MSE 2001 Exam 2
1/71
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
72 Terms
The bond-energy curve can be used to gain information about Vaporization temperature?
True
False
True
The bond-energy curve can be used to gain information about Primary bond type?
True
False
False
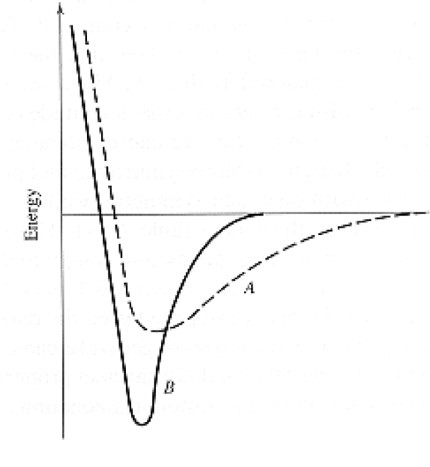
Which materials (A or B) exhibits a higher elastic modulus?
a. A
b. B
b
The steeper the slope of the force curve (at equilibrium distance), the lower the value of E
True
False
False
The value of the inter-atomic distance corresponds to
a. the bond energy is a minimum
b. when the slope of the force distance diagram is zero.
c. The radius of curvature is a minimum
No answer text provided.
a
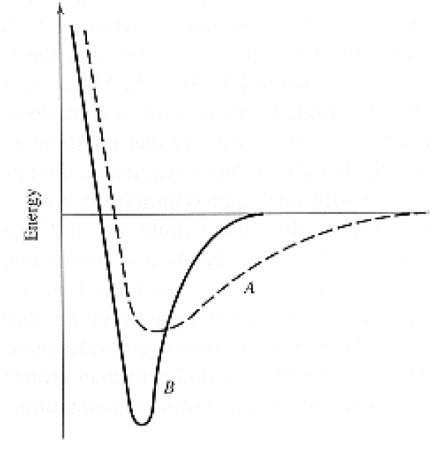
Which materials expands more under the same temperature (higher thermal coefficient of expansion)?
A
B
None of the above
A
Amorphous solids show long range order
a. correct
b. incorrect
b
Thermosets are
A. 3D structures that do not form a melt
B. They are thermoplastics
C. The can stretch to hundreds of their
D. All of the above
A
Primitive cell is a unit cell that contains one atom per cell
a) Correct
b) Incorrect
a
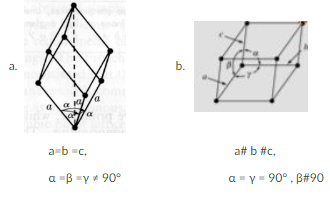
A Rhombohedral crystal structure is
a. a
b. b
c. Neither
a
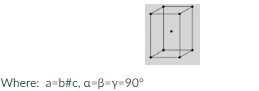
What is the lattice structure in the following diagram?
a. Simple tetragonal
b. Body-centered tetragonal
c. Simple orthorhombic
d. Body-centered orthorhombic
b
In the BCC atomic crystal structure, the only atomic contact is along the face diagonal.
a. True
b. False
b
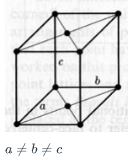
What is the Bravais Lattice for the following
a) cubic
b) hexagonal
c) Base centered Tetragonal
d) Base Centered Orthorhombic
d
What is the number of atoms per unit cell in FCC structures?
