BC 15
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms

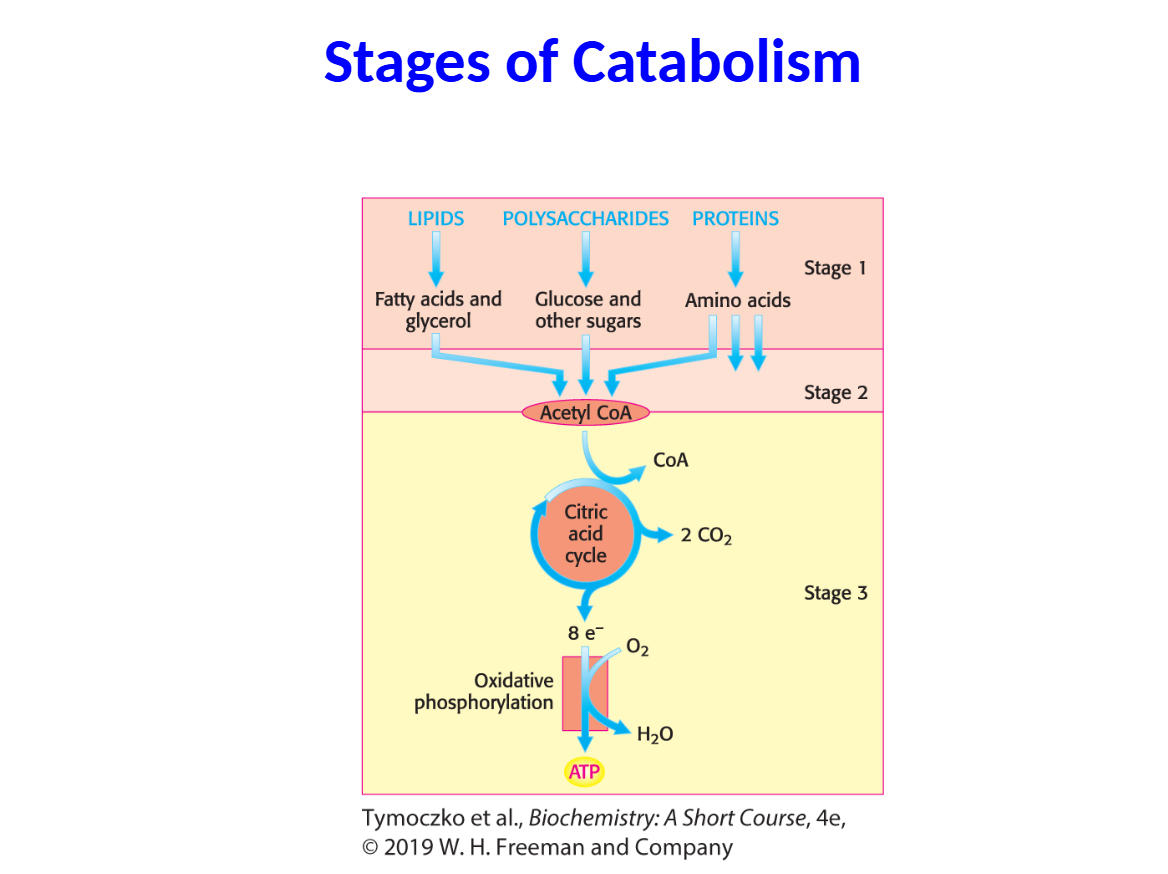
1.Large molecules in food are broken down into smaller molecules in the process of digestion.
2. The many small molecules are processed into key molecules of metabolism, most notably acetyl CoA.
3. ATP is produced from the complete oxidation of acetyl CoA.
Energy is required to power muscle contraction, cell
movement, and biosynthesis.
• Phototrophs obtain energy by capturing sunlight.
• Chemotrophs obtain energy through the oxidation of carbon
fuels

1.Molecules are degraded or synthesized stepwise in a
series of reactions termed metabolic pathways.
2. ATP is the energy currency of life.
3. ATP can be formed by the oxidation of carbon fuels.
4. Although many reactions occur inside a cell, a limited
number of reaction types that involve particular
intermediates are common to all metabolic pathways.
5. Metabolic pathways are highly regulated.
6. The enzymes involved in metabolism are organized into
large complexes.

Metabolism is a series of linked reactions that convert a specific reactant into a specific product.
• The entire set of cellular metabolic reactions are called intermediary metabolism

Metabolic pathways can be divided into two types:
1. Catabolic pathways combust carbon fuels to
synthesize ATP or ion gradients.
2. Anabolic pathways use ATP and reducing power to
synthesize large biomolecules.
NAD+ energy carrier
NADP energy carrier

Some pathways, called amphibolic pathways, can function anabolically or catabolically.
• Although anabolic and catabolic pathways may have reactions in common, the regulated, irreversible reactions are always distinct.

In order to construct a metabolic pathway, two criteria must be met:
1. The individual reactions must be specific.
2. The pathway in total must be thermodynamically favorable.
A thermodynamically unfavorable reaction in a pathway can be
made to occur by coupling it to a more favorable reaction.

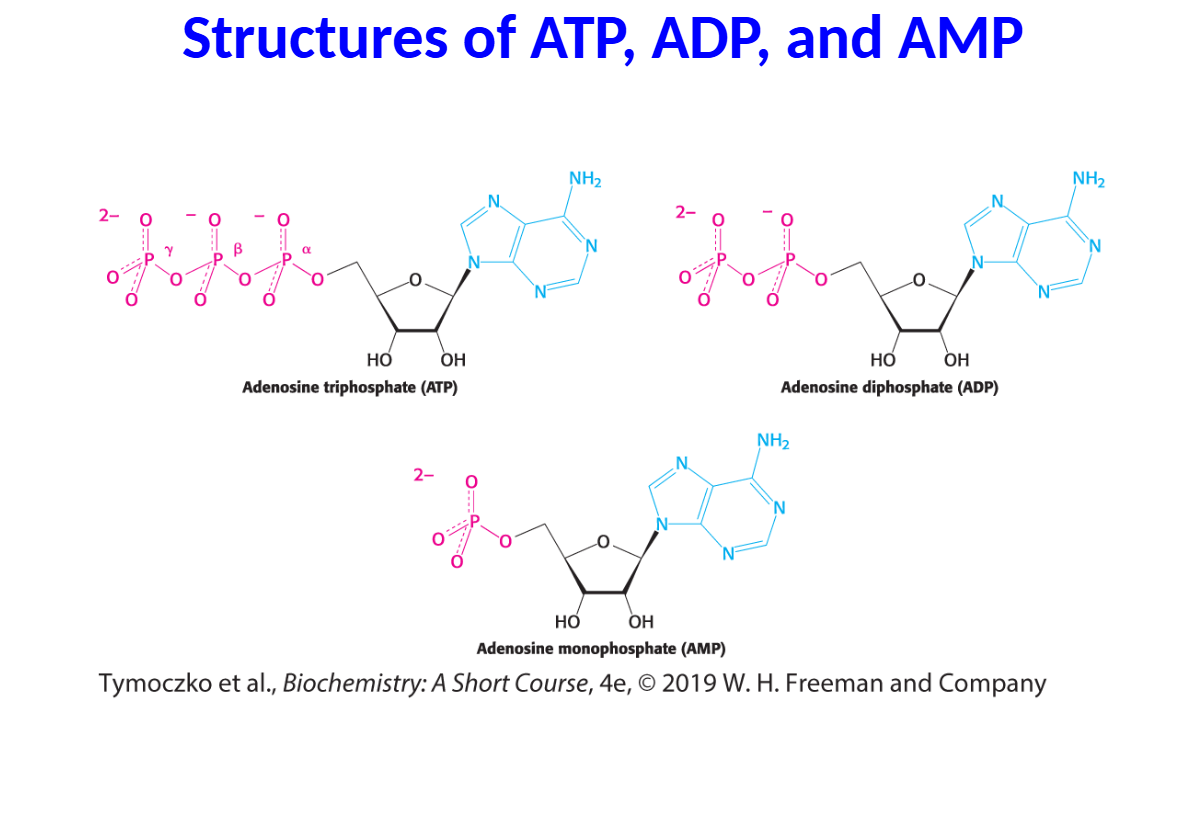
Energy derived from fuels or light is converted into adenosine triphosphate (ATP), the cellular energy currency.

The hydrolysis of ATP is exergonic because the triphosphate unit contains two phosphoanhydride bonds that are unstable.
• The energy released on ATP hydrolysis is used to power a host of cellular functions.


