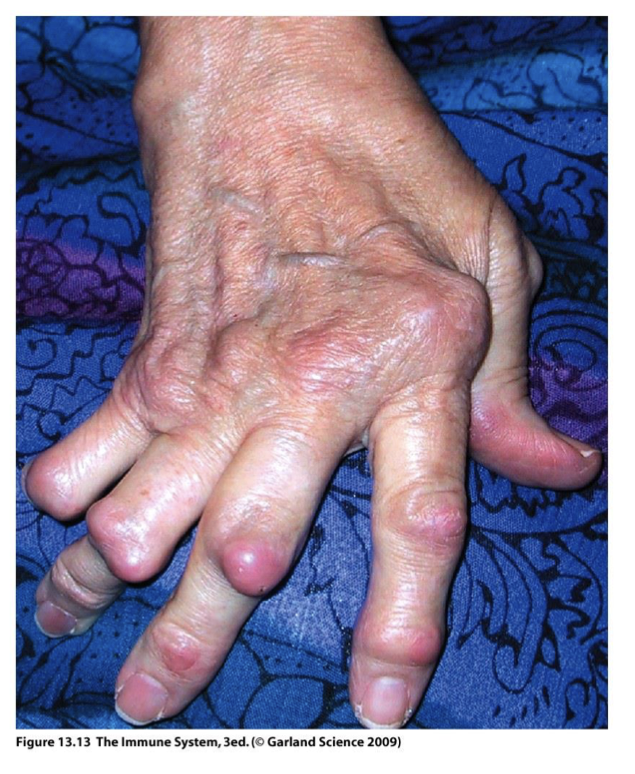
80% of patients suffering from Type IV AI: Rheumatoid Arthritis (RA) have? what is it?
Rheumatoid Factor (anti-Ig auto-Ab)
Rheumatoid Factor
(anti-Ig auto-Ab)
specific for fc IgG regions → making Ab that binds to the Fc fragment of any IgG in the body
90% of SLE patients suffer from?
Type IV AI: Rheumatoid Arthritis (RA)
why is Type IV AI: Rheumatoid Arthritis (RA) type four if its antibody mediated?
20% of patients dont have Rheumatoid Factor but still present with the disease → suggested T cell mediation is cause → may shift back to HS III due to ab presence but more research is needed
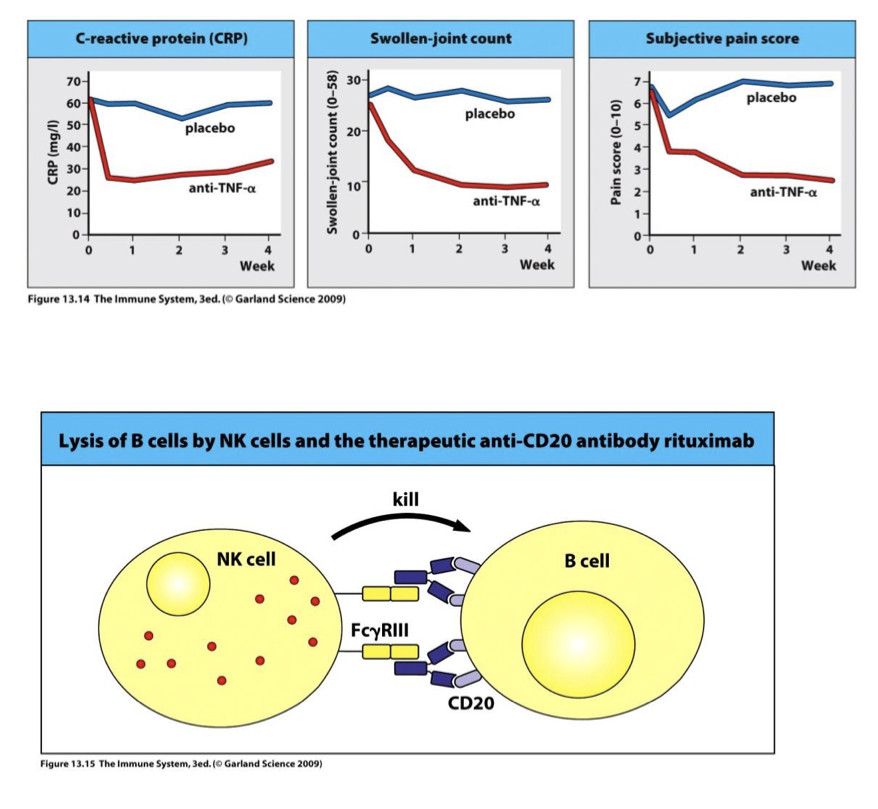
Type IV AI: Rheumatoid Arthritis (RA) treatment possibilities
Treatment with (anti) TNFa(alpha) mAb → reduced swelling & pain (top)
Treatment with anti CD20 mAb → deplete B cells via ADCC NK(alpha) mAb → reduced swelling & pain (bottom)
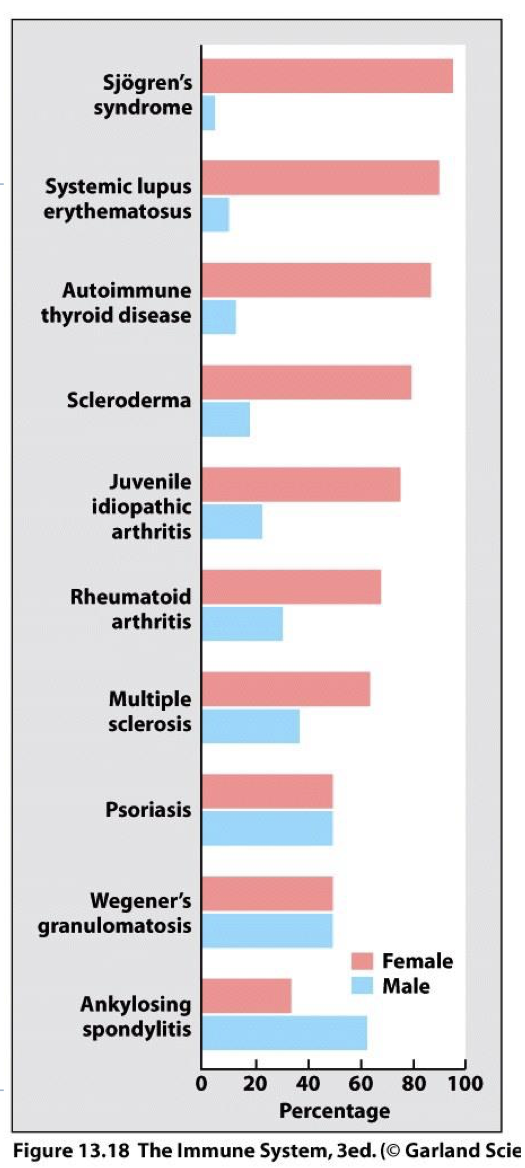
is gender a possible predisposition for autoimmune disease?
yes, it often occurs more in females then males (8% of the population who suffers from AI → 78% are women but we are not sure why)
are HLAs a possible predisposition to AI?