a) 1
b) 2
c) 4
c
![<p><span>What is the Miller indices representation of Vector in the z-axis </span></p><p><span>a) [0,0,1] </span></p><p><span>b) (001)</span></p><p><span>c) [001] </span></p><p><span>d) [2,1,1]</span></p>](https://knowt-user-attachments.s3.amazonaws.com/9225b91d-1397-4f82-874e-d9830aec69a5.jpeg)
What is the Miller indices representation of Vector in the z-axis
a) [0,0,1]
b) (001)
c) [001]
d) [2,1,1]
c
![<p><span>What is the Miller indices representation of Vector OF </span></p><p><span>a) [221] </span></p><p><span>b) (112)</span></p><p><span>c) [112] </span></p><p><span>d) 2,1,1</span></p>](https://knowt-user-attachments.s3.amazonaws.com/01e3de2b-c1f5-4874-bcd7-d5934cf2bb5d.jpeg)
What is the Miller indices representation of Vector OF
a) [221]
b) (112)
c) [112]
d) 2,1,1
c
![<p><span>What is the Miller indices representation of Vector OB Vector </span></p><p><span>a) [110] </span></p><p><span>b) (100) </span></p><p><span>c) [111] </span></p><p><span>d) 0,1,1</span></p>](https://knowt-user-attachments.s3.amazonaws.com/3761b69d-cb60-4541-a62d-4c3ccd285453.jpeg)
What is the Miller indices representation of Vector OB Vector
a) [110]
b) (100)
c) [111]
d) 0,1,1
a
![<p>What is the Miller indices representation for Plane A</p><p>a) [001]</p><p>b) (100)</p><p>c) [112]</p><p>d) (001)</p>](https://knowt-user-attachments.s3.amazonaws.com/e32d9473-f40a-489e-9b0f-a55ba1d2ffe3.jpeg)
What is the Miller indices representation for Plane A
a) [001]
b) (100)
c) [112]
d) (001)
d
![<p>What is the Miller indices representation for Plane B</p><p>a) [001]</p><p>b) (111)</p><p>c) [111]</p><p>d) (110)</p>](https://knowt-user-attachments.s3.amazonaws.com/e0fdab98-58cd-4a02-88fb-0cdf44abf98a.jpeg)
What is the Miller indices representation for Plane B
a) [001]
b) (111)
c) [111]
d) (110)
b
![<p>What is the Miller indices representation for Plane C</p><p>a) [001]</p><p>b) (111)</p><p>c) [011]</p><p>d) (110)</p>](https://knowt-user-attachments.s3.amazonaws.com/f0a8b1d3-b75a-43ab-bdd9-4c5031c73f9b.jpeg)
What is the Miller indices representation for Plane C
a) [001]
b) (111)
c) [011]
d) (110)
d
![<p>What is the Miller indices representation for Plane E?</p><p>a) [001]</p><p>b) (102)</p><p>c) [211]</p><p>d) (120)</p>](https://knowt-user-attachments.s3.amazonaws.com/5d257edd-3930-46fc-b4ad-6105355c490c.jpeg)
What is the Miller indices representation for Plane E?
a) [001]
b) (102)
c) [211]
d) (120)
b
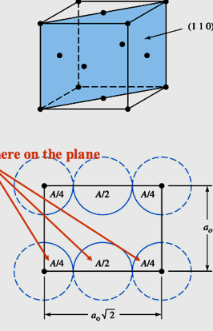
How many atoms do we have within the unit cell in the (110) shown below
a) 1
b) 2
c) 3
d) 4
b
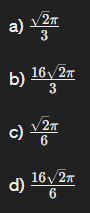
The Atomic Packing Factor of FCC crystal?:
c
The closest packed plane in a FCC Crystal is
a. (111)
b. (110)
c. [110]
d. (010)
a
The volume of a BCC unit cell in terms of the radius r of the atom within the unit cell
a. (4r / √3)³
b. (√3r / 2)³
c. √3 (r/4)³
d. 4r³ / √3
a
The length of the face diagonal [110] in a BCC cell with atomic radius of r is:
a. 4r√6 / 3
b. 2r√2 / √3
c. 2r√3 / √2
a
The linear density along 111 direction in the FCC single crystal
a. √3 / 3
b. √2 / (4√3r)
c. √3 / (4√2r)
d. √3 / (12r)
b
The planar density for the 111 closest packed plane in FCC structure
a. 2√3 / r²
b. 1 / (2√3r²)
c. 2 / (√3r²)
b
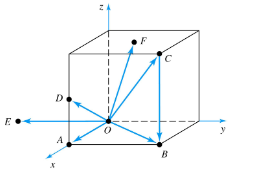
What is the cos θ between point OB and OF vectors?
a. √3/2
b. √3
c. 1/√3
d. -1/√3
c
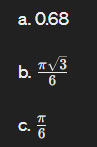
Calculate the APF of SC
c
The classification of crystal structures is based on atoms of same kind when replace the lattice
True
False
True
Which of the following is a closest packed plane in a HCP structure?
a. (0 1 1)
b. (1 0 1)
c. (2 1 0)
d. (0 0 1)
d
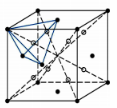
What is the interstitial position shown in the diagram?
a. Octahedral position
b. hexahedral position
c. tetrahedral position
c
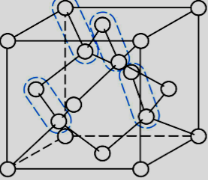
How many atoms are there in a unit cell of a diamond structure?
