3.1.9.2 determination of rate equation
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
some reactions take place in one stage or over several stages, what are all stages collectively referred to?
as the mechanism
each stage in the mechanism will have what?
its own equation + rate of reaction
in a mechanism,
a. what is a catalyst?
b. what is an intermediate?
a. a catalyst is used up in one step + re-made in a later step
b. an intermediate is made in one step + used up in a later step

what is linked to the mechanism?
the rate equation of a reaction
how can you use the mechanism to determine the rate for the overall reaction?
by considering the rate determining step
what is the rate determining step?
this is also known as the slow step in the mechanism

explain the process to using the mechanism to work out the rate equation
from the mechanism, the reactants in all the steps up to + including the slow step are the ones which appear in the rate equation, excluding any intermediates (as these are not added at the beginning of a reaction)
if there is 1 molecule of a particular reactant up to + including the RDS, it will be first order with respect to that reactant
if there are 2 molecules of a particular reactant up to + including the RDS, it will be second order
none of the species after the slow step will be in the rate equation as they have no effect on the rate of equation → zero order species will always appear in the stages after the RDS
(essentially: remove anything after slow step. remove intermediates. remove products.)
outline the presence of catalysts + intermediates in the rate equation + overall equation
catalysts can appear in the rate equation but do not appear in the overall equation
intermediates never appear in the rate equation or the overall equation

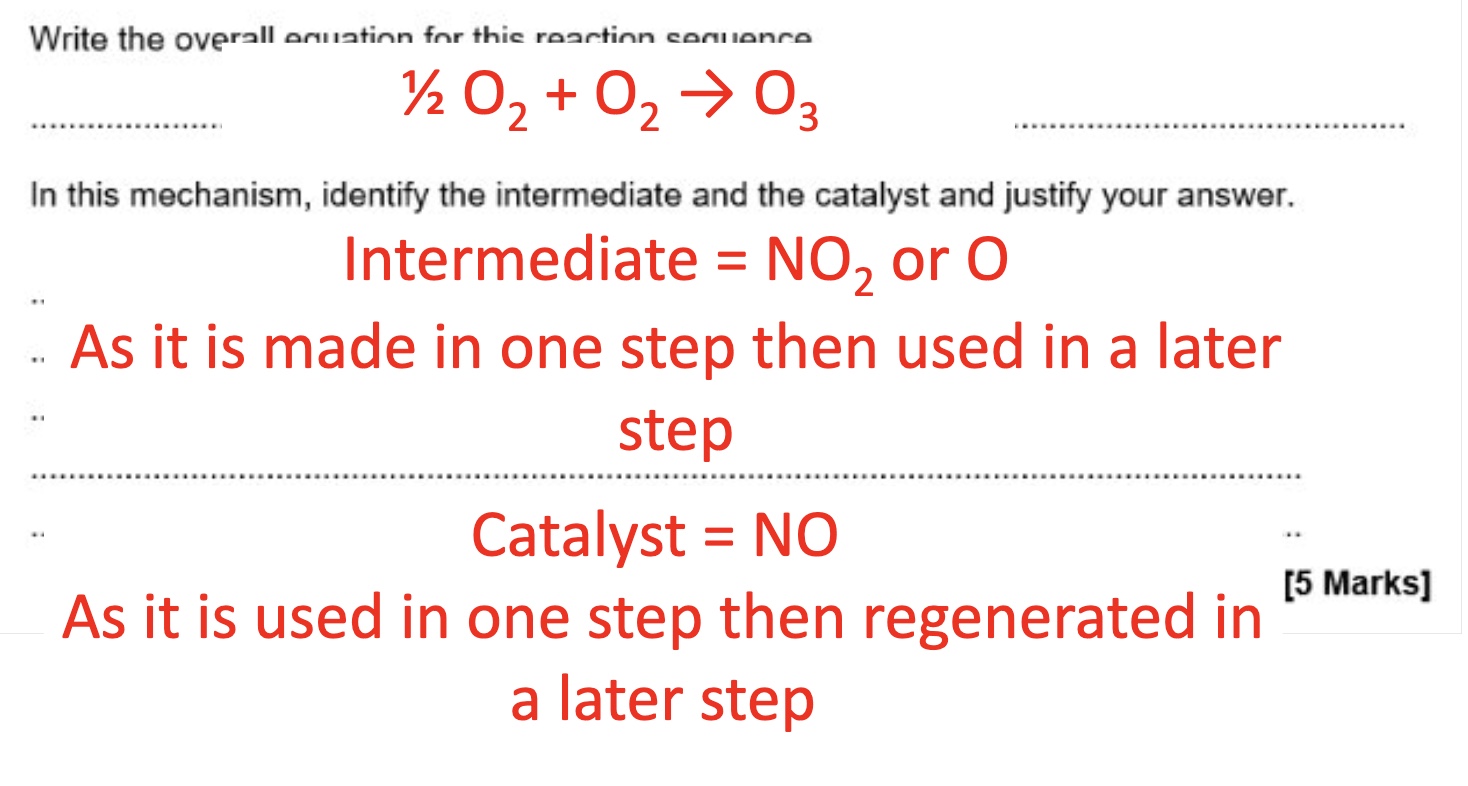
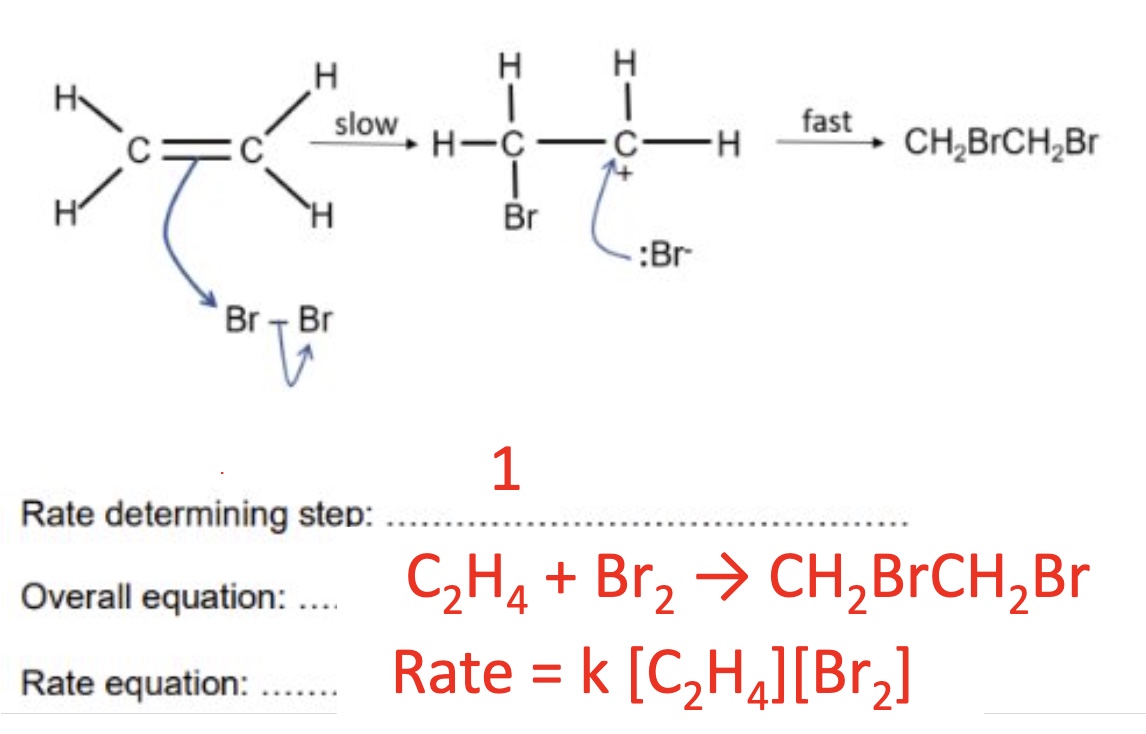
for this example, what will the rate equation be?