The Standard Free-Energy Change of a Reaction Is Related to the Equilibrium Constant

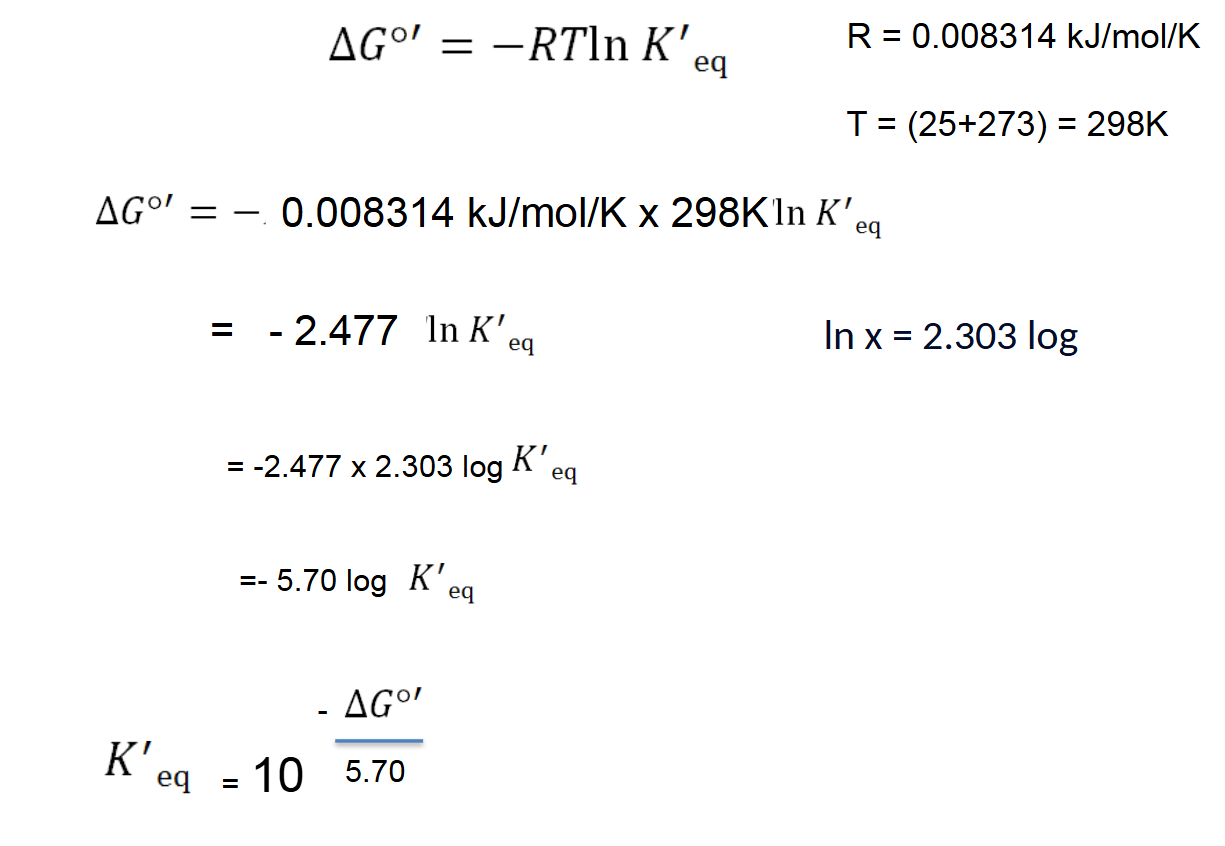


Phosphoryl-transfer potential―the standard free energy of
hydrolysis―is a means of comparing the tendency of
organic molecules to transfer a phosphoryl group to an
acceptor molecule.


ATP has a high phosphoryl-transfer potential because of
four key factors:
1. Charge repulsion
2. Resonance stabilization
3. Increase in entropy
4. Stabilization by hydration
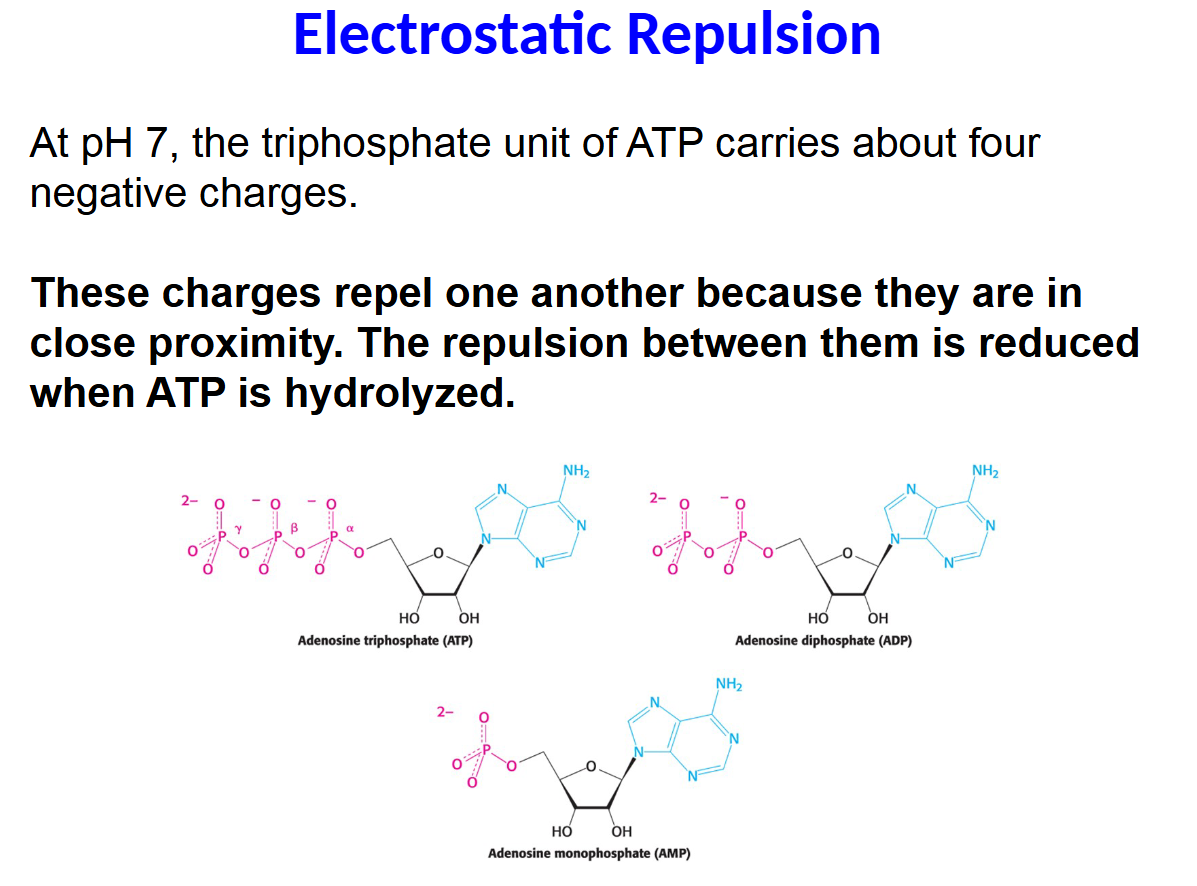
At pH 7, the triphosphate unit of ATP carries about four
negative charges.
These charges repel one another because they are in
close proximity. The repulsion between them is reduced
when ATP is hydrolyzed.
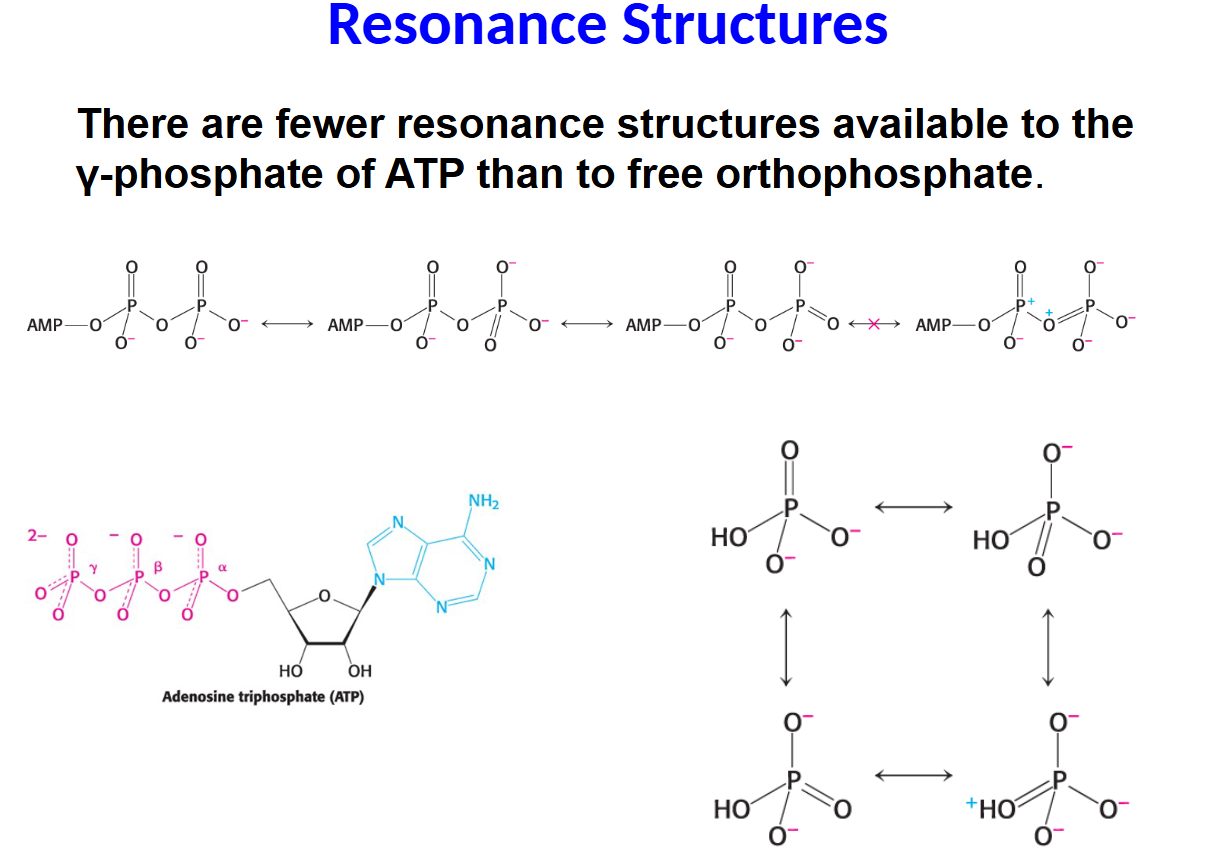
There are fewer resonance structures available to the γ-phosphate of ATP than to free orthophosphate
The entropy of the products (ADP+ Pi) is greater in that there are now two molecules instead of a single ATP molecule.
We disregard the molecule of water used to hydrolyze the ATP because given the high concentration (55.5 M), there is effectively no change in the concentration of water during the reaction