a. 4 atoms
b. 6 atoms
c. 8 atoms
d. 4-10 atoms
c
Diffractions occur when the scattered waves are:
a) In-phase
b) Out of phase
c) Have a phase lag
d) None of the above
a) In-phase
What is Bragg's law?
a) 2d=λsin2θ
b) nλ=dsin2θ
c) nλ=2dsinθ
d) None of the above
c) nλ=2dsinθ
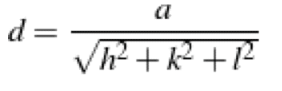
What is the interplanar spacing for the (321) family of planes in a cubic system with a lattice parameter of 2Å?
a) "2Å" /√14
b) "2Å" /√7
c) "2Å" /14
d) "2Å" /7
a) "2Å" /√14
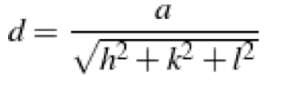
The first plane that may be observed in a diffraction for a cubic structure is:
nλ=2dsinθ
a) (110)
b) (100)
c) (200)
d) None of the above
b) (100)
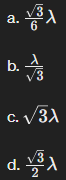
In an x-ray diffraction experiment for a single crystal, a diffraction peak for a specific plane can be obtained by having the incident beam making the proper angle with the plane of interest according to Bragg's law. For a single crystal of an FCC structure with a lattice parameter of 3Å.
The corresponding sin of the angle of diffraction for the (111) family of planes.
a. √3/6 λ
b. λ/√3
c. √3λ
d. √3/2 λ
a. √3/6 λ
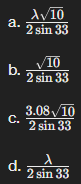
A cubic crystal shows a diffraction maximum from copper radiation, λ at θ=33, corresponding to diffraction from (130):
The lattice parameter is:
a
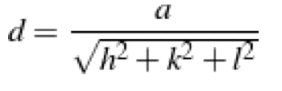
The second plane that may be observed in a diffraction for a cubic structure is:
nλ=2dsinθ
a) (110)
b) (100)
c) (200)
d) (111)
a) (110)
(Multiple Answers) The second plane that can be observed in a tetragonal structure, a=b=1, c=2 A is
a. (100)
b. (010)
c. (001)
d. (110)
e. (101)
f. 200
a and b
(Multiple Answers) The third plane that can be observed in a tetragonal structure is
a. (100)
b. (010)
c. (001)
d. (110)
e. (101)
f. 011
e and f
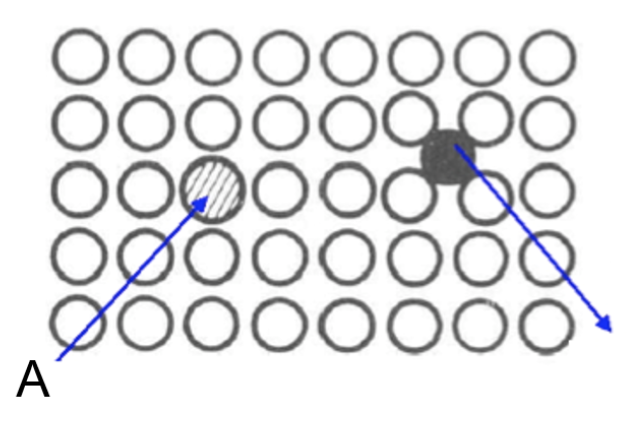
What is the type of defect in A?