yes, HLA Complex Variations account for 50% of genetic predisposition to AI disease. (HLA I and HLA II)
Linkage Disequilibrium
– alleles of two or more polymorphic genes are inherited together at frequencies higher than by chance. (8.1 haplotype associated with AI disease)
(essentially, genes inherited as groups making it difficult to pinpoint what genes cause AI)
what genes make you more succeseptiable to type I diabetes
Carrying both HLA-DQ2 and HLA-DQ8
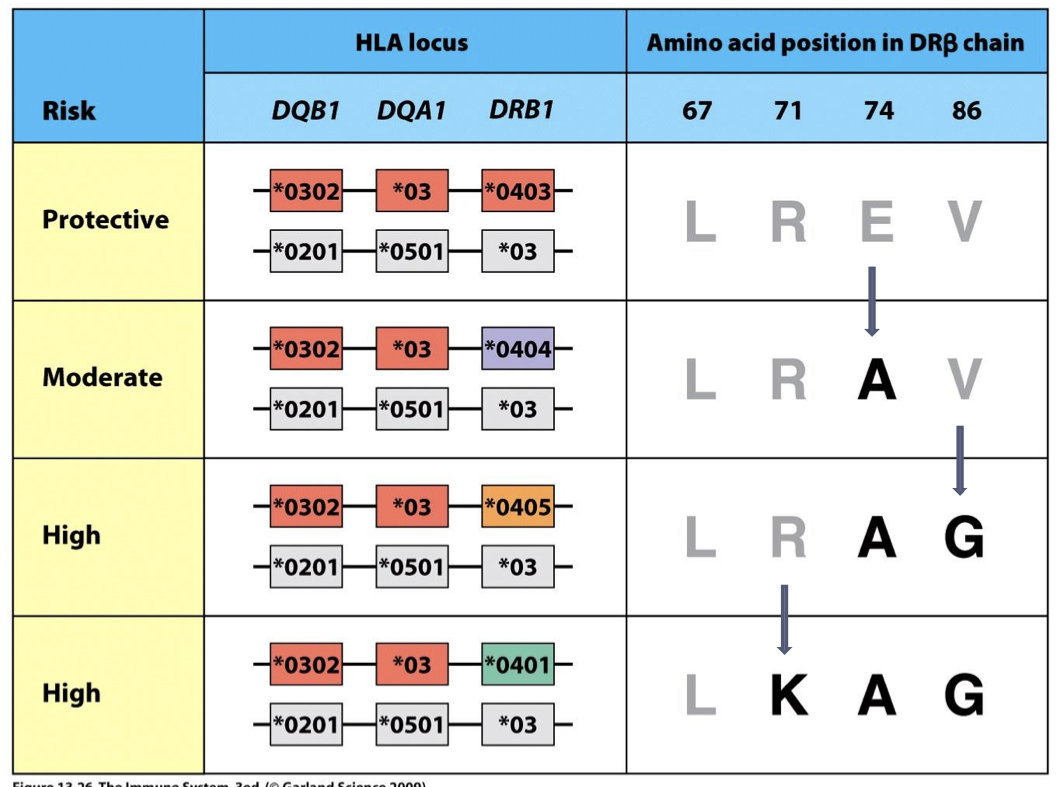
what can a single AA substitution determine?
Single AA substitutions can determine whether a HLA- DRB1*04 subtype confers susceptibility to or protection from Type I diabetes.
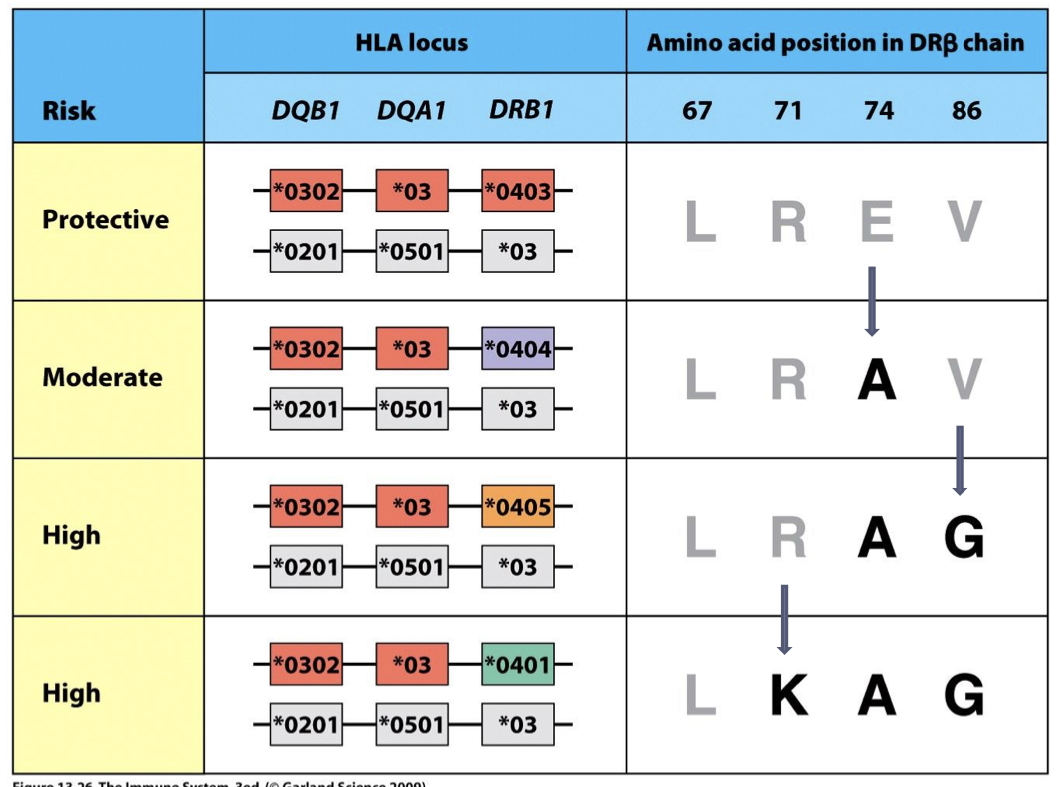
what gene provides dominant resistance to type I diabetes? (even the paired with susceptible allele)
HLA-DQ6
describe how HLA Single Amino Acid Substitutions provide Susceptibility vs. Protection
(see image) A person can go from being protected from Type I Diabetes to being susceptible with a single AA substitution in the DR(alpha) chain.
what environmental factors can be predispositions to autoimmune diseases?
smoking (ie. goodpastures syndrome)
physical trauma
infection (ie. rheumatic fever)
how is smoking a pre dispassion to AI?
AutoAb against collagen of basement membranes (kidney disease in all patients)
Pulmonary hemorrhage only in habitual smokers (because basement membrane of lug alveoli are damaged, giving circulating Ab access to B.M where immune complexes deposit)
how is physical trauma a pre dispassion to AI?
Gives Auto-Ab access to immune privileged site (brain, eyes, uterus)
Eye – blow to Anterior chamber causes immune response- blindness
how is infection a pre dispassion to AI?