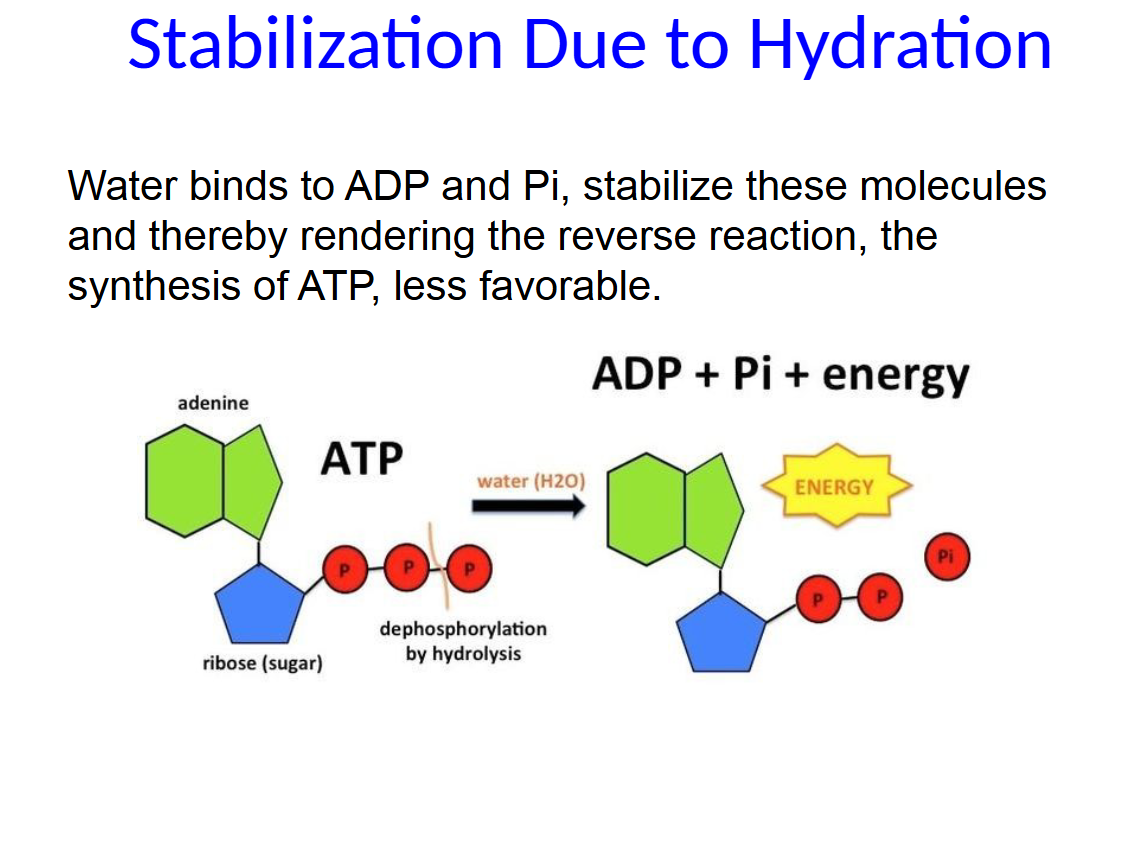
Water binds to ADP and Pi, stabilize these molecules and thereby rendering the reverse reaction, the synthesis of ATP, less favorable.
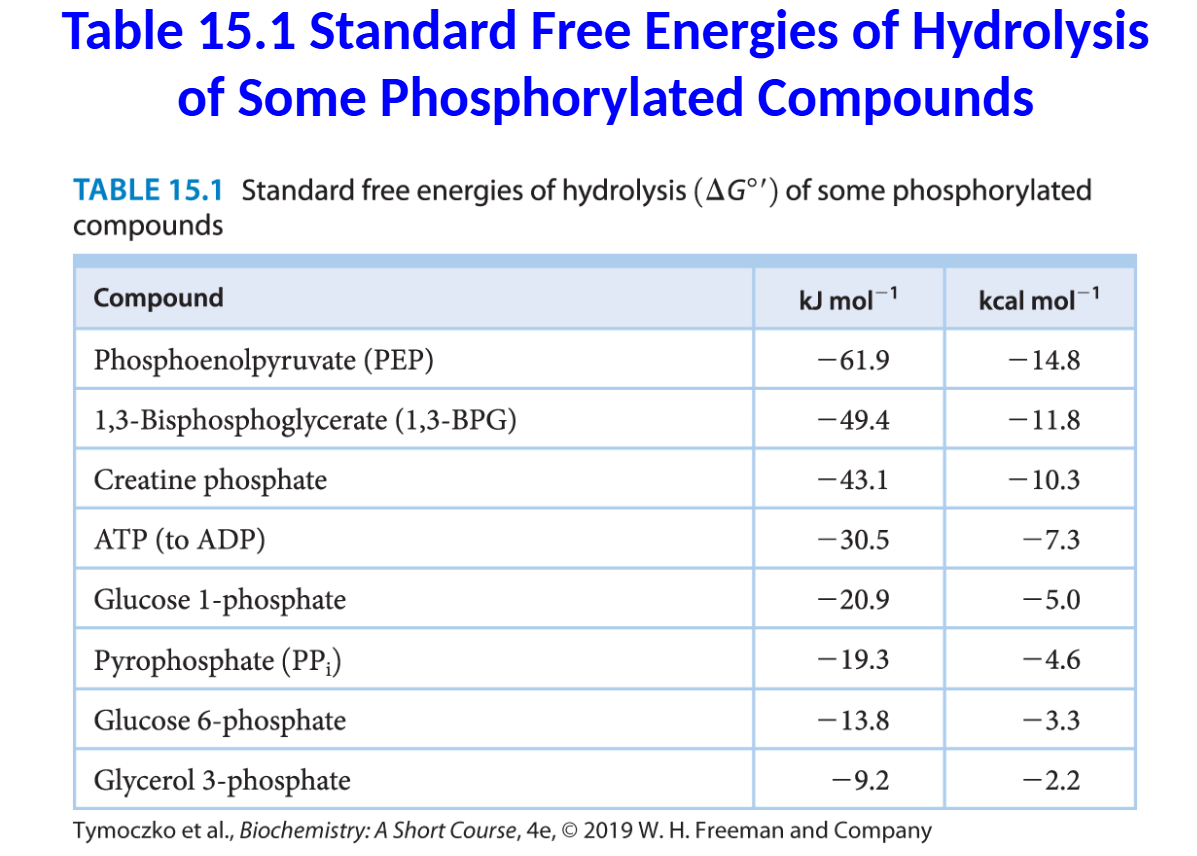
The standard free energies of hydrolysis provide a
convenient means of comparing the phosphoryl-transfer
potential of phosphorylated compounds.
Such comparisons reveal that ATP is not the only compound
with a high phosphoryl transfer potential.
In fact, some compounds in biological systems have a higher
phosphoryl-transfer potential than that of ATP. These
compounds include phosphoenolpyruvate (PEP), 1,3-
bisphosphoglycerate (1,3-BPG), and creatine phosphate