a) Substitutional
b) Interstitial
a) Substitutional
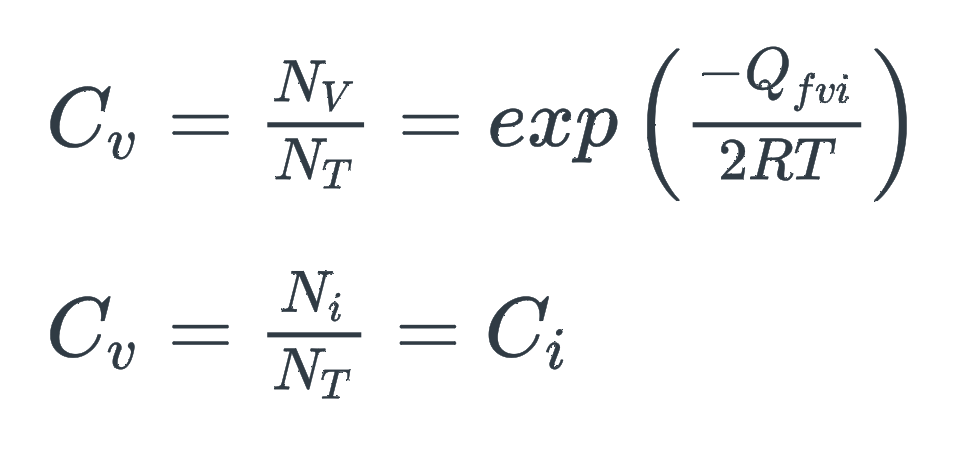
What is C in the following equations
a) The mass flux
b) Concentration
c) Density rate
b) Concentration

Atoms colored as blue are
a) Defect
b) Interstitial solid solution
c) Solvent
d) Solute
c) Solvent
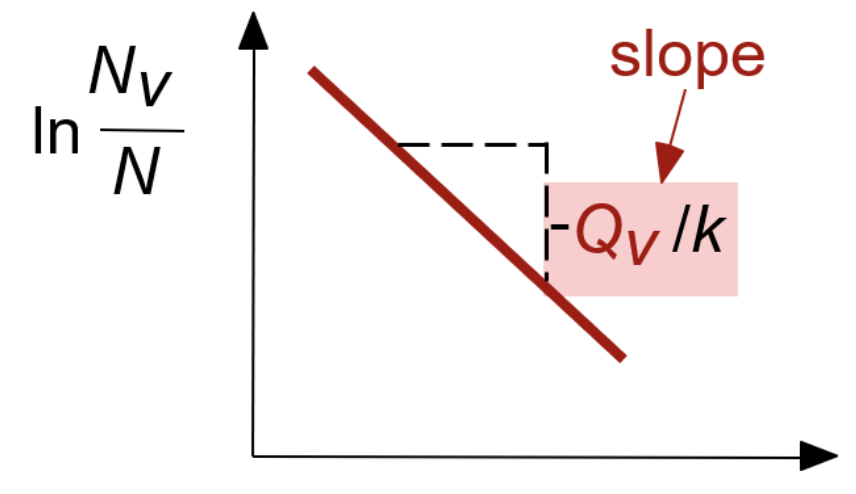
What is the x-axis in the following plot?
a) Temperature (T)
b) 1/T
c) time (t)
d) 1/t
b) 1/T
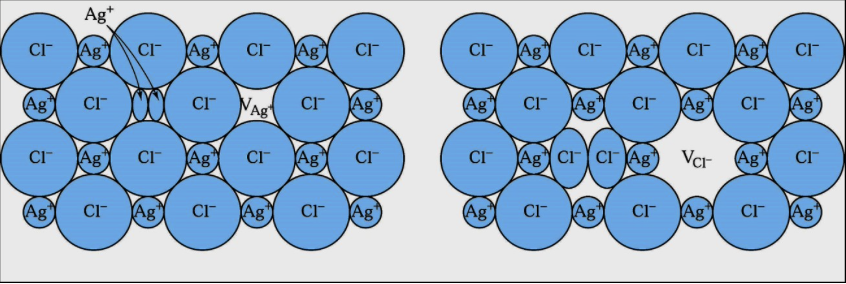
For the following configuration decide the type of defect
a. Schottky
b. Frenkel
b. Frenkel
To form a substitutional solid solution, the size difference between the solute and solvent must be no greater than
a. 1.5%
b. 5%
c. 15%
d. 30%
c. 15%
For Interstitial solid solution, we should have comparable
a. Electro-negativity
b. Valence
c. Crystal structure
d. None of the above
a. Electro-negativity
It is common that cations transfer into interstitial sites since they are smaller in size compared to anions
a. True
b. False
a. True
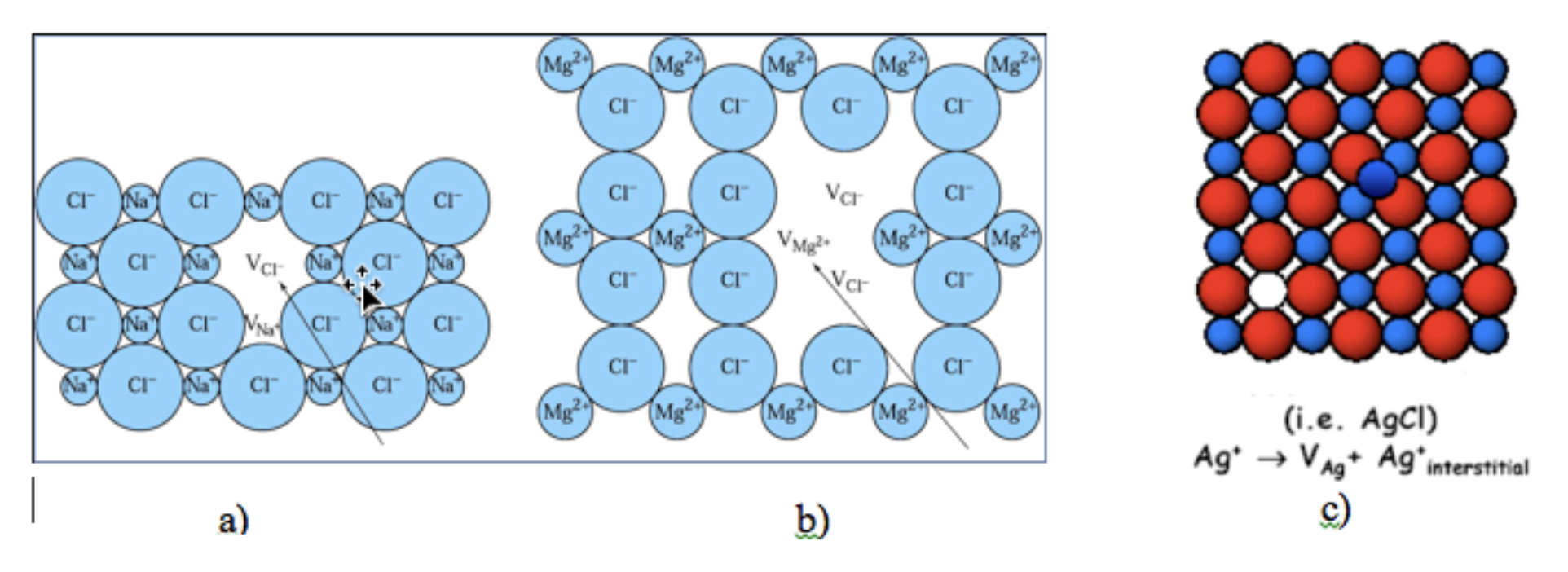
In the following identify the type of defect is presented in a:
a. Schottky
b. Frenkel
a. Schottky
For the flux J in Fick's first law, what does the negative sign denote?
a. Mass flow from high to low concentration
b. Mass flow from low to high concentration
c. Negative Temperature
d. Pressure lower than the standard atmospheric pressure
a.