AI can be byproduct of an infection
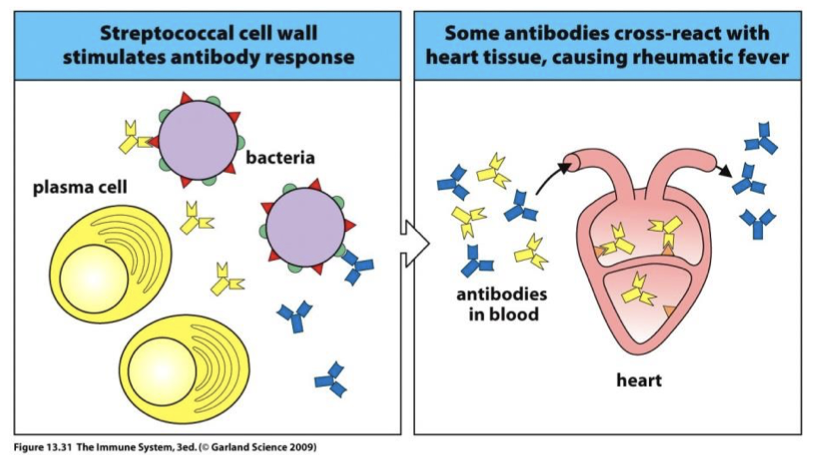
Throat infection with S. pyogenes. 2-3 wks later inflammation of heart, joints, kidneys (pathogen and host antigen have similar structures)
Ab to S. pyogenes self react to heart tissue, causing widespread inflammation (Rheumatic Fever) and can cause heart failure. (molecular mimicry)
molecular mimicry
pathogen and host antigen have similar structures (VERY BAD)
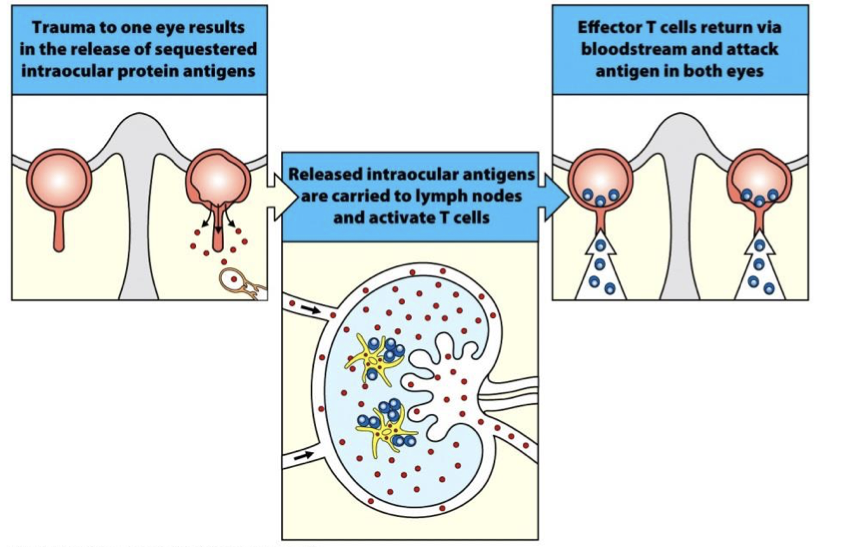
Physical Trauma & Infection Affect AI Disease Predisposition (explain eyes)
Damage to the eye permits T cells to enter area of immune privilege.
Physical Trauma & Infection Affect AI Disease Predisposition (explain infection)
Infection elicits immune response. Molecular mimicry cause Ab to cross-react with self- tissue.
three transplant types
1) Blood Transfusion
2) Solid Organs
3) HSC in Bone Marrow
3 barriers to overcome during transplantation
1) Perform Function After Transplant
2) Health of Recipient & Donor
3) Avoid Rejection (needs work)
what disease concerns can we seen in solid organ, bone marrow transplants as well as genetic differences?
Solid Organ – Hyperacute, Acute, & Chronic
Bone Marrow – Graft vs. Host Disease
Caused by genetic differences in donor & recipient HLA I & II
what is an antigen and response called during reactions to transplants?
antigen: alloantigen
reaction: alloreaction
what are two complications from transplantations?
organ transplant rejection
graft vs host disease
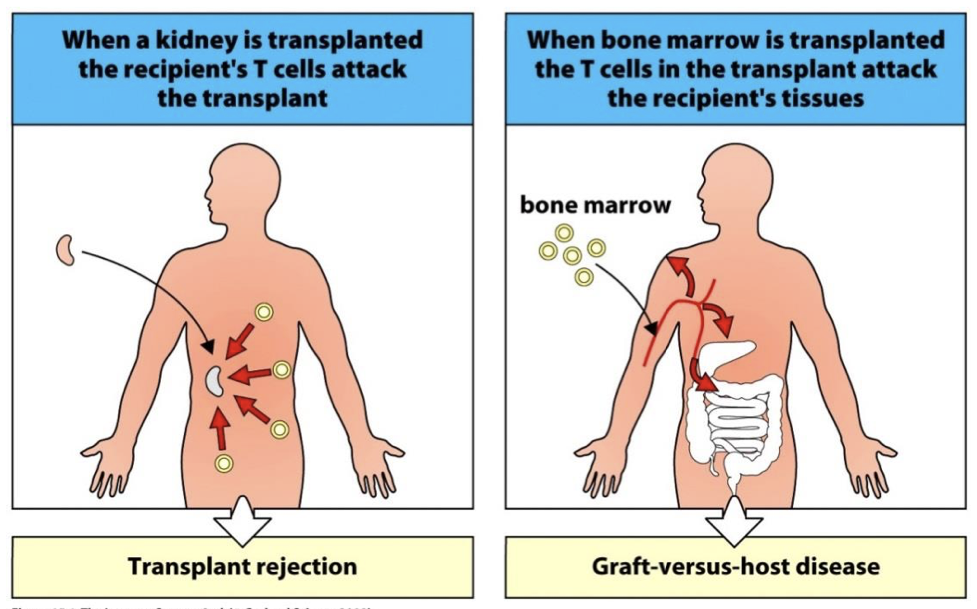
organ transplant rejection
seen in Solid Organ Transplant
Recipient immune response kills cells of the Donor organ
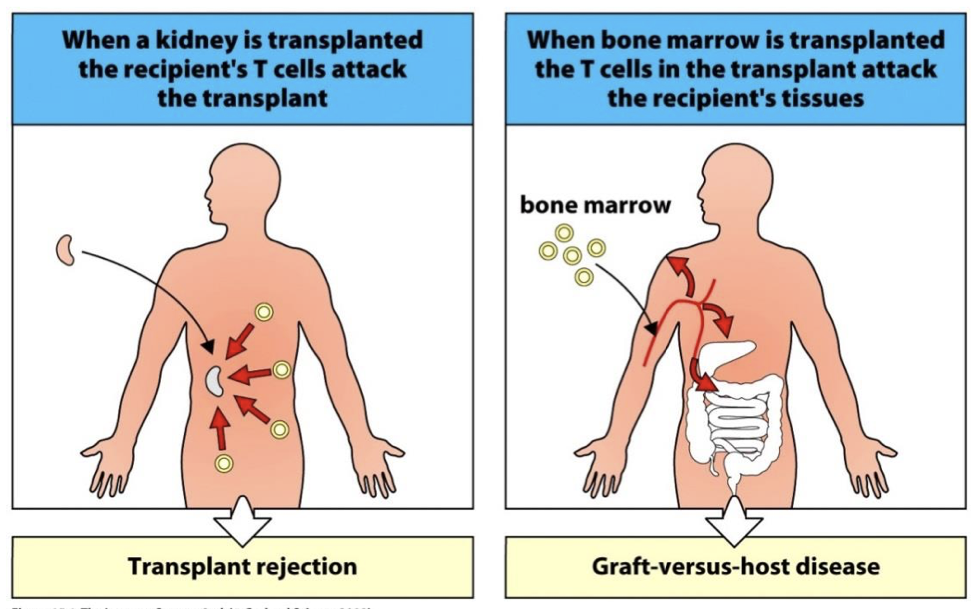
Graft vs host disease (GVHD)
seen in Bone Marrow Transplant
Mature T cells in marrow attack recipients healthy tissue.
donor vs recipient?
donor gives oran and recipient gets it
histroy of blood transfusions
1st blood transfusion to save a life was in 1812
Today 1 in 4 people will receive a blood transfusion (trauma, surgery, childbirth, severe blood loss)
3 types of donated blood (and their uses)
RBCs – to improve respiration & metabolism
Platelets – prevent bleeding & help clotting
Plasma – for fluid replacement & prevent bleeding
do RBCs require matching HLA?