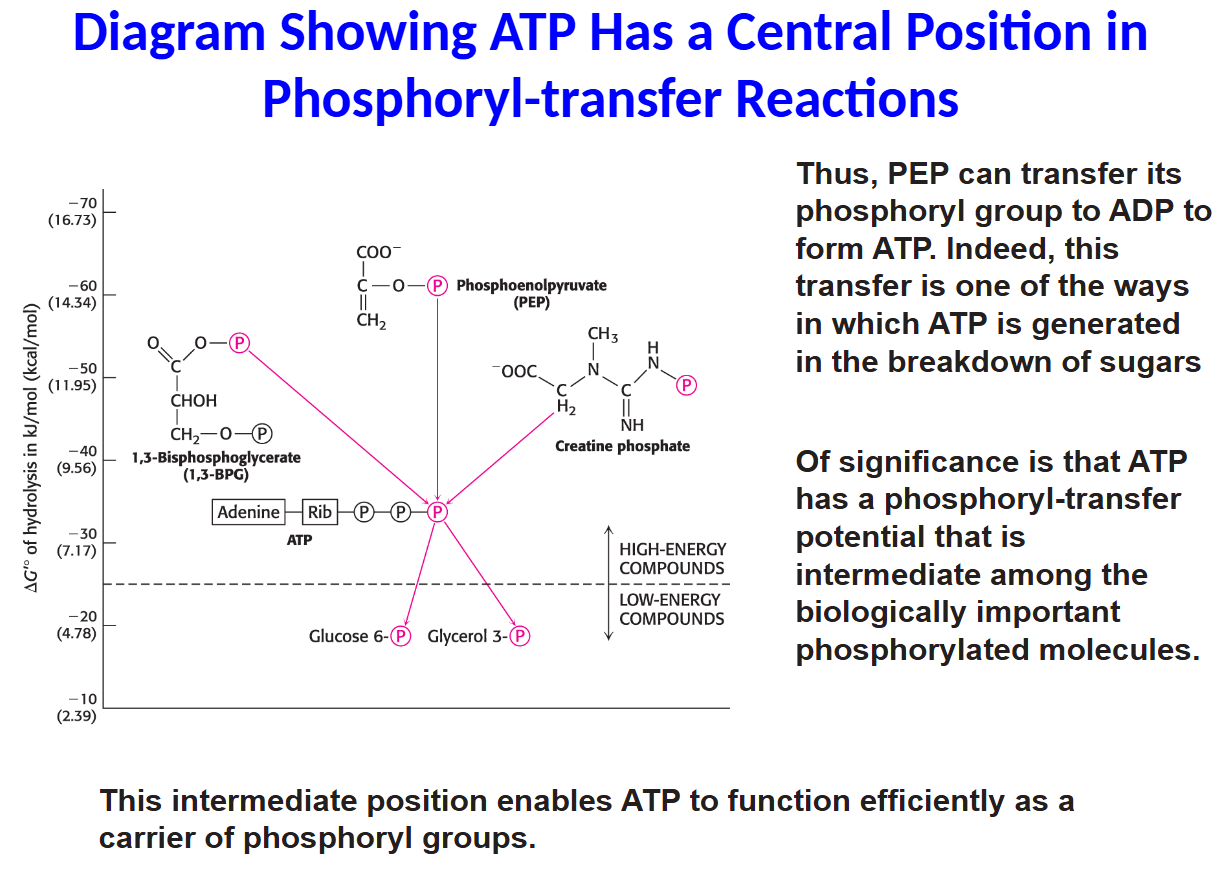
Thus, PEP can transfer its
phosphoryl group to ADP to
form ATP. Indeed, this
transfer is one of the ways
in which ATP is generated
in the breakdown of sugars
Of significance is that ATP
has a phosphoryl-transfer
potential that is
intermediate among the
biologically important
phosphorylated molecules.
This intermediate position enables ATP to function efficiently as a
carrier of phosphoryl groups.
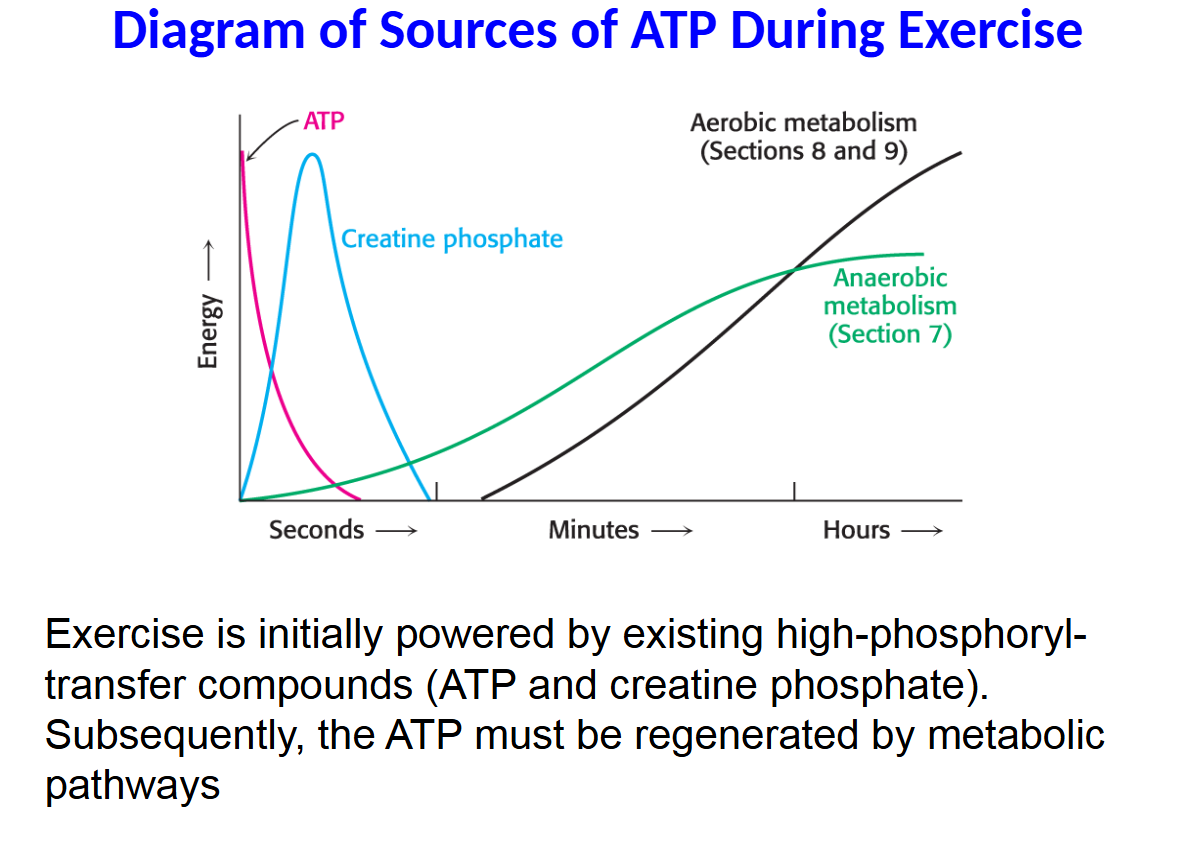
Muscle contains only enough ATP to power muscle contraction for less than a second.
Creatine phosphate can regenerate ATP from ADP, allowing a short burst of activity as in a sprint.
Once the creatine phosphate stores are depleted, ATP must be generated by metabolic pathways.