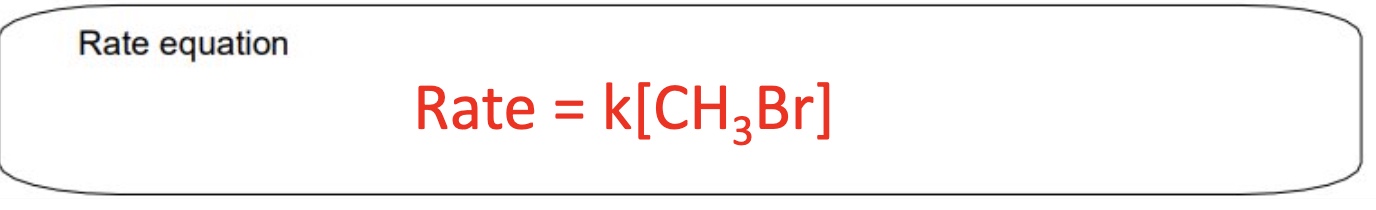
from this mechanism, give the:
a. rate determining step (RDS)
b. overall equation
c. rate equation
how will a catalyst affect the rate of reaction if its added after the RDS?
it will have no effect as its added after the RDS (therefore it will affect the rate if its added before or in the RDS)
explain the process/rules to identifying the RDS from the mechanism + rate equation
once all the species from the rate equation are used up as reactants in the mechanism, in the ratio/proportion as in the rate equation, then the later step is the RDS
intermediates never appear in the rate equation
if a species appears in the rate equation but does not appear in the overall equation, it is a catalyst

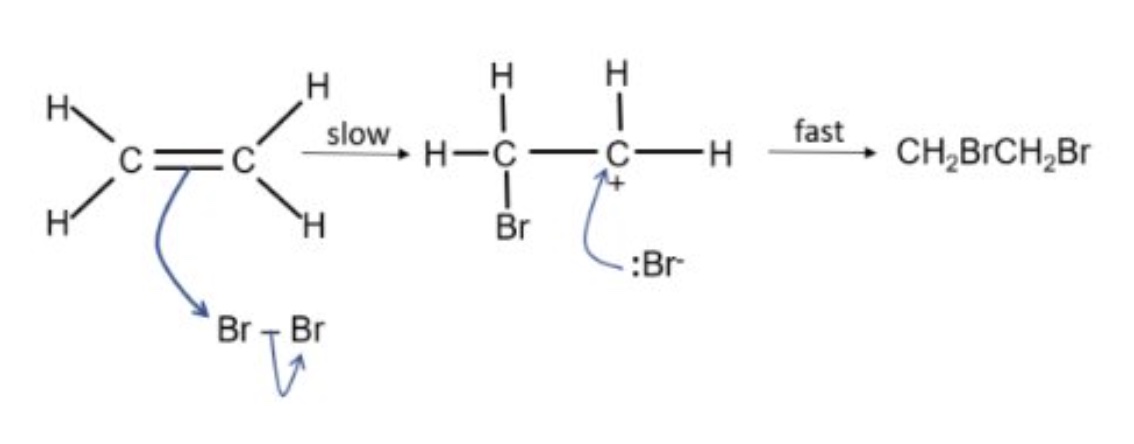
example:
identify the RDS + explain your answer

what is the criteria for measuring a reaction rate?
the concentration of one of the reactants or products must be measured over a period of time
the temperature must be kept constant as rate varies with temperature
the method chosen for measuring rate of reaction depends on what two things?
on the substance whose concentration is being measured
also on the speed of the reaction
required practicals 7a and 7b are strongly linked to what other practical?
required practical 3: investigation of how the rate of reaction changes with temperature
what are the 2 ways to measure the rate of a chemical reaction?
