RP 5 - organic
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
RP 5
distillation of product from a reaction
preparation of cyclohexene by dehydration and distillation of cyclohexanol
pour cyclohexanol into pear shaped flask thats been weighed, reweigh and record mass
use plastic graduated dropping pipette to add H2SO4 to flask
add anti bumping granules
assemble distillation apparatus and heat gently distilling any liquid which boils below 100 degrees
pour distillate into separating funnel and add saturated NaCl solution. shake and allow layers to separate
run lower level into beaker and transfer upper layer (contains crude cyclohexene) into small conical flask
add anhydrous CaCl2 to crude cyclohexene to remove water
stopper flask, shake and allow to stand until liquid becomes clear
decant into sample container thats been weighed
reweigh container, calculate mass produced and determine % yield
testing distillate
use bromine water to confirm alkene - goes colourless
why can the cyclohexene be separated in this way (step 3)
has a lower boiling point
what does saturated NaCl act as
drying agent
what does step 5 do
removes water, a little cyclohexanol and H3PO4
assumption made when calculating mass
all dry distillate is cyclohexene
common mark scheme method
acidify potassiu managanate soilution combining volume of it with equal volume of dilute H2SO4
pour mixture into pear shaped flask with a still head containing a thermometer
attach to condenser with ice cooled collecting vessel
add few anti bumping granules
heat flask gently
collect sample at bp of desired product
cooled collection vessel is essential to reduce evaporatation of product
diagram
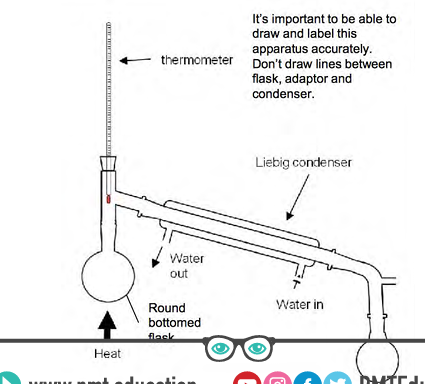
whys a water bath used
if there are flammable substances present
what do anti bumping granules do
prevent large bubbles forming
ensure liquid does not boil too vigorously - mixture would boil over into condenser and undesired impurities would contaminate product
condenser angle
tilted slightly down so any liquid can run into collection flask
where should thermometer bulb be
at the T junction connecting to the condenser to measure the correct bp
condenser water flow
water enters lowest point and leaves highest to go against gravity to
ensure water fills the condenser (prevents backflow of water)
maximise heat transfer for condensation (more efficient cooling)
collection flask
must not be sealed to condenser
system should not be air tight as it is heated the air inside expands
if air tight the air cannot escape and may cause apparatus to crack
preparation of ethanal by oxidation and distillation of ethanol
make oxidising agent by dissolving potassium dichromate in dilute H2SO4 - conc should be 1g in every 10cm3
use measuring cylinder to measure it and pour into boiling tube
cool in beaker of cold water
use teat pipette to add ethanol drop wise shaking to mix contents
add anti bumping granules
attach to a bung fitted with a right angled glass delivery tube
clamp boiling tube in water so delivery tube goes to a test tube immersed in cold water
gently heat to slowly distill some liquid distillate
use tollens to test
whys cold water used
keep test tube cool to avoid loss of volatile ethanal
tollens silver mirror test
add NaOH to AgNO3 in test tube
add just enough dilute ammonia solution to dissolve the brown ppt completely
using beaker of hot water (50-60 degrees) gently warm in test tube
add distillate
wait a few mins and record observations - silver mirror should form
safety precautions
make sure tollens reagent is disposed of thoroughly by rinsing it away with plenty of water
rinse glassware that has contained reagent with little dilute H2SO4 when finished