no only worry about surface antigens
surface antigens of RBCs
A,B, Rhesus
Other surface Ag that recipient may react to
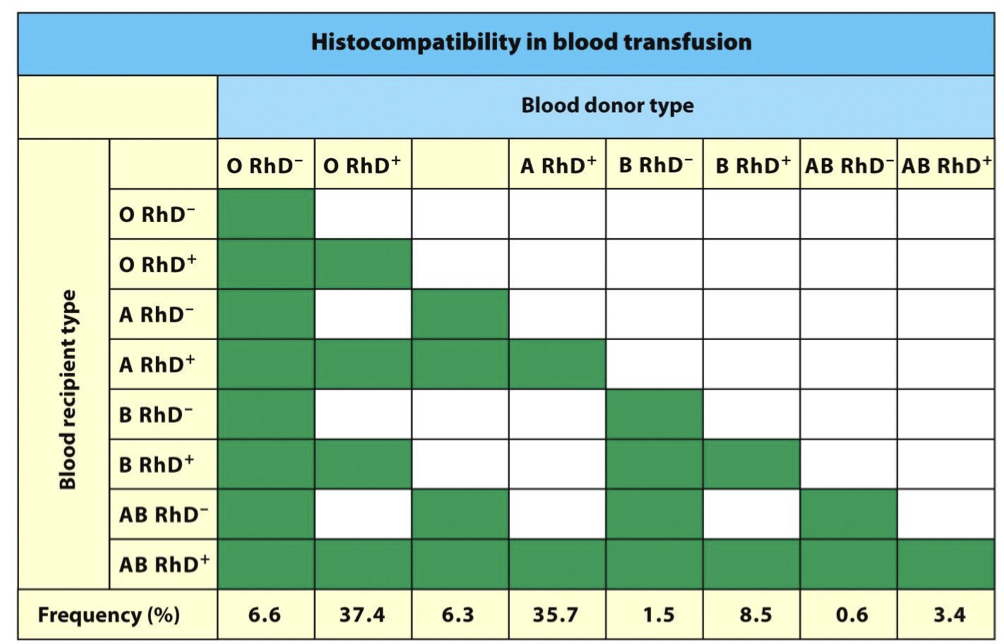
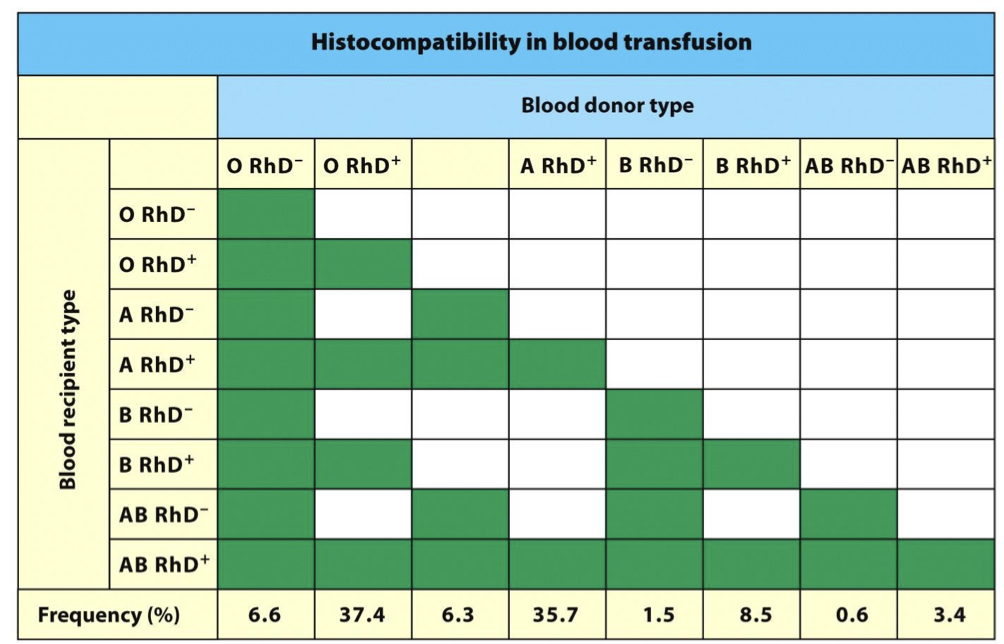
universal donor blood type
O Rh-
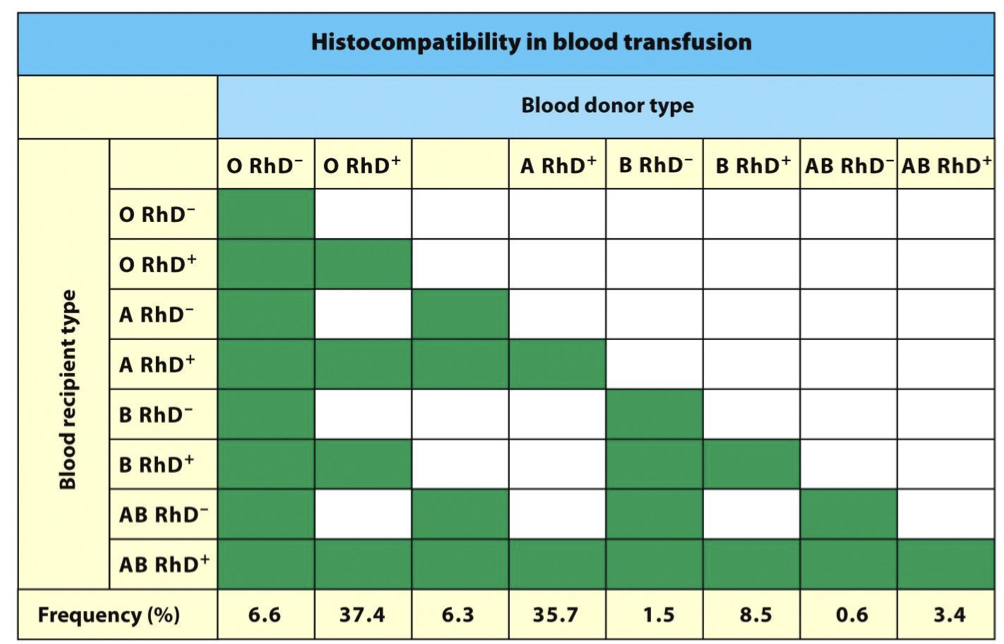
universal recipient blood type
AB Rh+
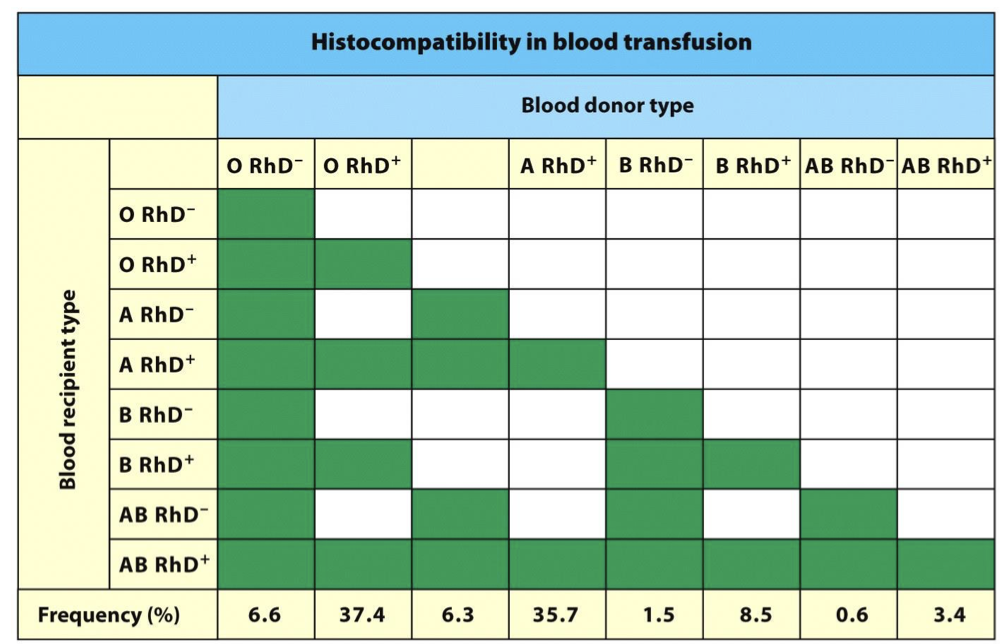
matching a blood donor requires?