using a continuous monitoring method (7b)
using an initial rates method (7a)
continuous monitoring method (required practical 7b)
describe the method for using the method above to calculate rate of reaction
using this method you would measure either the ‘amount’ of a product formed or the reactant lost continuously throughout the reaction
we could therefore measure:
concentration of a species reacted/formed
volume of gas formed
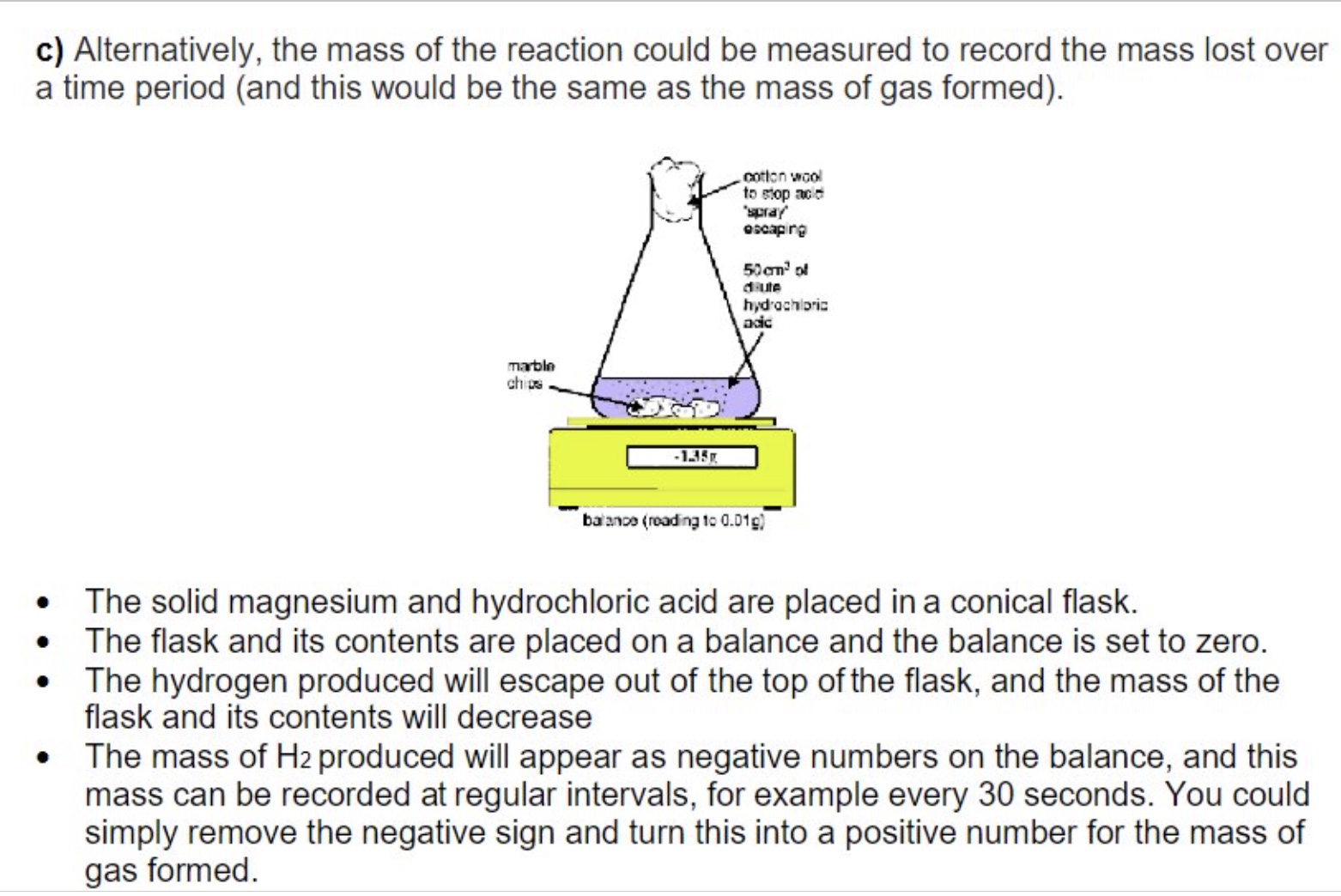
mass lost during a reaction (+ therefore mass of gas formed)
although the units of rate are normally moldm⁻³s⁻¹, we can still use volume gas formed or mass of gas formed as these are proportional to concentration (in these cases our units for rate will therefore be different)
by plotting concentration/volume/mass against time, you can then deduce the rate + also the order of reaction
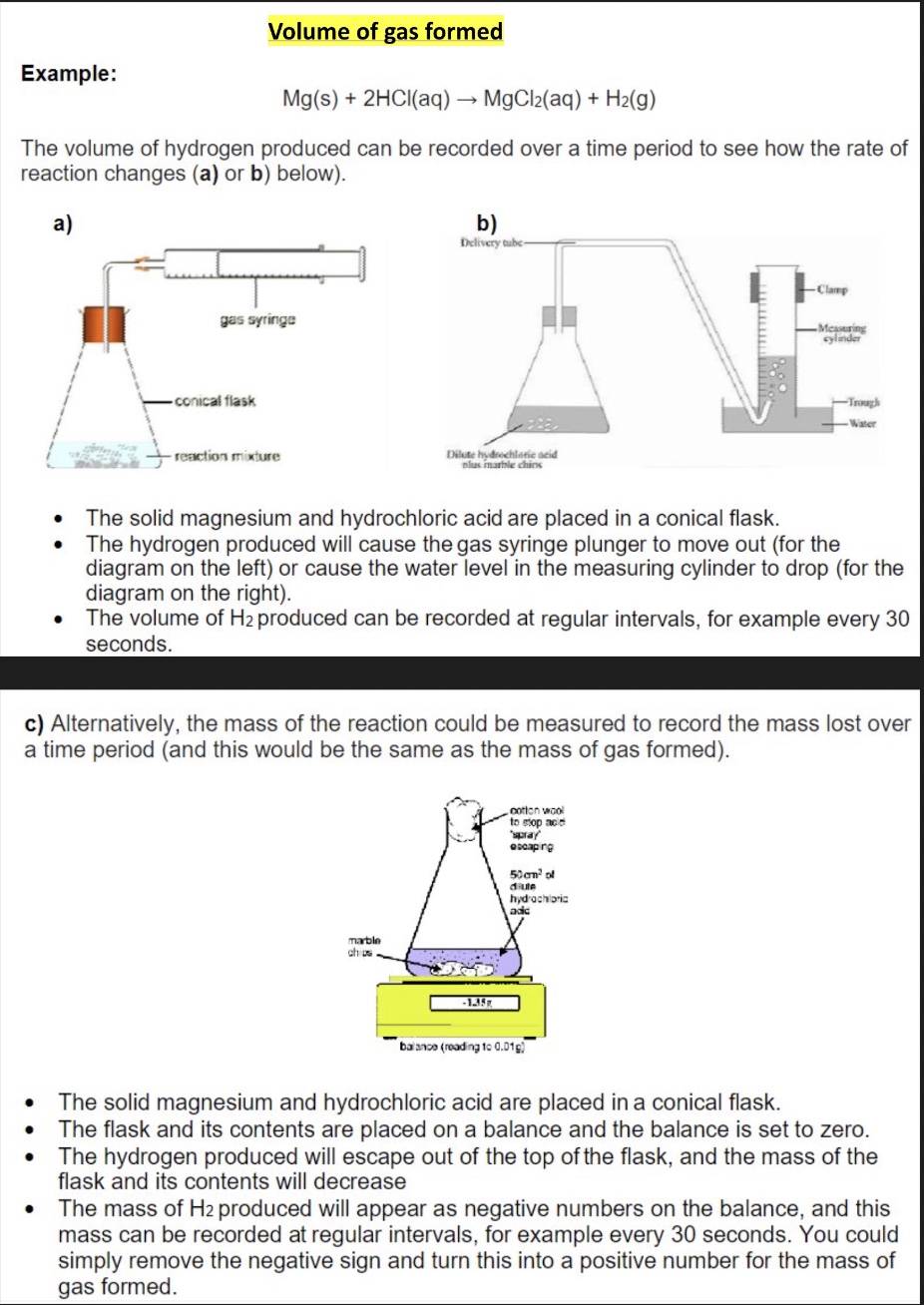
why is using a gas syringe to measure rate better?