The coefficient of diffusion for Cu in Ag for a given temperature is 0.8 cm²/s. Two planes in an Ag crystal are 2 mm apart. The concentration of Cu is 7.4 x 10^-18 atoms/cm³ on the first plane and 5.2 x 10^-20 atoms/cm³ on the second plane. What is the flux of Cu atoms between the two planes? Assume that the concentrations are constant.
a. (0.8 cm²/s)*((7.4x10^-18)-(5.2x10^-20)) atoms/cm³/(0.2 cm)
b. (5.2 cm²/s)*((7.4x10^-18)-(5.2x10^-20)) atoms/cm³/(0.2 cm)
c. (0.8 cm²/s)*((5.2x10^-18)-(7.4x10^-20)) atoms/cm³/(0.2 cm)
d. (0.8 cm²/s)*((7.4x10^-18)) atoms/cm³/(0.2 cm)
a.
In Fick's first Law, J has units of:
a. atoms/(cm² - sec)
b. cm²/sec
c. atoms/sec
d. atoms/cm³ - L
a.
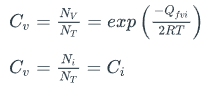
What is C in the following equations
a. The mass flux
b. Concentration
c. Density rate
b.
Fick’s First law is:
a. D = -JC
b. J = -C ∂D/∂x
c. J = -D ∂C/∂x
d. J = -D ∂²C/∂x²
c.
Effective Diffusion Penetration Distance is at which
a) the conc. of diffusing species = average of initial conc. and the surface conc.
b) The material stops diffusing
c) 2 mm below the surface
a)
When using Fick's Second Law, concentration is produced as a function of:
a. Temperature and Time
b. Temperature and Flux
c. Position and Time
d. Position and Flux
c.

In the following equation, τ is the:
a. Normal stress
b. Resolved shear stress
c. Applied force
b.
![<p>In the following slip occurs in the direction</p><p>a. [110]</p><p>b. [111]</p><p>c. [1<sup>-</sup>10]</p>](https://knowt-user-attachments.s3.amazonaws.com/0dbfc764-d2fb-42d9-b549-137b6a1d0406.jpeg)
In the following slip occurs in the direction
a. [110]
b. [111]
c. [1-10]
a.