KNOW IMAGE
ABO & Rh match
Cross Match Test determines if patient Ab will cross react with the donor RBCs.
cross test match
blood samples from each person combind to see if HS II ran occurs (NOTE: -/+ RH react with one another)
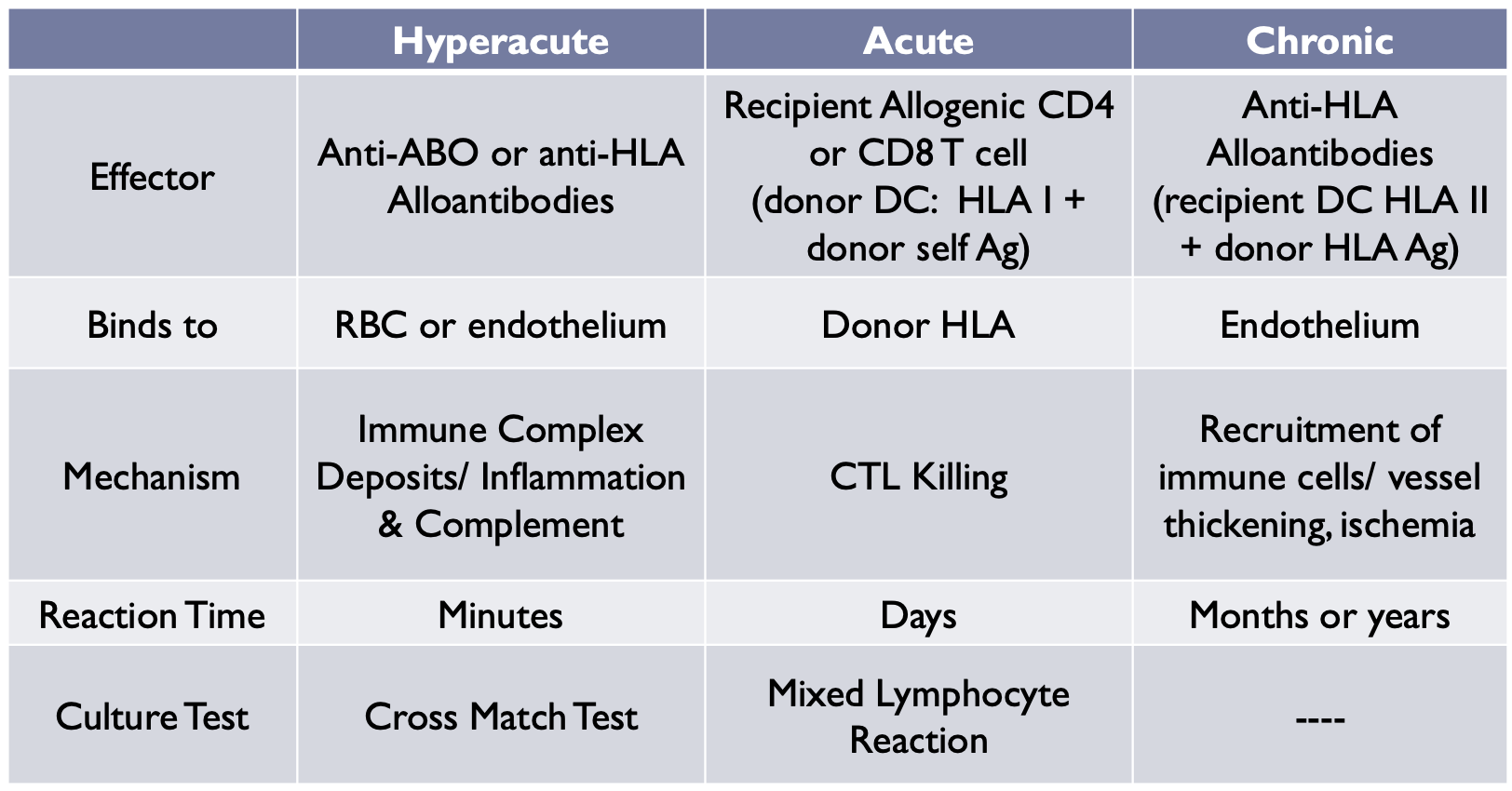
3 types of organ rejections
hyper acute, acute, chronic
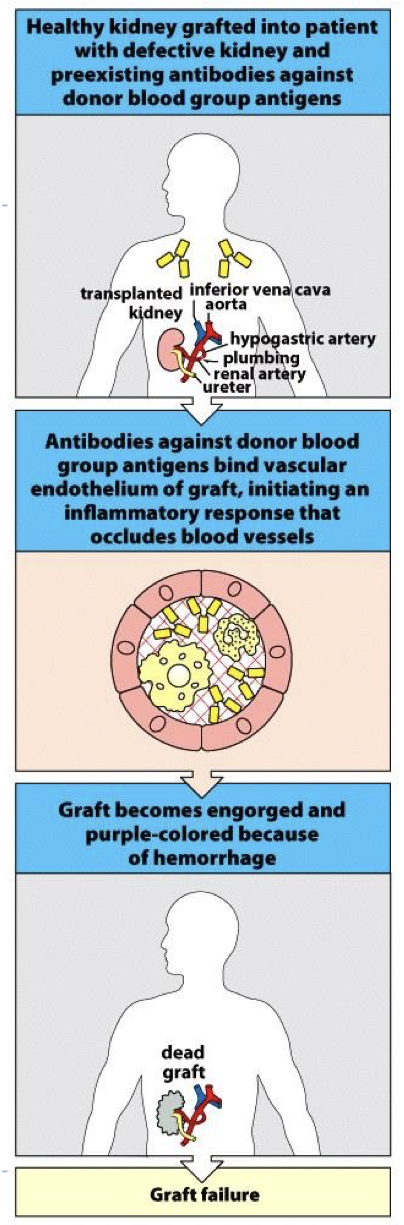
hyper acute rejection is due to? how is it prevented
pre- existing Ab in recipient.
Immune complexes form on vessels → inflammation → complement → rejection/organ loss
Prevented by HLA and ABO matching and Cross Match Test
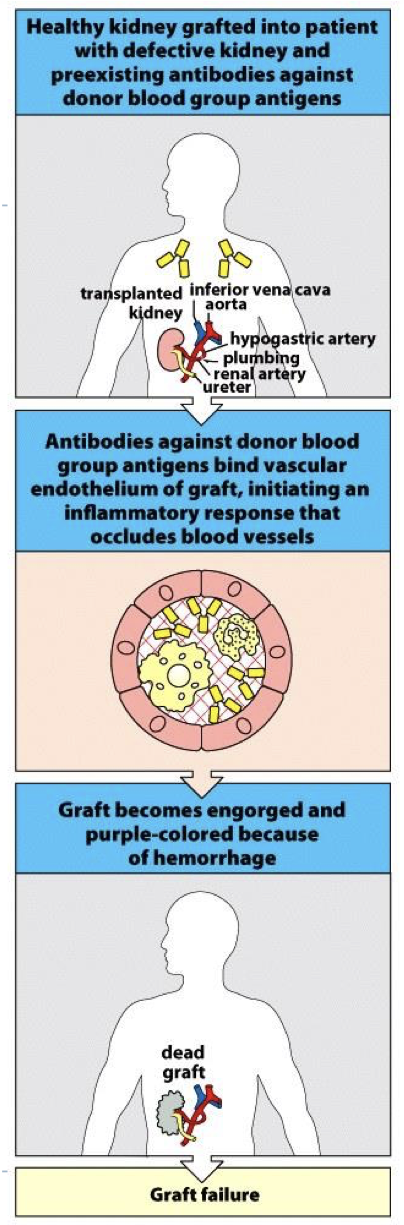
why Is it known as hyper acute rejection?