it is more accurate
what do you need to make sure you draw when drawing the diagram of required practical 7b eg the method of recording changes in volume of hydrogen?
a gas string will be in the diagram so make sure to draw the scale on the syringe
draw + label the bung on the conical flask as well
why is cotton wool used in the conical flask when measuring the mass of gas formed?
to prevent acid spraying/splashing out whilst still allowing gas to escape
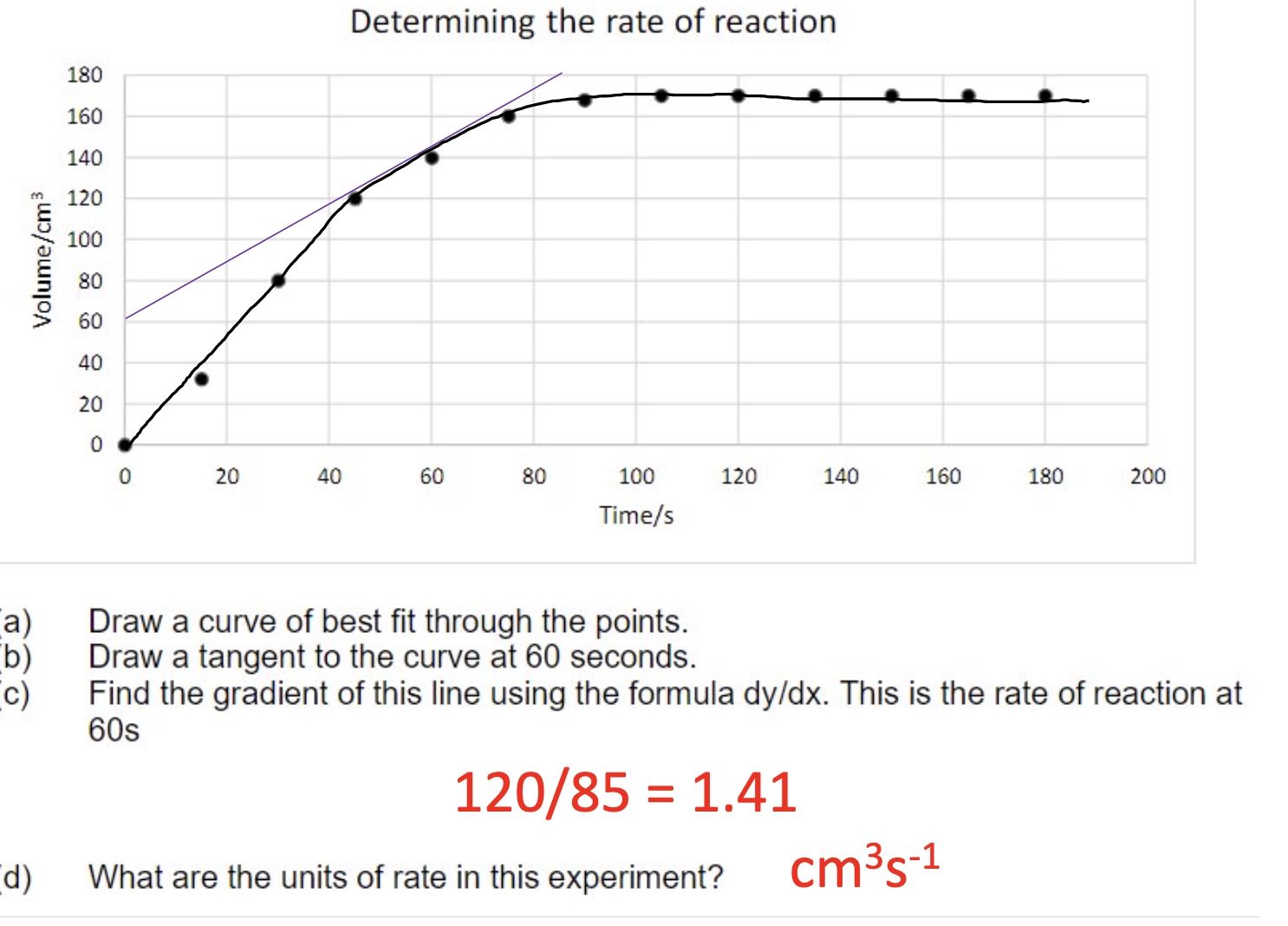
describe the steps to finding the rate of reaction from a concentration/volume/mass against time graph
