Chemistry | Test 1 and Exam 1 - Pointers
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
Periodic table arrangement
The arrangement of elements in the periodic table is in order of increasing atomic number
Locate metals and non-metals on the periodic table
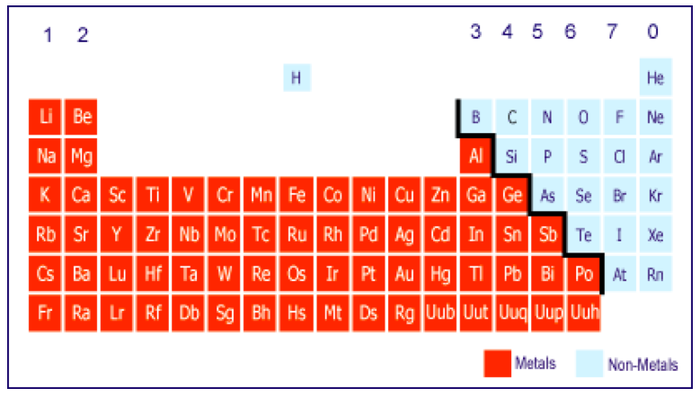
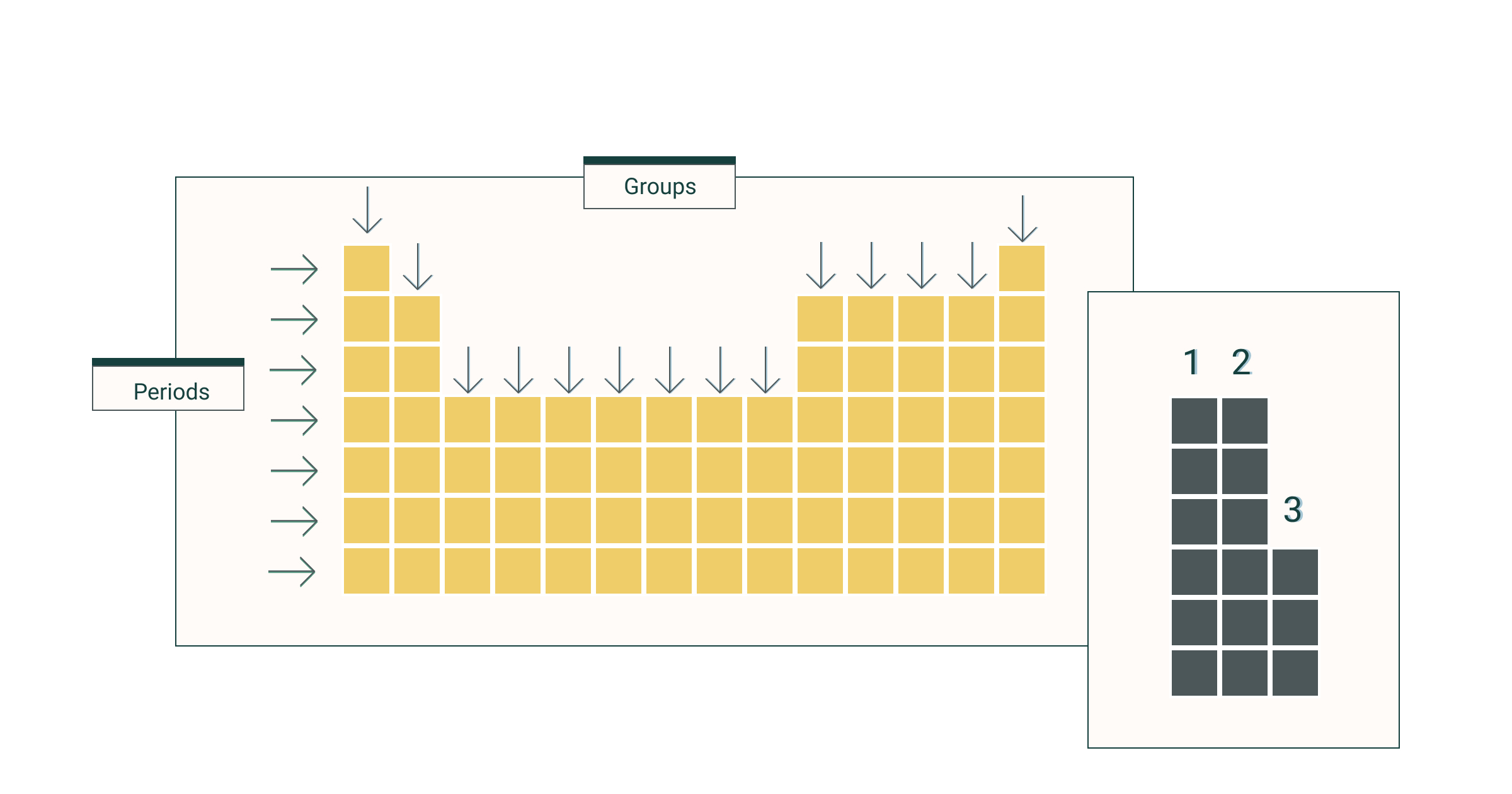
Distinguish between groups and periods on the periodic table
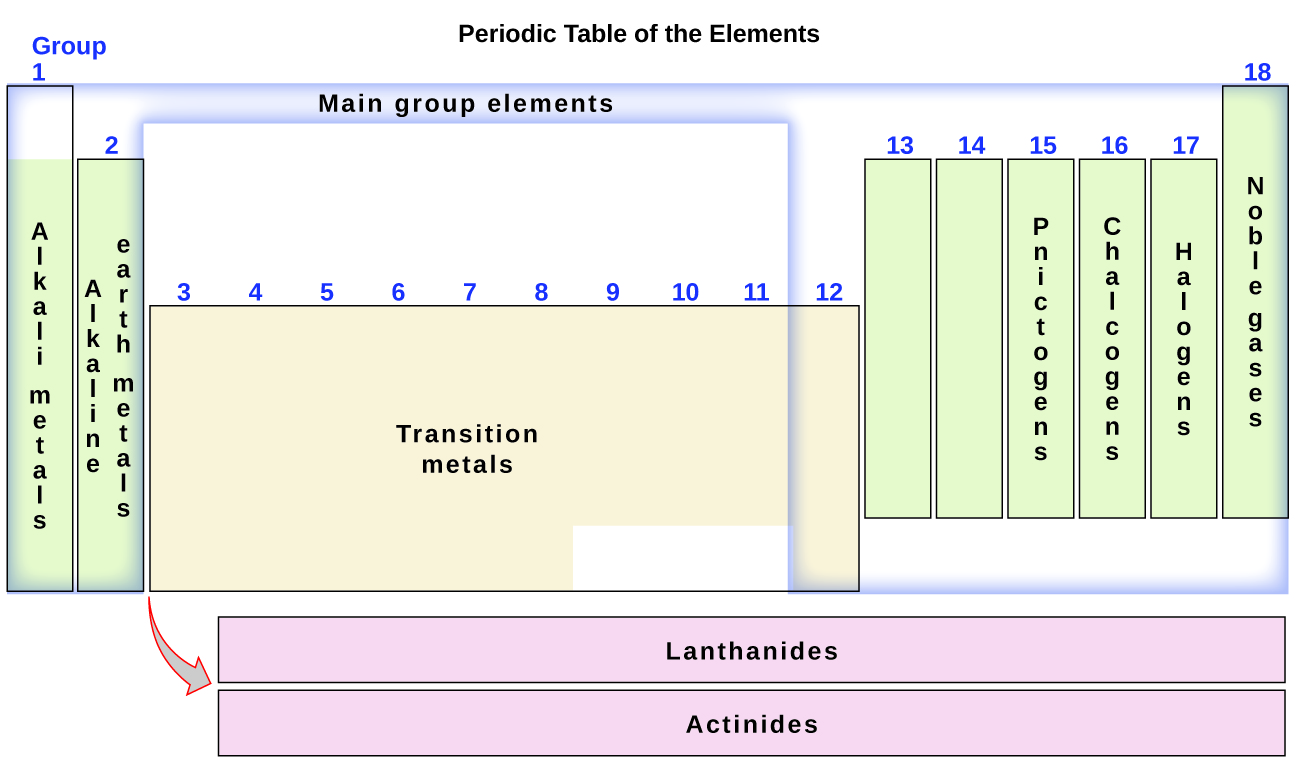
Locate and label the alkali metals, alkaline earth metals, transition metals, halogens and noble gases on the periodic table
List the relative charges and masses of protons, neutrons, and electrons
Proton | 1 | 1+ |
Neutron | 1 | 0/no charge |
Electron | 0 | 1- |
Structure of an atom
The atom has a dense, positively charged nucleus consisting of protons and neutrons, around which move negatively charged electrons.