In resting muscle, typical concentrations of these metabolites are [ATP] = 4 mM,
[ADP] = 0.013 mM, [creatine phosphate] = 25 mM, and [creatine] = 13 mM.
Because of its abundance and high phosphoryl-transfer potential relative to
that of ATP, creatine phosphate is a highly effective phosphoryl buffer.
Indeed, creatine phosphate is the major source of phosphoryl groups for ATP
regeneration for activities that require quick bursts of energy or short, intense
sprints
![<p>In resting muscle, typical concentrations of these metabolites are [ATP] = 4 mM,</p><p>[ADP] = 0.013 mM, [creatine phosphate] = 25 mM, and [creatine] = 13 mM.</p><p>Because of its abundance and high phosphoryl-transfer potential relative to</p><p>that of ATP, creatine phosphate is a highly effective phosphoryl buffer.</p><p>Indeed, creatine phosphate is the major source of phosphoryl groups for ATP</p><p>regeneration for activities that require quick bursts of energy or short, intense</p><p>sprints</p>](https://assets.knowt.com/user-attachments/7d504bbc-3beb-4db5-b002-dc3aedea0abc.png)
Exercise is initially powered by existing high-phosphoryl- transfer compounds (ATP and creatine phosphate). Subsequently, the ATP must be regenerated by metabolic pathways

Assume that the complete combustion of one mole of glucose to carbon dioxide and water liberates 2870 kJ/mol. If one contraction cycle in muscle requires 67 kJ, and the energy from the combustion of glucose is converted with an efficiency of 45% to contraction, how many contraction cycles could theoretically be fueled by the complete combustion of one mole of glucose? Round your answer to the nearest whole number.

Phosphate and its esters are especially prominent in biology for several reasons:
1. Phosphate esters are kinetically stable. Phosphate esters are thus molecules whose energy release can be manipulated by enzymes.
2. Phosphate esters are stable because the inherent negative charges resist hydrolysis in absence of enzyme. This accounts for the presence of phosphate in the backbone of DNA.
3. Because phosphate esters are kinetically stable, they are ideal regulatory molecules, added to molecules by kinases and removed by phosphatases.

Cells maintain a very high concentration of ATP.
• Recent research suggests that ATP may function as a biological hydrotrope.
• ATP prevents the formation of protein aggregates and dissolves those that form, implying that another role of ATP is maintaining protein solubility.


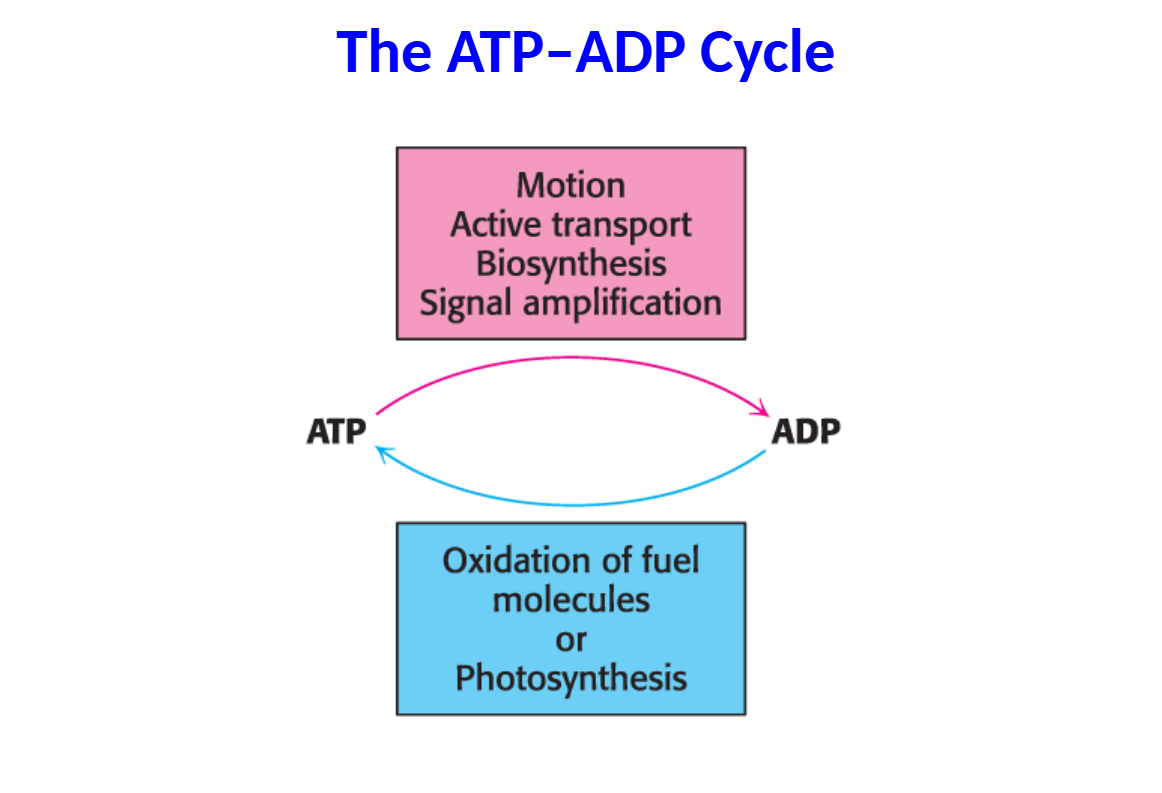
ATP is the immediate donor of free energy for biological activities.
• However, the amount of ATP is limited.
• Consequently, ATP must be constantly recycled to provide energy to power the cell.
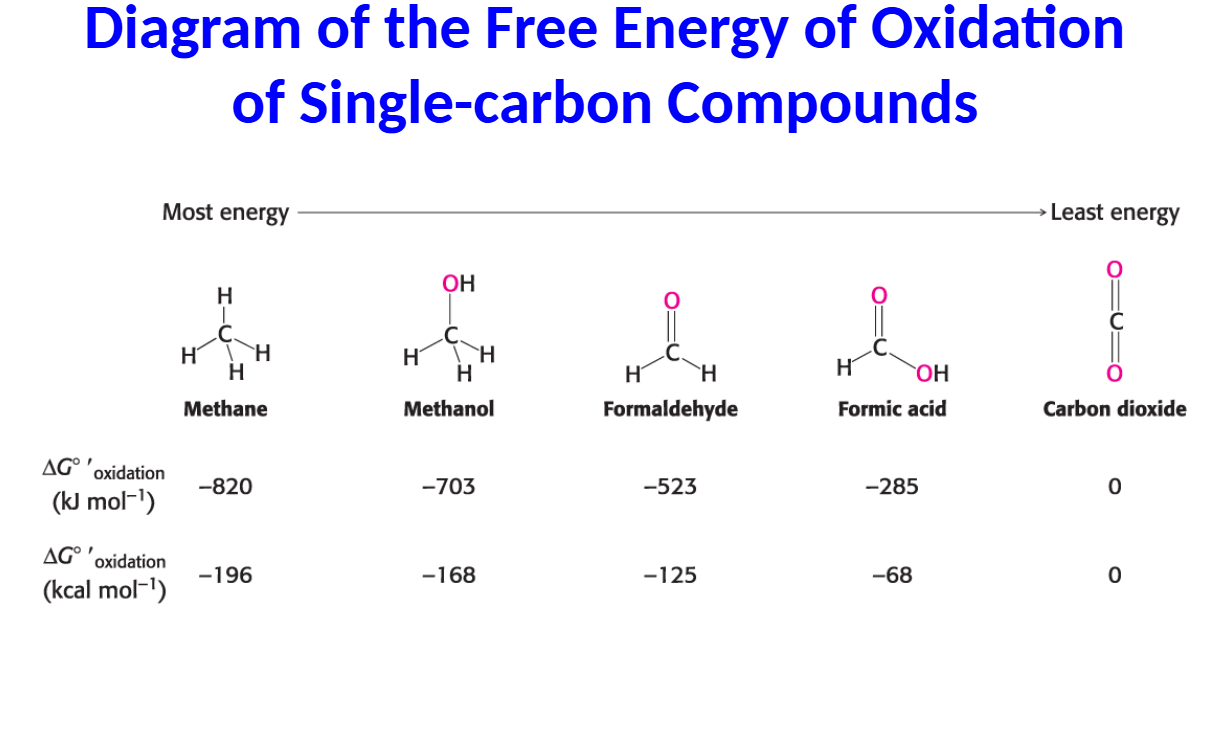
Oxidation reactions involve loss of electrons. Such reactions must be coupled with reactions that gain electrons. The paired reactions are called oxidation–reduction reactions, or redox reactions.
• The carbon atoms in fuels are oxidized to yield CO2, and the electrons are ultimately accepted by oxygen to form H2O.
• The more reduced a carbon atom is, the more free energy is released upon oxidation.
• Fats are a more efficient food source than glucose because fats are more reduced.