the rate of reaction is often calculated by:
collecting data + plotting a graph
drawing a line of best fit through the points
drawing a tangent at a particular time eg t = 0s
find the gradient of the tangent using the formula △y/△𝑥 eg change in conc/change in time
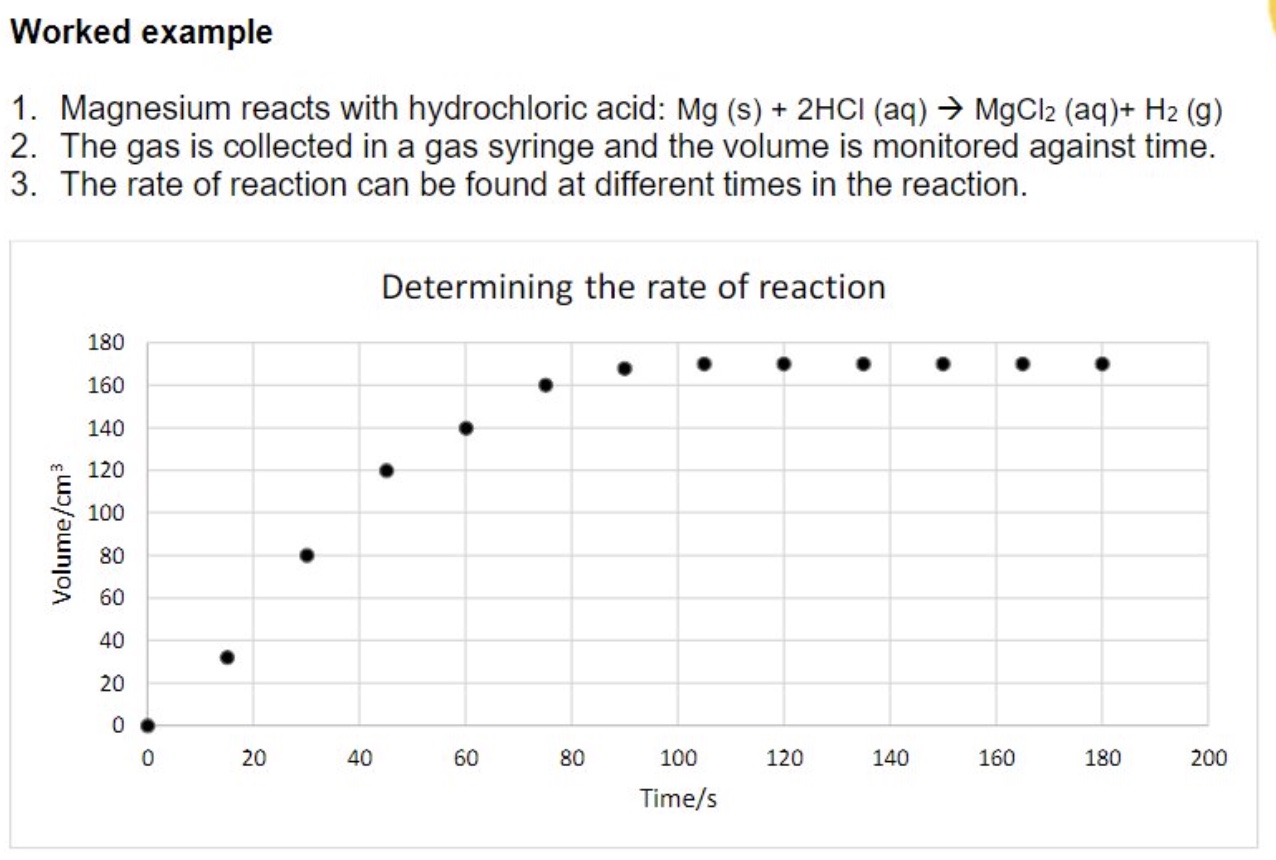
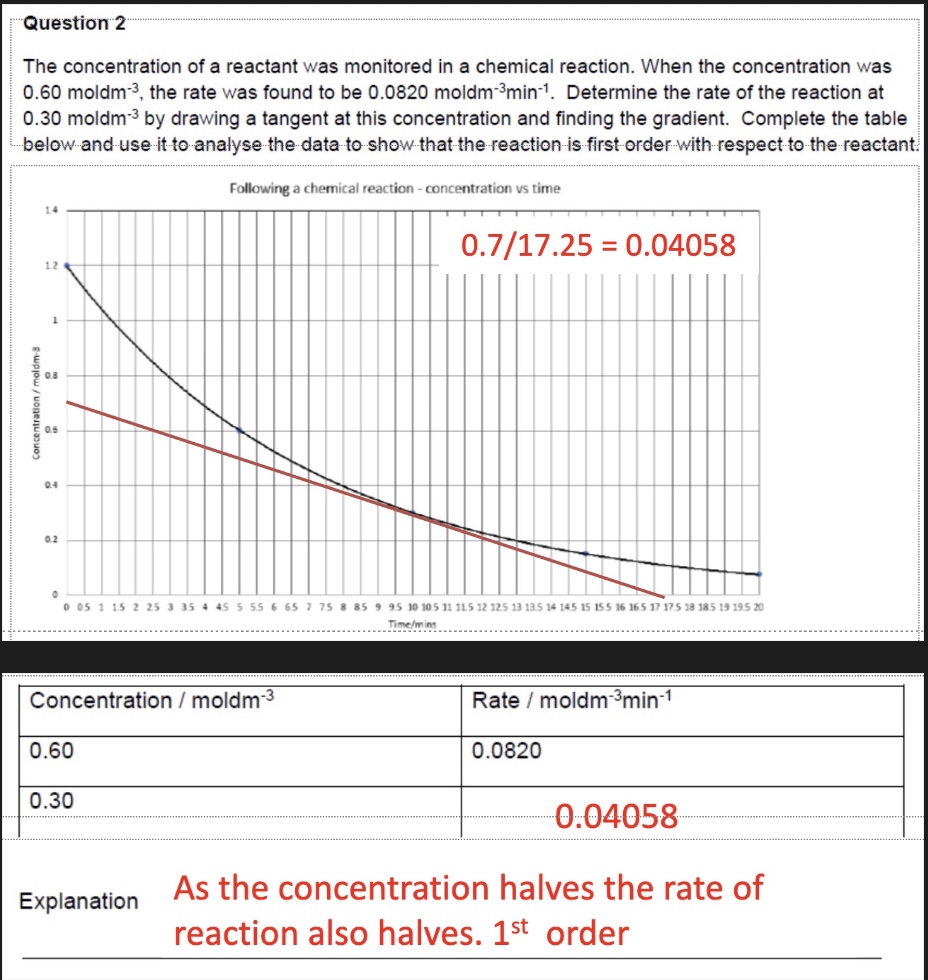
example of finding rate of reaction from graph
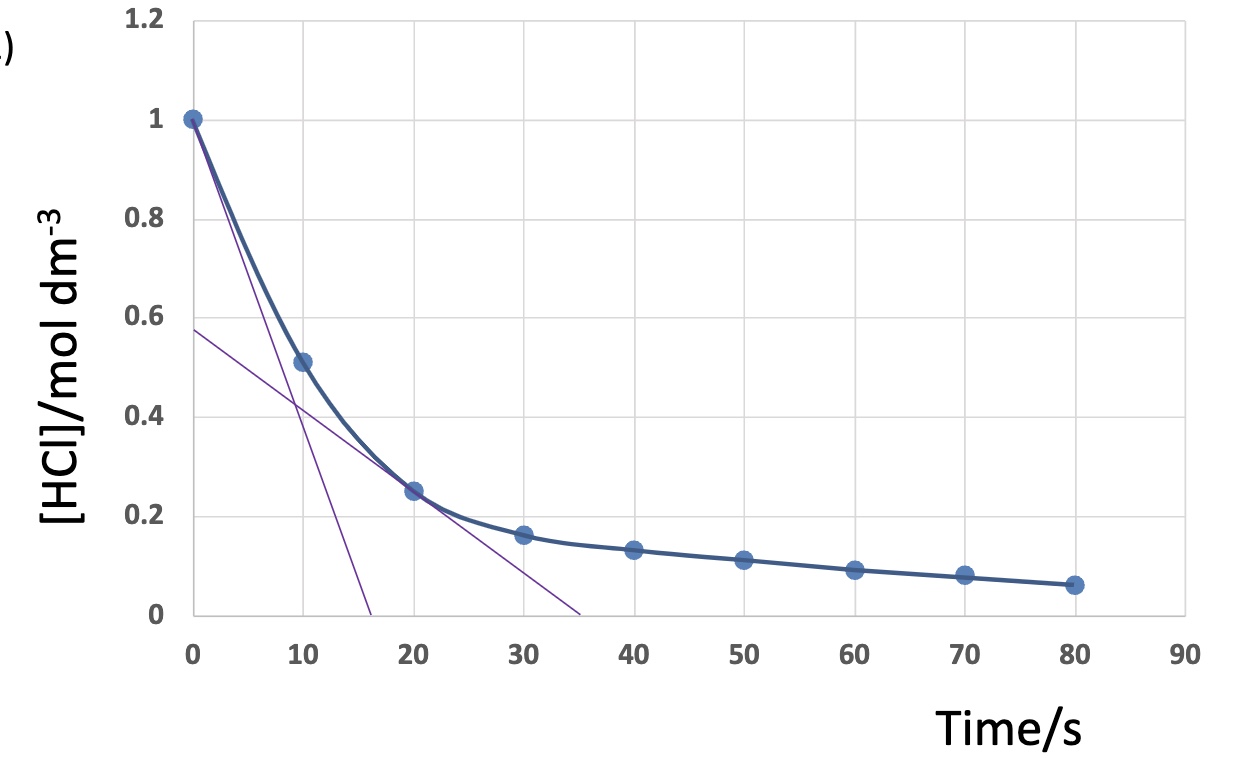
explain how + why the rate changes during this chemical reaction