How an element is identified
By its atomic number, which determines the number of protons (and electrons in a neutral atom) of the element
Define the mass number of an atom
The mass number of an atom is the total number and protons and neutrons in the nucleus of an atom
How the Periodic Table is arranged
It is arranged in order of increasing atomic number.
Determine the number of protons, neutrons, and electrons in a polyatomic or monatomic ion from the relationship between the atomic number and mass number
Mon/atomic ion
Protons = atomic number
Neutrons = mass number - atomic number
Electrons = atomic number
Polyatomic ion
Protons = sum of all atomic numbers
Neutrons = mass number - atomic number
Electrons = charge (e.g. +1 charge loses one electron → more positive)
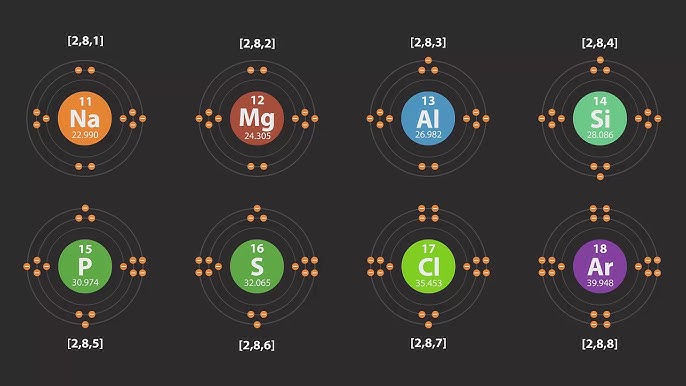
Identify the valence electrons of an atom
Valence electrons are the electrons located in the outermost shell (energy level) of an atom
Describe where electrons are located in an atom
The electrons in an atom are located in shells
Explain how positive ions and negative ions are formed by the loss and gain of valence electrons
Positive ions form when atoms lose valence electrons, leaving them with more positive protons than electrons.
Negative ions form when atoms gain valence electrons, resulting in more negative electrons than protons.
Relate the charges of the monatomic ions formed by the main group elements to their position on the Periodic Table
Main group elements form monatomic ions to achieve a stable valence shell, with charges directly related to their group number. Metals on the left (Groups 1, 2, 13) lose electrons to form positive ions with charges of +1, +2, or +3, while nonmetals on the right (Groups 15, 16, 17) gain electrons to form negative ions with charges of -3, -2, or -1
Stable outer shell electron configuration
An outer shell electron configuration of the noble gases is a stable configuration
Write the electron configuration of the first twenty elements and their monatomic ions
How valence electrons achieve a stable electron arrangement
Valence electrons are involved in electron transfer or sharing to achieve a stable electron arrangement. Reduces energy and reactivity
Write symbols and names of monatomic ions
Aluminium | Al | 3+ |
Barium | Ba | 2+ |
Bromide | Br | 1- |
Calcium | Ca | 2+ |
Chloride | Cl | 1- |
Chromium | Cr | 3+ |
Cobalt | Co | 2+ |
Copper (I) | Cu | 1+ |
Copper (II) | Cu | 2+ |
Fluoride | F | 1- |
Hydrogen | H | 1+ |
Iodide | I | 1- |
Iron (II) | Fe | 2+ |
Iron (III) | Fr | 3+ |
Lead | Pb | 2+ |
Lithium | Li | 1+ |
Magnesium | Mg | 2+ |
Nickel | N | 3- |
Nitride | N | 3- |
Oxide | O | 2- |
Phosphide | P | 3- |
Potassium | K | 1+ |
Silver | Ag | 1+ |
Sodium | Na | 1+ |
Sulfide | S | 2- |
Zinc | Zn | 2+ |
Describe how the law of conservation of mass applies to chemical reaction
The law of conservation of mass states that in a chemical reaction, mass is neither created nor destroyed; it is only rearranged. The total mass of the reactants must equal the total mass of the products. This ensures that the number of atoms of each element remains constant, with atoms simply changing how they are combined.