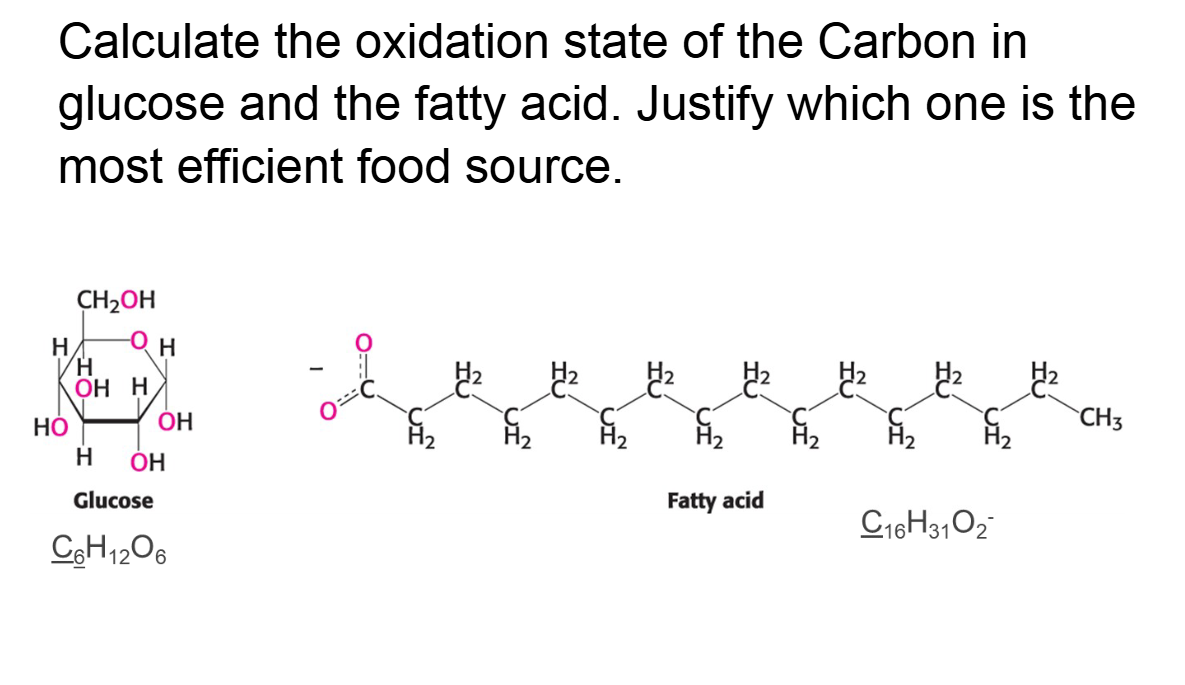
Assign oxidation states to the elements whose atoms are
underlined in each of the following compounds or ions.
Which carbon is in the most reduced form and ranks these
compounds highest to lowest source of energy

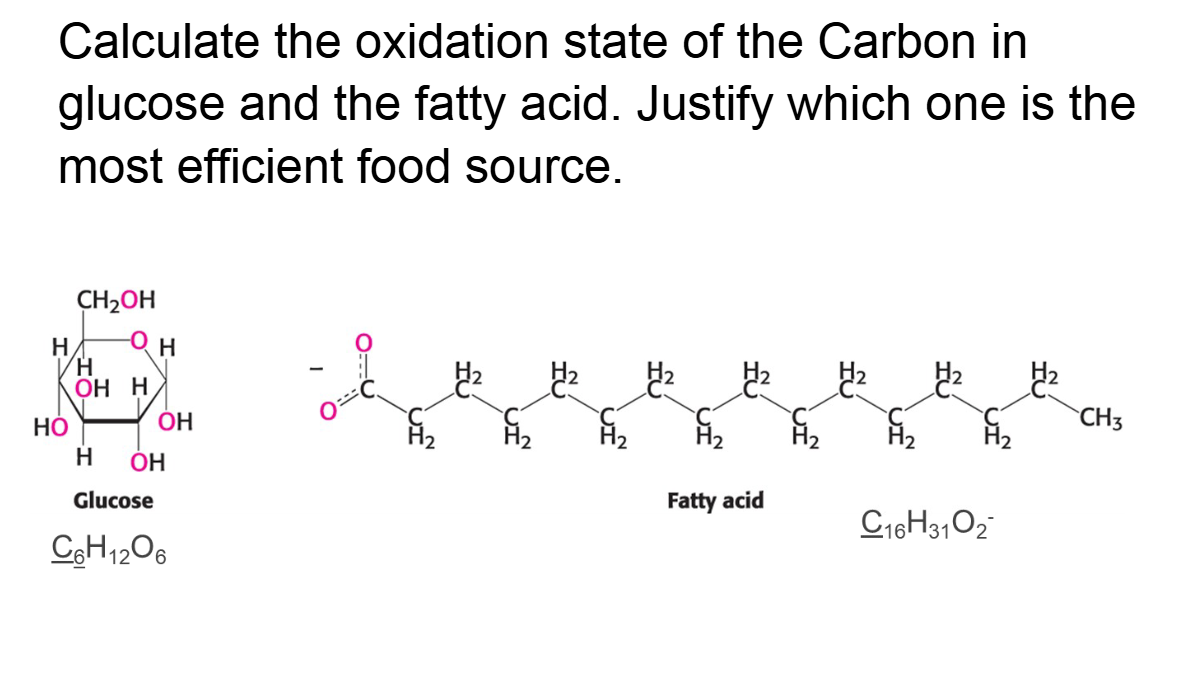
Calculate the oxidation state of the Carbon in glucose and the fatty acid. Justify which one is the most efficient food source.
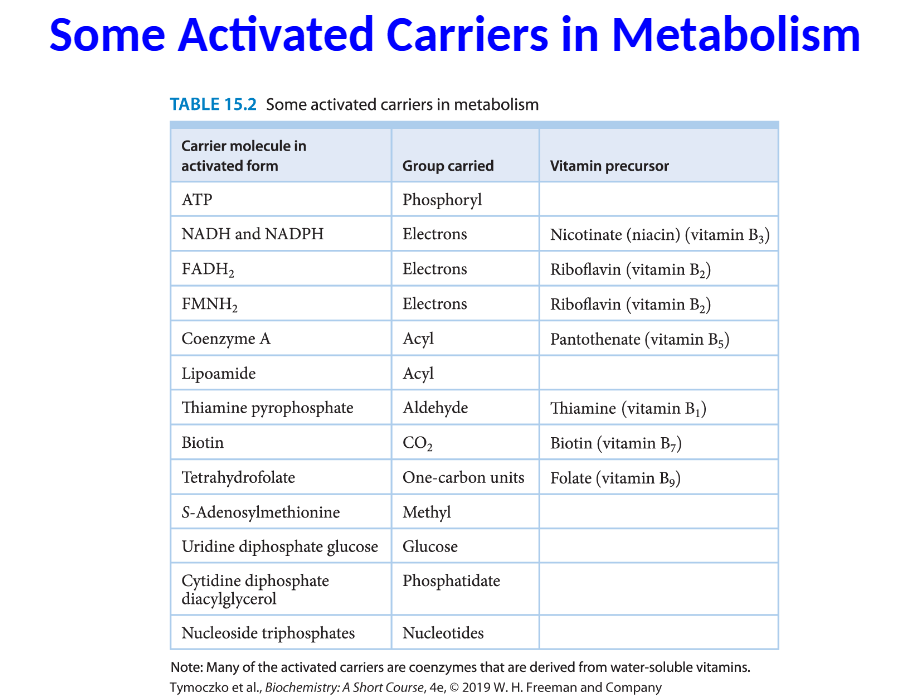
Two characteristics are common to activated carriers:
1. The carriers are kinetically stable in the absence of
specific catalysts.
2. The metabolism of activated groups is accomplished with
a small number of carriers.