The closest packed planes and directions in BCC materials are
a) {111} <110>
b) {110} <111>
c) {100} <110>
d) {110} <100>
b)
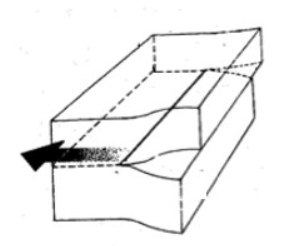
This is an example of
a) Screw dislocation
b) Edge dislocation
a)
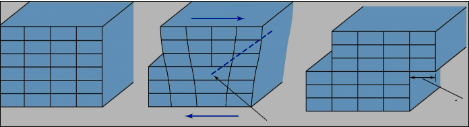
The Dashed line below is
a) Dislocation line
b) Burger's vector
c) Line of dislocation motion
a)
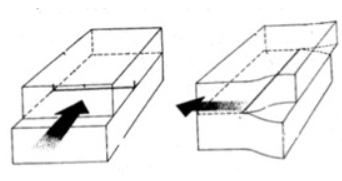
In Screw dislocations
a) Burger's vector is parallel to dislocation line
b) Burger's vector is perpendicular to dislocation line
a)
![<p>Suppose you have an FCC metal single crystal which is known to have a critical resolved shear stress of 2 MPa. Assume that a force is applied to a bar of this material in the [112] direction. The largest shear stress occurs when dislocations begin to move in the [10 -1] direction on the (111) plane. Calculate the cosine of the angle between the external force and the dislocation direction</p>](https://knowt-user-attachments.s3.amazonaws.com/9d875188-da1f-4fec-87ea-2c0016d244f5.jpeg)
Suppose you have an FCC metal single crystal which is known to have a critical resolved shear stress of 2 MPa. Assume that a force is applied to a bar of this material in the [112] direction. The largest shear stress occurs when dislocations begin to move in the [10 -1] direction on the (111) plane. Calculate the cosine of the angle between the external force and the dislocation direction
a
![<p><span>Suppose you have an FCC metal single crystal which is known to have a critical resolved shear stress of 2 MPa. Assume that a force is applied to a bar of this material in the [112] direction. The largest shear stress occurs when dislocations begin to move in the [10 -1] direction on the (111) plane. The cosine of the angle between the external force and the slip plane is </span></p>](https://knowt-user-attachments.s3.amazonaws.com/0ece1f7b-04de-4e2e-b1c7-90133fb4ea6e.jpeg)
Suppose you have an FCC metal single crystal which is known to have a critical resolved shear stress of 2 MPa. Assume that a force is applied to a bar of this material in the [112] direction. The largest shear stress occurs when dislocations begin to move in the [10 -1] direction on the (111) plane. The cosine of the angle between the external force and the slip plane is
c
![<p><span>Suppose you have an FCC metal single crystal which is known to have a critical resolved shear stress of 2 MPa. Assume that a force is applied to a bar of this material in the [112] direction. The largest shear stress occurs when dislocations begin to move in the [10 -1] direction on the (111) plane. </span></p><p><span>The normal (applied) stress to cause for the dislocation to move in the direction on the (111) plane. </span></p>](https://knowt-user-attachments.s3.amazonaws.com/504751d3-2fcc-4a3f-9f49-3cbbdbd47944.jpeg)
Suppose you have an FCC metal single crystal which is known to have a critical resolved shear stress of 2 MPa. Assume that a force is applied to a bar of this material in the [112] direction. The largest shear stress occurs when dislocations begin to move in the [10 -1] direction on the (111) plane.
The normal (applied) stress to cause for the dislocation to move in the direction on the (111) plane.
a
List the secondary bonds from strongest to weakest
a. Van der Waals, Hydrogen Bond, Permanent Dipoles
b. Hydrogen Bond, Van der Waals
b
Which of the following statements are correct
a) If Na atoms bond to form a solid, we get an ionic bond
b) A solid of Na atoms is more conducting than a solid compound containing NaCl atoms
c) A covalent bonding material is more ductile than a metallic bonding material
b
The difference between Van Der Waals and Hydrogen bond is that
a. Van Der Waals bonding is stronger
b. Van Der Waals Bonding is more permanent
c. None of the above
c
-ΔEN equal to 1.7 means:
A. Ionic bonding
B. Covalent bonding
C. Metallic bonding
D. Not enough information
D