Use solubility rules to classify ionic solids as being soluble or insoluble in water
(s) = solid
(l) = liquid (something pure, e.g. pure water - H2O)
(g) = gas
(aq) = aqueous (when something is dissolved in water to create a solution)
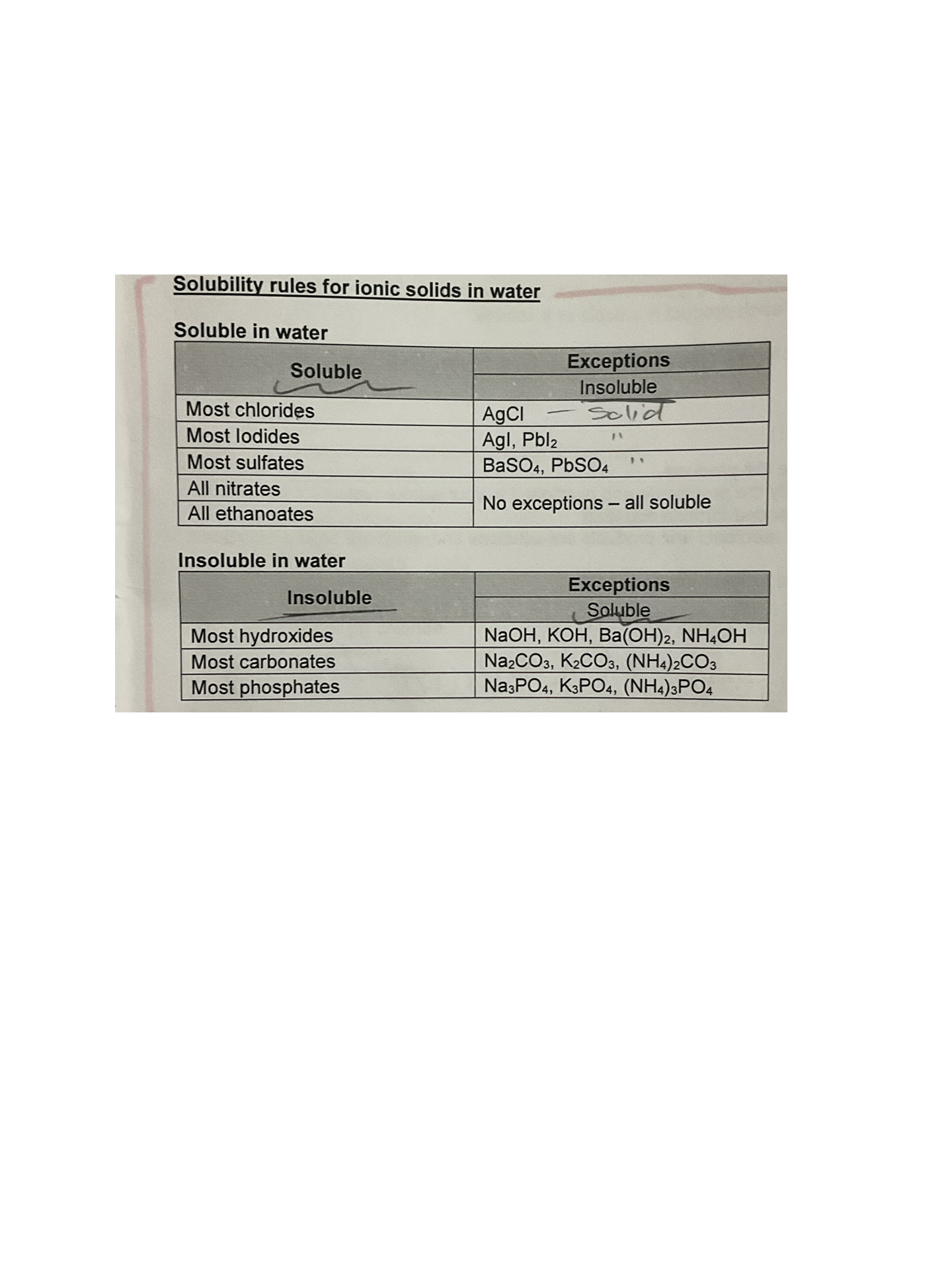
Write balanced chemical equations for the reaction between two ionic solutions, producing a precipitate, including state symbols in your equation

List the name and formulae of the following acids: HCl, HNO3, H2SO4, H3PO4, and CH3COOH
hydrochloric acid (1 molecule of hydrochloric acid consists of 1 atom of hydrogen and 1 atom of chloride)
nitric acid (1 molecule of nitric acid consists of 1 atom of hydrogen, 1 atom of nitrogen and 3 atoms of oxygen)
sulfuric acid (1 molecule of sulfuric acid consists of 2 atoms of hydrogen, 1 atom of sulfur and 4 atoms of oxygen)
phosphoric acid (1 molecule of phosphoric acid consists of 3 atoms of hydrogen, 1 atom of phosphorus and 4 atoms of oxygen)
ethanoic acid (1 molecule of ethanoic acid consist of 2 atoms of carbon, 4 atoms of hydrogen and 2 atoms of oxygen)
List the name and formulae of the following acids: hydrochloric acid, nitric acid, sulfuric acid, phosphoric acid, and ethanoic acid
HCl
HNO3
H2SO4
H3PO4
CH3COOH
List the pH ranges for acidic, basic and neutral substances
Acidic substances have a pH below 7 (0–6.9)
Neutral substances have a pH exactly at 7
Basic (alkaline) substances have a pH above 7 (7.1–14)
Give the expected colours for litmus paper and universal indicator in acids, bases and neutral substances
Universal indicator changes color based on pH, showing red/orange in acids, yellow/green in weak acids/neutrals, and blue/purple in bases.
Litmus turns red in acidic solutions (pH below 7) and blue in alkaline/basic solutions (pH above 7).

Predict the products of an acid/base and compose a word equation and balanced formulae equation