ATP is an activated carrier of phosphoryl groups. Other
activated carriers are common in biochemistry, and often
they are derived from vitamins.
• Nicotinamide adenine dinucleotide (NAD+) and flavin
adenine dinucleotide (FAD) carry activated electrons
derived from the oxidation of fuels.
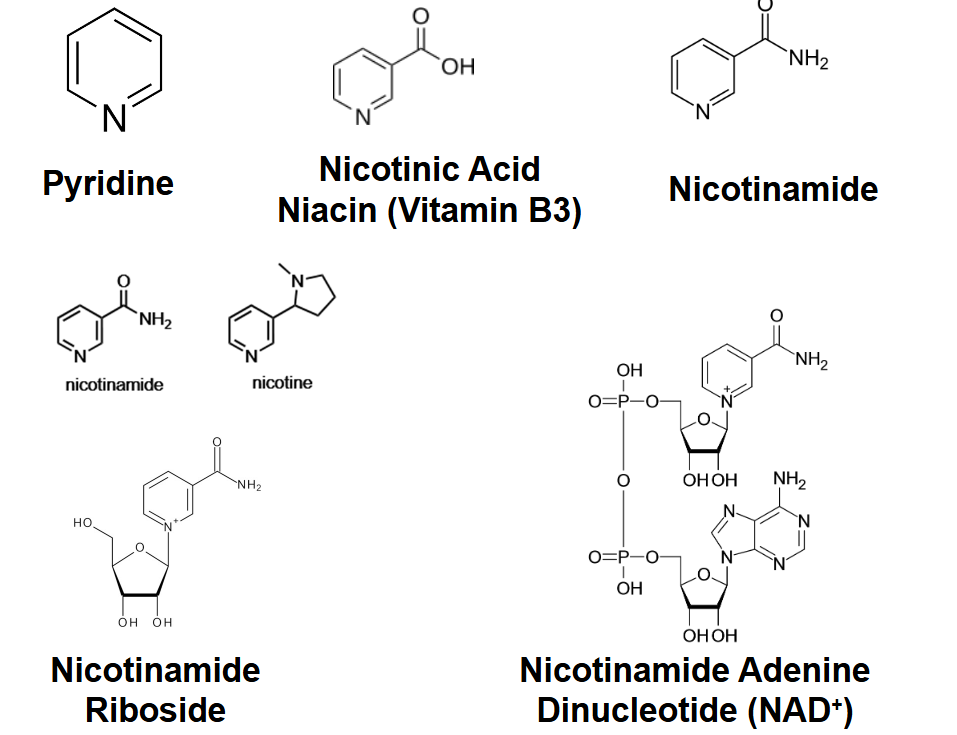
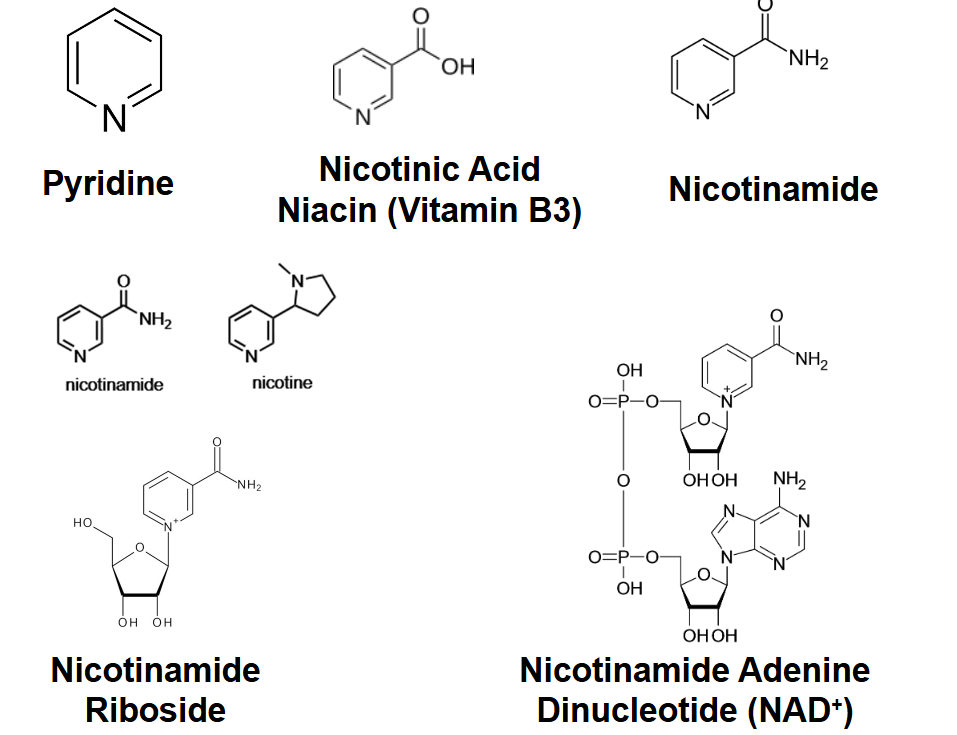
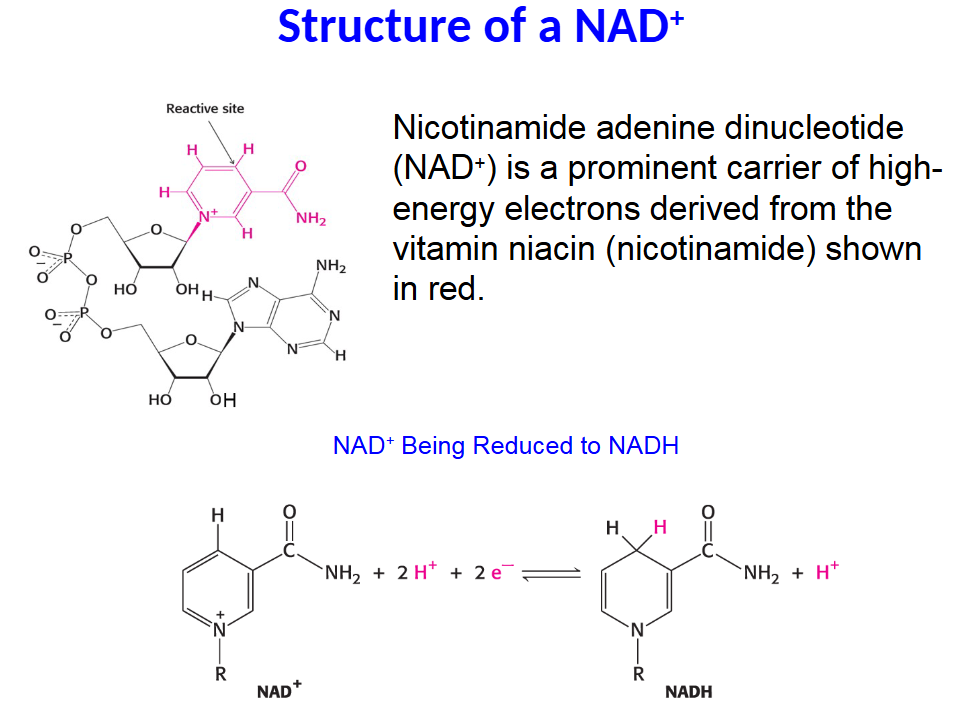
Nicotinamide adenine dinucleotide
(NAD+) is a prominent carrier of high-
energy electrons derived from the
vitamin niacin (nicotinamide) shown
in red.
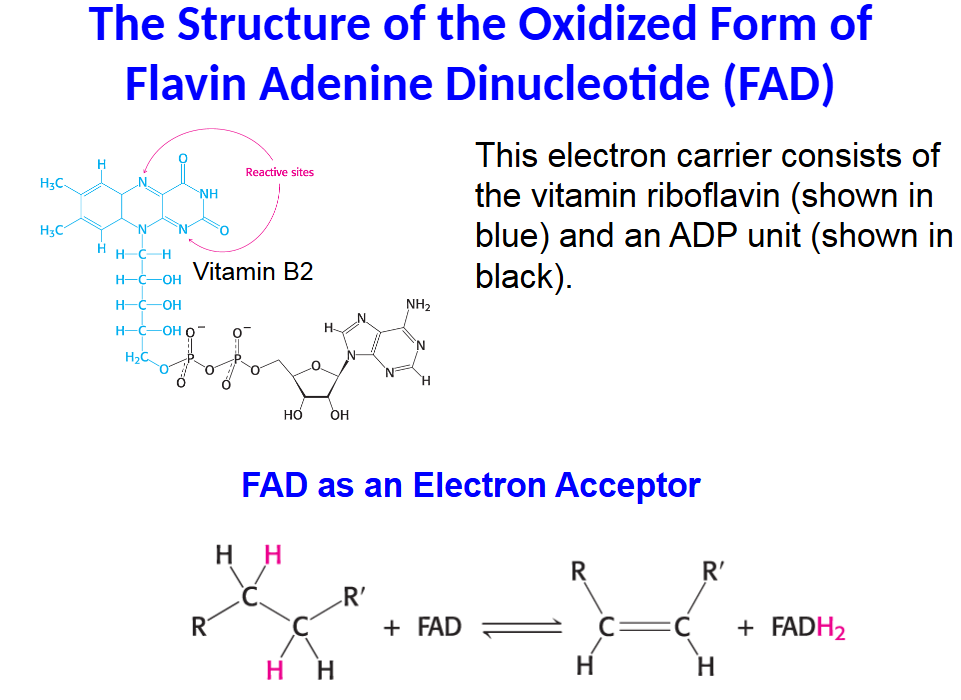
This electron carrier consists of the vitamin riboflavin (shown in blue) and an ADP unit (shown in black).