rate decreases over time as concentration of reactants decreases
there are less reactant particles in given volume
there is a lower frequency of successful collisions
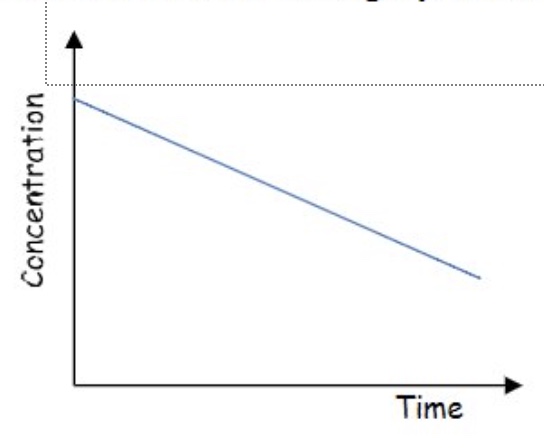
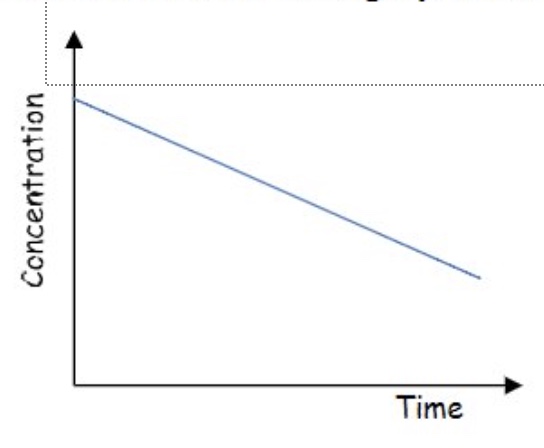
determine the order of reaction from this concentration-time graph + explain your answer
zero order:
we can see it is a straight line
this means as concentration changes, the gradient + therefore the rate does not change → therefore the order with respect to this reactant is zero
from this graph (order is zero) how can we calculate the rate constant, k