occurs immediately (on the table quick) → AB in body specific for AG on organ stimulant complement/rejection reactions
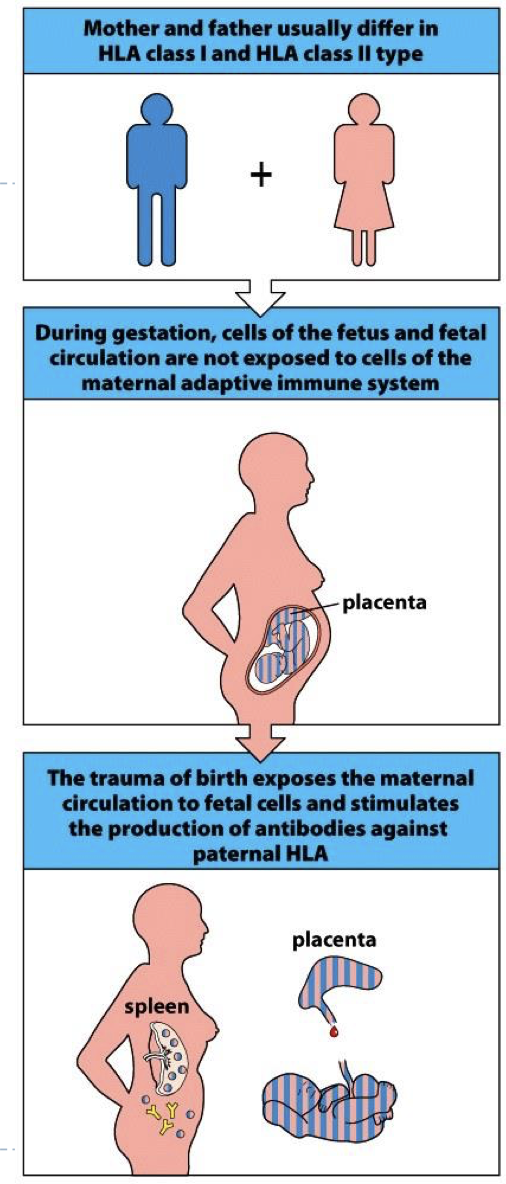
Where do anti HLA Ab come from in recipient?
1) Pregnancy
2) Blood Transfusion (possible)
3) Previous Organ Transplant (multiple transplants = more AB)
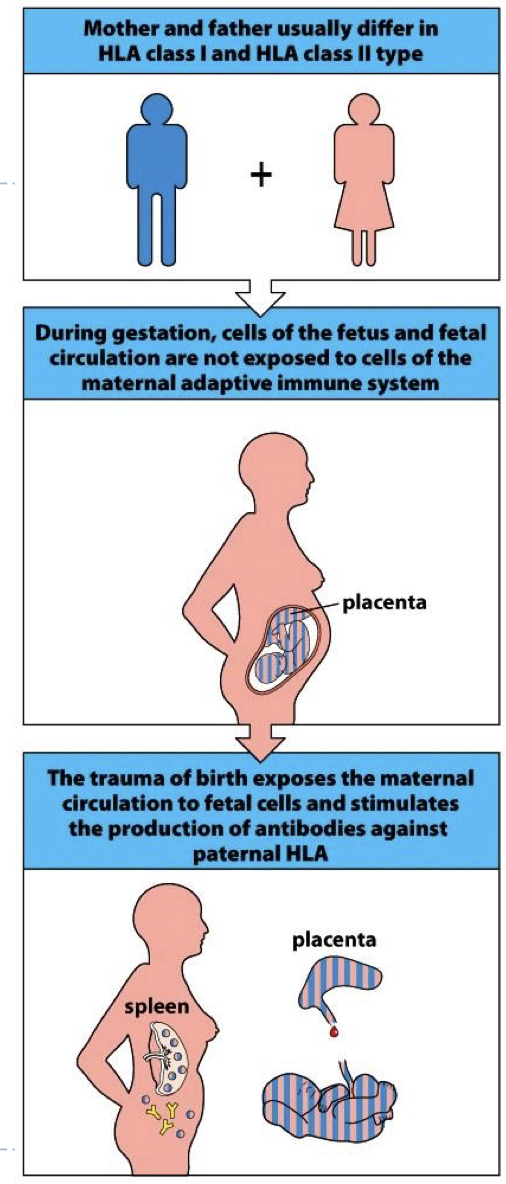
how does pregnancy cause anti HLA ab in recipient?
baby has one HLA from mom and dad (more pregnancies menas more AB)
acute rejection is caused by?
Acute rejection is due to recipient alloreactive T cells attacking graft by allorecognition
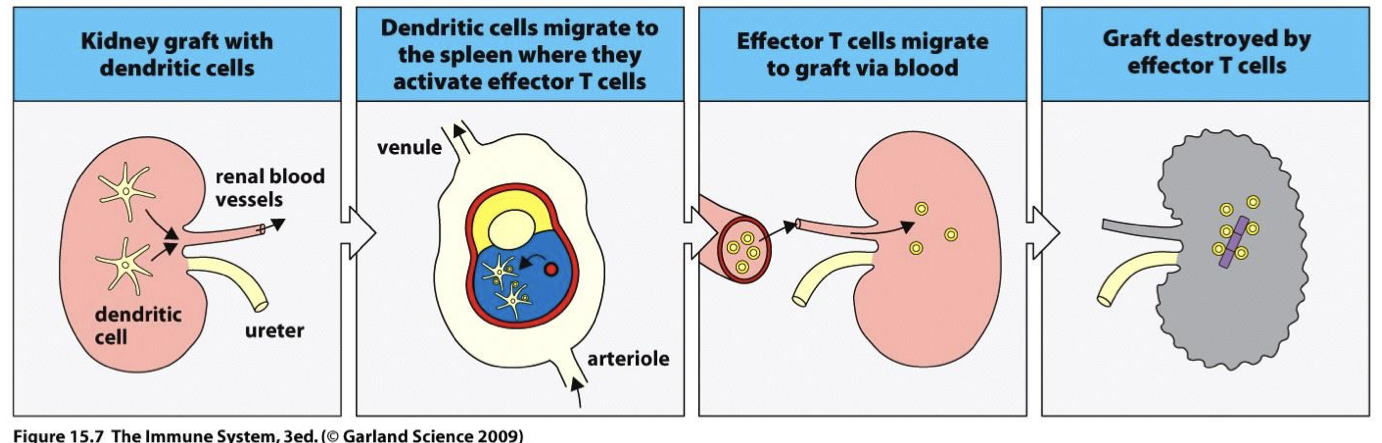
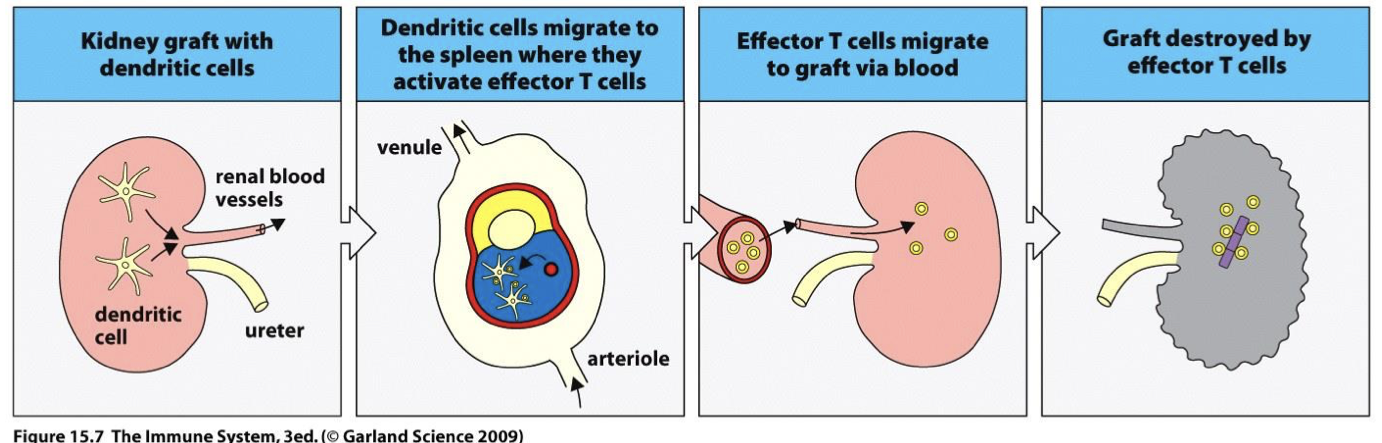
acute rejection process (and how long it takes)
Inflamed organ activates donor DCs → Donor self Ag presentation to recipient T cells in lymph node → CTL killing of organ (aka allogenic T cells) (takes days)