Metal + Acid → Salt + Hydrogen gas
Acid-base neutralisation typically produces a salt and water.
The H+ from the acid and OH- from the base combine to form, H2) while the remaining ions form the ionic salt.
Predict the products of an acid/carbonate and compose a word equation and balanced formulae equation
Carbonate + Acid → Water + CO2 + Salt
The reaction between an acid and a carbonate produces a salt, water, and carbon dioxide gas.
Describe how we can test for the presence of carbon dioxide gas, and give expected observations
The standard laboratory test for the presence of carbon dioxide (CO2) gas is the limewater (CA(OH)2) test
It involves passing the gas through a solution of calcium hydroxide, commonly known as limewater
Use the periodic table to calculate the molar mass of compounds and elements
Molar mass is calculated using the periodic table by summing the average atomic masses (in g/mol) of all atoms in a formula
e.g. Calcium Chloride (CaCl2) = Ca + Cl x 2 = 40 + 35.5 × 2 = 111 g/mol
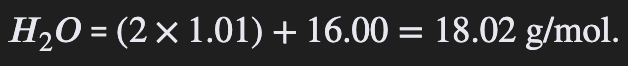
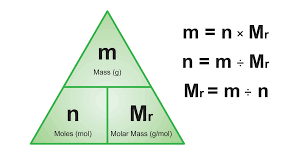
Use (and rearrange) the formula n = m/M
Number of moles of known substance → number of moles = mass ÷ molar mass (n = m/M)
Number of moles of unknown substance → coefficient of want ÷ coefficient of have X moles have
Calculate the mass of your unknown substance → mass = number of moles X molar mass (m = n X m)
Comparison of Ionic and Covalent Compounds
Characteristic | Ionic | Covalent |
Types of elements | metal and non-metal | non-metal and non-metal |
Electrons | lost & gained | shared |
Forms molecules? | no (lattics/crystals) | yes |
Side note: If a substance is covalent then it forms a molecule, however if it is ionic, it forms an ionic lattice
Element e.g. H, Zn, Cl.
Compound e.g. NaI, CuCl2