for zero order reactions, the rate equation is rate = k
therefore k can be determined by calculating the gradient
determine the order of reaction from this concentration-time graph + explain the method for finding this
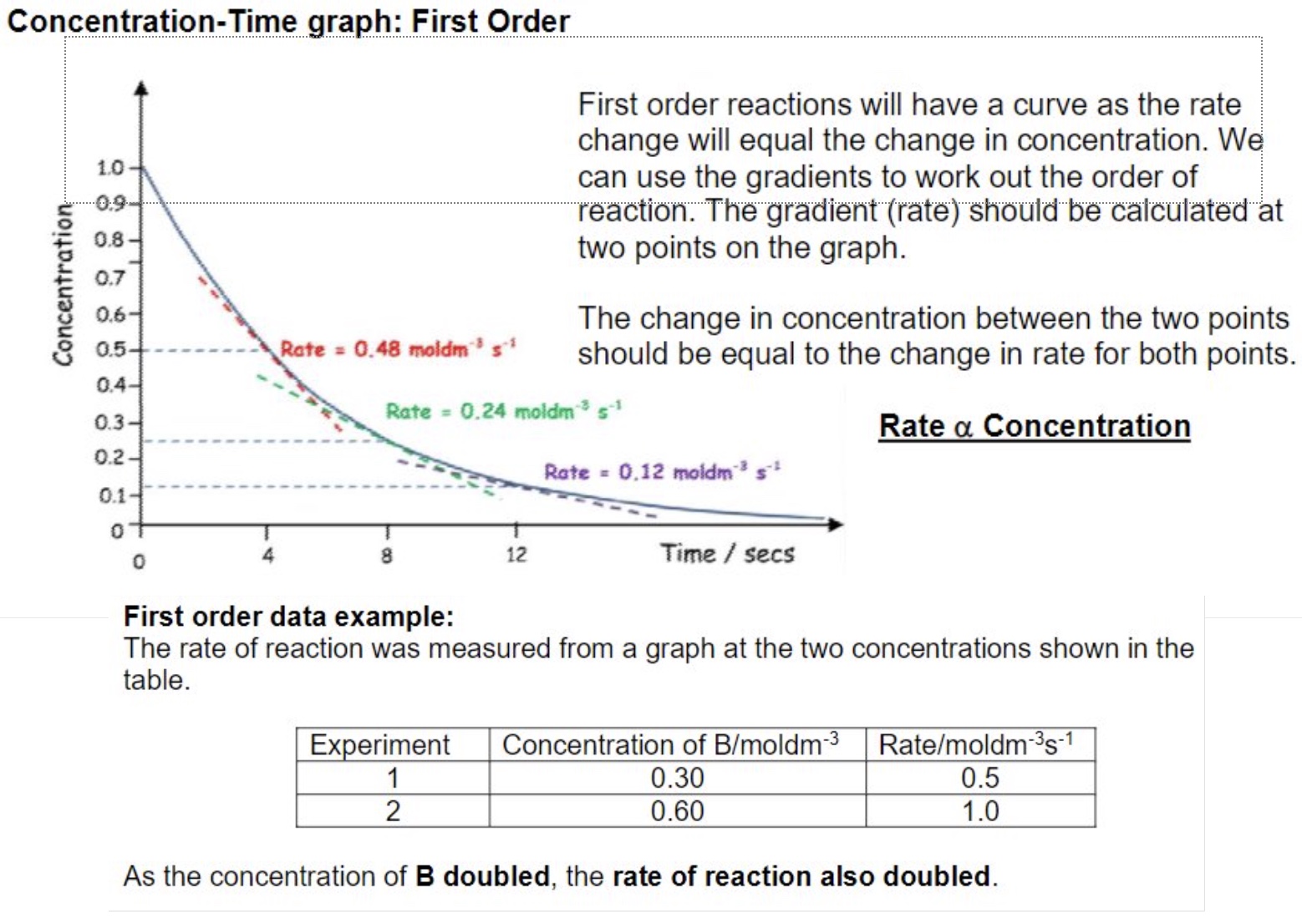
first order:
first order reactions will have a curve as the rate change will equal the change in concentration
we can use the gradients to work out the order of reaction — the gradient (rate) should be calculated at two points on the graph