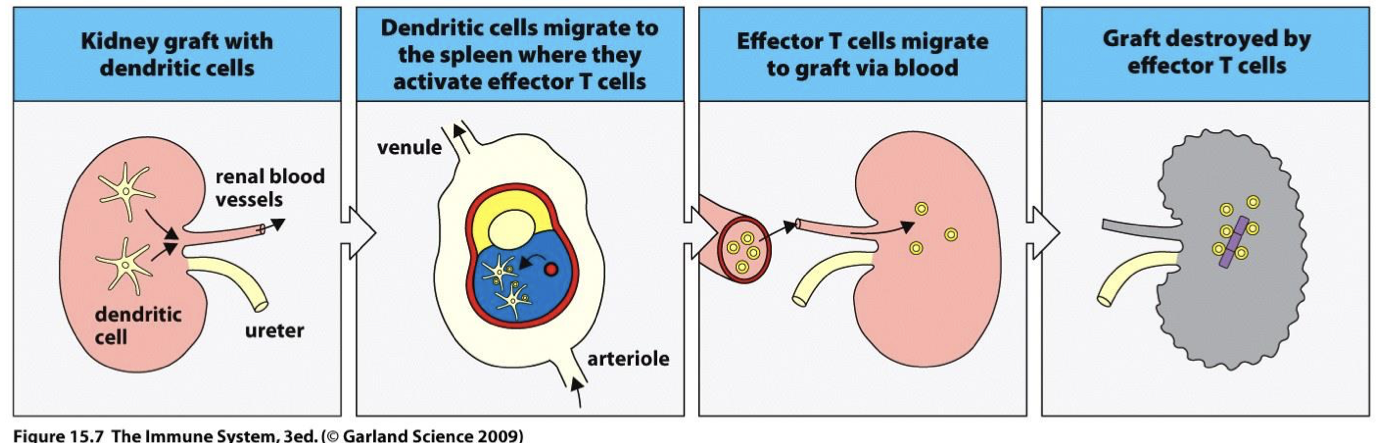
how is acute rejection prevented?
at first immunosuppressants are possible but Prevented by Mixed Lymphocyte Reaction (MLR) later on
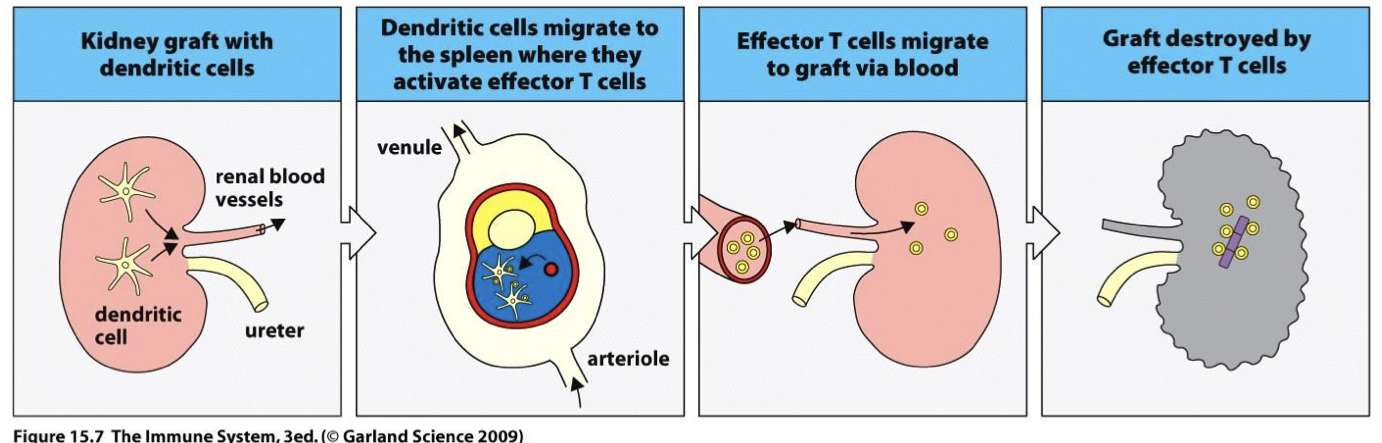
Mixed Lymphocyte Reaction (MLR)
cross match for alloreactive T cells by seeing if they response to donor tissue (in vitro)
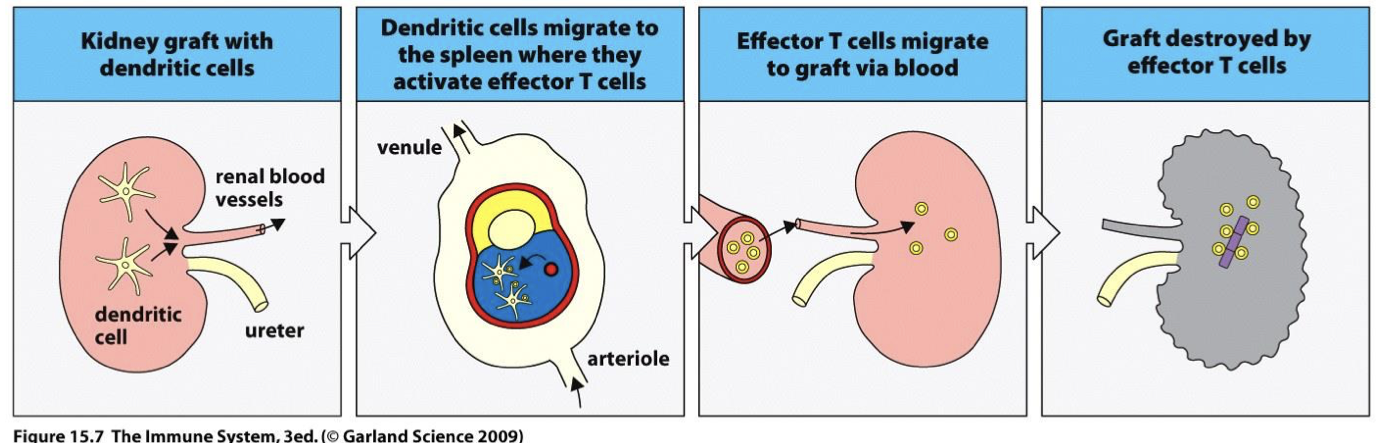
why do acute organ rejections occur?
most are part transplants (not everyone has a twin)
-T cells in recipient can activate and mount cytotoxic response against organ tissues → can cause total organ rejection → handling/small movements can cause inflammation +DC stimulation → once in the body DC go to L.N and T cell activation occurs)
chronic rejections take how long
slow, takes months to years after transplant
chronic rejection is due to?
Chronic rejection is due to recipient alloantibodies (anti donor HLA class I).