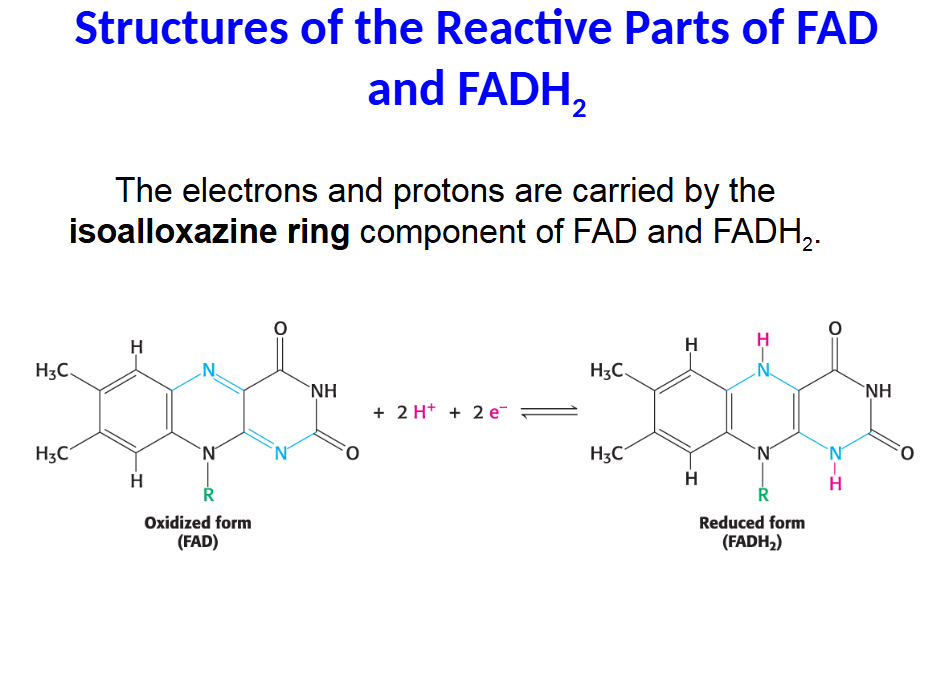
The electrons and protons are carried by the isoalloxazine ring component of FAD and FADH2.
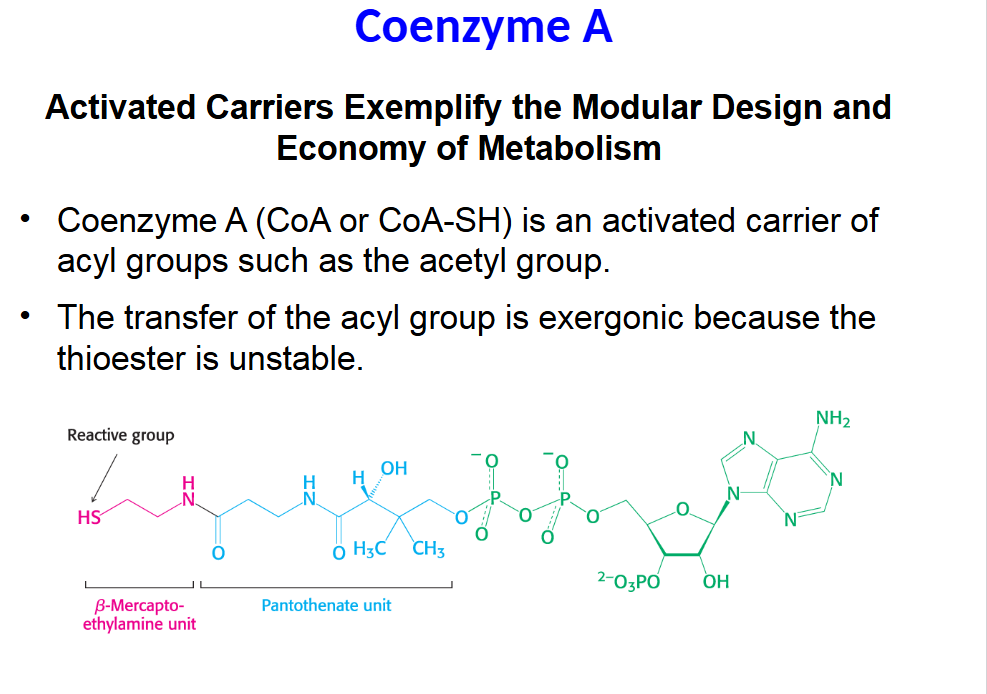
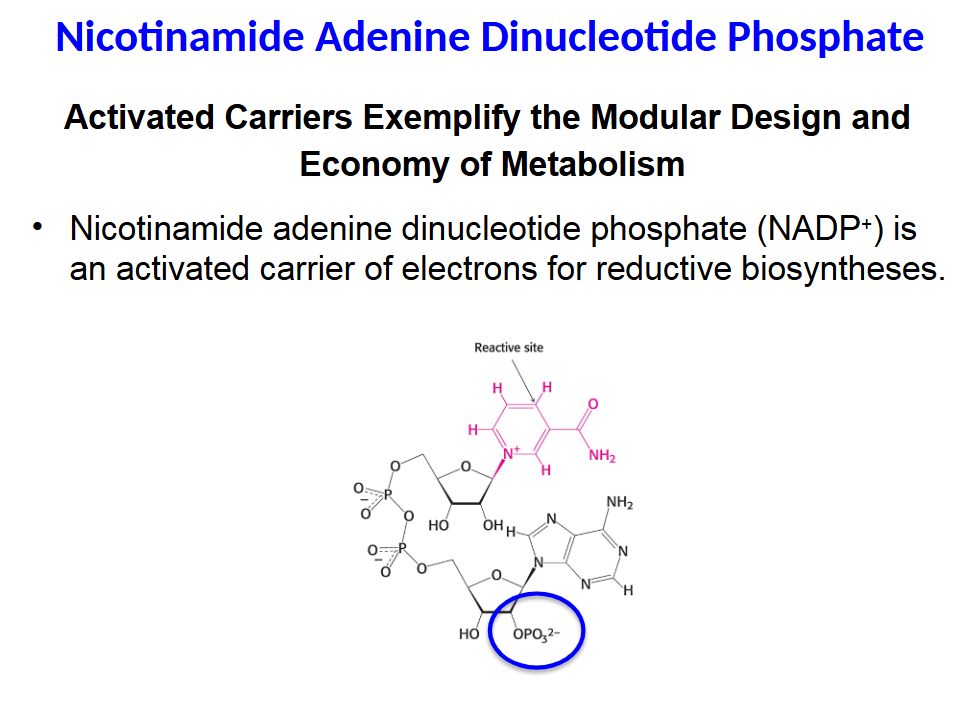
Activated Carriers Exemplify the Modular Design and
Economy of Metabolism
• Nicotinamide adenine dinucleotide phosphate (NADP+) is
an activated carrier of electrons for reductive biosyntheses.
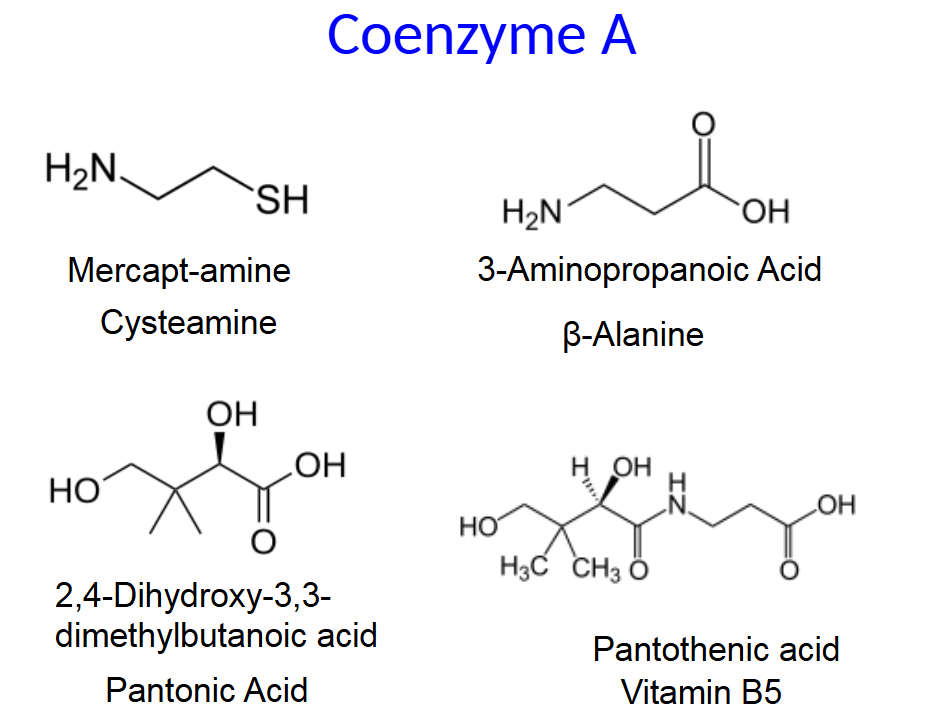
Coenzyme A (CoA or CoA-SH) is an activated carrier of
acyl groups such as the acetyl group.
• The transfer of the acyl group is exergonic because the
thioester is unstable