the change in concentration between the two points should be equal to the change in rate for both points
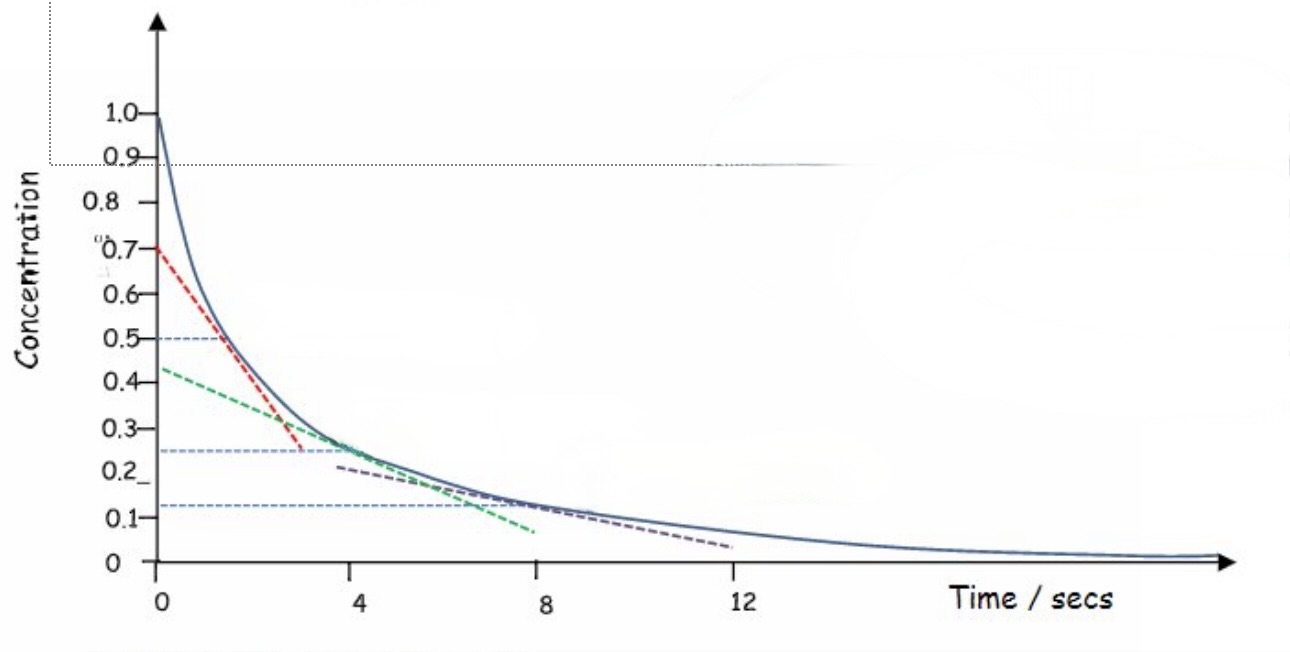
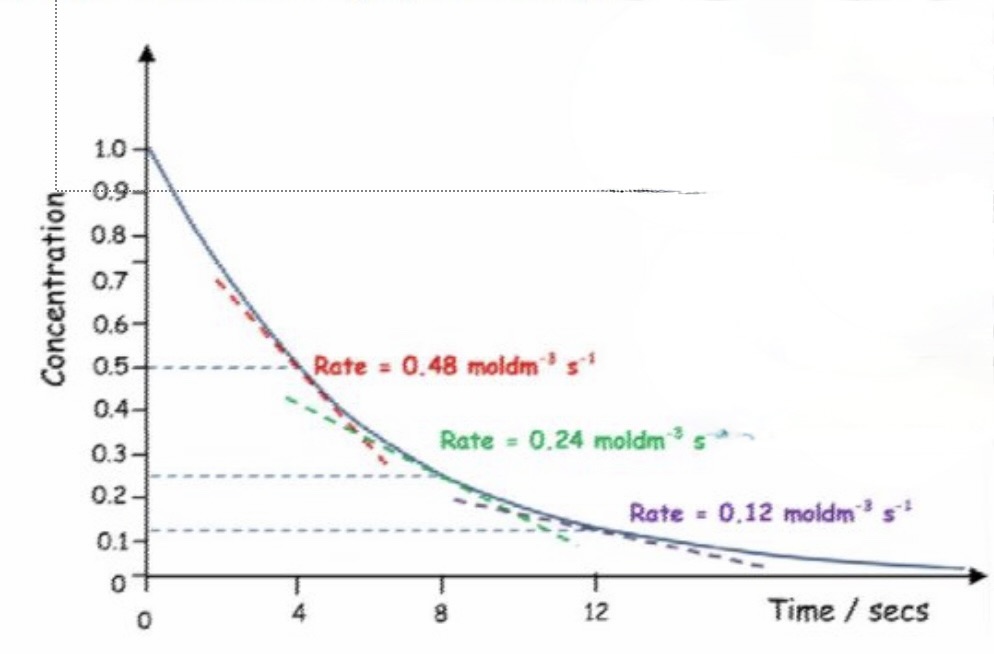
determine the order of reaction from this concentration-time graph + explain the method for finding this
second order reactions will also have a curve
when the gradient of two points on the curve are compared, the change in rate should equal the change in concentration squared
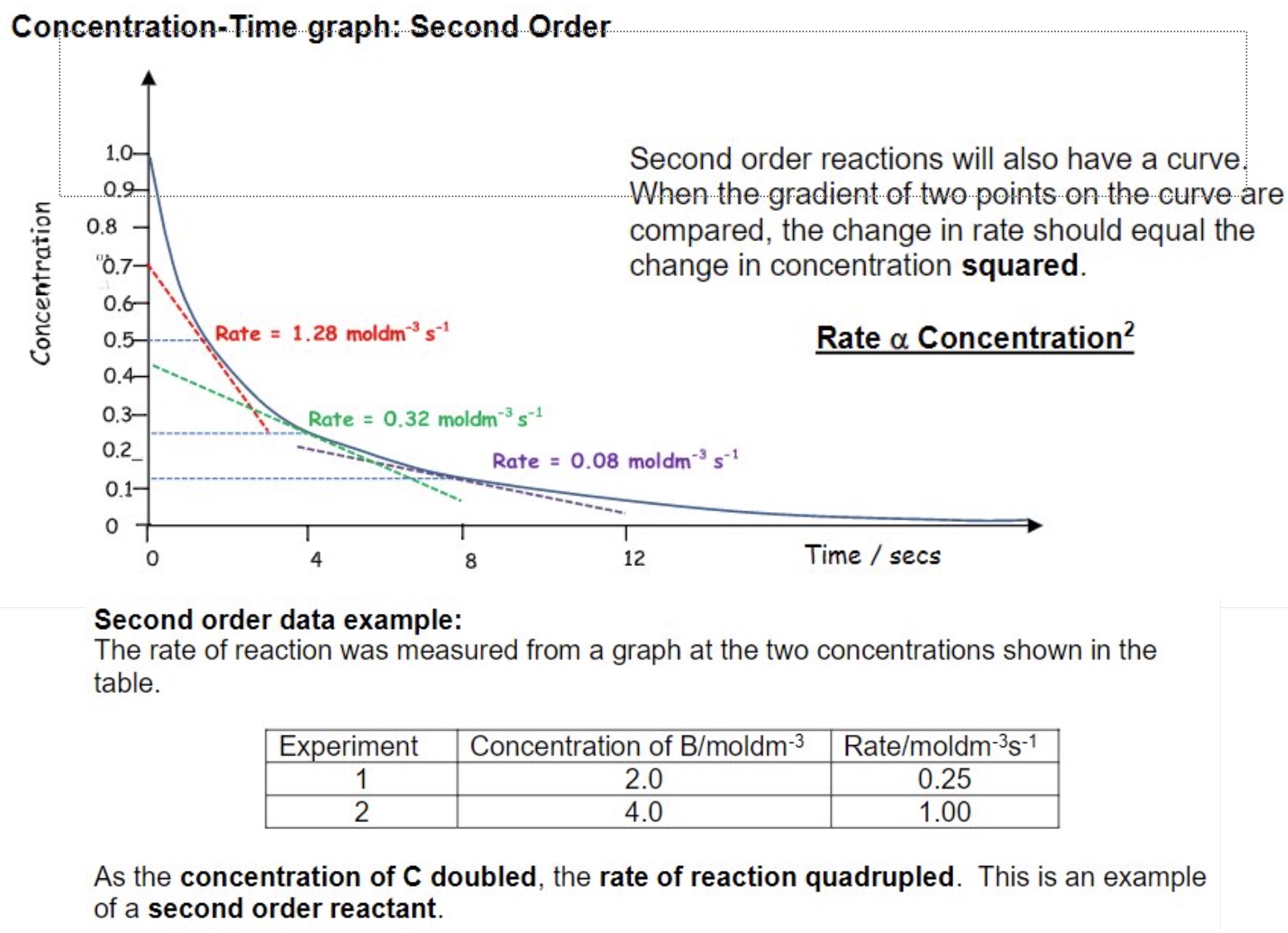
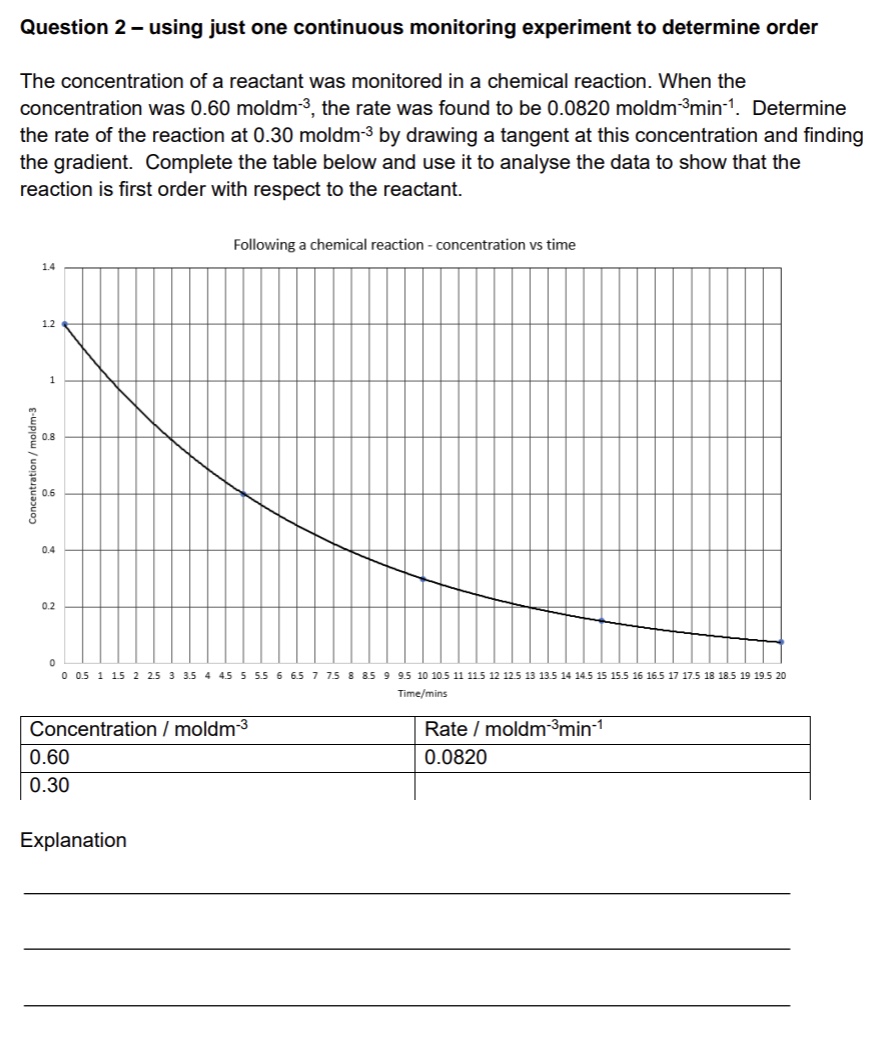
example: calculating order from graph
give an explanation for answer