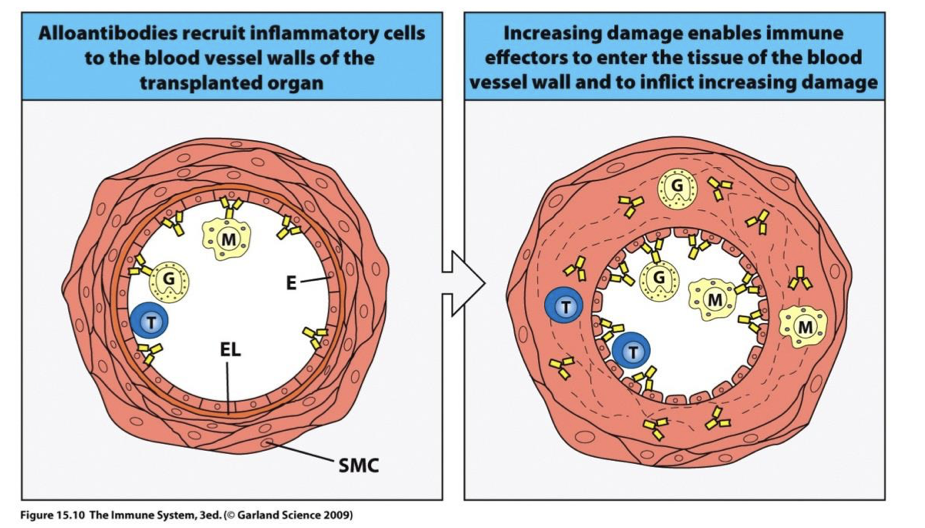
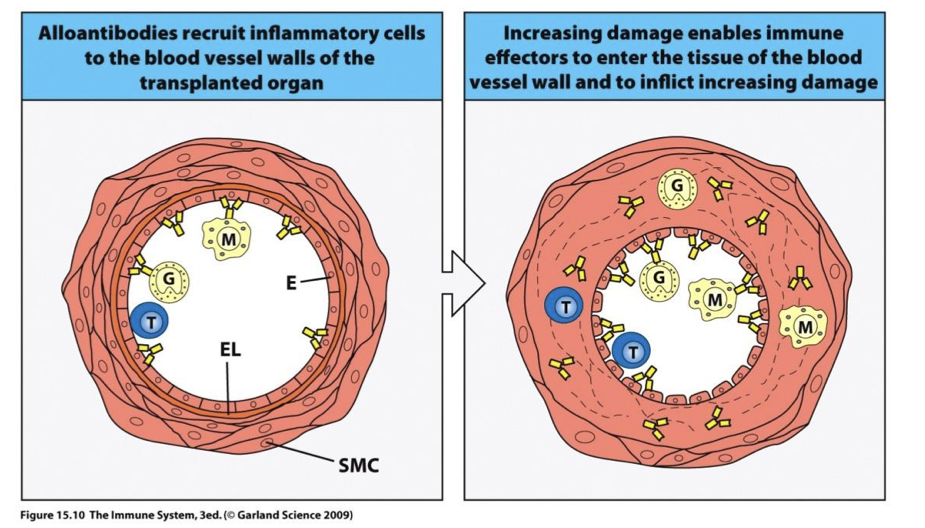
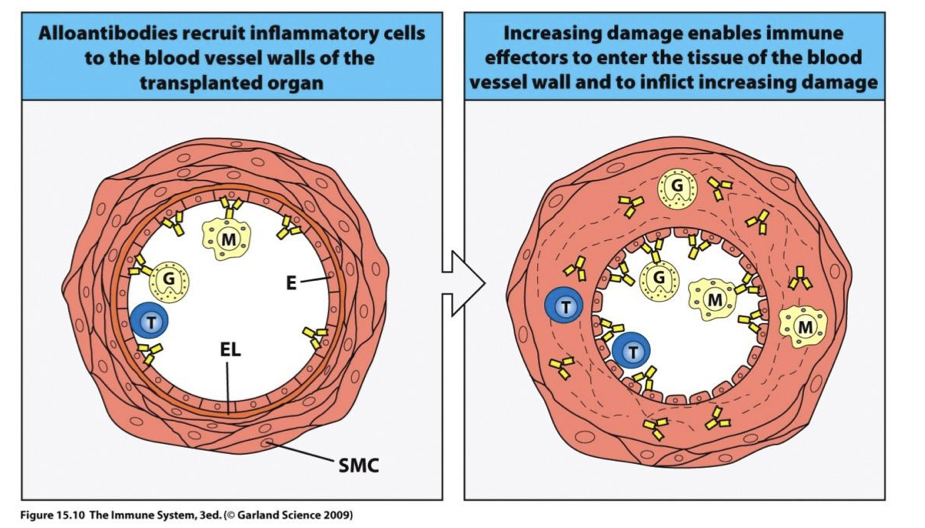
chronic rejection process
Recipient DC in L-node presents HLA peptide from donor DC → Ab produced against donor HLA → Ab bind endothelium and attract immune cells → vessel thickening/occlusion → ischemia
reaction in blood vessels of graft cause? explain
this is a major failure of what a % of which organs?
thickening vessel walls and narrowing lumen → decreases blood flow to organ which eventually dies
→ some DC die in the lymph node → pieces of cells taken up by recipient DC in LN and AG (donor cells) presented on recipient HLA 2 → TH2 B cell response in donor tissue → Ab produced → obstruction over time
→ cause of 50% of failure of kidney and heart transplants within ten years
Chronic rejection is the result of? (direct/indirect?)
the indirect pathway of allorecognition
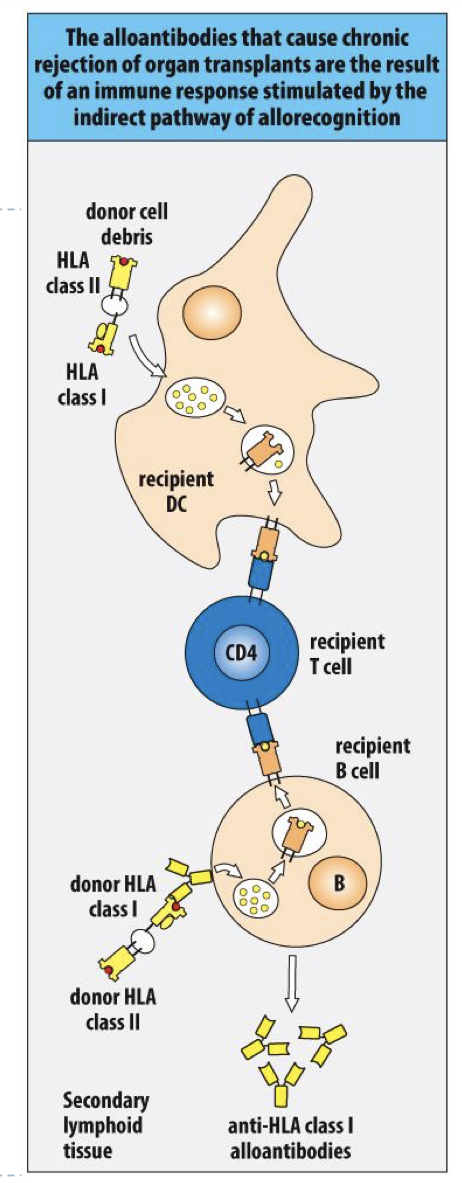
direct vs indirect pathway of allorecognition
direct= present Ag and response to it
indirect= recipient DC processing HLA 1-2 from Donor
ways to prevent organ failure
HLA matching improves survival of transplanted kidneys
Graft performance and health of patient increase with the degree of HLA match
Immunosuppressive drugs are given routinely to prevenrejection (eg. Rabbit antithymocyte globulin, anti-CD52, prednisone, tacrolimus, immunophilins, etc)
transplant rejection summary
see image
describe the process of transplanting Bone marrow (HSC)
Delete deficient immune cells and replace them with healthy immune cells.
Patient bone marrow destroyed by toxic chemo & radiation (myeloablative therapy).
Donor bone marrow transfused IV from living donor
Donor marrow HSCs reconstitute patient’s hematopoietic & produce new blood cells 2-3 wks post transplant (=engraftment)